Abstract
Background
With growing interest in focal therapy (FT) of prostate cancer (PCa) there is an increasing armamentarium of treatment modalities including high-intensity focused ultrasound (HIFU), cryotherapy, focal laser ablation (FLA), irreversible electroporation (IRE), vascular targeted photodynamic therapy (VTP), focal brachytherapy (FBT) and stereotactic ablative radiotherapy (SABR). Currently there are no clear recommendations as to which of these technologies are appropriate for individual patient characteristics. Our intention was to review the literature for special aspects of the different technologies that might be of advantage depending on individual patient and tumour characteristics.
Methods
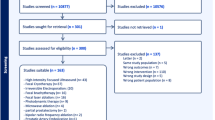
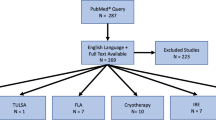
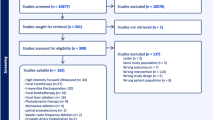
The current literature on FT was screened for the following factors: morbidity, repeatability, tumour risk category, tumour location, tumour size and prostate volume and anatomical issues. The ESUT expert panel arrived at consensus regarding a position statement on a structured pathway for available FT technologies based on a combination of the literature and expert opinion.
Results
Side effects were low across different studies and FT modalities with urinary continence rates of 90–100% and erectile dysfunction between 5 and 52%. Short to medium cancer control based on post-treatment biopsies were variable between ablative modalities. Expert consensus suggested that posterior lesions are better amenable to FT using HIFU. Cryotherapy provides best possible outcomes for anterior tumours. Apical lesions, when treated with FBT, may yield the least urethral morbidity.
Conclusions
Further prospective trials are required to assess medium to long term disease control of different ablative modalities for FT. Amongst different available FT modalities our ESUT expert consensus suggests that some may be better for diffe`rent tumour locations. Tumour risk, tumour size, tumour location, and prostate volume are all important factors to consider and might aid in designing future FT trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Abern MR, Tsivian M, Polascik TJ. Focal therapy of prostate cancer: evidence-based analysis for modern selection criteria. Curr Urol Rep. 2012;13:160–9.
Polascik TJ, Mouraviev V. Focal therapy for prostate cancer is a reasonable treatment option in properly selected patients. Urology. 2009;74:726–30.
Murphy DG, Loeb S. Prostate cancer: growth of AS in the USA signals reduction in overtreatment. Nat Rev Urol. 2015;12:604–5.
Welty CJ, Cooperberg MR, Carroll PR. Meaningful end points and outcomes in men on active surveillance for early-stage prostate cancer. Curr Opin Urol. 2014;24:288–92.
van den Bergh RC, Murphy DG, Costello AJ. Identification of candidates for observation. Curr Opin Urol. 2015;25:252–7.
Tsivian M, Abern MR, Polascik TJ. Evolution of the concept of focal therapy for prostate cancer. Oncology (Williston Park). 2013;27:64–68. 70
van der Poel H, Klotz L, Andriole G, Azzouzi AR, Bjartell A, Cussenot O, et al. Role of active surveillance and focal therapy in low- and intermediate-risk prostate cancers. World J Urol. 2015;33:907–16.
Ahmed HU, Emberton M. Active surveillance and radical therapy in prostate cancer: can focal therapy offer the middle way? World J Urol. 2008;26:457–67.
Bahn D, Silverman P, Lee F, Badalament R, Bahn ED, Rewcastle JC. Focal prostate cryoablation: initial results show cancer control and potency preservation. J Endourol. 2006;20:688–92.
Mouraviev V, Johansen TE, Polascik TJ. Contemporary results of focal therapy for prostate cancer using cryoablation. J Endourol. 2010;24:827–34.
Eggener SE, Scardino PT, Carroll PR, Zelefsky MJ, Sartor O, Hricak H, et al. Focal therapy for localized prostate cancer: a critical appraisal of rationale and modalities. J Urol. 2007;178:2260–7.
de la Rosette J, Ahmed H, Barentsz J, Johansen TB, Brausi M, Emberton M, et al. Focal therapy in prostate cancer-report from a consensus panel. J Endourol. 2010;24:775–80.
Ahmed HU, Akin O, Coleman JA, Crane S, Emberton M, Goldenberg L, et al. Transatlantic Consensus Group on active surveillance and focal therapy for prostate cancer. BJU Int. 2012;109:1636–47.
Donaldson IA, Alonzi R, Barratt D, Barret E, Berge V, Bott S, et al. Focal therapy: patients, interventions, and outcomes--a report from a consensus meeting. Eur Urol. 2015;67:771–7.
Ahmed HU, Berge V, Bottomley D, Cross W, Heer R, Kaplan R, et al. Can we deliver randomized trials of focal therapy in prostate cancer? Nat Rev Clin Oncol. 2014;11:482–91.
van den Bos W, Muller BG, Ahmed H, Bangma CH, Barret E, Crouzet S, et al. Focal therapy in prostate cancer: international multidisciplinary consensus on trial design. Eur Urol. 2014;65:1078–83.
Postema AW, De Reijke TM, Ukimura O, Van den Bos W, Azzouzi AR, Barret E, et al. Standardization of definitions in focal therapy of prostate cancer: report from a Delphi consensus project. World J Urol. 2016;34:1373–82.
Coleman JA, Scardino PT. Targeted prostate cancer ablation: energy options. Curr Opin Urol. 2013;23:123–8.
Salgaonkar VA, Prakash P, Rieke V, Ozhinsky E, Plata J, Kurhanewicz J, et al. Model-based feasibility assessment and evaluation of prostate hyperthermia with a commercial MR-guided endorectal HIFU ablation array. Med Phys. 2014;41:033301.
Ahmed HU, Moore C, Emberton M. Minimally-invasive technologies in uro-oncology: the role of cryotherapy, HIFU and photodynamic therapy in whole gland and focal therapy of localised prostate cancer. Surg Oncol. 2009;18:219–32.
Rischmann P, Gelet A, Riche B, Villers A, Pasticier G, Bondil P, et al. Focal high intensity focused ultrasound of unilateral localized prostate cancer: a prospective multicentric hemiablation study of 111 patients. Eur Urol. 2016;71:267–73.
Ganzer R,Hadaschik B,Pahernik S,Koch D,Baumunk D,Kuru T, et al. Prospective multicenter phase II-study on focal therapy (hemiablation) of the prostate with High Intensity focused ultrasound (HIFU). J Urol. 2017;199:983–989.
Babaian RJ, Donnelly B, Bahn D, Baust JG, Dineen M, Ellis D, et al. Best practice statement on cryosurgery for the treatment of localized prostate cancer. J Urol. 2008;180:1993–2004.
Ward JF, Jones JS. Focal cryotherapy for localized prostate cancer: a report from the national Cryo On-Line Database (COLD) registry. BJU Int. 2012;109:1648–54.
Truesdale MD, Cheetham PJ, Hruby GW, Wenske S, Conforto AK, Cooper AB. An evaluation of patient selection criteria on predicting progression-free survival after primary focal unilateral nerve-sparing cryoablation for prostate cancer: recommendations for follow up. Cancer J. 2010;16:544–9.
Onik G,Vaughan D,Lotenfoe R,Dineen M,Brady J, “Male lumpectomy”: focal therapy for prostate cancer using cryoablation. Urology. 2008;70:16–21.
Lindner U, Lawrentschuk N, Weersink RA, Davidson SR, Raz O, Hlasny E, et al. Focal laser ablation for prostate cancer followed by radical prostatectomy: validation of focal therapy and imaging accuracy. Eur Urol. 2010;57:1111–4.
Lindner U, Weersink RA, Haider MA, Gertner MR, Davidson SRH, Atri M. Image guided photothermal focal therapy for localized prostate cancer: Phase I trial. J Urol. 2009;182:1371–7.
Oto A, Sethi I, Karczmar G, McNichols R, Ivancevic MK, Stadler WM, et al. MR imaging-guided focal laser ablation for prostate cancer: Phase I trial. Radiology. 2013;267:932–40.
Raz O, Haider MA, Davidson SR, Lindner U, Hlasny E, Weersink R, et al. Real-time magnetic resonance imaging-guided focal laser therapy in patients with low-risk prostate cancer. Eur Urol. 2010;58:173–7.
Eggener SE, Yousuf A, Watson S, Wang S, Oto A. Phase II evaluation of magnetic resonance imaging guided focal laser ablation of prostate cancer. J Urol. 2016;196:1670–5.
Lepor H, Llukani E, Sperling D, Futterer JJ. Complications, recovery, and early functional outcomes and oncologic control following in-bore focal laser ablation of prostate cancer. Eur Urol. 2015;68:924–6.
Natarajan S, Jones TA, Priester AM, Geoghegan R, Lieu P, Delfin M, et al. Focal laser ablation of prostate cancer: feasibility of magnetic resonance imaging-ultrasound fusion for guidance. J Urol. 2017;198:839–47.
Taneja SS,Bennett J,Coleman J,Grubb R,Andriole G,Reiter RE, et al. Final results of a Phase I/II Multicenter Trial of WST11 vascular targeted photodynamic therapy for hemi-ablation of the prostate in men with unilateral low risk prostate cancer performed in the United States. J Urol. 2016;196:1096–104.
Azzouzi AR,Barret E,Moore CM,Villers A,Allen C,Scherz A, et al. TOOKAD((R)) soluble vascular-targeted photodynamic (VTP) therapy: determination of optimal treatment conditions and assessment of effects in patients with localised prostate cancer. BJU Int. 2013;112:766–74.
Moore CM, Azzouzi AR, Barret E, Villers A, Muir GH, Barber NJ, et al. Determination of optimal drug dose and light dose index to achieve minimally invasive focal ablation of localised prostate cancer using WST11-vascular-targeted photodynamic (VTP) therapy. BJU Int. 2015;116:888–96.
Azzouzi A-R, Vincendeau S, Barret E, Cicco A, Kleinclauss F, van der Poel HG, et al. Padeliporfin vascular-targeted photodynamic therapy versus active surveillance in men with low-risk prostate cancer (CLIN1001 PCM301): an open-label, phase 3, randomised controlled trial. Lancet Oncol. 2016;18:181–91.
Cosset JM, Cathelineau X, Wakil G, Pierrat N, Quenzer O, Prapotnich D, et al. Focal brachytherapy for selected low-risk prostate cancers: a pilot study. Brachytherapy. 2013;12:331–7.
Barret E, Ahallal Y, Sanchez-Salas R, Galiano M, Cosset JM, Validire P, et al. Morbidity of focal therapy in the treatment of localized prostate cancer. Eur Urol. 2013;63:618–22.
Kamrava M, Chung MP, Kayode O, Wang J, Marks L, Kupelian P, et al. Focal high-dose-rate brachytherapy: a dosimetric comparison of hemigland vs. conventional whole-gland treatment. Brachytherapy. 2013;12:434–41.
Kovacs G, Cosset JM, Carey B. Focal radiotherapy as focal therapy of prostate cancer. Curr Opin Urol. 2014;24:231–5.
King CR, Lehmann J, Adler JR, Hai J. CyberKnife radiotherapy for localized prostate cancer: rationale and technical feasibility. Technol Cancer Res Treat. 2003;2:25–30.
Macdougall ND, Dean C, Muirhead R. Stereotactic body radiotherapy in prostate cancer: is rapidarc a better solution than cyberknife? Clin Oncol (R Coll Radiol). 2014;26:4–9.
Lu DS, Kee ST, Lee EW. Irreversible electroporation: ready for prime time? Tech Vasc Interv Radiol. 2013;16:277–86.
Bozzini G, Colin P, Nevoux P, Villers A, Mordon S, Betrouni N. Focal therapy of prostate cancer: energies and procedures. Urol Oncol. 2013;31:155–67.
Onik G, Rubinsky B. Irreversible electroporation. Berlin, Heidelberg: Springer-Verlag; 2010. p. 235–47.
van den Bos W, Muller BG, de la Rosette JJ. A randomized controlled trial on focal therapy for localized prostate carcinoma: hemiablation versus complete ablation with irreversible electroporation. J Endourol. 2013;27:262–4.
Valerio M, Stricker PD, Ahmed HU, Dickinson L, Ponsky L, Shnier R, et al. Initial assessment of safety and clinical feasibility of irreversible electroporation in the focal treatment of prostate cancer. Prostate Cancer Prostatic Dis. 2014;17:343–7.
Valerio M, Dickinson L, Ali A, Ramachadran N, Donaldson I, Mccartan N et al. Nanoknife Electroporation Ablation Trial (NEAT): a prospective development study investigating focal irreversible electroporation for localised prostate cancer. J Urol. 2016.
TOOKAD (Padeliporfin) Vascular-Targeted Photodynamic Therapy versus Active Surveillance in men with low risk prostate cancer. A randomized phase 3 clinical trial.. Munich, Germany: European Association of Urology; 2016.
Marien A, Gill I, Ukimura O, Betrouni N, Villers A. Target ablation--image-guided therapy in prostate cancer. Urol Oncol. 2014;32:912–23.
Crouzet S, Murat FJ, Pasticier G, Cassier P, Chapelon JY, Gelet A. High intensity focused ultrasound (HIFU) for prostate cancer: current clinical status, outcomes and future perspectives. Int J Hyperth. 2010;26:796–803.
Shah TT, Ahmed H, Kanthabalan A, Lau B, Ghei M, Maraj B, et al. Focal cryotherapy of localized prostate cancer a systematic review of the literature. Expert Rev Anticancer Ther. 2014;14:1337–47.
Ahmed HU, Hindley RG, Dickinson L, Freeman A, Kirkham AP, Sahu M, et al. Focal therapy for localised unifocal and multifocal prostate cancer: a prospective development study. Lancet Oncol. 2012;13:622–32.
Bahn D, de Castro Abreu AL, Gill IS, Hung AJ, Silverman P, Gross ME, et al. Focal cryotherapy for clinically unilateral, low-intermediate risk prostate cancer in 73 men with a median follow-up of 3.7 years. Eur Urol. 2012;62:55–63.
Lambert EH, Bolte K, Masson P, Katz AE. Focal cryosurgery: encouraging health outcomes for unifocal prostate cancer. Urology. 2017;197:647–654.
Ellis DS, Manny TBJ, Rewcastle JC. Focal cryosurgery followed by penile rehabilitation as primary treatment for localized prostate cancer: initial results. Urology. 2007;70:9–15. 6 Suppl
Shah TT, Kasivisvanathan V, Jameson C, Freeman A, Emberton M, Ahmed HU. Histological outcomes after focal high-intensity focused ultrasound and cryotherapy. World J Urol. 2015;33:955–64.
Muto S, Yoshii T, Saito K, Kamiyama Y, Ide H, Horie S. Focal therapy with high-intensity-focused ultrasound in the treatment of localized prostate cancer. Jpn J Clin Oncol. 2008;38:192–9.
Orczyk C, Emberton M, Ahmed HU. What tumours should we treat with focal therapy based on risk category, grade, size and location? Curr Opin Urol. 2015;25:212–9.
Rouvière O, Glas L, Girouin N, Mège-Lechevallier F, Gelet A, Dantony E, et al. Prostate cancer ablation with transrectal high-intensity focused ultrasound: assessment of tissue destruction with contrast-enhanced US. Radiology. 2011;259:583–91.
Merrick GS, Wallner KE, Butler WM. Prostate cryotherapy: more questions than answers. Urology. 2005;66:9–15.
Crouzet S, Rouviere O, Martin X, Gelet A. High-intensity focused ultrasound as focal therapy of prostate cancer. Curr Opin Urol. 2014;24:225–30.
Sonacare. Sonafuse platform. 2016.
Shoji S, Uchida T, Nakamoto M, Kim H, de Castro Abreu AL, Leslie S, et al. Prostate swelling and shift during high intensity focused ultrasound: implication for targeted focal therapy. J Urol. 2013;190:1224–32.
Ghai S, Trachtenberg J, In-bore MRI. interventions: current status and future applications. Curr Opin Urol. 2015;25:205–11.
Le Nobin J, Rosenkrantz AB, Villers A, Orczyk C, Deng FM, Melamed J, et al. Image guided focal therapy for magnetic resonance imaging visible prostate cancer: defining a 3-dimensional treatment margin based on magnetic resonance imaging histology co-registration analysis. J Urol. 2015;194:364–70.
Sivaraman A, Barret E. Focal therapy for prostate cancer: an “A la Carte” approach. Eur Urol. 2016;69:973–5.
Ahmed HU, Dickinson L, Charman S, Weir S, McCartan N, Hindley RG, et al. Focal ablation targeted to the index lesion in multifocal localised prostate cancer: a prospective development study. Eur Urol. 2015;68:927–36.
Van Velthoven R, Aoun F, Limani K, Narahari K, Lemort M, Peltier A. Primary zonal high intensity focused ultrasound for prostate cancer: results of a prospective Phase IIa feasibility study. Prostate Cancer. 2014;2014:756189.
Author information
Authors and Affiliations
Contributions
RM: protocol development, data collection and management, manuscript writing/editing. VKAA: protocol development, data collection and management, manuscript writing/editing. HUA: data collection and management. AC: data collection and management. PR: data collection and management. GS: data collection and management. DT: data collection and management. EL: data collection and management. J-US: data collection and management. EB: protocol development, data collection and management, manuscript writing/editing
Corresponding author
Ethics declarations
Conflict of interest
PR is a consultant for EDAP-TMS, Vaulx-en-Velin, France and stock owner of EDAP TMS shares; HUA receives funding from SonaCare Medical (previously USHIFU) for clinical trials and has received funding from SonaCare Medical for travel to conferences. RG, VKAA, AC, GS, DT, EL, J-US and EB declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ganzer, R., Arthanareeswaran, V.K.A., Ahmed, H.U. et al. Which technology to select for primary focal treatment of prostate cancer?—European Section of Urotechnology (ESUT) position statement. Prostate Cancer Prostatic Dis 21, 175–186 (2018). https://doi.org/10.1038/s41391-018-0042-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0042-0
This article is cited by
-
Clinical application of single-shot echo-planar diffusion-weighted imaging with compressed SENSE in prostate MRI at 3T: preliminary experience
Magnetic Resonance Materials in Physics, Biology and Medicine (2022)
-
Making a case “against” focal therapy for intermediate-risk prostate cancer
World Journal of Urology (2021)
-
Focal ablation of apical prostate cancer lesions with irreversible electroporation (IRE)
World Journal of Urology (2021)
-
Fokale Therapie beim Prostatakarzinom – 3,5 Jahre Follow-up mit 92 % Rebiopsierate
Der Urologe (2020)
-
Kritische Bestandsaufnahme zur fokalen Therapie
Uro-News (2020)