Abstract
Study design
Longitudinal.
Objective
With an increased risk of depression in spinal cord injury, the study longitudinally examines depression to understand how post-injury autonomic regulation and coping might be related to somatic and cognitive manifestations of depression after 3 years.
Setting
Indian Spinal Injuries Center.
Methods
Twenty-eight spinal cord injury participants completed the follow-up assessment of the Patient Health Questionnaire 3 years post-injury. The participants were grouped based on post-injury autonomic regulation (high and low HRV) and the somatic and cognitive manifestation of depression reflected in a depression ratio. Wilcoxson signed-rank test tested the post-injury (T1) and 3 year follow–up (T2) depression scores.
Results
Depression score reduced after 3 years of injury (p ≤ 0.05). Only the high HRV group showed a higher depression ratio (somatic/ cognitive) atfollow-up (T2) (p ≤ 0.05). No difference was observed in post-injury coping between high and low HRV groups.
Conclusion
The reduced depression score at follow-up (T2) aligns with the ‘disability paradox’ and mightindicate cognitive adaptation, specifically for those who showed autonomic adaptability in the form of post-injury high autonomic variability.
Similar content being viewed by others
Introduction
Depression with an incidence of 22% [1] in spinal cord injury has significantmental health implications [2,3,4]. Given the variability and heterogeneity in the post-injury life of those with spinal cord injury, longitudinal studies aim to offer insights into the negative consequences of the injury such as vulnerability to depression. Further, literature regarding changes in the risk of depression over time in spinal cord injury has not been conclusive. Hoffmann et al. [4] observed that more than half of depressed SCI were not depressed at the end of 5 years. Similarly,a study by Arango et al. [5] showed a significant decrease in the odds of depression over 5 years. In their study, Bonanno et al. [6] showed two prominent trajectories of depression, an initial increase in depression followed by a decrease, and the other showed delayed onset of depression with a longitudinal increase. On the other hand, some studies show that depression scoresremain consistent over time [7, 8]. Saunders et al. [7] observed that depression scoresremained unaltered over 5 years. The study by Bombardier et al. [8] observed low depression score after 1 year. In another observation by Craig et al. [9], there was no change in depression 6 months post-discharge. Thus, literature has been ambiguous regarding changes in depression scores over time in spinal cord injury. The risk of developing depression in spinal cord injury might differ across contexts;significantly fewer studies have documented depression risk in spinal cord injury in developing countries with poor resources and infrastructural support for those with disabilities [10,11,12]. However, the factors contributing to the trajectory of depression over time remain poorly understood, with a dearth of studies examining depression longitudinally in spinal cord injury in the Asian population.
Depression has somatic and cognitive manifestations,thus, variability in depression reports is possible, and depression trajectory may be heterogeneous for individuals with spinal cord injury(SCI). Krause et al. observed that the cognitive manifestation is predictive of depression longitudinally [13, 14], indicating that cognitive adaptation to the injury might be an essential factor in the post-injury depression trajectory.Richardson and Richards [15], in a follow-up study on depression in individuals with SCI, observed both somatic and cognitive contributions to the manifestation of depression in a period of 25-year.
Understanding depression with somatic and cognitive manifestations in spinal cord injury might offer insights into the extent of post-injury adaptability. For instance, a widely explored post-injury response is coping with injury, an adjustment to injury that is a significant predictor of depression over time. Coping strategies of acceptance are associated with a reduced risk of developing depression longitudinally [4, 5, 16, 17]. A 10-year longitudinal study done by Pollard and Keneddy [17] observed that acceptance based coping predicted depression,and the coping strategy adapted by individuals with SCI was consistent over time [17].
Depression is associated with a physiological component of cardiac autonomic regulation, esp. heart rate variability [18, 19]. Heart rate variability (HRV) denotes variability between two successive heartbeats and measures autonomic regulation.HRV has time and frequency domains.The frequency domain of HRV has low frequency (LF) and high Frequency (HF) components. Depression is associated with low parasympathetic nervous system activity, indicated by low HF-HRV [19, 20]. In our earlier work relating HRVwith depression in individuals with SCI, probable depressed individuals with SCI had low HF-HRV than the non-depressed [21]. Our next work explored the psychophysiological aspect of depression in spinal cord injury [22] and depression was associated with lower odds in individuals with SCI with high HRV. The sympathetic dominance in form of high LF/ HF ratio was negatively associated with somatic- cognitive manifestations of depression in individuals with SCI [22]. Similarly, acceptance based coping was negatively related to sympathetic dominance in LF/HF ratio [22]. These results [22] indicate that the widely reported phenomenon of ‘Disability Paradox’ which is good quality of life and absence or reduced mental health problems despite disability [23, 24]. It is possible that factors such as depression manifestations, coping methods, and autonomic regulation in the form of heart rate variability contribute to counter-intuitive observation of ‘Disability Paradox’. Thus,the current study first examines change in depression after 3 years,andexplores whether post-injury (at discharge) HRV and coping is linked with depression after 3 years.
Methods
Participants
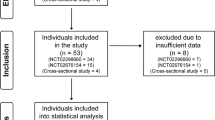
A total of twenty-eight participants participated in the current study. The participants from a previous study cohort [22] were telephonically contacted for participation in the current study. Out of the 91 participants of the previous study cohort, 32 participants (35.16%) could not be contacted for follow-up. Out of the remaining 59 participants who responded, 15 did not consent to participation. For the rest, 16 participants,the caregivers wanted to respond on behalf of the participant, thus were excluded. Finally, responses of 28 participants (Mean age = 28.68 years, SD = 5.70) were included for the current study. The initial cohort had participants with a level of injury from C-4 to T-12 with AIS levels A, B, and C. All participants were above 18 years of age. The participants diagnosed with any other neurological or psychiatric conditions other than spinal cord injury were excluded from the initial cohort.
Procedure
All participants were telephonically contacted and consent for participation was obstained. Following the consent for participation, the PHQ-9 questions were asked to the participant and the response was recorded.
Research ethics
All applicable institutional and governmental regulations concerning human volunteers’ ethical use were followed during this research. The follow-up was done for the participants who volunteered to participate in the study approved by the Ethical Committee (ISIC/IIRS/RP/2015/081). The study is a part of the doctoral work of the corresponding author.
Measures used
Depression
Patient health questionnaire- 9
PHQ- 9 [25] was used to screen depression in spinal cord injury participants.Patient Health questionnaire-9 is a self-administered nine-item depression questionnaire to screen depression.The questions are marked by how persistent the symptoms have been in the past 2 weeks: 0—not at all, 1—several days, 2—more than half of the days, 3—nearly every day.The total score of the questionnaire ranges from 0–27.PHQ-9 was studied as somatic and cognitivemanifestations [14].
Coping
Spinal cord lesion coping strategies questionnaire
Spinal Cord Lesion Coping Strategies Questionnaire (SCL-CSQ) [26] has been used to study coping.The scale has the following factors: Acceptance (four items), Fighting spirit (five items), and Social reliance (three items). The data used in the analysis was from the previous cohort [22]. Acceptance and Fighting Spirit as coping factors are considered in the current work.
Heart rate variability- Kubios Heart rate variability software (standard version 2.1)
Polar RS 800 CX and Kubios Heart rate variability software (standard version 2.1) calculated the heart rate variability. As per norms of recording of HRV [27], a 5-min recording at a sampling rate of 1000 Hz was used. The frequency domain of HRV has been associated with depression [20, 28] and is highly reproducible in individuals with SCI [29]. Thus, specifically, frequency-domain components of H.F.were used for the study.
Data analysis
To derive insights into understandingcopingand depression, the coping and depression ratios were computed. Acceptance-based coping is associated with understanding the injury, indicating the cognitive adjustments post-injury, whereas fighting spirit denotes the bodily adaptions post-injury, indicating the somatic adjustments [26]. Thus, a ratio of fighting spirit and acceptance-based coping was computed [22], indicating a somatic/cognitive coping ratio post-injury(T1). Depression has somatic and cognitive manifestations. Thus, the depression ratio was computed as a somatic/ cognitive manifestation ratio of depression. Depression and coping ratios reflect the sympathetic dominance in the LF/HF ratio [22].
Separate ratios for depression were computed, each for T1 (first year) and T2(follow-up). Variables that contained a value of ‘0’ were uniformly added a ‘1’ to ensure a non-zero denominator in the ratio. A cut-off score of 10 grouped the participants as probable depressed and non-depressed [30].
Data analysis was done using the Windows version of SPSS 21. Shapiro Wilk test assessed the normality of PHQ-9 and Spinal Cord Lesion Coping Strategies Questionnaire(SCL-CSQ) scores. Participants were grouped into high and low HRV groups according to their HF-HRV at T1, using median split method [22]. Since normality was not present, Wilcoxson Signed rank test was done to test the difference in depression ratio at T1 and T2 within the high and low HRV groups. Mann–Whitney U tests were done to test the difference in T1coping ratio (fighting spirit/ acceptance) between HRV groups and between probable depressed and non-depressed groups. The level of significance was set at p ≤ 0.05.
Results
Participants’ characteristics
The participants’ characteristics are in Table 1. The participants were divided into – High and Low HRV groups.
Change in depression between T1 and T2
Compared to the depression scores of twenty-eight spinal cord injury participants after 1 year of injury (T1 median = 12.50), depression scores reduced at 3 years follow-up (T2 median = 10.00) with W = 74.50, p = 0.02 (C.I. = −4.00 to −0.50), indicating variability in the trajectory of post-injury depression.
Like others, we used a cut-off score of 10 in PHQ-9 [30] to identify probable depression and to understand how autonomic variability might be linked with depression as observed in our earlier work [22]. In the high HRV group, nine participants had scores reflecting probable depression at T1, and at T2,there were seven, whereas, in the low HRV group, thirteen participants showed scores reflecting probable depression at T1, and ten participants at T2 (Table 2).
To understand the change in somatic and cognitive manifestations of depression, we took a ratio of depression scores to reflect a balance of somatic and cognitive manifestations of depression in the form of a ratio. A comparison of depression ratio(somatic–cognitive manifestation ratio) at T1 and T2 was undertaken using Wilcoxsin signed-rank test for the HRV groups. Results of the high HRV group showed a significant difference between depression ratio(somatic–cognitive manifestation ratio) at T1(Median = 0.80) and T2(Median = 1.00) with W = 75.00, p = 0.04 (Table 3). In the low HRV group, there was no significant difference observed between depression ratio(somatic–cognitive manifestation ratio) at T1(Median = 0.80) and T2(Median = 0.83) with W = 66.00, p = 0.15. These results indicate that the risk of depression in individuals with SCI varies with time, and high autonomic variability might be associated with an altered balance between the cognitive and somatic manifestations of depression.
Comparison of coping ratio between HRV groups
The coping ratio (fighting spirit/ acceptance) at T1 was compared between HRV groups using the Mann–Whitney U test. There was no difference observed between high HRV (Median = 1.00, N = 15) and low HRV (Median = 1.00, N = 13) with U = 86.00, p = 0.62.
Comparison of coping ratio between probable depressed and non-depressed at T2
Similarly T1 coping ratio did not differ between follow up (T2) probable depressed (Median = 1.00, N = 17) and non-depressed (Median = 1.00, N = 11) with U = 74.00, p = 0.38.
Discussion
The current study documented reduced depression scores after 3 years of injury (T2) compared to depression scores at 1 year (T1). Although counter-intuitive, these results align with Hoffman et al. [4] and Arango et al. [5], wherein a decrease in depression was observed after 5 years of injury. Others have also observed a trajectory of decline in depression over time [6]. These results align with the “Disability Paradox,” wherein studies have documented normal or high well-being despite a disability such as spinal injury. Migliorini et al. [23] observed this paradoxical normal to high well-being in spinal cord injury, emphasizing cognitive adaptation to injury.
Depression is significant psychological morbidity following spinal cord injury [3]. In the present study, results indicate that autonomic variability, post-injury coping might be linked tocognitive and somatic aspects of depressionafter 3 years of injury.Depression is a complex construct with somatic and cognitive manifestation [13, 15]; thus, a somatic and cognitive manifestation ratio was computed for the study.Higher depression ratio (somatic /cognitive) at T2 indicated somatic dominanceincreased in the high HRV group (W = 75.00, p = 0.04) but remained unaltered in the low HRV group (W = 66.00, p = 0.15). The cognitive manifestationof influences follow-up depression scoresmore than the somatic manifestation of depression [13, 14]. High HRV is associated with better cognitive abilities [31], indicating greater adaptability, which may be why individuals with SCI with high HRV in the first year post-injury might be at a lower risk of experiencing depression at the follow-up. Our earlier work observed that low HF-HRV at in spinal cord injury participants might be a risk for depression [24], and autonomic variability was linked with coping and depression risk [22].
Interestingly, there was no difference observed in the T1 coping ratio between the two HRV groups indicating a similar coping strategy irrespective of HRV groups. In contrast to the literature supporting better coping associated with less depression [32], the initial coping ratio (T1) did not differ between probable depressed and non-depressed at follow-up (T2). Thus, the post-injury autonomic regulation may contribute to follow-up depressionindependent of coping strategies as a psychological response to the injury.
Conclusion
Depression is often associated with poor well-being [23, 24], and therefore reduced depression scores in a follow-up might indicate cognitive adaptation to the injury. Given the low resources, policy and infrastructural support compared to developed countries, we observe potential for cognitive adaptation to spinal injury. This study explored the longitudinal risk of depression in spinal cord injury, to our knowledge, the first study to report disability paradox in a developing country of India, and to explore the link between autonomic regulation and depression trajectory.The HRV as a measure at discharge could be an important physiological marker for depression in spinal cord injury.
Limitations
The attrition rate has been a problem with previous longitudinal studies [5, 14]. The present results have to be interpreted within the context of a high attrition rate resulting in a small sample size. Although the results are preliminary, these might be useful in advocating the inclusion of autonomic measures such as HRV with self-reported questionnaires and surveys as measures that identify spinal cord injury participants at risk for developing depression. Future studies should examine the extent of generalizability of such paradoxical effects.
Data availability
The datasets generated during and analyzed during the current study are not publicly available to maintain confidentiality as they contain information regarding spinal cord injury patients’ mental well-being. However, these are available from the corresponding author on reasonable request.
References
Williams R, Murray A Prevalence of depression after spinal cord injury: A meta-analysis. Arch Phys Med Rehabil [Internet]. Elsevier Ltd; 2015;96:133–40. https://doi.org/10.1016/j.apmr.2014.08.016.
Craig AR, Hancock KM, Dickson HG. Spinal cord injury: A search for determinants of depression two years after the event. Br J Clin Psychol. 1994;33:221–30.
Craig A, Tran Y, Middleton J. Psychological morbidity and spinal cord injury: A systematic review. Spinal Cord. 2009;47:108–14.
Hoffman JM, Bombardier CH, Graves DE, Kalpakjian CZ, Krause JS. A longitudinal study of depression from 1 to 5 years after spinal cord injury. Arch Phys Med Rehabil [Internet]. Elsevier Inc. 2011;92:411–8. https://doi.org/10.1016/j.apmr.2010.10.036.
Arango-Lasprilla JC, Ketchum JM, Starkweather A, Nicholls E, Wilk AR. Factors predicting depression among persons with spinal cord injury 1 to 5 years post injury. NeuroRehabilitation. 2011;29:9–21.
Bonanno GA, Kennedy P, Galatzer-Levy IR, Lude P, Elfström ML. Trajectories of resilience, depression, and anxiety following spinal cord injury. Rehabil Psychol. 2012;57:236–47.
Saunders LL, Krause JS, Focht KL. A longitudinal study of depression in survivors of spinal cord injury. Spinal Cord. 2012;50:72–7. https://pubmed.ncbi.nlm.nih.gov/21808257/
Bombardier CH, Adams LM, Fann JR, Hoffman JM. Depression trajectories during the first year after spinal cord injury. Arch Phys Med Rehabil. 2016;97:196–203.
Craig A, Nicholson Perry K, Guest R, Tran Y, Dezarnaulds A, Hales A, et al. Prospective Study of the Occurrence of Psychological Disorders and Comorbidities after Spinal Cord Injury. Arch Phys Med Rehabil [Internet]. Elsevier Ltd; 2015;96:1426–34. https://doi.org/10.1016/j.apmr.2015.02.027.
Lim S-W, Shiue Y-L, Ho C-H, Yu S-C, Kao P-H, Wang J-J, et al. Anxiety and depression in patients with traumatic spinal cord injury: a nationwide population-based cohort study. PLoS One. 2017;12:e0169623. https://doi.org/10.1371/journal.pone.0169623.
Singh R, Rohilla RK, Siwach R, Dhankar SS, Kaur K. Understanding psycho-social issues in persons with spinal cord injury and impact of remedial measures. Int J Psychosoc Rehabil. 2011;16:104–11.
Arafat S, Ahmed S, Ahmed F, Zaman S, Andalib A. Depression in spinal cord injury patients: a cross-sectional observation with PHQ-9 in a rehabilitation center of Bangladesh. J Behav Heal. 2017;7:1.
Krause JS, Reed KS, McArdle JJ. Prediction of somatic and non-somatic depressive symptoms between inpatient rehabilitation and follow-up. Spinal Cord [Internet]. Spinal Cord. 2010;48:239–44. https://pubmed.ncbi.nlm.nih.gov/19736559/
Krause JS, Reed KS, McArdle JJ Factor structure and predictive validity of somatic and nonsomatic symptoms from the patient health questionnaire-9: A longitudinal study after spinal cord injury. Arch Phys Med Rehabil [Internet]. Elsevier Inc. 2010;91:1218–24. https://doi.org/10.1016/j.apmr.2010.04.015.
Richardson EJ, Richards JS. Factor structure of the PHQ-9 screen for depression across time since injury among persons with spinal cord. Injury 2008;53:243–9.
Elfström ML, Kreuter M, Rydén A, Persson LO, Sullivan M. Effects of coping on psychological outcome when controlling for background variables: A study of traumatically spinal cord lesioned persons. Spinal Cord. 2002;40:408–15.
Pollard C, Kennedy P. A longitudinal analysis of emotional impact, coping strategies and post-traumatic psychological growth following spinal cord injury: A 10-year review. Br J Health Psychol. 2007;12:347–62.
Beauchaine TP. Respiratory sinus arrhythmia: A transdiagnostic biomarker of emotion dysregulation and psychopathology. Curr Opin Psychol. 2015;3:43–7.
Koch C, Wilhelm M, Salzmann S, Rief W, Euteneuer F. A meta-analysis of heart rate variability in major depression. Psychol Med. 2019;49:1948–57. https://doi.org/10.1017/S0033291719001351.
Kemp AH, Quintana DS, Gray MA, Felmingham KL, Brown K, Gatt JM, et al. Impact of Depression and Antidepressant Treatment on Heart Rate Variability: A Review and Meta-Analysis. Biol Psychiatry [Internet]. Elsevier Inc. 2010;67:1067–74. https://doi.org/10.1016/j.biopsych.2009.12.012.
Mitra S, Singh V. Autonomic dysregulation and low heart rate variability in spinal cord injury(SCI): A marker for depression. Int J Physiother. 2020;7:108–13.
Singh V, Mitra S. Psychophysiological impact of spinal cord injury: Depression, coping and heart rate variability. J Spinal Cord Med [Internet]. 2022;0:1–9. https://doi.org/10.1080/10790268.2022.2052503.
Migliorini C, Tonge B. Reflecting on subjective well-being and spinal cord injury. J Rehabil Med. 2009;41:445–50.
Albrecht GL, Devlieger PJ. The disability paradox: highly qualified of life against all odds. Soc Sci Med. 1999;48:977–88.
Kalpakjian CZ, Toussaint LL, Albright KJ, Bombardier CH, Krause JK, Tate DG, et al. Patient health questionnaire-9 in spinal cord injury: An examination of factor structure as related to gender. J Spinal Cord Med. 2009;32:147–56.
Elfström ML, Rydén A, Kreuter M, Persson LO, Sullivan M. Linkages between coping and psychological outcome in the spinal cord lesioned: Development of SCL-related measures. Spinal Cord. 2002;40:23–9.
Malik M, Bigger JT, Camm AJ, Kleiger RE, Malliani A, Moss AJ. et al. Task force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability standards of measurement, physiological interpretation, and clinical use. Eur Heart J. 1996;17:354–81.
Koch C, Wilhelm M, Salzmann S, Rief W, Euteneuer F A meta-Analysis of heart rate variability in major depression. Psychol Med. Cambridge University Press; 2019;49:1948–57.
Ditor DS, Kamath MV, MacDonald MJ, Bugaresti J, McCartney N, Hicks AL. Reproducibility of heart rate variability and blood pressure variability in individuals with spinal cord injury. Clin Auton Res. 2005;15:387–93.
Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: Validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606–13.
Thayer JF, Hansen AL, Saus-rose E, Psychol C, Johnsen BH. Heart rate variability, prefrontal neural function, and cognitive performance: the neurovisceral integration perspective on self-regulation, adaptation, and health. Ann Behav Med. 2009;37:141–53.
Kennedy P, Marsh N, Lowe R, Grey N, Short E, Rogers B. A longitudinal analysis of psychological impact and coping strategies following spinal cord injury. Br J Health Psychol. 2000;5:157–72.
Acknowledgements
We acknowledge all the Spinal cord injury participants for their participation.
Author information
Authors and Affiliations
Contributions
Conception or design of the work, data analysis and interpretation, drafting the article, critical revision of the article, final approval of the version to be published: VS and SM. Data collection: SM.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, V., Mitra, S. Autonomic variability, depression and the disability paradox in spinal cord injury. Spinal Cord Ser Cases 8, 76 (2022). https://doi.org/10.1038/s41394-022-00542-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-022-00542-6