Abstract
Contrary to intuition, most individuals are resilient to psychological trauma and only a minority is vulnerable. Men and women are known to respond differently to trauma exposure, however, mechanisms underlying the relationship between sex differences and trauma resilience and vulnerability are not yet fully understood. Taking advantage of the Behavioral Profiling approach, which enables differentiating between ‘affected’ and ‘unaffected’ individuals, we examined sex-associated differences in stress exposure effects on hippocampal expression of selected stress-related GABA-A receptor targeting miRNAs. Levels of the miRNA-144 and miRNA-33 were measured in male and female affected (vulnerable, e.g., higher freezing time) and unaffected (resilient) rats. In male rats, increased levels of miRNA-144 and miRNA-33 were observed in the dorsal dentate gyrus (dDG) and ventral dentate gyrus (vDG) respectively, of stress-exposed but unaffected animals. In females, we observed an increased expression of miRNA-144 and miRNA-33 in the ventral cornu ammonis 1 (vCA1) of affected animals. Accordingly, we inhibited miRNAs expression selectively in hippocampal subregions using oligonucleotides containing locked nucleic acid bases, to examine the miRNAs’ causal contribution to either vulnerability or resilience to stress in each sex. Inhibition of miRNA-144 in dDG and miRNA-33 in vDG in males resulted in an increased prevalence of vulnerable animals, while inhibition of miRNA-144 and miRNA-33 in vCA1 in females increased the proportion of resilient animals. The current findings reveal a critical sex-associated difference in the role of miRNAs in stress vulnerability and resilience. This novel understanding of sex-associated epigenetic involvement in the mechanism of stress-related psychopathologies may help improve gender-specific diagnosis and effective treatment.
Similar content being viewed by others
Introduction
Sex differences in stress responses have been consistently demonstrated in human and animal models [1,2,3,4]. For instance, men are more likely to turn to substance abuse following a stressful event [5], while women are more likely to suffer from anxiety and trauma-related disorders [6,7,8]. Furthermore, there are non-sex related individual differences. Studies have shown that the majority of people are resilient to stress, and a relatively small proportion is vulnerable and may develop long-term stress-related psychopathologies [9, 10]. The underlying mechanisms by which individual and sex differences influence stress resilience and vulnerability are still largely unknown and are of fundamental importance to improving treatment of stress-related psychopathologies. It is possible that these mechanisms involve epigenetics, and specifically alterations in microRNAs (miRNAs) expression [11,12,13,14]. Accumulating evidence shows that dysregulation of miRNAs in body fluids and brain tissues may be associated with stress-related conditions in both patients and animal models [15,16,17,18,19,20].
Several in vivo studies indicate that miRNAs could be involved in vulnerability and resilience to stress-related mental disorders [21,22,23], as well as in stress-related sex differences [24, 25]. However, the contribution of miRNA to the mechanism of sex-related individual patterns of trauma response, was not yet systematically studied. Some miRNAs were demonstrated to contribute to the post-transcriptional regulation of gene expression of GABA-A receptors, following stress exposure [26]. For instance, exposure of mice to acute adult stress led to differential expressions of several GABA-A receptor targeting miRNAs in the hippocampus. Among them are miRNA-144 and miRNA-33 which target four or more mRNAs encoding GABA-A receptors [27]. A pilot study showed that similar changes to these miRNAs occurred in rats’ hippocampus following juvenile stress exposure (Fig. S2). Changes in miRNAs following stress exposure were not yet, to the best of our knowledge, studied in different subregions of the hippocampus.
Different hippocampal subregions are suggested to have distinct functions. While the dorsal hippocampus was found to be involved in cognition and spatial mapping, the ventral hippocampus is regarded as playing a role in stress responses, emotional memory formation and anxiety [28, 29]. Additionally, differences were found between the involvement of the dentate gyrus (DG) and cornu ammonis 1 (CA1), with regards to sensitivity and impact of exposure to stress [30, 31]. Having found hippocampal alterations in miRNA-144 and miRNA-33 following stress exposure, it would be interesting to pin down site-specific GABA-A related miRNA expression in the aforementioned subregions of the hippocampus and the extent to which sex influences the vulnerable or resilient response patterns.
Therefore, the first aim of the current study was to explore whether there is a difference in the expression pattern of the GABA-A receptor targeting miRNA-144 and miRNA-33 transcripts in the dorsal and ventral dentate gyrus (dDG, vDG, receptively) and dorsal and ventral CA1 (dCA1, vCA1, receptively) hippocampal areas, in the stress exposed affected rats compared to that of the stress exposed but unaffected, male and female rats. In the second part, aiming to directly examine the contribution of miRNA-144 and miRNA-33 to trauma vulnerability and resilience in males and females, we inhibited their functioning in the relevant subregions of the hippocampus.
Material and methods
Animals
For experiment 1, male and female Sprague–Dawley rats (postnatal day (PND) 22 on arrival, weight 30–50 g, Harlan Laboratories, Jerusalem, Israel), were group housed (22 ± 2 °C; light–dark cycle: 12/12 h), with water and food ad libitum. For experiment 2, male rats arrived on PND 45 (weight 175–199 g) and female rats arrived on PND 22 (weight 30–50 g) and were kept in the same conditions. Animals were randomly allocated to experimental groups. Sample size for each section was based on extensive previous experience [3, 4, 32,33,34,35,36,37,38,39]. Experimenters were blind to the group allocation during behavioral test performance. Animal care and experiments were performed in accordance with the NIH Guide for care and use of laboratory animals and approved by the University of Haifa ethical committee (610/19).
Experimental procedure
The experimental design is depicted in Fig. 1. Rats were assigned to two experimental paradigms:
Experiment 1
Following arrival and acclimation all rats were randomly assigned to one of four cohorts: Control (male: n = 20, female: n = 17); juvenile stress (JS, male: n = 23, female: n = 20); adult stress (AS, male: n = 23, female: n = 20); and combined JS + AS (male: n = 23, female: n = 20). JS and JS + AS rats were subjected to JS protocol. In adulthood, all rats were habituated to the water-associated zero maze (WAZM), and following this, only groups AS and JS + AS were immediately subjected to underwater trauma (UWT) stress. 14 days later, all rats were re-exposed to the elevated plus maze (EPM) and WAZM for the purpose of behavioral profiling. 24 h later, tissue samples were extracted and assessment of miRNA-144 and miRNA-33 levels in the different subregions of the hippocamps was conducted. In total, this experiment was replicated four times.
Experiment 2
miRNA inhibition. Since, based on results in experiment 1, we hypothesized that knocking down miRNA-144 and miRNA-33 would compromise resilience in males, we used a protocol with a relatively low baseline prevalence of affected animals, to more effectively detect a potential increase in the proportion of affected animals by our manipulation [32]. Locked nucleic acid (LNA) inhibition was performed for miRNA-144 in the dDG and for miRNA-33 in the vDG following exposure to UWT (see Fig. S6 for cannula placement, and additional supporting data in table S1 and Figs. S4 and S5). Male rats were randomly assigned to one of four cohorts: exposure to adult stress and LNA inhibition (AS-LNA: n = 7); exposure to adult stress without inhibition (AS-SCR: n = 10); LNA inhibition without stress exposure (CTR-LNA: n = 8); and no stress exposure/no inhibition (CTR-SCR: n = 7).
In females, since, based on results from experiment 1, we hypothesized that blocking the functioning of the miRNAS will support stress resilience, we employed the combined exposure to JS and UWT, a stress protocol with an observed initial high baseline prevalence of affected animals, in order to more effectively be able to detect an increase in the proportion of unaffected animals. Rats were randomly assigned to one of four cohorts: exposure to juvenile and adult stress and LNA inhibition (JS + AS-LNA: n = 8); exposure to juvenile and adult stress without inhibition (JS + AS-SCR: n = 10); LNA inhibition without stress exposure (CTR-LNA: n = 13); and no stress exposer and no inhibition (CTR-SCR: n = 10). LNA inhibition was performed for miRNA-144 and miRNA-33 in the vCA1 following exposure to the UWT.
Fourteen days later all rats were re-exposed to the EPM and WAZM and were subjected to behavioral profiling. In total, this experiment was replicated four times.
Juvenile stress (JS) exposure
JS rats were exposed to 3 different stressors on 3 consecutive days under full light condition (as described before [3, 33]): PND 27–15 min of forced swim stress (water temperature 22 ± 2 °C); PND 28 - elevated platform (12 × 12 cm, 70 cm above floor level) exposure for 3 × 30 min (1 h ITI in the home cage); PND 29—2 h restraint.
Water-associated zero maze (WAZM)
The WAZM is an integrated wet and dry context maze that enables an association of the maze with an UWT [34]. On PND 60, following 5 min habituation to the room, rats were habituated to the WAZM for additional 5 min, on the dry arena, facing the closed part of the WAZM, in one of its open arms. Then rates were removed from the dry platform and placed in the aquatic center of the apparatus, where they were subjected to UWT stress for 45 s, using a special metal net (AS). Following the procedure, the rats were dried and returned to their home cages. For the test, following 2 min habituation to the room, rats were placed on the dry arena at the same location. Rats were given 5 min to explore the arena, during which their behavior was recorded and analyzed by EthoVision XT10 video tracking system (Noldus, Wageningen, Netherlands) (For an elaborate description of this procedure see [35]).
Elevated plus maze (EPM)
For a different context measurement of emotional behavior not directly associated with the trauma, elevated plus maze (EPM) test was carried out on PND 74, 24 h before the WAZM test, as described before [40]. After 2 min habituation to the room, rats were placed in the center of the maze (110 × 110 cm, 70 cm above the floor; two opposing open arms/closed arms), facing an open arm and were allowed to freely explore the arena maze for 5 min. Behavior was recorded and analyzed by EthoVision XT10 video tracking system (Noldus, Wageningen, Netherlands).
Behavioral profiling
Analysis of individual behavior, indicating resilience or vulnerability, was used to establish the different effects of JS and AS conditions, as well as miRNA-144 and miRNA-33 LNA inhibition on rats’ behavioral phenotype. In this analysis the classification of animals’ behavioral profiles was based on the upper or lower 20th percentiles of anxiety-like behaviors (WAZM and EPM tests) of the control group distribution. Classification criteria were based on the lower 20th percentiles of control’s distribution, for time spent in open arms (WAZM and EPM), distance traveled in open arms (WAZM) and total distance traveled (WAZM) and on the upper 80th percentiles for freezing time (WAZM and EPM). All rats were individually discerned for each criterion. For the final classification, every rat that demonstrated a behavioral profile that fell within a minimum of 4 out of 6 criteria was classified as affected [36].
Surgery and cannulation
The procedures were performed under Ketamine anesthesia (10% Ketamine 100 mg/kg, and 2% dormitor, 10 mg/kg (Vetmarket, Petah-Tikva, Israel), both i.p.). Rats were restrained in a stereotaxic apparatus (Stoelting instruments) and implanted with a stainless-steel guide cannula (23 gauge, thin walled). Double guided cannulas were bilaterally implanted in the dDG (−3.7 mm posterior, ±2.3 mm lateral and −3.8 mm ventral to bregma) and vDG (−5.8 mm posterior, ±4.5 mm lateral and −7.1 mm ventral to bregma) in male, and in the vCA1 (−6 mm posterior, ±5.6 mm lateral and −7.6 mm ventral to bregma) in female. The cannulas were set in place using acrylic dental cement and secured by three skull screws. A stylus was placed in the guide cannula to prevent clogging. For the following first 3 days, rats were treated with analgesics (Dipyrone, 0.3 ml/kg, s.c.; Vetmarket) and antibiotics (15% Amoxycillin, 0.2 ml/kg, s.c.; Vetmarket, Petah Tikva, Israel) and were allowed to recover in their home cage for a total of one week before behavioral testing began. For microinjection, the stylus was removed, and a 14-gauge (for the dDG) or 16-gauge (for the vDG and vCA1) injection cannulae, extending 1.0 mm from the tip of the guide cannula, were inserted. The injection cannula was connected via polyethylene PE20 tubing to a Hamilton microsyringe driven by a microinfusion pump (PHD1000, Harvard Apparatus, USA). Bilateral microinjections of a 0.5 µl volume were delivered over 1 min. The injection cannula was left in each position for an additional 1 min before withdrawal to minimize dragging of the injected liquid along the injection tract [41].
Quantitative real-time PCR (qRT-PCR) for assessment miRNA expression
Total RNA was extracted using miRNeasy RNA Isolation Mini Kit (Qiagen, Hilden, Germany, Cat. No. 217004). Reverse transcription was carried out using qScript cDNA Synthesis Kit (Quanta Biosciences, Gaithersburg, USA, Cat. No. 95047) following the manufacturer’s protocol. miRNA-144-3p (Quanta Biosciences, Cat. No. MIR SET), miRNA-33-5p (Quanta Biosciences, Cat. No. MIR SET) and miRNA-381-5P (Quanta Biosciences, Cat. No. MIR SET) expression were assessed using SYBR Green qRT-PCR amplification (5 ng total RNA in 20 μl total reaction volume, Cat. No. 95074) using specific primers (0.4 μl each, Quanta Biosciences, Gaithersburg, USA) according to manufacturer’s instructions. Real-time PCR analysis was performed on a QuantStudio 3 real-time PCR system (Thermo Fisher Scientific Inc., Waltham, MA, USA), using the following PCR conditions: (95 °C for 3 min Holding stage, followed by 40 cycles (95 °C for 15 s, 60 °C for 45 s) [42].
For data analysis, the mean cycle threshold (CT) was determined for each triplicate assay and relative quantification of each target gene was conducted with the ddCT method [43], normalizing each sample to the overall content of cDNA using RNU6 as an internal control {dCT; dCT = [CT (target gene)] − [CT (RNU6)]}. Normalization of all ddCT values was done relative to Control group with ddCT = dCT(sample) − mean dCT (Control group). Transformation to RQ values for a specific target gene and area was done according to RQ = 2−ddCT with RQ(Control) = 1.
miRNA inhibition
miRCURY LNA miRNA144-3P inhibitors (sequence 3′-5′: AGTACATCATCTATACTGT, Qiagen, Hilden, Germany) and miRCURY LNA miRNA33-5p inhibitors (sequence 5′-3′: GCAATGCAACTACAATGCA, Qiagen, Hilden, Germany) were dissolved in water to 400 μM, heated at 65 °C for 10 min, cooled on ice, and diluted in artificial cerebrospinal fluid (ACSF) to a final working concentration of 40 μM. The inhibitors have a full phosphorothioate backbone modification (PS) (added for enhanced stability and potency). Scrambled LNA, which does not bind to any cellular miRNA, were used as a negative control (sequence 5′-3′: TAACACGTCTATACGCCA, Qiagen, Hilden, Germany) [27].
Statistical analysis
The sample sizes of each experiment were determined based on our previous studies of stress-emulating paradigms [32, 33, 35, 36]. Data were analyzed using the IBM SPSS Statistics Software (IBM, Armonk, NY, USA). For comparing anxiety like behavior (e.g., freezing time in the WAZM) following different stress exposures, a one-way ANOVA followed by Fisher’s LSD post hoc test was applied. The prevalence of affected vs. unaffected populations between the groups in experiment 1 and experiment 2 was compared using Pearson’s chi squared test. For miRNA level differences between type of stress exposures or behavioral profiles, and their interactions, a two-way ANOVA, followed by Fisher’s LSD post hoc test was applied when data was normally distributed. For non-normally distributed data were analyzed by Kruskal-Wallis test followed by pairwise two-sided comparisons. Homogeneity of variance was confirmed with Levene’s test for equality of variances. Significance was accepted when p < 0.05. Outliers were defined as two or more standard deviations from the mean and removed from the analysis. GraphPad Prism 7.0 (GraphPad Software, San Diego, CA), and BioRender (https://biorender.com/) were used to create figures.
Results
Group and individual differences in response to stress exposure in males and females
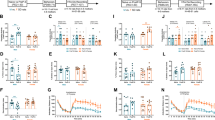
Freezing behavior as expressed in total freezing time in the WAZM was measured in males and females. In males, freezing time of the JS + AS group was significantly longer (Fig. 2A; F(3,85) = 6.19, p < 0.001) compared to all other groups (Control: P < 0.001, JS: p < 0.001, AS: p < 0.01). In females, both AS and JS + AS groups showed more freezing behavior (Fig. 2B; H(3) = 19.94, p < 0.001) compared to control (AS: P < 0.01, JS + AS: P < 0.001) and JS groups (AS: P < 0.05, JS + AS: P < 0.01), demonstrating an overall increase in anxiety-like behaviors after exposure to combined stress in males and to AS and combined stress in females.
A In males, the JS + AS group spent more time freezing in the WAZM compared to all other groups. Number of animals: Control: n = 20, JS: n = 23, AS: n = 23, JS + AS = 23. B In females, both AS and JS + AS groups showed increased freezing behavior in the WAZM compared to control and JS rats. Number of animals: Control: n = 17, JS: n = 20, AS: n = 20, JS + AS = 20. C Behavioral profiling according to anxiety-like behaviors in male rats, revealed an increase in the prevalence of affected animals in the JS + AS group compared to that of all other groups. D In females, behavioral profiling analysis uncovered a significantly higher prevalence of affected animals among JS + AS and AS groups equally, compared to that of the control group. A significantly higher proportion of affected animals, though less pronounced, was detected in the JS group compared to that of the control group. A, B Data presented as individual values, with bars and whiskers representing means and standard errors, respectively. C, D Values are the % and number of affected and unaffected animals in each group. *P < 0.05, **P < 0.01, ***P < 0.001.
Sex differences in response to trauma exposure between groups of males and females have been established in the past [44,45,46], however, due to the fact that some individuals are more susceptible to stress than others, a comparison of the different groups’ means does not expose differences in stress induced changes at the individual level between males and females. Hence, we used behavioral profiling in order to identify vulnerable and resilient individuals among the male and female groups following exposure to the various stressors.
Behavioral profiling revealed sex-related differences. In males, increased prevalence of affected animals was found in the JS + AS group compared to all other groups (Fig. 2C; Control: χ2 (1) = 11.85, p < 0.01; JS: χ2(1) = 9.13, p < 0.01; AS: χ2 (1) = 4.29, p < 0.05).
In females, increased prevalence of affected rats was found in both the AS and JS + AS groups compared to the control group (Fig. 2D; AS: χ2 (1) = 15.1, p < 0.001, JS + AS: χ2 (1) = 15.1, p < 0.001, receptively). In addition, though less pronounced, increased prevalence of affected animals was found also in the JS groups compared to the control group (Fig. 2D; JS: χ2 (1) = 6.09, p < 0.05).
miRNA levels in affected and unaffected male rats
For miRNA-144, a significant main effect was demonstrated for behavioral profile that was restricted to the dDG (Fig. 3A; H(2) = 7.84, P < 0.05). Post hoc comparisons revealed increased miRNA-144 levels specifically in unaffected rats compared to those in affected rats (p < 0.05) and unexposed control rats (p < 0.05). A similar trend, not statistically significant, was observed in the vDG (Fig. 3B; H(2) = 4.43, P = 0.11). No significant main effects for behavioral profile or type of stress exposure were observed in either the dCA1 or vCA1 (Fig. 3C, D).
A Increase in miRNA-144 levels occurred exclusively in the dorsal dentate gyrus (dDG) of unaffected rats with a history of JS, AS or combined JS + AS, whereas no main effects for stress exposure group or behavioral profile were observed in the ventral dentate gyrus (vDG) (B), dorsal Cornu Ammonis 1 (dCA1). C or ventral CA1 (vCA1) (D). Within the vDG, an increase in miRNA-33 expression was evident in stress-exposed, but unaffected rats (F). No main effects for group or profile were observed for miRNA-33 expression in the dDG (E) dCA1 (G) and vCA1 (H). Data presented as means and standard errors of fold-change, relative to control. *Significant difference between profiles, p < 0.05, **p < 0.01.
For miRNA-33, a significant main effect for behavioral profile was observed in the vDG (Fig. 3F; F(1,81) = 5.78, p < 0.05). Post hoc comparisons revealed a significant increase of miRNA-33 transcript level in stress exposed, unaffected rats compared to that of affected and control rats (p < 0.01, p < 0.01, receptively). No significant main effects of type of stress exposure or behavioral profile were observed in the dDG (Fig. 3E), dCA1 (Fig. 3G) or vCA1 (Fig. 3H).
For miRNA-381, no significant main effects for behavioral profile or type of stress exposure were observed in either the dDG, vDG or dCA1 (Fig. S3A–C). (Fig. SD) In the vCA1 a significant main effect for type of stress exposure was observed (F(2,74) = 3.65, p < 0.05). Post hoc comparisons revealed lower levels of miRNA-381 expression in JS + AS exposed animals compared to the AS group (p < 0.05).
miRNA levels in affected and unaffected female rats
For miRNA-144, a significant main effect for behavioral profile was observed in the vCA1 (Fig. 4D; F(1,66) = 7.66, P < 0.01), Post hoc comparisons revealed increased miRNA-144 levels specifically in affected compared to unaffected rats (P < 0.01) and unexposed control rats (p < 0.05). No significant main effects for behavioral profile, type of stress exposure or interactions were observed in the dDG, vDG or in the dCA1 (Fig. A–C).
A–C No main effects or interactions were observed in miRNA-144 levels in the dDG, vDG and dCA1. D Increased miRNA-144 levels occurred specifically in the vCA1 of affected compared to unaffected rats. E–G No main effects or interactions were observed in miRNA-33 levels in the dDG, vDG and dCA1. H Within the vCA1 increased miRNA-33 levels occurred in stress affected compared to those in unaffected rats. Data presented as means and standard errors of fold-change relative to control. *Significant difference between profiles, p < 0.05.
For miRNA-33, a significant main effect for behavioral profile was observed in the vCA1 (Fig. 4H; F(1,68) = 4.13, P < 0.05), post hoc comparisons showing increased transcript levels in stress affected compared to those in unaffected rats (P < 0.05). No significant differences were observed in the dDG, vDG or in the dCA1 (Fig. 4E–G).
For miRNA-381, no significant main effects for behavioral profile or type of stress exposure were observed in either the dDG, vDG or dCA1 (Fig. S3E–G). (Fig. S3H) In the vCA1 a significant main effect for type of stress exposure was observed (F(2,67) = 5.10, p < 0.001). Post hoc comparisons revealed lower levels of miRNA-381 expression in JS + AS exposed animals compared to that in the other groups (control: p < 0.01, JS: p < 0.001, AS: p < 0.001).
The proportions of affected/unaffected following LNA inhibition
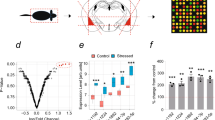
Next, following the initial findings in males (experiment 1), which demonstrated an association between increased levels of miRNA-144 (dDG) and miRNA-33 (vDG) and resilience, we inhibited these miRNAs following adult stress using LNA inhibition. A causal relation should lead to reduced resilience, and thus to increased proportion of affected subjects. To increase the likelihood of identifying effects of the manipulation, inhibition was conducted following AS exposure, which is expected, without manipulation, to lead to low proportion of affected animals. The results showed that indeed, inhibition of miRNA-144 in the dDG and of miRNA-33 in the vDG in male rats (for cannula location see Fig. 5A), significantly increased the prevalence of affected animals in the AS-LNA group (Fig. 5B; χ2(1) = 4.41, P < 0.05) compared to CTR-SCR (Fig. 5B; χ2(1) = 5.6, P < 0.01) (Fig. 2C).
A Schematic of cannula implantations in males. B In male rats, behavioral profiling revealed an increased prevalence of affected rats in the AS-LNA group compared to that in the AS-CTR group. Number of animals: CTR-SCR: n = 7, CTR-LNA: n = 8, AS-SCR: n = 10, AS-LNA = 7. All values are mean ± SEM. C Schematic of cannula implantations in females. D In female rats, behavioral profiling revealed an increased prevalence of unaffected rats in the JS + AS-LNA group compared to that of the JS + AS-SCR rats. Number of animals: CTR-SCR: n = 10, CTR-LNA: n = 13, JS + AS-SCR: n = 10, JS + AS-LNA = 8. Values are the % and number of affected and unaffected animals in each group. *Different from JS + AS-SCR, p < 0.005.
In contrast, in females, higher levels of miRNA-144 and miRNA-33 in the vCA1 were found to be associated with increased vulnerability to stress. Inhibiting these miRNAs in females could be thus expected to enhance resilience. Therefore, we examined the impact of inhibition of those miRNAs in vCA1 in animals exposed to JS + AS, a condition which is, by itself, associated with an initial high proportion of affected animals. Indeed, inhibition of miRNA144 and miRNA33 in the vCA1 in female rats (for cannula location see Fig. 5C), significantly increased the proportion of ‘unaffected’ rats in the AS-LNA group compared to that of AS-SCR (Fig. 5D; χ2(1) = 2.81, P < 0.05) and that of the control group (Fig. 5D; χ2(1) = 5.6, P < 0.05).
Discussion
The current results are a continuation of studies emphasizing the importance of shifting away from working with group averages and towards analyzing responses to stress and trauma at the individual level [3, 32, 33, 35, 36, 47,48,49]. The current results demonstrate significant differences in alterations in miRNAs in exposed animals between ‘affected’ and ‘unaffected’ individuals. Such differences would have been completely masked by averaging the results of the exposed groups.
The alterations found in miRNA expression were region-specific. These results join previous findings, emphasizing that the role of expression alterations of specific proteins should be discussed in the context of the specific region, in which they were identified [32, 36, 37]. This, of course, represents a significant challenge for planning translational targeted interventions. However, the first step in any such consideration is to identify region-specific alterations, interventions in which would be sufficient to induce a phenotypic effect at the behavioral level. Here, we could demonstrate that the region-specific alterations in expression of miRNAs identified following trauma exposure were indeed sufficiently involved in vulnerability or resilience, to the level that manipulating the functionality of these alterations was sufficient to alter the outcome of trauma exposure. Differential expression of neuromodulators such as serotonin, norepinephrine or dopamine, or the interneuron-localized neuropeptide Y (NPY), were found to mediate stress resilience and vulnerability in a trauma-exposed population, at the individual level [50,51,52], and epigenetic mechanisms were suggested to mediate such stress-related individual differences [53, 54].
But results did not only indicate individual differences and region specificity, but also sex-associated differences. Trauma-related psychopathologies, including post-traumatic stress disorder (PTSD) and post-traumatic depression, are more prevalent in women than in men [55,56,57,58]. Gonadal hormones such as testosterone and estrogen as well as interacting genetic factors, have long been believed to differentially affect the hypothalamic-pituitary-adrenal (HPA) axis or modulate hippocampal functioning both in humans and in animal models [59,60,61], potentially affecting the risk of developing pathology directly, or through epigenetic mechanisms [62]. Even though some reports could not demonstrate such association (see, e.g., refs. [63,64,65]), the majority of studies have demonstrated differences in stress responses between males and females following exposure to traumatic events, and have documented this difference in animal models and experimental tests of stress and trauma [3, 38, 39, 66,67,68,69]. In accordance with that, here, we found no difference between males and females in the prevalence of ‘affected’ individuals following exposure to JS + AS, but only in females there was a significant increase in the prevalence of ‘affected’ individuals following exposure to AS alone.
An important focus of the current study was the identification of sex-associated differences in miRNAs expression alterations. Specifically, in males, up-regulation of miRNA-144 in the dDG and miRNA-33 in the vDG, was related to resilience while in females, these miRNAs increased expression in the vCA1 was associated with greater vulnerability (Fig. 3A, F, D). In accordance with that, down-regulation of those miRNAs was sufficient to induce increased vulnerability in males and increased resilience in females. The population of rats profiled as ‘unaffected’ was reduced in males and increased in females following LNA inhibition in the relevant regions (Fig. 5B, D). To the best of our knowledge this is the first study demonstrating enhanced resilience in females following specific miRNA inhibition. Furthermore, alterations in two different miRNAs (males and females), and in two brain regions (males) have led us to employ a dual intervention, handling two miRNAs simultaneously. The success of this approach, as revealed by significant effects on stress vulnerability and resilience, encourages its employment in future studies. Identification of the downstream, possibly also sex-specific targets of miRNA-33 and miRNA-144 will further clarify the mechanisms of their differential effects as they relate to both gender and subregions. However, clearly, these are not the only molecules and the only regions involved. Future studies may investigate the specific target genes associated with these miRNAs. It may also be beneficial to examine hormonal involvement (e.g., corticosterone, estradiol, or progesterone) in sex-related resilience/vulnerability mechanisms.
In summary, the current results reveal critical sex-associated differences in hippocampal epigenetic responses to trauma exposure, which contribute to stress vulnerability in females, and to stress resilience in males. The results emphasize the significant value of dissociating neural mechanisms underlying coping with stress from those mechanisms underlying failure to cope, which could lead to the development of trauma-related psychopathologies. Towards that goal, it is critical to differentiate between ‘affected’ and ‘unaffected’ individuals. The findings here, that resilience is associated with significant alterations in expression of specific miRNAs in specific hippocampal regions, add to a body of findings indicating that stress resilience is not a passive quality but rather an active response which enables coping with the experience. Better understanding of the mechanisms at the basis of such active resilience, may eventually be translated to ways of enhancing stress resilience.
Change history
31 January 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41398-023-02333-5
References
Leach LS, Christensen H, Mackinnon AJ, Windsor TD, Butterworth P. Gender differences in depression and anxiety across the adult lifespan: the role of psychosocial mediators. Soc Psychiatry Psychiatr Epidemiol. 2008;43:983–98.
McLean CP, Anderson ER. Brave men and timid women? A review of the gender differences in fear and anxiety. Clin Psychol Rev. 2009;29:496–505.
Horovitz O, Tsoory MM, Yovell Y, Richter-Levin G. A rat model of pre-puberty (juvenile) stress-induced predisposition to stress-related disorders: sex similarities and sex differences in effects and symptoms. World J Biol Psychiatry. 2014;15:36–48.
Brydges NM, Moon A, Rule L, Watkin H, Thomas KL, Hall J. Sex specific effects of pre-pubertal stress on hippocampal neurogenesis and behaviour. Transl Psychiatry. 2018;8:1–12.
Grant BF, Dawson DA, Stinson FS, Chou SP, Dufour MC, Pickering RP. The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991–1992 and 2001–2002. Drug Alcohol Depend. 2004;74:223–34.
Breslau N, Davis GC, Andreski P, Peterson EL, Schultz LR. Sex differences in posttraumatic stress disorder. Arch Gen Psychiatry. 1997;54:1044–8.
Bale TL, Epperson CN. Sex differences and stress across the lifespan. Nat Neurosci. 2015;18:1413–20.
Sheikh JI, Leskin GA, Klein DF. Gender differences in panic disorder: findings from the National Comorbidity Survey. Am J Psychiatry. 2002;159:55–8.
Helzer JE, Robins LN, McEvoy L. Post-traumatic stress disorder in the general population. N. Engl J Med. 1987;317:1630–4.
Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593–602.
Klengel T, Binder EB. Epigenetics of stress-related psychiatric disorders and gene×environment interactions. Neuron. 2015;86:1343–57.
Issler O, Chen A. Determining the role of microRNAs in psychiatric disorders. Nat Rev Neurosci. 2015;16:201–12.
Narahari A, Hussain M, Sreeram V. MicroRNAs as biomarkers for psychiatric conditions: a review of current research. Innov Clin Neurosci. 2017;14:53–5.
Hunsberger JG, Austin DR, Chen G, Manji HK. MicroRNAs in mental health: from biological underpinnings to potential therapies. Neuromolecular Med. 2009;11:173–82.
Schmidt U, Keck ME, Buell DR. miRNAs and other non-coding RNAs in posttraumatic stress disorder: a systematic review of clinical and animal studies. J Psychiat Res. 2015;65:1–8.
Martin CG, Kim H, Yun S, Livingston W, Fetta J, Mysliwiec V, et al. Circulating miRNA associated with posttraumatic stress disorder in a cohort of military combat veterans. Psychiat Res. 2017;251:261–5.
Lee MY, Baxter D, Scherler K, Kim TK, Wu X, Abu-Amara D, et al. Distinct profiles of cell-free microRNAs in plasma of veterans with post-traumatic stress disorder. J Clin Med. 2019;8:963.
Balakathiresan NS, Chandran R, Bhomia M, Jia M, Li H, Maheshwari RK. Serum and amygdala microRNA signatures of posttraumatic stress: fear correlation and biomarker potential. J Psychiat Res. 2014;57:65–73.
Vreugdenhil E, Verissimo CS, Mariman R, Kamphorst JT, Barbosa JS, Zweers T, et al. MicroRNA 18 and 124a down-regulate the glucocorticoid receptor: implications for glucocorticoid responsiveness in the brain. Endocrinology. 2009;150:2220–8.
Schmidt U, Herrmann L, Hagl K, Novak B, Huber C, Holsboer F, et al. Therapeutic action of fluoxetine is associated with a reduction in prefrontal cortical miR-1971 expression levels in a mouse model of posttraumatic stress disorder. Front Psychiat. 2013;4:66.
Chen RJ, Kelly G, Sengupta A, Heydendael W, Nicholas B, Beltrami S, et al. MicroRNAs as biomarkers of resilience or vulnerability to stress. Neuroscience. 2015;305:36–48.
Lopizzo N, Zonca V, Cattane N, Pariante CM, Cattaneo A. miRNAs in depression vulnerability and resilience: novel targets for preventive strategies. J Neural Transm. 2019;126:1241–58.
Maurel OM, Torrisi SA, Barbagallo C, Purrello M, Salomone S, Drago. et al. Dysregulation of miR-15a-5p, miR-497a-5p and miR-511-5p Is Associated with Modulation of BDNF and FKBP5 in Brain Areas of PTSD-Related Susceptible and Resilient Mice. Int J Mol Sci. 2021;22:5157.
McKibben LA, Dwivedi Y. Early life and adult stress promote sex dependent changes in hypothalamic miRNAs and environmental enrichment prevents stress-induced miRNA and gene expression changes in rats. BMC Genomics. 2021;22:1–24.
Luo PX, Manning CE, Fass JN, Williams AV, Hao R, Campi KL, et al. Sex-specific effects of social defeat stress on miRNA expression in the anterior BNST. Behav Brain Res. 2021;401:113084.
Shan L, Ma D, Zhang C, Xiong W, Zhang Y. miRNAs may regulate GABAergic transmission associated genes in aged rats with anesthetics-induced recognition and working memory dysfunction. Brain Res. 2017;1670:191–200.
Jovasevic V, Corcoran KA, Leaderbrand K, Yamawaki N, Guedea AL, Chen HJ, et al. GABAergic mechanisms regulated by miR-33 encode state dependent fear. Nat Neurosci. 2015;18:1265–71.
Fanselow MS, Dong HW. Are the dorsal and ventral hippocampus functionally distinct structures? Neuron. 2010;65:7–19.
Strange BA, Witter MP, Lein ES, Moser EI. Functional organization of the hippocampal longitudinal axis. Nat Rev Neurosci. 2014;15:655–69.
Fa M, Xia L, Anunu R, Kehat O, Kriebel M, Volkmer H, et al. Stress modulation of hippocampal activity–spotlight on the dentate gyrus. Neurobiol Learn Mem. 2014;112:53–60.
Alkadhi KA. Cellular and molecular differences between area CA1 and the dentate gyrus of the hippocampus. Mol Neurobiol. 2019;56:6566–80.
Regev-Tsur S, Demiray YE, Tripathi K, Stork O, Richter-Levin G, Albrecht A. Region-specific involvement of interneuron subpopulations in trauma-related pathology and resilience. Neurobiol Dis. 2020;143:104974.
Ardi Z, Albrecht A, Richter-Levin A, Saha R, Richter-Levin G. Behavioral profiling as a translational approach in an animal model of posttraumatic stress disorder. Neurobiol Dis. 2016;88:139–47.
Ritov G, Richter-Levin G. Water associated zero maze: a novel rat test for long term traumatic re-experiencing. Front Behav Neurosci. 2014;8:1.
Ritov G, Richter-Levin G. Pre-trauma Methylphenidate in rats reduces PTSD-like reactions one month later. Transl Psychiatry. 2017;7:e1000.
Ritov G, Boltyansky B, Richter-Levin G. A novel approach to PTSD modeling in rats reveals alternating patterns of limbic activity in different types of stress reaction. Mol Psychiatry. 2016;21:630–41.
Tripathi K, Demiray YE, Kliche S, Jing L, Hazra S, Hazra JD, et al. Reducing glutamic acid decarboxylase in the dorsal dentate gyrus attenuates juvenile stress induced emotional and cognitive deficits. Neurobiol Stress. 2021;15:100350.
Jacobson-Pick S, Richter-Levin G. Differential impact of juvenile stress and corticosterone in juvenility and in adulthood, in male and female rats. Behav Brain Res. 2010;214:268–76.
Zitman FM, Richter-Levin G. Age and sex-dependent differences in activity, plasticity and response to stress in the dentate gyrus. Neuroscience. 2013;249:21–30.
Pellow S, Chopin P, File SE, Briley M. Validation of open: closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Meth. 1985;14:149–67.
Ganon-Elazar E, Akirav I. Cannabinoids and traumatic stress modulation of contextual fear extinction and GR expression in the amygdala-hippocampal-prefrontal circuit. Psychoneuroendocrinol. 2013;38:1675–87.
Zaidan H, Galiani D, Gaisler-Salomon I. Pre-reproductive stress in adolescent female rats alters oocyte microRNA expression and offspring phenotypes: pharmacological interventions and putative mechanisms. Transl Psychiatry. 2021;11:1–16.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods. 2001;25:402–8.
Bangasser DA, Valentino RJ. Sex differences in stress-related psychiatric disorders: neurobiological perspectives. Front Neuroendocrinol. 2014;35:303–19.
Rubinow DR, Schmidt PJ. Sex differences and the neurobiology of affective disorders. Neuropsychopharmacology. 2019;44:111–28.
Bangasser DA, Wicks B. Sex‐specific mechanisms for responding to stress. J Neurosci Res. 2017;95:75–82.
Cohen H, Zohar J, Matar MA, Zeev K, Loewenthal U, Richter-Levin G. Setting apart the affected: the use of behavioral criteria in animal models of post-traumatic stress disorder. Neuropsychopharmacology. 2004;29:1962–70.
Richter-Levin G, Sandi C. Title: “Labels matter: is it stress or is it trauma?”. Transl Psychiatry. 2021;11:385.
Richter-Levin G, Stork O, Schmidt MV. Animal models of PTSD: a challenge to be met. Mol Psychiatry. 2019;24:1135–56.
Charney DS. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am J Psychiat. 2004;161:195–216.
Wu G, Feder A, Cohen H, Kim JJ, Calderon S, Charney DS, et al. Understanding resilience. Front Behav Neurosci. 2013;7:10.
Haglund ME, Nestadt PS, Cooper NS, Southwick SM, Charney DS. Psychobiological mechanisms of resilience: relevance to prevention and treatment of stress-related psychopathology. Dev Psychopathol. 2007;19:889–920.
Dudley KJ, Li X, Kobor MS, Kippin TE, Bredy TW. Epigenetic mechanisms mediating vulnerability and resilience to psychiatric disorders. Neurosci Biobehav Rev. 2011;35:1544–51.
Zannas AS, West AE. Epigenetics and the regulation of stress vulnerability and resilience. Neuroscience. 2014;264:157–70.
Breslau N. Gender differences in trauma and posttraumatic stress disorder. J Gend Specif Med. 2002;5:34–40.
Kessler RC, Petukhova M, Sampson NA, Zaslavsky AM, Wittchen HU. Twelve‐month and lifetime prevalence and lifetime morbid risk of anxiety and mood disorders in the United States. Int J Methods Psychiatr Res. 2012;21:169–84.
Tolin DF, Foa EB. Sex differences in trauma and posttraumatic stress disorder: a quantitative review of 25 years of research. Psychol Trauma. 2008;S:37–85.
Stein MB, Walker JR, Forde DR. Gender differences in susceptibility to posttraumatic stress disorder. Behav Res Ther. 2000;38:619–28.
Maeng LY, Milad MR. Sex differences in anxiety disorders: interactions between fear, stress, and gonadal hormones. Hormones Behav. 2015;76:106–17.
Fenchel D, Levkovitz Y, Vainer E, Kaplan Z, Zohar J, Cohen H. Beyond the HPA-axis: The role of the gonadal steroid hormone receptors in modulating stress-related responses in an animal model of PTSD. Eur Neuropsychopharmacol. 2015;25:944–57.
Scharfman HE, MacLusky NJ. Differential regulation of BDNF, synaptic plasticity and sprouting in the hippocampal mossy fiber pathway of male and female rats. Neuropharmacology. 2014;76:696–708.
Raabe FJ, Spengler D. Epigenetic risk factors in PTSD and depression. Front Psychiat. 2013;4:80.
Lilly MM, Pole N, Best SR, Metzler T, Marmar CR. Gender and PTSD: what can we learn from female police officers? J Anxiety Disord. 2009;23:767–74.
Haskell SG, Gordon KS, Mattocks K, Duggal M, Erdos J, Justice A. et al. Gender differences in rates of depression, PTSD, pain, obesity, and military sexual trauma among Connecticut war veterans of Iraq and Afghanistan. J Women’s Health. 2010;19:267–71.
Crum-Cianflone NF, Jacobson I. Gender differences of postdeployment post-traumatic stress disorder among service members and veterans of the Iraq and Afghanistan conflicts. Epidemiol Rev. 2014;36:5–18.
Bangasser DA, Valentino RJ. Sex differences in molecular and cellular substrates of stress. Cell Mol Neurobiol. 2012;32:709–23.
Bowman RE, Zrull MC, Luine VN. Chronic restraint stress enhances radial arm maze performance in female rats. Brain Res. 2001;904:279–89.
Chow C, Epp JR, Lieblich SE, Barha CK, Galea LA. Sex differences in neurogenesis and activation of new neurons in response to spatial learning and memory. Psychoneuroendocrinology. 2013;38:1236–50.
Jacobson-Pick S, Audet MC, McQuaid RJ, Kalvapalle R, Anisman H. Stressor exposure of male and female juvenile mice influences later responses to stressors: modulation of GABAA receptor subunit mRNA expression. Neuroscience. 2012;215:114–26.
Acknowledgements
We would like to thank Dr. Maya Lebow from Prof. Chen laboratory at the Weizmann Institute of Science for her help with miRNA protocols, Dr. Amitai Mordechai from Ben-Gurion University for his help with miRNA analysis, and Dr. Silvia Mandel for her help in editing the MS. We would like to express our gratitude to the Israel Scholarship Education Foundation for partially funding this study.
Funding
This study was supported by a United States - Israel Binational Science Foundation (BSF) (#2019261) to GRL and JR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Krispil-Alon, M., Jovasevic, V., Radulovic, J. et al. Sex-specific roles of hippocampal microRNAs in stress vulnerability and resilience. Transl Psychiatry 12, 503 (2022). https://doi.org/10.1038/s41398-022-02267-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-022-02267-4
This article is cited by
-
Recent advances in the role of miRNAs in post-traumatic stress disorder and traumatic brain injury
Molecular Psychiatry (2023)