Abstract
Background
Current prostate biopsy (PBx) protocol for prostate cancer (PCa) diagnosis is to perform systematic biopsies (SBx) combined with targeted biopsies (TBx) in case of positive MRI (i.e. PI-RADS ≥ 3). To assess the utility of performing SBx in combination with TBx, we determined the added value of SBx brought to the diagnosis of PCa according to their sextant location and MRI target characteristics.
Methods
In our local prospectively collected database, we conducted a single-center retrospective study including all patients with a suspicion of PCa, who underwent transrectal ultrasound-guided (TRUS) prostate biopsies (PBx) with a prior MRI and a single lesion classified as PI-RADS ≥ 3. We have characterized the SBx according to their location on MRI: same sextant (S-SBx), adjacent sextant (A-SBx), ipsilateral side (I-SBx) and contralateral side (C-SBx). The added value of SBx and TBx was defined as any upgrading to significant PCa (csPCa) (ISUP ≥2).
Results
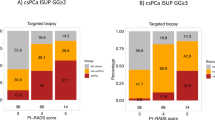
371 patients were included in the study. The added value of SBx was 10% overall. Regarding the lesion location and the SBx sextant, the added value of SBx was: 5.1% for S-SBx, 5.4% for A-SBx, 4.9% for I-SBx and 1.9% for C-SBx. The overall added value of SBx was 6.8% for PI-RADS 3 lesions, 14% for PI-RADS 4 lesions and 6.7% for PI-RADS 5 lesions (p = 0.063). The added value of SBx for contralateral side was 1.9% (2/103), 3.1% (5/163) and 0% (0/105) for PI-RADS 3, PI-RADS 4 and PI-RADS 5 lesions, respectively (p = 0,4). The added value of SBx was lower when the number of TBx was higher (OR 0.57; CI 95% 0.37–0.85; p = 0.007).
Conclusions
Our results suggest that the utility of performing SBx in the contralateral lobe toward the MRI lesion was very low, supporting that they might be avoided.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets used and/or analysed for the study are available from the corresponding author on request.
References
Uroweb - European Association of Urology [Internet]. [cited 2022 Jun 6]. EAU Guidelines on Prostate Cancer - Uroweb. Available from: https://uroweb.org/guidelines/prostate-cancer.
Rouvière O, Puech P, Renard-Penna R, Claudon M, Roy C, Mège-Lechevallier F, et al. Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol. 2019;20:100–9.
Van der Leest M, Cornel E, Israël B, Hendriks R, Padhani AR, Hoogenboom M, et al. Head-to-head comparison of transrectal ultrasound-guided prostate biopsy versus multiparametric prostate resonance imaging with subsequent magnetic resonance-guided biopsy in biopsy-naïve men with elevated prostate-specific antigen: a large prospective multicenter clinical study. Eur Urol. 2019;75:570–8.
Drost FJH, Osses D, Nieboer D, Bangma CH, Steyerberg EW, Roobol MJ, et al. Prostate magnetic resonance imaging, with or without magnetic resonance imaging-targeted biopsy, and systematic biopsy for detecting prostate cancer: a cochrane systematic review and meta-analysis. Eur Urol. 2020;77:78–94.
Loeb S, Bjurlin MA, Nicholson J, Tammela TL, Penson DF, Carter HB, et al. Overdiagnosis and overtreatment of prostate cancer. Eur Urol. 2014;65:1046–55.
Chowdhury R, Abbas A, Idriz S, Hoy A, Rutherford EE, Smart JM. Should warfarin or aspirin be stopped prior to prostate biopsy? An analysis of bleeding complications related to increasing sample number regimes. Clin Radiol. 2012;67:e64–70.
Eineluoto JT, Järvinen P, Kilpeläinen T, Lahdensuo K, Kalalahti I, Sandeman K, et al. Patient experience of systematic versus fusion prostate biopsies. Eur Urol Oncol. 2018;1:202–7.
Kalalahti I, Huotari K, Erickson AM, Petas A, Vasarainen H, Rannikko A. Infectious complications after transrectal MRI-targeted and systematic prostate biopsy. World J Urol. 2022;40:2261–5.
Bryk DJ, Llukani E, Taneja SS, Rosenkrantz AB, Huang WC, Lepor H. The Role of Ipsilateral and contralateral transrectal ultrasound-guided systematic prostate biopsy in men with unilateral magnetic resonance imaging lesion undergoing magnetic resonance imaging-ultrasound fusion-targeted prostate biopsy. Urology. 2017;102:178–82.
Freifeld Y, Xi Y, Passoni N, Woldu S, Hornberger B, Goldberg K, et al. Optimal sampling scheme in men with abnormal multiparametric MRI undergoing MRI-TRUS fusion prostate biopsy. Urol Oncol: Semin Orig Investig. 2019;37:57–62.
Cata E, Andras I, Ferro M, Kadula P, Leucuta D, Musi G, et al. Systematic sampling during MRI-US fusion prostate biopsy can overcome errors of targeting—prospective single center experience after 300 cases in first biopsy setting. Transl Androl Urol. 2020;9:2510–8.
Turkbey B, Rosenkrantz AB, Haider MA, Padhani AR, Villeirs G, Macura KJ, et al. Prostate imaging reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur Urol. 2019;76:340–51.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA, et al. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40:244–52.
Dell’Oglio P, Stabile A, Soligo M, Brembilla G, Esposito A, Gandaglia G, et al. There is no way to avoid systematic prostate biopsies in addition to multiparametric magnetic resonance imaging targeted biopsies. Eur Urol Oncol. 2020;3:112–8.
Ploussard G, Borgmann H, Briganti A, de Visschere P, Fütterer JJ, Gandaglia G, et al. Positive pre-biopsy MRI: are systematic biopsies still useful in addition to targeted biopsies? World J Urol. 2019;37:243–51.
Borghesi M, Ahmed H, Nam R, Schaeffer E, Schiavina R, Taneja S, et al. Complications after systematic, random, and image-guided prostate biopsy. Eur Urol. 2017;71:353–65.
Ahdoot M, Wilbur AR, Reese SE, Lebastchi AH, Mehralivand S, Gomella PT, et al. MRI-targeted, systematic, and combined biopsy for prostate cancer diagnosis. N. Engl J Med. 2020;382:917–28.
Nobin JL, Orczyk C, Deng FM, Melamed J, Rusinek H, Taneja SS, et al. Prostate tumour volumes: evaluation of the agreement between magnetic resonance imaging and histology using novel co-registration software. BJU Int. 2014;114:E105–12.
Padhani AR, Weinreb J, Rosenkrantz AB, Villeirs G, Turkbey B, Barentsz J. Prostate Imaging-Reporting and Data System Steering Committee: PI-RADS v2 Status Update and Future Directions. Eur Urol. 2019;75:385–96.
Brisbane WG, Priester AM, Ballon J, Kwan L, Delfin MK, Felker ER, et al. Targeted prostate biopsy: umbra, penumbra, and value of perilesional sampling. Eur Urol. 2022;82:303–10.
Lahoud J, Doan P, Kim L, Patel MI. Perilesional biopsies increase detection of significant prostate cancer in men with PI-RADS 4/5 Lesions: Validation of the PI-RADS Steering Committee Recommendation. Eur Urol. 2021;80:260–1.
Radtke JP. Multiparametric Magnetic Resonance Imaging (MRI) and MRI–Transrectal Ultrasound fusion biopsy for index tumor detection: correlation with radical prostatectomy specimen. Eur Urol. 2016;70;846–853.
Porpiglia F, Checcucci E, De Cillis S, Piramide F, Amparore D, Piana A, et al. A prospective randomized controlled trial comparing target prostate biopsy alone approach vs. target plus standard in naïve patients with positive mpMRI. Minerva Urol Nephrol. 2023;75;31–41.
Günzel K, Haas M, Maxeiner A, Stephan C, Buckendahl J, Asbach P, et al. Predictive parameters identifying men eligible for a sole MRI/Ultrasound fusion-guided targeted biopsy without an additional systematic biopsy. Urol Int. 2017;98:15–21.
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N. Engl J Med. 2018;378:1767–77.
Ploussard G, Beauval JB, Renard-Penna R, Lesourd M, Manceau C, Almeras C, et al. Assessment of the minimal targeted biopsy core number per MRI Lesion for improving prostate cancer grading prediction. J Clin Med. 2020;9:225.
Calio BP, Sidana A, Sugano D, Gaur S, Maruf M, Jain AL, et al. Risk of upgrading from prostate biopsy to radical prostatectomy pathology—does saturation biopsy of index lesion during multiparametric magnetic resonance imaging-transrectal ultrasound fusion biopsy help? J Urol. 2018;199:976–82.
Diamand R, Hollans M, Lefebvre Y, Sirtaine N, Limani K, Hawaux E, et al. The role of perilesional and multiparametric resonance imaging-targeted biopsies to reduce the risk of upgrading at radical prostatectomy pathology: A retrospective monocentric study. Urol Oncol: Semin Orig Investig. 2022;40:192.e11–192.e17.
Falagario U, Jambor I, Taimen P, Syvänen KT, Kähkönen E, Merisaari H, et al. Added value of systematic biopsy in men with a clinical suspicion of prostate cancer undergoing biparametric MRI-targeted biopsy: multi-institutional external validation study. World J Urol. 2021;39:1879–87.
Uleri A, Baboudjian M, Tedde A, Gallioli A, Long-Depaquit T, Palou J, et al. Is there an impact of transperineal versus transrectal magnetic resonance imaging–targeted biopsy in clinically significant prostate cancer detection rate? A systematic review and meta-analysis. Eur Urol Oncol. 2023;6:621–8.
Author information
Authors and Affiliations
Contributions
Concept and design, drafting of the manuscript: AZ, AV, TAN. Acquisition of data: AZ, TAN, KS. Statistical analysis: TAN. Interpretation of data, critical revisions to manuscript: AZ, AV, TAN. Manuscript revision: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent
All procedures in the study were performed with local ethics committee approval. Informed consent was obtained from all individual participants included in the study as requested by the ethical committee (29BRC21.0148-B2021CE.28).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zambon, A., Nguyen, TA., Fourcade, A. et al. Which protocol for prostate biopsies in patients with a positive MRI? Interest of systematic biopsies by sectors. Prostate Cancer Prostatic Dis (2023). https://doi.org/10.1038/s41391-023-00770-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41391-023-00770-3
This article is cited by
-
A novel biopsy scheme for prostate cancer: targeted and regional systematic biopsy
BMC Urology (2024)
-
Elucidating the need for prostate cancer risk calculators in conjunction with mpMRI in initial risk assessment before prostate biopsy at a tertiary prostate cancer center
BMC Urology (2024)
-
Best of 2023 in Prostate Cancer and Prostatic Diseases
Prostate Cancer and Prostatic Diseases (2024)