Abstract
Introduction
Osteochondromas are benign bone tumors which occur as solitary lesions or as part of the syndrome multiple hereditary exostoses. While most osteochondromas occur in the appendicular skeleton, they can also occur in the spine. Most lesions are asymptomatic however some may encroach on the spinal cord or the nerve roots causing neurological symptoms. While most patients with osteochondromas undergo laminectomy without fusion, laminectomy with fusion is indicated in appropriately selected cases of spinal decompression.
Case presentation
We present a case of a 32-year-old male with history of multiple hereditary exostoses who presented with symptoms of bilateral upper extremity numbness and complaints of gait imbalance and multiple falls. He reported rapid progression of his symptoms during the 10 days before presentation. Computed tomography of the cervical spine revealed a lobulated bony tumor along the inner margin of the cervical 4 lamina. He underwent cervical 3 and 4 laminectomies, partial cervical 2 and 5 laminectomies and cervical 3–5 mass screw placement. Pathology was consistent with osteochondroma. The patient’s symptoms had markedly improved at follow-up.
Conclusion
According to our literature review, osteochondromas most commonly occur at cervical 2 and cervical 5. We present a case of an osteochondroma at a less common level, cervical 4. While most osteochondromas are addressed with laminectomy without arthrodesis, the decision of whether arthrodesis is necessary should be considered in all patients with osteochondroma as with any cervical decompression.
Similar content being viewed by others
Introduction
Osteochondromas are benign bone tumors which can occur as solitary lesions or as part of the autosomal dominant hereditary syndrome, multiple hereditary exostoses (MHE). Osteochondromas develop when small fragments of epiphyseal growth plate cartilage herniate through the periosteum and continue to undergo endochondral ossification, resulting in the classic bony tumors with cartilaginous caps [1]. Fragmentation of the epiphyseal growth plate may be caused by trauma or by deficient periosteum. Osteochondromas typically occur in the appendicular skeleton, while only 1–9% of all osteochondromas occur in the spine [2]. In MHE, 7–9% of lesions occur in the spine, compared with 1–4% of solitary osteochondromas [3].
Between 50 and 58% of spinal osteochondromas are found in the cervical spine, most commonly the posterior elements (64%) [3, 4]. One theory for the higher prevalence of osteochondromas in the cervical spine is the increased mobility and microtrauma that occurs in the cervical spine, which may lead to displacement of cartilage and subsequent exostosis [5].
Here we present a case of a 32-year-old male with multiple hereditary exostosis with severely progressive myelopathy due to an osteochondroma involving the cervical 4 lamina as well as a review of the literature. Most cervical osteochondromas are addressed with laminectomy without arthrodesis. We present a case in which the patient underwent laminectomy and resection with arthrodesis and review the indications for cervical fusion in the setting of myelopathy.
Case presentation
A 32-year-old male presented for symptoms of bilateral upper extremity numbness and complaints of imbalance with multiple recent falls. His symptoms had progressed rapidly, worsening over the course of 10 days. Medical history was significant for multiple hereditary exostosis, generalized epilepsy, autism spectrum disorder, and bipolar disorder. Family history revealed his father had exostosis as well at an unspecified site. Physical examinations revealed spastic tone in the upper extremities with 4/5 weakness throughout bilateral upper extremities and 4+/5 throughout bilateral lower extremities. Sensation was decreased to light touch in bilateral hands, worse on the left side, consistent with an American Spinal Injury Association score D, and Nurick grade 4 [6, 7]. Deep tendon reflexes were exaggerated in the upper and lower extremities bilaterally with pathologic Hoffman’s reflex, upgoing Babinski sign, and clonus noted bilaterally. Gait was spastic and scissored. Patient ambulated with the assistance of a front-wheeled walker.
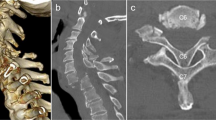
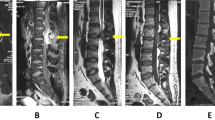
Preoperative imaging consisted of computerized tomography (CT) and magnetic resonance imaging (MRI) of the cervical spine. CT of the cervical spine demonstrated a lobulated, circumscribed bony excrescence along the inner margin of the cervical 4 (C4) lamina measuring 1.1 × 0.8 × 1.2 cm in the longitudinal, AP and transverse dimensions with extension anteriorly into the spinal canal and producing marked central stenosis (Fig. 1). MRI of the cervical spine demonstrated marked stenosis at the C4-5 level with marked mass effect upon the spinal cord and adjacent T2 signal hyperintensity (Fig. 2). Further imaging of the axial spine demonstrated multiple benign appearing bony excrescences involving the multiple thoracic transverse processes, right lamina of thoracic 11, lumbar 4 lamina, upper left posterior-lateral iliac bone, iliac side of the left sacroiliac joint, and upper right posterior iliac bone.
Given his rapid progression of symptoms and neurologic presentation, he was admitted to the hospital for urgent decompression. He underwent cervical 3 and 4 laminectomies, partial cervical 2 and 5 laminectomies, and cervical 3–5 lateral mass screw placement. Motor and somatosensory evoked potentials were monitored. Adhesions between the mass and dura were separated using microsurgical techniques and the mass was successfully removed en bloc without durotomy. Pathologic examination was consistent with osteochondroma.
There were no immediate postoperative adverse events after surgery. His postoperative course was uncomplicated, and he was discharged home on postoperative day two with improved strength in his upper extremities.
On outpatient follow-up, his symptoms had improved significantly with noted improvement in balance and decreased numbness in the fingers. Physical examination revealed no new focal deficits at 1 month post operatively. Routine cervical X-ray on follow-up demonstrated stable hardware placement (Fig. 3). MRI postoperatively showed gross total resection with decompression of the spinal cord (Fig. 4). The patient was seen subsequently for 9-month postoperative follow-up at which time he denied any neck pain or balance symptoms. He was neurologically intact on physical exam. He stated that he had regained strength and was dropping objects less frequently compared with prior to surgery. The patient was pleased with his postoperative recovery.
Discussion
Osteochondromas most commonly occur in the appendicular skeleton, while only 1–9% of patients have involvement of the spine [3]. However, spinal osteochondromas are likely underreported as they are usually asymptomatic [8]. Fifty to 58% of spinal osteochondromas are found in the cervical spine, C2 (29%), and C5 (24%) being the most commonly affected levels (Fig. 5). The osteochondroma in the current study was located at C4, a less commonly affected level (17%, Fig. 5) [1, 3, 9,10,11,12,13,14,15,16,17,18,19,20]. Osteochondromas frequently arise from the posterior elements, from spinous processes and lamina, where ossification centers are located [3, 21].
The average age at presentation of patients with spinal osteochondromas is 30 years with a higher incidence in males (2.5:1). Those with MHE are likely to present at younger ages [22]. Most spinal osteochondromas are asymptomatic because they grow outward and away from the spinal canal. When tumor growth is away from the spinal canal, those who become symptomatic most commonly present with oncological pain of the spine [21]. When tumor growth is toward the spinal canal or neural foramina, patients may present with myelopathy or radicular symptoms. In a study of solitary osteochondromas, 24 of 55 (43.6%) patients with cervical osteochondromas were myelopathic, while 14 (25.4%) had radiculopathy [23].
The rate of malignant transformation varies in the literature, with authors reporting 1–5% in those with solitary osteochondromas and 3–25% in patients with MHE [3, 21, 22, 24]. Asymptomatic lesions without malignant features may be observed with radiographic surveillance [23]. Features concerning for malignant transformation include a cartilaginous cap that is greater than 3 centimeters (cm) thick, recurrence after complete resection, growth after skeletal maturity, or rapid growth [22]. Some authors have suggested removal of asymptomatic lesions when there is displacement of the spinal cord or effacement of the thecal sac [2]. Treatment for symptomatic spinal osteochondromas is surgical excision with complete resection of the cartilaginous cap to prevent recurrence, and does not require adjuvant chemotherapy or radiation [21, 23]. Most symptomatic cases that have been reported in the literature have undergone laminectomy or hemilaminectomy given the posterior location of the exostoses [5]. In prior reports, laminectomy alone is the most common surgical technique used for cervical osteochondromas [1, 3, 24]. In a review of cervical osteochondromas by Veeravagu et al. 16 of 25 cases were treated with posterior decompression without fusion, while two patients underwent laminoplasty and the remainder were unspecified [1]. Similarly, in Gille et al. 33 of 36 patients with cervical osteochondromas underwent laminectomies while two did not undergo surgery and one was unspecified [24]. None of the patients with cervical osteochondroma underwent fusion. However, as with any other setting of cervical stenosis that requires decompression, indications for fusion should be carefully considered.
For posterior pathology, options include cervical laminectomy alone, cervical laminectomy with arthrodesis, and cervical laminoplasty. The appropriate operation in the subaxial cervical spine should take into consideration several key elements including the stability of the spine, compressive location, and sagittal balance [25]. In the setting of osteochondroma involving the posterior cervical spinal elements, intralesional excision or incomplete resection is associated with a higher recurrence and as such it is recommended to remove the osteochondroma as well its cartilaginous cap [26, 27]. Recurrence after resection of solitary osteochondromas has been reported to be <4% [22]. Considering these goals, posterior surgical options in most cases of cervical osteochondromas involves a decision between laminectomy alone or laminectomy with arthrodesis.
Considerations for laminectomy with arthrodesis in our patient included his young age with the cumulative lifetime risk of progressive kyphosis requiring additional operations. Further intraoperative findings of a mobile segment further supported the utilization of laminectomy with arthrodesis. En bloc, as opposed to intralesional tumor resection of the involved lamina, also widens the extent of the laminectomy and contributes to the risk of subsequent kyphosis. The risk of postlaminectomy increases substantially if foraminotomies and medial facetectomies are added to the laminectomy.
Conclusion
We presented a case of a young patient with a spinal osteochondroma, review the literature regarding cervical osteochondromas as well as briefly review surgical considerations. We found that cervical osteochondromas most commonly occur at C2 and C5. Our patient had an osteochondroma at a less common level, C4. While most osteochondromas are addressed with laminectomy without arthrodesis, the decision of whether arthrodesis is necessary should be considered in all patients with osteochondroma as with any cervical decompression. Most importantly, a wide laminectomy may be required for total resection, necessitating arthrodesis to prevent postlaminectomy kyphosis. Cervical alignment should also be considered. Our patient recovered well after three and four laminectomies, partial cervical 2 and 5 laminectomies, and cervical 3–5 lateral mass screw placement.
References
Veeravagu A, Li A, Shuer LM, Desai AM. Cervical osteochondroma causing myelopathy in adults: management considerations and literature review. World Neurosurg. 2017;97:752.e5–13.
Roach JW, Klatt JW, Faulkner ND. Involvement of the spine in patients with multiple hereditary exostoses. J Bone Joint Surg Am. 2009;91:1942–8.
Giudicissi-Filho M, de Holanda CV, Borba LA, Rassi-Neto A, Ribeiro CA, de Oliveira JG. Cervical spinal cord compression due to an osteochondroma in hereditary multiple exostosis: case report and review of the literature. Surg Neurol. 2006;66 (Suppl 3):S7–11.
Sinelnikov A, Kale H. Osteochondromas of the spine. Clin Radiol. 2014;69:e584–90.
Lotfinia I, Vahedi A, Aeinfar K, Tubbs RS, Vahedi P. Cervical osteochondroma with neurological symptoms: literature review and a case report. Spinal Cord Ser Cases. 2017;3:16038.
Kirshblum SC, Waring W, Biering-Sorensen F, Burns SP, Johansen M, Schmidt-Read M, et al. Reference for the 2011 revision of the International Standards for Neurological Classification of Spinal Cord Injury. J Spinal Cord Med. 2011;34:547–54.
Nurick S. The pathogenesis of the spinal cord disorder associated with cervical spondylosis. Brain. 1972;95:87–100.
Bess RS, Robbin MR, Bohlman HH, Thompson GH. Spinal exostoses: analysis of twelve cases and review of the literature. Spine. 2005;30:774–80.
Roblot P, Alcalay M, Cazenave-Roblot F, Levy P, Bontoux D. Osteochondroma of the thoracic spine. Report of a case and review of the literature. Spine. 1990;15:240–3.
Vahedi P, Rymarczuk G, Gillick JL, Prasad SK, Lotfinia I. Letter to the editor: intraspinal cervical osteochondroma. World Neurosurg. 2017;101:805–10.
Ezra N, Tetteh B, Diament M, Jonas AJ, Dickson P. Hereditary multiple exostoses with spine involvement in a 4-year-old boy. Am J Med Genet A. 2010;152A:1264–7.
Han IH, Kuh SU. Cervical osteochondroma presenting as brown-sequard syndrome in a child with hereditary multiple exostosis. J Korean Neurosurg Soc. 2009;45:309–11.
Aldea S, Bonneville F, Poirier J, Chiras J, George B, Carpentier A. Acute spinal cord compression in hereditary multiple exostoses. Acta Neurochir (Wien). 2006;148:195–8.
Scher N, Panje WR. Osteochondroma presenting as a neck mass: a case report. Laryngoscope. 1988;98:550–3.
Rao A, Abraham RG, Rajshekhar V. Osteochondroma of rib with neural foraminal extension and cord compression. Neurol India. 2007;55:428–9.
Tahasildar N, Sudesh P, Goni V, Tripathy SK. Giant osteochondroma of axis in a child with multiple hereditary exostoses: case report and review of literature. J Pediatr Orthop B. 2012;21:280–5.
Raswan US, Bhat AR, Tanki H, Samoon N, Kirmani AR. A solitary osteochondroma of the cervical spine: a case report and review of literature. Childs Nerv Syst. 2017;33:1019–22.
Sofka CM, Saboeiro GR, Schneider R. Multiple hereditary exostoses. HSS J. 2005;1:49–51.
Sultan M, Khursheed N, Makhdoomi R, Ramzan A. Compressive myelopathy due to osteochondroma of the atlas and review of the literature. Pediatr Neurosurg. 2016;51:99–102.
Wang V, Chou D. Anterior C1-2 osteochondroma presenting with dysphagia and sleep apnea. J Clin Neurosci. 2009;16:581–2.
Sciubba DM, Macki M, Bydon M, Germscheid NM, Wolinsky JP, Boriani S, et al. Long-term outcomes in primary spinal osteochondroma: a multicenter study of 27 patients. J Neurosurg Spine. 2015;22:582–8.
Srikantha U, Bhagavatula ID, Satyanarayana S, Somanna S, Chandramouli BA. Spinal osteochondroma: spectrum of a rare disease. J Neurosurg Spine. 2008;8:561–6.
Yakkanti R, Onyekwelu I, Carreon LY, Dimar JR 2nd. Solitary osteochondroma of the spine-a case series: review of solitary osteochondroma with myelopathic symptoms. Glob Spine J. 2018;8:323–39.
Gille O, Pointillart V, Vital JM. Course of spinal solitary osteochondromas. Spine. 2005;30:E13–9.
McAllister BD, Rebholz BJ, Wang JC. Is posterior fusion necessary with laminectomy in the cervical spine? Surg Neurol Int. 2012;3 (Suppl 3):S225–31.
Bernard SA, Murphey MD, Flemming DJ, Kransdorf MJ. Improved differentiation of benign osteochondromas from secondary chondrosarcomas with standardized measurement of cartilage cap at CT and MR imaging. Radiology. 2010;255:857–65.
Chang DG, Park JB. Osteochondroma arising from the transverse process of the lower cervical spine in an elderly patient. World Neurosurg. 2019;130:450–3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fowler, J., Takayanagi, A., Siddiqi, I. et al. Cervical osteochondroma: surgical planning. Spinal Cord Ser Cases 6, 44 (2020). https://doi.org/10.1038/s41394-020-0292-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-020-0292-7