Abstract
Background
To assess the diagnostic added value of sampling secondary lesions at prostate mpMRI (SL) in addition to index lesion (IL) in detecting significant prostate cancer (csPCa) when also systematic biopsy (SBx) is performed.
Methods
We relied on a cohort of 312 men with two suspicious lesions at prostate mpMRI who underwent subsequent targeted biopsy of each lesion (TBx) and concomitant SBx at two tertiary-referral centers between 2013 and 2019. The study outcome was the added value of targeting SL (i.e., the one with a lower PI-RADS score and/or the smaller size compared to IL) in the detection of csPCa. To this aim, we compared different biopsy strategies (SBx + overall TBx vs SBx + IL-targeted biopsy vs SBx + SL-targeted biopsy) and assessed whether SL features could be correlated with detection of csPCa at overall TBx in a multivariable logistic regression model (MVA).
Results
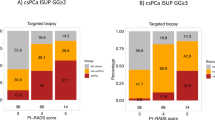
Overall, 44% of men had csPCa at TBx of all lesions while 39% and 23% of men had csPCa found in IL and SL, respectively. The rate of csPCa found at SBx, IL-TBx, and SL-TBx only was 5%, 6%, and 2%, respectively. The detection rate of csPCa for SBx + IL-TBx was 47%. The addition of SL-TBx increased csPCa detection by only 2% (p = 0.12). At MVA, neither PI-RADS of SL nor the number of cores targeting SL was associated with an increased detection of csPCa (all p > 0.3). Conversely, age (OR: 1.07), PSA (OR: 1.07), prostate volume (OR: 0.98), and PI-RADS of the IL (OR: 2.36) were independently associated with csPCa detection at TBx (all p < 0.01).
Conclusions
There is no significant benefit in terms of csPCa detection when an adequate SBx is performed in combination with IL-TBx in patients with multiple mpMRI lesions. In these men target biopsy of secondary lesions can be safely omitted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Stabile A, Giganti F, Rosenkrantz AB, Taneja SS, Villeirs G, Gill IS, et al. Multiparametric MRI for prostate cancer diagnosis: current status and future directions. Nat Rev Urol. 2019;17:41–61.
Ljungberg B, Albiges L, Bensalah K, Bex A, Giles RH, Hora M, et al. EAU Guidelines. Presented at the EAU Annual Congress Amsterdam 2020. 2020.
Turkbey B, Rosenkrantz AB, Haider MA, Padhani AR, Villeirs G, Macura KJ, et al. Prostate Imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur Urol. 2019;0232:1–12.
Kasivisvanathan V, Stabile A, Neves JB, Giganti F, Valerio M, Shanmugabavan Y, et al. Magnetic resonance imaging-targeted biopsy versus systematic biopsy in the detection of prostate cancer: a systematic review and meta-analysis (figure presented.). Eur Urol. 2019;76:284–303.
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N. Engl J Med. 2018;378:1767–77.
Stabile A, Giganti F, Emberton M, Moore CM. MRI in prostate cancer diagnosis: do we need to add standard sampling? A review of the last 5 years. Prostate Cancer Prostatic Dis. 2018;21:473–87.
Rouvière O, Puech P, Renard-penna R, Claudon M, Roy C, Mège-lechevallier F, et al. Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol. 2019;20:100–9.
van der Leest M, Cornel EB, Israel B, Hendriks R, Padhani AR, Hoogenboom M. et al. Head-to-head comparison of transrectal ultrasound-guided prostate biopsy versus multiparametric prostate resonance imaging with subsequent magnetic resonance-guided biopsy in biopsy-naïve men with elevated prostate-specific antigen: a large prospective Multicenter Clinical Study. Eur Urol. 2019;75:570–8.
Gandaglia G, Ploussard G, Valerio M, Mattei A, Fiori C, Roumiguié M, et al. The key combined value of multiparametric magnetic resonance imaging, and magnetic resonance imaging–targeted and concomitant systematic biopsies for the prediction of adverse pathological features in prostate cancer patients undergoing radical prostatect. Eur Urol. 2019;77:P733–41.
Dell’Oglio P, Stabile A, Soligo M, Brembilla G, Esposito A, Gandaglia G, et al. There is no way to avoid systematic prostate biopsies in addition to multiparametric magnetic resonance imaging targeted biopsies. Eur Urol Oncol. 2019;3:112–8.
Stabile A, Dell’Oglio P, Cobelli De F, Esposito A, Gandaglia G, Fossati N, et al. Association between prostate imaging reporting and data system (PI-RADS) score for the index lesion and multifocal, clinically significant prostate cancer. Eur Urol Oncol. 2018;1:1–8.
Dell’Oglio P, Stabile A, Dias BH, Gandaglia G, Mazzone E, Fossati N, et al. Impact of multiparametric MRI and MRI-targeted biopsy on pre-therapeutic risk assessment in prostate cancer patients candidate for radical prostatectomy. World J Urol. 2018;3:P739–47.
Johnson DC, Raman SS, Mirak SA, Kwan L, Bajgiran AM, Hsu W, et al. Detection of individual prostate cancer foci via multiparametric magnetic resonance imaging. Eur Urol. 2018;75:712–20.
Patel N, Halpern JA, Kasabwala K, Cricco-Lizza E, Herman M, Margolis D, et al. Multiple regions of interest on multiparametric magnetic resonance imaging are not associated with increased detection of clinically significant prostate cancer on fusion biopsy. J Urol. 2018;200:559–63.
Radtke JP, Schwab C, Wolf MB, Freitag MT, Alt CD, Kesch C, et al. Multiparametric magnetic resonance imaging (MRI) and MRI – transrectal ultrasound fusion biopsy for index tumor detection: correlation with radical prostatectomy specimen. Eur Urol. 2016;70:846–53.
Drost F-JH, Osses D, Nieboer D, Bangma CH, Steyerberg EW, Roobol MJ, et al. Prostate magnetic resonance imaging, with or without magnetic resonance imaging-targeted biopsy, and systematic biopsy for detecting prostate cancer: a cochrane systematic review and meta-analysis. Eur Urol. 2019;77:78–94.
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, et al. ESUR prostate MR guidelines 2012. Eur Radio. 2012;22:746–57.
Barentsz JO, Weinreb JC, Verma S, Thoeny HC, Tempany CM, Shtern F, et al. Synopsis of the PI-RADS v2 Guidelines for Multiparametric Prostate Magnetic Resonance Imaging and Recommendations for Use. Eur Urol. 2015;69:41–9.
European Urology Association. EAU guidelines on prostate cancer [Internet]. 2020.
Tewes S, Hueper K, Hartung D, Imkamp F, Herrmann TRW, Weidemann J, et al. Targeted MRI/TRUS fusion-guided biopsy in men with previous prostate biopsies using a novel registration software and multiparametric MRI PI-RADS scores: first results. World J Urol. 2015;33:1707–14.
Logan JK, Rais-Bahrami S, Turkbey B, Gomella A, Amalou H, Choyke PL, et al. Current status of magnetic resonance imaging (MRI) and ultrasonography fusion software platforms for guidance of prostate biopsies. BJU Int. 2014;114:641–52.
Shoji S, Hiraiwa S, Endo J, Hashida K, Tomonaga T, Nakano M, et al. Manually controlled targeted prostate biopsy with real-time fusion imaging of multiparametric magnetic resonance imaging and transrectal ultrasound: an early experience. Int J Urol. 2015;22:173–8.
Monda SM, Dall’Era MA. What are we missing? Magnetic resonance imaging–negative clinically significant prostate cancer in PROMIS. Eur Urol. 2020;78:171–2.
Eineluoto JT, Järvinen P, Kilpeläinen T, Lahdensuo K, Kalalahti I, Sandeman K, et al. Patient experience of systematic versus fusion prostate biopsies. Eur Urol Oncol. 2018;1:202–7.
Wegelin O, Exterkate L, van der Leest M, Kelder JC, Bosch JLHR, Barentsz JO. et al. Complications and adverse events of three magnetic resonance imaging-based target biopsy techniques in the diagnosis of prostate cancer among men with prior negative biopsies: results from the FUTURE trial, a multicentre randomised controlled trial. Eur Urol Oncol. 2019;2:617–24.
Acknowledgements
Study conception and design: AS and AB. Acquisition of data: FB, GM, FP, GS, EM, SS, and GOC. Analysis and interpretation of data: AS, FB, GM, FP, GS, EM, SS, GOC, VC, AE, GG, NF, FDC, FM, RJK, and AB. Drafting of the manuscript: AS and FB. Critical revision of the manuscript for important intellectual content: AS, GG, NF, FM, RJK, and AB. Statistical analysis: AS. Obtaining funding: None. Administrative, technical, or material support: None. Supervision: FM, RJK, and AB. Other: None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Stabile, A., Barletta, F., Motterle, G. et al. Optimizing prostate-targeted biopsy schemes in men with multiple mpMRI visible lesions: should we target all suspicious areas? Results of a two institution series. Prostate Cancer Prostatic Dis 24, 1137–1142 (2021). https://doi.org/10.1038/s41391-021-00371-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-021-00371-y
This article is cited by
-
Novel sampling scheme with reduced cores in men with multiparametric MRI-visible lesions undergoing prostate biopsy
Abdominal Radiology (2023)
-
MRI-based analysis of different clinically significant prostate cancer detection rate of prostate imaging reporting and data system score 4 in the peripheral zone
Abdominal Radiology (2022)