Abstract
Background
Prediction of side-specific extraprostatic extension (EPE) is crucial in selecting patients for nerve-sparing radical prostatectomy (RP). Multiple nomograms, which include magnetic resonance imaging (MRI) information, are available predict side-specific EPE. It is crucial that the accuracy of these nomograms is assessed with external validation to ensure they can be used in clinical practice to support medical decision-making.
Methods
Data of prostate cancer (PCa) patients that underwent robot-assisted RP (RARP) from 2017 to 2021 at four European tertiary referral centers were collected retrospectively. Four previously developed nomograms for the prediction of side-specific EPE were identified and externally validated. Discrimination (area under the curve [AUC]), calibration and net benefit of four nomograms were assessed. To assess the strongest predictor among the MRI features included in all nomograms, we evaluated their association with side-specific EPE using multivariate regression analysis and Akaike Information Criterion (AIC).
Results
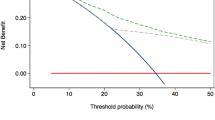
This study involved 773 patients with a total of 1546 prostate lobes. EPE was found in 338 (22%) lobes. The AUCs of the models predicting EPE ranged from 72.2% (95% CI 69.1–72.3%) (Wibmer) to 75.5% (95% CI 72.5–78.5%) (Nyarangi-Dix). The nomogram with the highest AUC varied across the cohorts. The Soeterik, Nyarangi-Dix, and Martini nomograms demonstrated fair to good calibration for clinically most relevant thresholds between 5 and 30%. In contrast, the Wibmer nomogram showed substantial overestimation of EPE risk for thresholds above 25%. The Nyarangi-Dix nomogram demonstrated a higher net benefit for risk thresholds between 20 and 30% when compared to the other three nomograms. Of all MRI features, the European Society of Urogenital Radiology score and tumor capsule contact length showed the highest AUCs and lowest AIC.
Conclusion
The Nyarangi-Dix, Martini and Soeterik nomograms resulted in accurate EPE prediction and are therefore suitable to support medical decision-making.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Feng TS, Sharif-Afshar AR, Wu J, Li Q, Luthringer D, Saouaf R, et al. Multiparametric MRI improves accuracy of clinical nomograms for predicting extracapsular extension of prostate cancer. Urology. 2015;86:332–7.
Rayn KN, Bioom JB, Gold SA, Hale GR, Baiocco JA, Mehralivand S, et al. Added value of multiparametric magnetic resonance lmaging to clinical nomograms tor predicting adverse pathology in prostate cancer. J Urol. 2019;200:1041–7.
Gandaglia G, Ploussard G, Valerio M, Mattei A, Fiori C, Roumiguié M, et al. The key combined value of multiparametric magnetic resonance imaging, and magnetic resonance imaging–targeted and concomitant systematic biopsies for the prediction of adverse pathological features in prostate cancer patients undergoing radical prostatect. Eur Urol. 2020;77:733–41.
Martini A, Gupta A, Lewis SC, Cumarasamy S, Haines KG, Briganti A, et al. Development and internal validation of a side-specific, multiparametric magnetic resonance imaging-based nomogram for the prediction of extracapsular extension of prostate cancer. BJU Int. 2018;122:1025–33.
Nyarangi-Dix J, Wiesenfarth M, Bonekamp D, Hitthaler B, Schütz V, Dieffenbacher S, et al. Combined clinical parameters and multiparametric magnetic resonance imaging for the prediction of extraprostatic disease—a risk model for patient-tailored risk stratification when planning radical prostatectomy. Eur Urol Focus. 2020;6:1205–12.
Soeterik TFW, Van Melick HHE, Dijksman LM, Küsters-Vandevelde HVN, Stomps S, Schoots IG, et al. Development and external validation of a novel nomogram to predict side-specific extraprostatic extension in patients with prostate cancer undergoing radical prostatectomy. Eur Urol Oncol. 2020;S2588-9311.
Wibmer AG, Kattan MW, Alessandrino F, Baur ADJ, Boesen L, Franco FB, et al. International multi-site initiative to develop an mri-inclusive nomogram for side-specific prediction of extraprostatic extension of prostate cancer. Cancers. 2021;13:2627.
Bleeker SE, Moll HA, Steyerberg EW, Donders ART, Derksen-Lubsen G, Grobbee DE, et al. External validation is necessary in prediction research: A clinical example. J Clin Epidemiol. 2003;56:826–32.
Siontis GCM, Tzoulaki I, Castaldi PJ, Ioannidis JPA. External validation of new risk prediction models is infrequent and reveals worse prognostic discrimination. J Clin Epidemiol 2015;68:25–34.
Soeterik TFW, van Melick HHE, Dijksman LM, Küsters-Vandevelde H, Stomps S, Schoots IG, et al. Development and external validation of a novel nomogram to predict side-specific extraprostatic extension in patients with prostate cancer undergoing radical prostatectomy. Eur Urol Oncol. 2022;5:328–37.
Diamand R, Roche JB, Lievore E, Lacetera V, Chiacchio G, Beatrici V, et al. External validation of models for prediction of side-specific extracapsular extension in prostate cancer patients undergoing radical prostatectomy. Eur Urol Focus. 2023;9:309–16.
Veerman H, Heymans MW, van der Poel HG. External validation of a prediction model for side-specific extraprostatic extension of prostate cancer at robot-assisted radical prostatectomy. Eur Urol Open Sci. 2022;37:50–52.
Blas L, Shiota M, Nagakawa S, Tsukahara S, Matsumoto T, Lee K, et al. Validation of user-friendly models predicting extracapsular extension in prostate cancer patients. Asian J Urol. 2023;10:81–88.
Soeterik TFW, van Melick HHE, Dijksman LM, Küsters-Vandevelde HVN, Biesma DH, Witjes JA, et al. External validation of the Martini nomogram for prediction of side-specific extraprostatic extension of prostate cancer in patients undergoing robot-assisted radical prostatectomy. Urologic Oncol: Semin Original Investig. 2020;38:372–8.
Baco E, Rud E, Vlatkovic L, Svindland A, Eggesbø HB, Hung AJ, et al. Predictive value of magnetic resonance imaging determined tumor contact length for extracapsular extension of prostate cancer. J Urol. 2015;193:466–72.
Eurboonyanun K, Pisuchpen N, O’Shea A, Lahoud RM, Atre ID, Harisinghani M. The absolute tumor-capsule contact length in the diagnosis of extraprostatic extension of prostate cancer. Abdom Radiol. 2021;46:4014–24.
Li W, Dong A, Hong G, Shang W, Shen X. Diagnostic performance of ESUR scoring system for extraprostatic prostate cancer extension: a meta-analysis. Eur J Radiol. 2021;143:109896.
Asfuroğlu U, Asfuroğlu BB, Özer H, Gönül İI, Tokgöz N, İnan MA, et al. Which one is better for predicting extraprostatic extension on multiparametric MRI: ESUR score, Likert scale, tumor contact length, or EPE grade? Eur J Radiol. 2022;149:110228.
Ahdoot M, Wilbur AR, Reese SE, Lebastchi AH, Mehralivand S, Gomella PT, et al. MRI-targeted, systematic, and combined biopsy for prostate cancer diagnosis. N Engl J Med 2020;382:917–28.
Padhani AR, Weinreb J, Rosenkrantz AB, Villeirs G, Turkbey B, Barentsz J. Prostate imaging-reporting and data system steering committee: PI-RADS v2 status update and future directions. Eur Urol. 2019;75:385–96.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA. The 2014 international society of urological pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40:244–52.
Turkbey B, Rosenkrantz AB, Haider MA, Padhani AR, Villeirs G, Macura KJ, et al. Prostate imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur Urol. 2019;76:340–51.
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, et al. ESUR prostate MR guidelines 2012. Eur Radiol. 2012;22:746–57.
Collins GS, Reitsma JB, Altman DG, Moons KGM. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD Statement. Eur Urol. 2015;67:1142–51.
Van Calster B, Wynants L, Verbeek JFM, Verbakel JY, Christodoulou E, Vickers AJ, et al. Reporting and interpreting decision curve analysis: a guide for investigators. Eur Urol. 2018;74:796–804.
Van Buuren S, Groothuis-Oudshoorn K. Multivariate imputation by chained equations in R. J Stat Softw 2011;45:1–67.
Hou Y, Zhang YH, Bao J, Bao ML, Yang G, Shi HB, et al. Artificial intelligence is a promising prospect for the detection of prostate cancer extracapsular extension with mpMRI: a two-center comparative study. Eur J Nucl Med Mol Imaging. 2021;48:3805–16.
Cuocolo R, Stanzione A, Faletti R, Gatti M, Calleris G, Fornari A, et al. MRI index lesion radiomics and machine learning for detection of extraprostatic extension of disease: a multicenter study. Eur Radiol. 2021;31:7575–83.
Solari EL, Gafita A, Schachoff S, Bogdanović B, Villagrán Asiares A, Amiel T, et al. The added value of PSMA PET/MR radiomics for prostate cancer staging. Eur J Nucl Med Mol Imaging. 2022;49:527–38.
Nguyen LN, Head L, Witiuk K, Punjani N, Mallick R, Cnossen S, et al. The risks and benefits of cavernous neurovascular bundle sparing during radical prostatectomy: a systematic review and meta-analysis. J Urol. 2017;198:760–9.
Soeterik TFW, van Melick HHE, Dijksman LM, Stomps S, Witjes JA, van Basten JPA. Nerve sparing during robot-assisted radical prostatectomy increases the risk of ipsilateral positive surgical margins. J Urol. 2020;204:91–95.
Heetman JG, Soeterik TFW, Wever L, Meyer AR, Nuininga JE, van Soest RJ, et al. A side-specific nomogram for extraprostatic extension may reduce the positive surgical margin rate in radical prostatectomy. World J Urol. 2022;40:2919–24.
Christophe C, Montagne S, Bourrelier S, Roupret M, Barret E, Rozet F, et al. Prostate cancer local staging using biparametric MRI: assessment and comparison with multiparametric MRI. Eur J Radiol. 2020;132:109350.
Author information
Authors and Affiliations
Consortia
Contributions
Conception and design: TFWS. Acquistion of data: JGH, EJRJvan derH, PR, FZ, CK, SS, FDM, GN, GLB, FS, NvonO, NP, PATB, LW, HHEvM, RCNvandenB, GG, TFWS. Statistical analysis: JGH. Analysis and interpretation of data: JGH. Drafting of the manuscript: TFWS, JGH. Critical revision of the manuscript for important intellectual content: JGH, EJRJvanderH, PR, FZ, CK, SS, FDM, GN, GLB, FS, NvonO, NP, PATB, LW, JPAVB, HHEvanM, RCNvandenB, GG, TFWS. Supervision: TFWSoeterik.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
The Medical research Ethics Committee United (METC-U) registered the study protocol under W18.055 and concluded that the study protocol was not subjected to the Human Subject Act. The METC-U concluded that informed consent was not needed.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heetman, J.G., van der Hoeven, E.J.R.J., Rajwa, P. et al. External validation of nomograms including MRI features for the prediction of side-specific extraprostatic extension. Prostate Cancer Prostatic Dis (2023). https://doi.org/10.1038/s41391-023-00738-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41391-023-00738-3
This article is cited by
-
Applying prediction models in clinical practice: the importance of fine details
Prostate Cancer and Prostatic Diseases (2023)