Abstract
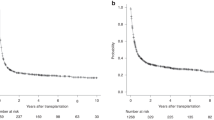
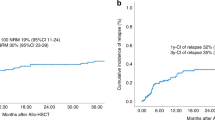
Autologous hematopoietic stem cell transplantation (auto-HSCT) is the standard of care for patients with diffuse large B-cell lymphoma (DLBCL) who relapse/progress after first line chemoimmunotherapy. Long-term outcome of those who relapse after transplant is poor. We present the results of a retrospective study of 256 adult patients reported to the EBMT registry with DLBCL who relapsed after auto-HSCT performed between 2003 and 2013, and who received active salvage strategies. One hundred and fifty-four (60%) were male; median age was 53 years. Median time to relapse was 7 months, 65% relapsed during the first year. Overall response rate after salvage therapy was 46%. Median follow-up after first salvage therapy was 40 months (IQR 23–63 months). Overall survival (OS) at 3 years was 27% (95% CI 22–33). OS at 3 years of patients relapsing longer than 1 year after auto-HSCT was 41% (95% CI 31–53) compared with 20% (95% CI 14–24) in those who relapsed in less than 1 year. Eighty-two patients (32%) had a second HSCT, an allogeneic HSCT (allo-HSCT) in 69 cases, at a median time of 6.5 months after relapse. OS at 3 years after allo-HSCT was 36% (95% CI 25–51). In conclusion, the prognosis of patients with DLBCL that relapse after auto-HSCT is dismal. Patients who relapse in less than 1 year remain an unmet need, and should be considered for CAR T cell therapy or clinical trials. Patients who relapse after 1 year can be rescued with salvage therapies and a second HSCT. These results provide a benchmark to compare data of new prospective studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO Classification of Tumours of Hematopoietic and lymphoid Tissue. IARC: Lyon 2008.
Salar A, Fernández de Sevilla A, Romagosa V, Domingo-Claros A, González-Barca E, de Sanjosé S, et al. Distribution and incidence rates of lymphoid neoplasms according to the REAL classification. A prospective study of 940 cases. Eur J Haematol. 1997;59:231–237.
Coiffier B, Lepage E, Briere J, Herbrecht R, Tilly H, Bouabdallah R, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:235–242.
Pfreundschuh M, Trümper L, Osterborg A, Pettengell R, Trneny M, Imrie K, et al. MabThera International TrialGroup: CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006;7:379–391.
Philip T, Guglielmi C, Hagenbeek A, Somers R, Van der Lelie H, Bron D, et al. Autologous bone marrow transplantation as compared with salvage chemotherapy in relapses of chemotherapy-sensitive non-Hodgkin’s lymphoma. N Engl J Med. 1995;333:1540–1545.
Prince HM, Imrie K, Crump M, Stewart AK, Girouard C, Colwill R, et al. The role of intensive therapy and autologous blood and marrow transplantation for chemotherapy-sensitive relapsed and primary refractory non-Hodgkin’s lymphoma: identification of major prognostic groups. Br J Haematol. 1996;92:880–889.
Nagle SJ, Woo K, Schuster SJ, Nasta SD, Stadtmauer E, Mick R, et al. Outcome of patients with relapsed/refractory diffuse large B-cell lymphoma with progression of lymphoma after autologous stem cell transplantation in the rituximab era. Am J Hematol. 2013;88:890–894.
Van Den Neste E, Schmitz N, Mounier N, Gill D, Linch D, Trneny M, et al. Outcomes of diffuse large B-cell lymphoma patients relapsing after autologous stem cell transplantation: an analysis of patients included in the CORAL study. Bone Marrow Transplant. 2017;52:216–221.
Kim JW, Kim SW, Tada K, Fukuda T, Lee JH, Lee JJ, et al. Allogeneic stem-cell transplantation in patients with de novo diffuse large B-cell lymphoma who experienced relapse or progression after autologous stem cell transplantation: a Korea–Japan collaborative study. Ann Hematol. 2014;93:1345–1351.
Rigacci L, Puccini B, Dodero A, Iacopino P, Castagna L, Bramanti S, et al. Allogeneic hematopoietic stem cell transplantation in patients with diffuse large B-cell lymphoma relapsed after autologous stem cell transplantation: a GITMO study. Ann Hematol. 2012;91:931–939.
Van Kampen RJ, Canals C, Schouten HC, Nagler A, Thomson KJ, Vernant JP, et al. Allogeneic stem-cell transplantation as salvage therapy for patients with diffuse large B-cell non-Hodgkin’s lymphoma relapsing after an autologous stem-cell transplantation: an analysis of the European Group for Blood and Marrow Transplantation Registry. J Clin Oncol. 2011;29:1342–1348.
Fenske TS, Ahn KW, Graff TM, DiGilio A, Bashir Q, Kamble RT, et al. Allogeneic transplantation provides durable remission in a subset of DLBCL patients relapsing after autologous transplantation. Br J Haematol. 2016;174:235–248.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene Ciloleucel CAR T-Cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531–2544.
Schuster SJ, Svoboda J, Chong EA, Nasta SD, Mato AR, Anak Ö, et al. Chimeric antigen receptor T cells in refractory B-cell lymphomas. N Engl J Med. 2017;377:2545–2554.
Sureda A, Bader P, Cesaro S, Dreger P, Duarte RF, Dufour C, et al. Indications for allo- and auto-SCT for haematological diseases, solid tumours and immune disorders: current practice in Europe, 2015. Bone Marrow Transplant. 2015;50:1037–1056.
Gisselbrecht C, Glass B, Mounier N, Singh Gill D, Linch DC, Trneny M, et al. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J Clin Oncol. 2010;28:4184–4190.
Vose JM, Habermann TM, Czuczman MS, Zinzani PL, Reeder CB, Tuscano JM, et al. Single agent lenalidomide is active in patients with relapsed or refractory aggressive non-Hodgkin lymphoma who received prior stem cell transplantation. Br J Haematol. 2013;162:639–647.
Ohmachi K, Niitsu N, Uchida T, Kim SJ, Ando K, Takahashi N, et al. Multicenter phase II study of bendamustine plus rituximab in patients with relapsed or refractory diffuse large B-cell lymphoma. J Clin Oncol 2013;31:2103–2109.
Zinzani PL, Pellegrini C, Argnani L, Broccoli A. Prolonged disease-free survival in elderly relapsed diffuse large B-cell lymphoma patients treated with lenalidomide plus rituximab. Haematologica 2016;101:e385–386.
Hernandez-Ilizaliturri FJ, Deeb G, Zinzani PL, Pileri SA, Malik F, Macon WR, et al. Higher response to lenalidomide in relapsed/refractory diffuse large B-cell lymphoma in non germinal center B-cell-like than in germinal center B-cell-like phenotype. Cancer 2011;117:5058–5066.
Martelli M, Ferreri AJ, Agostinelli C, Di Rocco A, Pfreundschuh M, Pileri SA. Diffuse large B-cell lymphoma. Crit Rev Oncol Hematol. 2013;87:146–171.
Pettengell R, Coiffier B, Narayanan G, de Mendoza FH, Digumarti R, Gomez H, et al. Pixantrone dimaleate versus other chemotherapeutic agents as a single-agent salvage treatment in patients with relapsed or refractory aggressive non-Hodgkin lymphoma: a phase 3, multicentre, open-label, randomised trial. Lancet Oncol. 2012;13:696–706.
Crump M, Neelapu SS, Farooq U, Van Den Neste E, Kuruvilla J, Westin J, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study. Blood. 2017;130:1800–1808.
Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, et al. Long-term safety and activity ofaxicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a singlearm, multicentre, phase 1-2 trial. Lancet Oncol 2019;20:31–42.
Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med 2019;380:45–56.
Acknowledgements
We acknowledge the kind support of all physicians and data managers of the participating transplantation centers.
Author information
Authors and Affiliations
Contributions
E.G.-B. proposed the study, interpreted the data, and wrote the article. A.S. was the study coordinator, participated in the study design, interpreted the data, and edited the article. S.M. interpreted the data and edited the article. A.B. helped with the design and was responsible for statistical analysis. D.B., M.T., T.M., H.F., M.G.M., J.T.B., G.G., J.A.S, M.B., B.B., S.G.V., A.J., X.L., A.A., X.P., M.C., Z.N.Ö., E.V., M.I.-R., J.Y.C., E.J., W.S., J.M., A.E., P.D. reported updated patient data, and read and commented on the article. All the authors proofread the article and agreed on the data presented.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
González-Barca, E., Boumendil, A., Blaise, D. et al. Outcome in patients with diffuse large B-cell lymphoma who relapse after autologous stem cell transplantation and receive active therapy. A retrospective analysis of the Lymphoma Working Party of the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 55, 393–399 (2020). https://doi.org/10.1038/s41409-019-0650-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0650-x
This article is cited by
-
The impact of allogeneic hematopoietic cell transplantation on the mortality of poor-risk non-Hodgkin lymphoma: an intent-to-transplant analysis
Bone Marrow Transplantation (2021)
-
Immunotherapies Old and New: Hematopoietic Stem Cell Transplant, Chimeric Antigen Receptor T Cells, and Bispecific Antibodies for the Treatment of Relapsed/Refractory Diffuse Large B Cell Lymphoma
Current Hematologic Malignancy Reports (2021)
-
Prognostic impact of total metabolic tumor volume in large B-cell lymphoma patients receiving CAR T-cell therapy
Annals of Hematology (2021)
-
Hypomagnesemia at the time of autologous stem cell transplantation for patients with diffuse large B-cell lymphoma is associated with an increased risk of failure
Blood Cancer Journal (2021)