Abstract
Early life stress can result in depression in humans and depressive-like behaviour in rodents. In various animal models of depression, the lateral habenula (LHb) has been shown to become hyperactive immediately after early life stress. However, whether these pathological changes persist into adulthood is less well understood. Hence, we utilised the maternal separation (MS) model of depression to study how early life stress alters LHb physiology and depressive behaviour in adult mice. We find that only a weak depressive phenotype persists into adulthood which surprisingly is underpinned by LHb hypoactivity in acute slices, accompanied by alterations in both excitatory and inhibitory signalling. However, while we find the LHb to be less active at rest, we report that the neurons reside in a sensitised state where they are more responsive to re-exposure to stress in adulthood in the form of acute restraint, thus priming them to respond to aversive events with an increase in neuronal activity mediated by changes in glutamatergic transmission. These findings thus suggest that in addition to LHb hyperactivity, hypoactivity likely also promotes an adverse phenotype. Re-exposure to stress results in the reappearance of LHb hyperactivity offering a possible mechanism to explain how depression relapses occur following previous depressive episodes.
Similar content being viewed by others
Introduction
The lateral habenula (LHb) is an evolutionarily conserved brain structure located within the epithalamus which encodes aversive events [1, 2], and depressive behaviour [3,4,5,6]. Specifically, the LHb becomes hyperactive in depression [3, 4, 7,8,9], thus enhancing output to the midbrain reward circuitry, for which the LHb acts as an inhibitory modulator [10,11,12].
Indeed, many studies have employed the use of a variety of different animal models of depressive behaviour including chronic mild stress [13], chronic restraint stress [4, 6], social defeat [14, 15], learned helplessness [3] and various models of early life stress [8, 16,17,18], and have independently reached this conclusion that the LHb becomes hyperactive in depression. However, the majority of these studies have carried out experimentation shortly after exposure to the relevant stressor, and as such there is comparatively little evidence as to the long-term persistence of depressive phenotype, and the corresponding synaptic and physiological alterations within the LHb [18].
Hence, in this study we aimed to assess how early life stress in the form of maternal separation influences depressive behaviour, and alters lateral habenular physiology and synaptic connectivity in adult mice. Furthermore, we then sought to ask how early life stress influences subsequent re-exposure to stress in adulthood.
Materials and methods
Brief summaries of methods can be found below. Complete materials and methods can be found in the supplementary materials.
Animals
All procedures were approved by the Ethics committee of the University of Strathclyde, Glasgow, and carried out in accordance with the relevant UK legislation (the Animals (Scientific Procedures) Act, 1986). C57BL/6J and SOM-IRES-Cre mutant mice were kept on a 12:12 light/dark cycle under standard group housing conditions with unlimited access to water and normal mouse chow, unless otherwise stated. Both males and females were used in this work, and unless otherwise stated, data were pooled between genders.
Maternal separation procedure
The maternal separation (MS) procedure was adapted from a previously published protocol [8]. Briefly, at postnatal day 6 (P6) litters were separated into groups of either control (CTRL) pups, which remained with the mother, or MS pups, which were separated into individually isolated compartments of a heated cage in a separate room for 6 h per day from P6-16. MS pups were then weaned early at P17.
Behavioural testing and acute restraint procedure
Mice underwent behavioural testing at approximately 8–10 weeks, and were single housed for these experiments. 3 behavioural tests were implemented in this study. These were sucrose preference, the open field test, and the splash test. Mice were then returned to group-housing conditions upon completion of behavioural testing. For animals which underwent acute restraint, this was carried out for a period of 1 h in a modified handling tube.
Stereotaxic viral injections
Stereotaxic viral injections were carried out as previously described [19]. Briefly, SOM-IRES-Cre heterozygous mice were deeply anaesthetized with inhaled isoflurane and transferred to a stereotaxic frame where viral injections (either AAV9-EF1a-DIO-hChR2(H134R)-EYFP, titre 1.8 × 1013 vg/mL, 200 nL; or AAV9-pCAG-FLEX-EGFP-WPRE, titre 2.5 × 1013 vg/mL, 100 nL) were performed bilaterally at the following stereotaxic coordinates (from Bregma): AP 0.45; ±1.3; depth 5.8. Following completion of surgery, animals were given at least two weeks to allow expression of the virus before acute slice preparation for electrophysiology.
Acute brain slice preparation and in vitro electrophysiological recordings
Mice were humanely euthanized by cervical dislocation and immediately decapitated. Brains were rapidly removed, brains slices were prepared following standard procedures and electrophysiological recordings were performed as previously described [19, 20].
Transcardial perfusion, immunohistochemistry and confocal microscopy
Mice were terminally anaesthetized by intraperitoneal injection of 50% lidocaine and 50% pentobarbital, and then perfused through the left ventricle with 0.1 M PBS followed by perfusion with 4% PFA dissolved in PBS. Brains were then removed and fixed overnight in 4% PFA in PBS, after which they were cryoprotected in a solution containing 30% sucrose in PBS where they were kept until required for histology. Immunohistochemical staining against c-Fos was then performed as previously described [19], and slices were imaged on a Leica SP8 confocal microscope.
Results
Early life stress induces mild depressive symptoms in adult mice, and alters LHb physiology
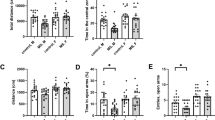
We first sought to validate that MS for 6 h per day with early weaning (methods) can induce depressive behaviour in adult mice. Hence, following MS, mice were allowed to develop to adulthood (≈8–10 weeks of age), when they underwent behavioural testing. Mice were subjected to a series of 3 behavioural paradigms to test for anhedonia, anxiety-like behaviour and motivation which were sucrose preference, the open field test and the splash test respectively (Fig. 1A; N = 37 CTRL; N = 39 MS mice). Sucrose preference testing was carried out over 3 consecutive days, and indeed we saw a reduction in sucrose preference in MS mice over these 3 days (Fig. 1B; p = 0.038; 2-way ANOVA column factor). However, a particularly striking effect was that this deficit was most prominent on day 2 of testing (p = 0.028; Sidak’s test). This was an interesting observation, as on day 2 of our paradigm, we switched the position of the sucrose and water bottles (Methods), hence suggesting that our model also induces a possible deficit in reversal learning [21]. We did not observe any change in anxiety-like behaviour (Fig. 1C; p = 0.670; unpaired T-test), locomotor activity (Fig. 1D; p = 0.593; unpaired T-test) or motivation (Fig. 1E; p = 0.286; unpaired T-test). However, MS mice exhibited an increased latency to first grooming bout in the splash test (Fig. 1F; p = 0.050; Mann-Whitney test), and interestingly spent more time rearing in the splash test (Fig. 1G; p = 0.020; unpaired T-test), which may be indicative of social contact seeking [22]. To account for variability between behavioural tests within individual mice, we also calculated an integrated Z score [23], which gives an arbitrary score of emotionality for each mouse, by normalising and integrating readouts from each behavioural test. This indeed revealed that MS mice had overall greater emotionality than CTRL mice (Fig. 1H; p = 0.019; unpaired T-test). However, overall these results led us to conclude that the observed phenotype was relatively mild in the adult mice.
A Schematic illustrating experimental timeline. B Behavioural data from sucrose preference test across 3 days of testing. C Comparison of % of test time in borders and of D mean locomotor activity from open field test. E Comparison plot of % of test time spent grooming; F of latency to first grooming bout and G of total time spent rearing on hind legs in splash test. H Averaged z scores of behavioural traits for each mouse.
We next sought to assess how MS altered LHb neuronal physiology. Mice were sacrificed shortly after behavioural testing, and whole-cell recordings were carried out in acute brain slices (Fig. 2A; n/N = 36/6 CTRL; n/N = 39/6 MS neurons/mice). MS induced no changes in passive physiological properties (Fig. S1A; input resistance p = 0.984; unpaired T-test; and resting membrane potential p = 0.235; Mann-Whitney test), and induced only a weak trend towards an increase in intrinsic excitability (Fig. S1B, C; p = 0.083; 2-way ANOVA column factor). However, the most striking difference we observed was a reduction in spontaneous neuronal activity (Fig. 2B, C; p = 0.0009; Chi-square test), which appeared to be specific for tonically active neurons. This was an interesting observation as bursting activity of LHb neurons is believed to be the primary driver of depressive behaviour [4, 6]. Moreover, of those neurons which were spontaneously active, we observed that they were active at a lower frequency following MS (Fig. 2D; p = 0.014; Mann-Whitney test). We did not however observe any correlation between the mean spontaneous activity for each mouse and corresponding behavioural Z score, likely as 3 out of 6 mice which we recorded from had mean spontaneous activity close to 0 (Fig. 2E). We also observed no difference in sag potential between conditions (Fig. 2F, G; p = 0.678; 2-way ANOVA column factor). However, although neurons from MS mice did not display more bursting activity at rest, we found that they did fire rebound bursts with a reduced latency following hyperpolarising current injection (Fig. 2H, I; p < 0.0001; 2-way ANOVA column factor), leading us to speculate that the neurons may be in a state where they are more primed to fire in response to synaptic input. Hence, we also recorded spontaneous excitatory postsynaptic currents (sEPSC’s) in a separate cohort of mice (Fig. S2; n/N = 44/4 CTRL; n/N = 46/4 MS neurons/mice). We did not observe any differences in frequency (Fig. S2A; p = 0.342; Mann-Whitney test) but did observe a slight but significant reduction in current amplitude following MS (Fig. S2A; p = 0.012; Mann-Whitney test). However, this dataset was likely confounded by the fact that the CTRL mice for this particular cohort appeared to display a phenotype more similar to the MS mice than other CTRL mice, and as such we attempted to account for this by plotting the behavioural Z score of each mouse against the mean sEPSC frequency for all cells recorded from each individual mouse (Fig. S2C). Indeed, when we did this we observed a negative correlation between Z score and mean sEPSC frequency (Fig. S2C; p = 0.016; simple linear regression), indicating that mice which existed in a more depressed state apparently exhibited reduced excitatory drive onto LHb neurons. Altogether, these results point to a scenario whereby MS reduces spontaneous firing of LHb neurons in brain slices, possibly by reducing presynaptic excitatory drive.
A Schematic illustrating experimental timeline. (B–D) Spontaneous activity comparison for both conditions. B Pie charts depicting percentages of recorded neurons which were classed as spontaneously active (spontaneous AP discharge > 0.5 Hz). C Example spontaneous activity recordings from both conditions. D Comparison plot of mean spontaneous activity frequency, for neurons which were spontaneously active (>0.5 Hz frequency). E XY plot of behavioural z score against mean spontaneous activity frequency calculated for each individual mouse from which electrophysiological recordings were taken. Mean spontaneous activity scores are calculated as the mean frequency of all cells recorded from each mouse. F Example traces and G plot of input current against sag potential in both conditions. Sag potential was calculated as the difference of the peak (+) and the steady state (++) of the membrane hyperpolarisation induced in response to hyperpolarising current steps. H Example traces of rebound bursts induced in both conditions following hyperpolarising current injection. I Plot of input current against latency to initial spike of the rebound burst induced upon current step end.
MS weakens inhibitory synaptic transmission onto LHb neurons
Multiple other works have shown that a reduction in inhibitory signalling is associated with a depressive phenotype [7, 8, 24], and as such we sought to test how MS influenced inhibitory signalling in adult mice. We first recorded spontaneous IPSCs (sIPSCs) throughout the LHb in acute slices (Fig. 3; n/N = 46/6 CTRL; n/N = 49/6 MS neurons/mice). We observed no overall difference in either frequency (Fig. 3A; p = 0.296; Mann-Whitney test) or amplitude (Fig. 3A; p = 0.245; Mann-Whitney test). However, we did observe a striking gradient across the mediolateral axis of the LHb in CTRL mice (Fig. 3B, C; p = 0.005; simple linear regression) with sIPSC frequency being greatest in the medial LHb, which was not present in MS mice (Fig. 3B, C; p = 0.325; simple linear regression). This thus lead us to suspect that there may be subregional differences in spontaneous inhibitory synaptic input to LHb neurons. Indeed, when we broke our analysis down into the medial (<0.45 mm from midline) and lateral (>0.45 mm from midline) LHb sub-regions, we observed a reduction in sIPSC frequency specifically in the medial LHb (Fig. 3D; p = 0.048; Mann-Whitney test). Furthermore, we found that there was a negative correlation of the behavioural Z score with IPSC frequency (Fig. 3E; p = 0.013; simple linear regression), thus suggesting that more depressed animals exhibited lower levels of inhibitory synaptic drive. Overall, these results suggest that MS results in a reduction in spontaneous inhibitory input, specifically in the medial LHb.
A Comparison plots of sIPSC frequency and amplitude between conditions. B XY plot with simple linear regression of sIPSC frequency against approximate distance from midline for both conditions. C Example sIPSC recordings from neurons recorded in both the medial and lateral LHb for both conditions. D Comparison plots of sIPSC frequency in both the medial and lateral LHb. E XY plot of behavioural z score against mean sIPSC frequency calculated for each individual mouse recorded from. Mean sIPSC scores are calculated as the mean sIPSC frequency of all cells recorded from each mouse.
In our previous work we reported on a strong inhibitory projection to the LHb which appeared to arise from somatostatin-positive (SOM) neurons in the ventral pallidum of the basal forebrain [19]. Other work has shown that excitatory pallidal projections to the LHb can promote depressive behaviour [15], and that inhibitory LHb-projecting pallidal projections promote reward [25]. As such, we hypothesised that inhibitory drive onto the LHb from SOM-positive pallidal neurons may be lost following MS. We tested this by injecting a Cre-dependent virus encoding channelrhodopsin (ChR2) into the basal forebrain of SOM-Cre mice and recorded light-induced postsynaptic events in LHb neurons (Fig. 4A; n/N = 18/4 CTRL; n/N = 20/4 MS neurons/mice). At least 2 weeks post-injection, we could observe eYFP-positive terminal fields within the LHb (Fig. 4B). We first recorded inhibitory currents induced following a single 1 ms pulse at various intensities, and tested for differences between CTRL and MS mice by fitting a one-phase association exponential curve to each dataset [26]. CTRL and MS groups were found to have very different curve fits (Fig. 4C; p < 0.0001; non-linear curve fit), which specifically was found to be a reduction in the curve plateau for MS mice (Fig. 4C, D; p = 0.03; non-linear curve fit), thus suggesting a reduction in postsynaptic current amplitude without a corresponding change in kinetics. We also tested presynaptic release probability in current clamp configuration and observed no differences (Fig. 4E; p = 0.457, 2-way ANOVA). However, an interesting observation was that in a sub-fraction of responsive neurons, the inhibitory drive was found to be strong enough to induce rebound firing (Fig. S3A, B), which interestingly was observed to be strongest at 10 Hz stimulation frequency (Fig. S3C).
A Schematic illustrating experimental timeline for optogenetic experiments. B Example image of eYFP terminal fields within the LHb following viral injection. C Intensity-response curve of LED intensity plotted against mean peak amplitude of the oIPSC, with one phase exponential curve fitted for both conditions. P value is a comparison of plateaus from both fitted curves. D Example traces of oIPSCs from both conditions. (E) Plot of oIPSP peaks from 20 Hz LED stimulation normalised to the amplitude of the 1st peak in each recording. Inset is an example recording from a neuron from an MS mouse.
Altogether, the above observations indicate a weakening of inhibitory synaptic drive from inhibitory SOM-positive pallidal forebrain neurons onto LHb neurons following MS. Due to the observed change in plateau without corresponding change in rise constant of the curve (Fig. 4C), and the apparent lack of change in presynaptic release probability (Fig. 4E), the case could be made that this may be due to a down-regulation of postsynaptic GABA receptors. However, this does not innately rule out a change in the intrinsic excitability of the presynaptic neurons. As such, we aimed to test this by recording from presynaptic SOM-positive pallidal neurons. We first injected a retrograde Cre-dependent virus encoding tdTomato into the LHb of SOM-Cre mice. However, while we could clearly observe terminals in the LHb, and tdTomato-positive soma in the entopeduncular nucleus, where SOM-positive neurons project to the LHb, labelling in the entire basal forebrain region was very sparse (data not shown). Instead, we therefore injected a Cre-dependent anterograde virus encoding eGFP into the basal forebrain and recorded from putative presynaptic eGFP-positive neurons (Fig. S4A; n/N = 35/4 CTRL; n/N = 34/4 MS neurons/mice). As with our optogenetic experiments, this resulted in robust terminal labelling within the LHb (Fig. S4B). Neurons were found to be spontaneously active at a similar frequency within both conditions (Fig. S4C; p = 0.977; Mann-Whitney test) and had similar resting membrane potentials (Fig. S4D; p = 0.865; unpaired T-test). However, neurons from MS mice exhibited a reduction in input resistance (Fig. S4D; p = 0.031; unpaired T-test), and a slight but statistically significant reduction in action potential firing following depolarising current injection (Fig. S4E, F; p = 0.040; 2-way ANOVA column factor). Additionally, these neurons exhibited a strong reduction in sag potential (Fig. S4H, I; p < 0.0001; 2-way ANOVA column factor). Hence, all of these observations point to a reduction in excitability in SOM-positive pallidal neurons in adult mice following MS. Altogether with our optogenetic experiments, these results indicate that MS reduces inhibitory connectivity between the pallidum and forebrain via postsynaptic modifications which may also be accompanied by a presynaptic reduction in neuronal excitability.
MS sensitises LHb neurons to acute stress
Thus far we have shown that MS induces a seemingly mild depressive phenotype within the adult mouse, which is accompanied by reduced activity within the LHb in slices and various synaptic alterations. While the changes we observed in inhibitory transmission (Figs. 3 and 4) appear to fit relatively well with the current literature, our data for the physiological properties of LHb neurons (Fig. 2) apparently goes against the central hypothesis that depression is driven by hyperactivity within the LHb. Referring again to our behavioural data, we speculated that a possible explanation for this may be that rather than being in a strongly depressed state as adults, the mice are in a state where they are only mildly depressed but rather do not respond particularly well to emotional challenge. Several observations led us to this hypothesis: firstly, the observation that the sucrose preference deficit is strongest on day 2 when the positions of the bottles are switched (Fig. 1B), which may indicate that the MS mice do not respond as well to changes in learned behaviours. Secondly, that the MS mice exhibited an increased latency to groom in the splash test (Fig. 1F). This is a reflection of the fact that the initial reaction of the mice to being sprayed with sucrose solution is to panic and flee, and the MS mice seemingly do this for longer and hence take longer to relax and start grooming. Thirdly, the observation that although not more spontaneously active at rest, the neurons from MS mice fire rebound bursts with a shorter latency (Fig. 2H, I), possibly indicative that they are more primed to fire in response to synaptic drive.
To test this, we therefore submitted both CTRL and MS mice to an acute stressor in the form of 1 hour restraint, then immediately sacrificed them and performed acute slice recordings (Fig. 5A; n/N = 56/5 CTRL; n/N = 51/5 MS neurons/mice). As with our previous recordings, we observed no difference in passive physiological properties of LHb neurons (Fig. 5B; input resistance p = 0.958; T-test; RMP p = 0.793; Mann-Whitney test). However, here we observed a more prominent increase in intrinsic excitability in MS neurons (Fig. 5C; p < 0.0001; 2-way ANOVA column factor). Surprisingly, we observed a lesser difference in the latency to rebound burst, although still significant (Fig. 5D; p = 0.042; 2-way ANOVA column factor). As hypothesised, we found that a greater fraction of the neurons from MS mice were spontaneously active at rest following acute restraint (Fig. 5E, F; p < 0.0001; Chi square test). Moreover, although the average frequency of neurons which were spontaneously active was not found to be different (Fig. 5G; p = 0.764; Mann-Whitney test), neurons from the MS mice displayed a differing distribution of spontaneous activity frequencies (Fig. 5H; p = 0.045; Kolmogorov-Smirnov test). Interestingly, we also observed a trend towards a positive correlation between the behavioural Z score of the mice and the mean spontaneous activity frequency for each mouse (Fig. 5I; p = 0.063; simple linear regression), indicating that the emotional state of the mouse is a reasonably valid predictor of the response of the LHb neurons to stress. We further tested our hypothesis that the LHb neurons are more sensitive to stress histologically, by quantifying expression of the immediate early gene cFos in a separate cohort of both CTRL and MS mice either with or without exposure to acute restraint (Fig. 5J, K; N = 8 CTRL; N = 9 MS; N = 10 CTRL restraint; N = 9 MS restraint mice). Restraint was able to reliably drive cFos expression in both CTRL and MS mice (Fig. 5J, K; p < 0.0001; one-way ANOVA), with MS mice exhibiting a trend towards a greater numbers of cFos-positive neurons than CTRL mice following restraint (Fig. 5J; p = 0.081; Sidak’s test) but not in non-restrained animals (Fig. 5J; p = 0.718; Sidak’s test), thus further confirming that LHb neurons in MS animals are more sensitised to stress.
A Schematic illustrating experimental timeline. B Comparison plots of passive physiological properties in both conditions. C Input-output plot of input current against mean number of induced action potentials. D Plot of input current against latency to initial spike of the rebound burst induced upon current step end. Spontaneous activity comparison for both conditions. E Pie charts depicting percentages of recorded neurons which were classed as spontaneously active (>0.5 Hz frequency). F Example spontaneous activity recordings from both conditions. G Comparison plot of mean spontaneous activity frequency, for neurons which were spontaneously activity (>0.5 Hz frequency). H Probability distribution histogram comparing mean spontaneous activity distribution for all recorded neurons between conditions. Data are 0.5 Hz bins. I XY plot of behavioural z score against mean spontaneous activity frequency calculated for each individual mouse recorded from. Mean spontaneous activity scores are calculated as the mean spontaneous frequency of all cells recorded from each mouse. J cFos cell counts calculated in 4 test conditions. Data are mean ± SEM of cFos counts/area calculated as averages of all counted slices for each mouse. K Example confocal images of cFos immunoreactivity from each of the 4 test conditions.
Finally, we asked if this increase in activity was synaptically driven. We tested this by recording sEPSC’s in slices from CTRL and MS mice following restraint (n/N = 56/5 CTRL; n/N = 55/5 MS neurons/mice). We observed no difference in sEPSC frequency (Fig. S5A; p = 0.950; Mann-Whitney test) or amplitude (Fig. S5A; p = 0.974; Mann-Whitney test), with no obvious correlation to behavioural phenotype (Fig. S5B; p = 0.223; simple linear regression). However, we did observe a difference in kinetics, with many neurons in slices from CTRL mice exhibiting distinctive currents with an increased rise time (Fig. S5D; p = 0.013; Mann-Whitney test) and a strong trend towards an increased decay (Fig. S5E; p = 0.051; Mann-Whitney test) which were less prevalent in MS mice. We also observed a clearly different data distributions for both rise time (Fig. S5D; p < 0.0001; Kolmogorov-Smirnov test) and decay (Fig. S5E; p < 0.0001; Kolmogorov-Smirnov test). These distinctive currents appeared to be AMPA-mediated in that they were sensitive to the AMPA antagonist NBQX (Figs. S5C and S6; p < 0.0001; Dunn’s multiple comparisons test), but not the NMDA antagonist AP5 (Fig. S6; p = 0.324; Dunn’s multiple comparisons test). A possible explanation for this may be differences in AMPA subunit composition between MS and CTRL mice, which reflects differences in ongoing plasticity [27]. Interestingly, AMPA currents with extended kinetics have recently been shown to be involved in the induction of synaptic plasticity in hippocampal principal cells [28], and as such we can speculate that this may be ongoing to a greater extent in CTRL mice, which may be related to the induction of depressive symptoms following restraint stress [6]. Indeed, it would make sense that these events are less prominent in MS mice, where it can be assumed that synaptic potentiation has already occurred to a greater extent. Altogether, these data are indicative of a scenario whereby LHb neurons are more responsive to stress following MS likely via differences in postsynaptic AMPA receptor subunit composition.
Discussion
In this work we have implemented the maternal separation model of depression, in tandem with behavioural assays and in vitro electrophysiological recording techniques to dissect how early life stress influences the behavioural state and the underlying physiology of the LHb in adult mice. We report that the depressive phenotype is relatively weak in the adults. We also find that this is accompanied by a decrease in spontaneous neuronal activity, with a weakening of synaptic input from inhibitory SOM-positive forebrain neurons. However, rather than being more active at rest, in our model we find that the LHb neurons have a heightened sensitivity to respond to the re-exposure of stressful events. This may offer a neurobiological explanation as to why relapses occur following remission of depressive episodes.
Relating to our conclusions, arguably the most important aspect of our work that must be discussed is the validity of the MS model in successfully inducing depressive symptoms. It is important to note that historically, works implicating the MS model of depression have reported very variable results [29]. Indeed, while some studies have reported that early life stress can reliably induce depressive symptoms and drive LHb hyperactivity [8, 16,17,18], others have reported a failure of early life stress to induce a depressive phenotype [30, 31], and some evidence has even reported that it can induce resilience in the adult animals [32]. This inherent variability in reliability of MS is likely at least partially explained by inconsistencies in protocols, with longer periods of separation generally being thought to more reliably induce aberrant phenotypes [33]. Furthermore, it has also been shown that variations of the MS protocol in which MS is performed unpredictably (ie. at varying timepoints each day, and not necessarily on consecutive days) are more reliable in inducing depressive symptoms [32, 34]. A further confounding factor is the propensity for MS protocols to induce compensatory maternal care from the dam upon return to the home cage [35, 36], which can alleviate the depressive phenotype. Another possible explanation is species and strain difference: it is believed that mice are generally more resistant to the adverse effects of MS than rats [29, 37], and C57 mice are thought to be particularly resilient [37]. Hence, we employed the maternal separation with early weaning variant of the protocol [38], a variant of the protocol with an extended separation period (6 hours per day) and early weaning at postnatal day 17 to negate the possibility for compensatory care at this critical developmental period which has previously been shown to induce a phenotype in C57 mice, and even with this optimized protocol we could only observe relatively few behavioural deficits. Importantly however, of the behavioural deficits we did observe, these all point in the direction of the MS mice exhibiting a more susceptible phenotype than the CTRL mice, hence ruling out the possibility that our model has also promoted resilience (Fig. 1). Nonetheless, our findings reinforce the need for more robust MS protocols, which we speculate could possibly be optimized by combining unpredictable MS protocols with early weaning to simultaneously enhance the impact of the MS phase of the protocol while also negating compensatory care pre-weaning.
The next key question that must be addressed is why our model induces a reduction in spontaneous activity within the LHb. It is now very well accepted that LHb hyperactivity promotes depressive behaviour [3, 4, 7,8,9], and our data does not superficially support this. Firstly, to address this question, we would point to the fact that in healthy animals, the LHb is active to serve an important purpose: that is to encode reward prediction error and prevent reinforcement of behaviours with negative outcomes [39]. Therefore, the relationship between LHb activity level and behavioural phenotype is likely not as simple as heightened activity equalling a more pronounced depressive phenotype, and reduced activity equalling a less depressed phenotype. Hence it may be the case that a reduction in LHb activity is also indicative of an aberrant phenotype. Indeed, a recent hypothesis has proposed that LHb hypoactivity in childhood may promote attention deficit hyperactivity disorder, which in turn primes the LHb to be more responsive to stress in adulthood [40]. Experimental evidence has also shown that LHb inactivation induces a reversal learning deficit [41], which may explain why we see a prominent deficit in sucrose preference when the position of the bottles are switched (Fig. 1B). Thus, LHb hypoactivity is not likely to promote a healthy phenotype. Secondly, and in line with our first point, our recordings in CTRL mice (Fig. 2B) largely agree with the existing literature [4, 17, 18] in the observed distribution of tonic, bursting and silent neurons, thus supporting the claim that background LHb activity is important within healthy animals, and also ruling out the possibility of a recording artefact. Finally, although it is now well accepted that excitatory synaptic drive is potentiated onto the LHb in depression [3], it should also be noted that there is somewhat conflicting evidence in that recent work has also observed a decrease in postsynaptic LHb AMPA receptor expression following exposure to stress [42]. Indeed, our data also suggest that a more depressed phenotype correlates with a reduction in spontaneous excitatory postsynaptic current frequency (Fig. S2B). Accounting for all of this evidence we would therefore conclude that in addition to LHb hyperactivity, LHb hypoactivity may also promote an aberrant phenotype and as such the reduction in spontaneous activity we observe is not mutually exclusive with the central hypothesis that LHb hyperactivity drives depression.
In terms of inhibitory signalling within the LHb, the literature is relatively consistent in that this promotes behavioural reinforcement [25, 43, 44], and that inhibition of the LHb has an antidepressant effect [45, 46]. Consistent with this, inhibitory signalling has been shown to be perturbed in various models of depression [7, 8, 24], and indeed our data lend further support to this hypothesis. It is interesting to note that the loss of spontaneous inhibitory input we observed appears to be specific for the medial portion of the LHb (Fig. 3B–D). Previous work has identified a population of inhibitory LHb neurons which resides specifically within the medial LHb [47, 48], and hence one could speculate that activity of this population may be down-regulated following MS, therefore resulting in a loss of local inhibition. Additionally, we also report a reduction in connectivity between inhibitory pallidal neurons and the LHb, which our data also suggest involves a possible presynaptic reduction in excitability (Fig. S4) although we must note that as an anterograde labelling approach was used here, we cannot say with absolute certainty that these neurons project directly to the LHb. While previous work has characterised inhibitory projections from various pallidal regions to the LHb relatively extensively [14, 25, 43, 49], to our knowledge this is the first time that such a projection has been shown to be implicated in the pathogenesis of a model of depression.
As a final note, here we have focused specifically on early life stress-induced alterations in excitatory and inhibitory signalling within the LHb. However, it should also be noted that there is growing evidence that other neurotransmitters influence LHb activity [50], and conversely that the LHb can influence other transmitter systems downstream. As such it may be the case that alterations in other transmitter systems are driving the physiological changes within our model. Interestingly recent evidence has indicated that early life stress alters Kappa opioid receptor (KOR) signalling at LHb synapses both pre- and postsynaptically, resulting in a loss of LHb excitability in a subset of neurons [17]. Furthermore, KOR signalling was also shown to perturb both GABAergic and glutamatergic drive onto LHb neurons. As we see a loss of both excitatory and inhibitory drive, paired with a reduction in LHb excitability it may be the case that our model is also inducing alterations in KOR signalling within the LHb, thus altering LHb output and control over downstream reward pathways. Assuming this as a possibility, then it is also likely that alterations in other transmitters systems are induced in our model, and further work should serve to elucidate these.
Conclusions
Depression is a complex disease, with hugely variable aetiology [51]. Our work further complements the existing literature in that we provide evidence that LHb hypoactivity can also be associated with a depressive phenotype, and may be representative of a state where the animal is hypersensitive to stressful events. This work challenges the classical view that within the LHb, hyperactivity is the sole driver state of depressive behaviour. Further work into the specific molecular mechanisms by which these changes occur may shed new light onto the pathogenesis of depression and may unveil novel molecular targets for future therapies.
References
Matsumoto M, Hikosaka O. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature. 2007;447:1111–5.
Lecca S, Meye FJ, Trusel M, Tchenio A, Harris J, Schwarz MK, et al. Aversive stimuli drive hypothalamus-to-habenula excitation to promote escape behavior. Elife. 2017;6:1–16.
Li B, Piriz J, Mirrione M, Chung C, Proulx CD, Schulz D, et al. Synaptic potentiation onto habenula neurons in the learned helplessness model of depression. Nature. 2011;470:535–9.
Yang Y, Cui Y, Sang K, Dong Y, Ni Z, Ma S, et al. Ketamine blocks bursting in the lateral habenula to rapidly relieve depression. Nature. 2018;554:317–22.
Hu H, Cui Y, Yang Y. Circuits and functions of the lateral habenula in health and in disease. Nat Rev Neurosci. 2020;21:277–95. https://doi.org/10.1038/s41583-020-0292-4. at
Zheng, Z, Guo, C, Li, M, Yang, L, Liu, P, Zhang, X, et al. Hypothalamus-habenula potentiation encodes chronic stress experience and drives depression onset. Neuron 1–16 (2022) https://doi.org/10.1016/j.neuron.2022.01.011.
Lecca S, Pelosi A, Tchenio A, Moutkine I, Lujan R, Hervé D, et al. Rescue of GABAB and GIRK function in the lateral habenula by protein phosphatase 2A inhibition ameliorates depression-like phenotypes in mice. Nat Med. 2016;22:254–61.
Tchenio A, Lecca S, Valentinova K, Mameli M. Limiting habenular hyperactivity ameliorates maternal separation-driven depressive-like symptoms. Nat Commun. 2017;8:1135.
Cui Y, Yang Y, Ni Z, Dong Y, Cai G, Foncelle A, et al. Astroglial Kir4.1 in the lateral habenula drives neuronal bursts in depression. Nature. 2018;554:323–7.
Wang RY, Aghajanian GK. Physiological evidence for habenula as major link between forebrain and midbrain raphe. Science. 1977;197:89–91.
Ji H, Shepard PD. Lateral habenula stimulation inhibits rat midbrain dopamine neurons through a GABA(A) receptor-mediated mechanism. J Neurosci. 2007;27:6923–30.
Jhou TC, Fields HL, Baxter MG, Saper CB, Holland PC. The Rostromedial Tegmental Nucleus (RMTg), a major GABAergic afferent to midbrain dopamine neurons, encodes aversive stimuli and inhibits motor responses. Neuron. 2009;61:786–800.
Cerniauskas I, Winterer J, Jong JW, De, Lukacsovich D, Yang H, et al. Chronic Stress Induces Activity, Synaptic, and Transcriptional Remodeling of the Lateral Habenula Associated with Deficits in Motivated Behaviors. Neuron. 2019;104:899–915.e8.
Golden SA, Heshmati M, Flanigan M, Christoffel DJ, Guise K, Pfau ML, et al. Basal forebrain projections to the lateral habenula modulate aggression reward. Nature. 2016;534:688–92.
Knowland D, Lilascharoen V, Pham Pacia C, Shin S, Hou-Jen Wang E, Kook Lim B, et al. Distinct ventral pallidal neural populations mediate separate symptoms of depression. Cell. 2017;170:284–297.e18.
Authement ME, Langlois LD, Shepard RD, Browne CA, Lucki I, Kassis H, et al. A role for corticotropin-releasing factor signaling in the lateral habenula and its modulation by early-life stress. Sci Signal 2018;11:eaan6480.
Simmons SC, Shepard RD, Gouty S, Langlois LD, Flerlage WJ, Cox BM, et al. Early life stress dysregulates kappa opioid receptor signaling within the lateral habenula. Neurobiol Stress. 2020;13:100267.
Langlois LD, Berman RY, Shepard RD, Simmons SC, Tsuda MC, Gouty S, et al. Potentiation of glutamatergic synaptic transmission onto lateral habenula neurons following early life stress and intravenous morphine self-administration in rats. Addict Biol. 2022;27:e13064.
Webster JF, Vroman R, Balueva K, Wulff P, Sakata S, Wozny C. Disentangling neuronal inhibition and inhibitory pathways in the lateral habenula. Sci Rep. 2020;10:1–17.
Webster JF, Vroman R, Beerens S, Sakata S, Wozny C. NDNF is selectively expressed by neocortical, but not habenular neurogliaform cells. Eur J Neurosci. 2021;53:3561–75.
Baker PM, Mizumori SJY. Control of behavioral flexibility by the lateral habenula. Pharmacol Biochem Behav. 2017;162:62–68.
Fukumitsu K, Kaneko M, Maruyama T, Yoshihara C, Huang AJ, McHugh TJ, et al. Amylin-Calcitonin receptor signaling in the medial preoptic area mediates affiliative social behaviors in female mice. Nat Commun. 2022;13:709.
Guilloux JP, Seney M, Edgar N, Sibille E. Integrated behavioral z-scoring increases the sensitivity and reliability of behavioral phenotyping in mice: Relevance to emotionality and sex. J Neurosci Methods. 2011;197:21–31.
Shabel SJ, Proulx CD, Piriz J, Manilow R. Mood regulation. GABA/glutamate co-release controls habenula output and is modified by antidepressant treatment. Science. 2014;345:1494–8.
Stephenson-Jones M, Bravo-Rivera C, Ahrens S, Furlan A, Xiao X, Fernandes-Henriques C, et al. Opposing contributions of GABAergic and Glutamatergic Ventral pallidal neurons to motivational behaviors. Neuron. 2020;105:921–933.e5.
Libovner Y, Fariborzi M, Tabba D, Ozgur A, Jafar T, Lur G. Repeated exposure to multiple concurrent stresses induce circuit specific loss of inputs to the posterior parietal cortex. J Neurosci. 2020;40:1849–61.
Henley JM, Wilkinson KA. Synaptic AMPA receptor composition in development, plasticity and disease. Nat Rev Neurosci 2016;17:337–250.
Pampaloni NP, Riva I, Carbone AL, Plested AJR. Slow AMPA receptors in hippocampal principal cells. Cell Rep. 2021;36:109496.
Tractenberg SG, Levandowski ML, de Azeredo LA, Orso R, Roithmann LG, Hoffmann ES, et al. An overview of maternal separation effects on behavioural outcomes in mice: Evidence from a four-stage methodological systematic review. Neurosci Biobehav Rev. 2016;68:489–503.
Millstein RA, Holmes A. Effects of repeated maternal separation on anxiety- and depression-related phenotypes in different mouse strains. Neurosci Biobehav Rev. 2007;31:3–17.
Tan S, Ho HS, Song AY, Low J, Je HS. Maternal separation does not produce a significant behavioral change in mice. Exp Neurobiol 2017;26:390–8.
Shi D-D, Zhang Y-D, Ren Y-Y, Peng S-Y, Yuan T-F, Wang Z. Predictable maternal separation confers adult stress resilience via the medial prefrontal cortex oxytocin signaling pathway in rats. Mol Psychiatry. 2021;26:7296–307.
Nylander I, Roman E. Is the rodent maternal separation model a valid and effective model for studies on the early-life impact on ethanol consumption? Psychopharmacology. 2013;229:555–69.
Franklin TB, Russig H, Weiss IC, Grff J, Linder N, Michalon A, et al. Epigenetic transmission of the impact of early stress across generations. Biol Psychiatry. 2010;68:408–15.
Macrí S, Mason GJ, Würbel H. Dissociation in the effects of neonatal maternal separations on maternal care and the offspring’s HPA and fear responses in rats. Eur J Neurosci. 2004;20:1017–24.
Orso R, Creutzberg KC, Wearick-Silva LE, Wendt Viola T, Tractenberg SG, Benetti F, et al. How Early Life Stress Impact Maternal Care: A Systematic Review of Rodent Studies. Front Behav Neurosci. 2019;13:197.
Own LS, Patel PD. Maternal behavior and offspring resiliency to maternal separation in c57bl/6 mice. Horm Behav. 2013;63:411–7.
George ED, Bordner KA, Elwafi HM, Simen AA. Maternal separation with early weaning: A novel mouse model of early life neglect. BMC Neurosci. 2010;11:123.
Hikosaka O. The habenula: from stress evasion to value-based decision-making. Nat Rev Neurosci. 2010;11:503–13.
Lee YA, Goto Y. The Habenula in the link between ADHD and mood disorder. Front Behav Neurosci. 2021;15:699691.
Baker PM, Oh SE, Kidder KS, Mizumori SJY. Ongoing behavioral state information signaled in the lateral habenula guides choice flexibility in freely moving rats. Front Behav Neurosci. 2015;9:1–22.
Nuno-Perez A, Trusel M, Lalive AL, Congiu M, Gastaldo D, Tchenio A, et al. Stress undermines reward-guided cognitive performance through synaptic depression in the lateral habenula. Neuron. 2021;109:1–10.
Faget L, Zell V, Souter E, McPherson A, Ressler R, Gutierrez-reed N, et al. Opponent control of behavioral reinforcement by inhibitory and excitatory projections from the ventral pallidum. Nat Commun. 2018;9:1–14.
Lalive AL, Congiu M, Lewis C, Groos D, Clerke JA, Tchenio A, et al. Synaptic inhibition in the lateral habenula shapes reward anticipation. Curr Biol. 2022;32:1829–1836.E4.
Winter C, Vollmayr B, Djodari-Irani A, Klein J, Sartorius A. Pharmacological inhibition of the lateral habenula improves depressive-like behavior in an animal model of treatment resistant depression. Behav Brain Res. 2011;216:463–5.
Huang L, Xi Y, Peng Y, Yang Y, Huang X, Fu Y, et al. A visual circuit related to habenula underlies the antidepressive effects of light therapy. Neuron. 2019;102:128–142.e8.
Zhang L, Hernández VS, Swinny JD, Verma AK, Giesecke T, Emery AC, et al. A GABAergic cell type in the lateral habenula links hypothalamic homeostatic and midbrain motivation circuits with sex steroid signaling. Transl Psychiatry. 2018;8:50.
Flanigan ME, Aleyasin H, Li L, Burnett CJ, Chan KL, LeClair KB, et al. Orexin signaling in GABAergic lateral habenula neurons modulates aggressive behavior in male mice. Nat Neurosci. 2020;23:638–50.
Pribiag H, Shin S, Wang EHJ, Sun F, Datta P, Okamoto A, et al. Ventral pallidum DRD3 potentiates a pallido-habenular circuit driving accumbal dopamine release and cocaine seeking. Neuron. 2021;109:2165–2182.e10.
Webster JF, Lecca S, Wozny C. Inhibition Within the Lateral Habenula—Implications for Affective Disorders. Front Behav Neurosci. 2021;15:786011.
Otte C, Gold SM, Penninx BW, Pariante CR, Etkin A, Fava M, et al. Major depressive disorder. Nat Rev Dis Prim. 2016;2:16065.
Acknowledgements
We are grateful to the BPU staff for expert technical assistance. We also thank Dr Salvatore Lecca for discussions on a previous version of the manuscript.
Funding
This work was funded by an EPSRC Doctoral Prize to JFW, a NARSAD Young Investigator Award to CW (Grant number 28217, named P&S Fund Investigator) and by the European Union’s Horizon 2020 Research and Innovation Program under Grant Agreement No. ICT-36-2020-101016787, DEEPER to CW.
Author information
Authors and Affiliations
Contributions
JFW performed the experiments, SB contributed to experiments, CW and JFW designed the study, CW supervised the work. JFW wrote manuscript with help of CW All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Webster, J.F., Beerens, S. & Wozny, C. Effects of early life stress and subsequent re-exposure to stress on neuronal activity in the lateral habenula. Neuropsychopharmacol. 48, 745–753 (2023). https://doi.org/10.1038/s41386-022-01493-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-022-01493-0