Abstract
The prefrontal cortex (PFC) supports a diversity of cognitive processes. Impairment in PFC-dependent cognition is associated with multiple psychiatric disorders, including those known to display sex differences. Our ability to treat this impairment is limited, in part due to an incomplete understanding of the neural mechanisms that support PFC-dependent cognition. In previous studies in male rats, we demonstrated that corticotropin-releasing factor (CRF) receptors and neurons in caudal dorsomedial PFC (dmPFC) regulate PFC-dependent working memory. Subcortically, CRF can exert sex-specific actions, a subset of which are ovarian steroid dependent. To date, the cognitive actions of dmPFC CRF neurotransmission in females are unknown. To address this gap, the current studies examined the effects of chemogenetic and pharmacological manipulations of CRF receptors and neurons within the dmPFC of female rats tested in a spatial working memory task. Outside of proestrus, activation of both CRF receptors and neurons in the caudal, but not rostral, dmPFC impaired working memory. Meanwhile, blockade of CRF receptors in the caudal dmPFC or globally in the brain, improved working memory performance, similar to that seen in males. In contrast, these effects were not observed during proestrus. These observations demonstrate that while CRF neurotransmission in the PFC regulates working memory similarly in males and females, these actions are not observed in females when ovarian steroids are at peak levels.
Similar content being viewed by others
Introduction
The prefrontal cortex (PFC) supports cognitive processes needed for the attainment of distal goals, particularly under distracting conditions [1]. Impaired PFC-dependent cognition is a hallmark feature of multiple psychiatric disorders. The incidence and phenotype of many of these disorders, including depression, schizophrenia, and attention deficit hyperactivity disorder (ADHD), vary across men and women [2,3,4]. Currently available pharmacological treatments for PFC cognitive dysfunction suffer from significant limitations [5]. Our incomplete understanding of the neural mechanisms that support PFC-dependent cognition, particularly across the broader population, limits the development of more effective treatments for PFC-related cognitive impairment.
Prior studies in male rats demonstrate that corticotropin-releasing factor (CRF) receptors in the caudal dorsomedial PFC (dmPFC) impair PFC-dependent working memory [6]. Subsequent chemogenetic studies demonstrated that activation of CRF neurons in this region also impairs working memory, an action that was dependent on local CRF receptors [7]. Conversely, suppression of caudal dmPFC CRF neuronal activity and blockade of CRF receptors in this region, or globally in the brain, improved working memory [6, 7]. Collectively, these observations demonstrate that, in males, CRF neurotransmission within the PFC regulates PFC-dependent cognition. However, subcortically, CRF has been demonstrated to exert sex-specific actions, a subset of which are ovarian steroid-dependent [8, 9]. Moreover, as noted, there exist significant sex differences in behavioral disorders associated with PFC cognitive dysfunction. Lastly, while global distribution of CRF in the brain impaired PFC-dependent sustained attention in males and females, this was only observed in females during portions of the estrus cycle associated with lower ovarian steroids [10]. To date, the degree to which CRF neurotransmission in the PFC affects cognitive function in females and whether these actions fluctuate across estrus cycle stages associated with high vs. low ovarian steroids are unknown.
To address these issues, the current studies examined the effects of pharmacological and chemogenetic manipulations of PFC CRF receptors and neurons in female rats tested in a delayed response task of spatial working memory. To activate CRF neurons in the PFC, we selectively expressed ‘excitatory’ (hM3Dq) DREADDs in rostral or caudal dmPFC CRF neurons, similar to earlier studies in males [7]. To better compare to prior studies in males [6, 10, 11] and to better assess the potential clinical utility of CRF receptor antagonists, we also examined the working memory effects of CRF and a CRF antagonist distributed globally in the brain. To determine estrus cycle dependency, treatments were delivered at distinct stages of the estrus cycle associated with high (proestrus) vs. lower levels of ovarian steroids (outside proestrus) [12].
Outside of proestrus, activation of CRF receptors and neurons in the caudal dmPFC impaired, while inhibition of caudal dmPFC CRF neurotransmission improved, working memory. Within proestrus, when ovarian steroids reach peak levels, these actions were not observed. These studies demonstrate that in females tested outside the relatively short period of proestrus, PFC CRF neurotransmission impacts working memory similar to that seen in males. These observations further our understanding of the neurobiology of PFC-dependent cognition and suggest that circulating ovarian steroids modulate the cognitive actions of PFC CRF neurotransmission.
Methods and materials
With the exception of vaginal cytology, all methods are as described previously [6, 7, 13]. Additional details can be found in the Supplemental Methods section.
Animals
Female Sprague-Dawley rats (200-300 grams; Charles River, Wilmington, Massachusetts) were pair-housed in frosted polycarbonate cages on a 13/11-hour light/dark cycle. Animals were fed ad libitum for the first 4–7 days after arrival and then titrated for each pair (10–13 g of standard chow/day/animal) to maintain motivation for food reward while avoiding weight loss. Rats were handled extensively prior to behavioral testing and weighed 2x/week. Training/testing was conducted between 0800 and 1600 h (5–6 days/week). All procedures were in accordance with the National Institutes of Health of the United States guidelines and approved by the university’s Institutional Animal Care and Use Committee.
Surgery
Stereotaxic surgery was performed under isoflurane anesthesia (1–2%). Intra-brain infusion cannulae (25 ga.) were positioned ~200 µm below the dura to minimize cortical damage (bilaterally for intra-PFC, unilaterally for ICV). For intra-PFC infusions, needle projection length varied as needed [6]. Stainless steel stylets prevented occlusion of cannulae and were replaced as needed. For chemogenetic activation, a cocktail of two viruses was infused bilaterally into the caudal or rostral dmPFC via 33 ga. infusion needles [7]. This cocktail expresses hM3Dq and mCherry in a Cre-dependent manner selectively in CRF neurons (AAV8-CRF-Cre; Vector Biolabs, Malvern, PA + AAV8-hSyn-DIO-hM3Dq-mCherry; Addgene, Cambridge, MA). Viral controls lacked the DREADD transgene (AAV8-hSyn-DIO-mCherry; Addgene, Cambridge, MA). 1.65 µl of viral cocktail mixed in a 1:2 ratio of CRF-Cre:2nd virus was infused at a rate of 0.25 µl/min.
Behavioral Training and Testing
Working memory training and testing was conducted in rooms devoid of external spatial cues. Briefly, animals were trained to enter the arm of a T-maze not chosen on the previous trial to receive a food reward in 20-trial sessions/day (45 mg sucrose pellet/trial, Bio-Serv, Flemington, NJ). Rats were placed in a start box at the base of the maze between trials and prevented from exiting by a removable gate, with an initial delay of 10-seconds and a maximum of 60-seconds. Treatments were only given if performance during the two days prior to testing did not differ by more than 10%. Baseline performance levels of 70–95% or 62.5–87.5% were used to detect working memory impairing or improving effects, respectively. Treatment effects were measured as a within-subjects percent change from baseline performance, measured as the average of the 2 days preceding treatment.
Drug treatments
CRF (ovine, Bachem, Torrance, CA) was dissolved in buffered artificial extracellular fluid (AECF; 147 mmol/L NaCl, 1.3 mmol/L CaCl, 0.9 mmol/L MgCl, 2.5 mmol/L KCl; pH = 7.4). The non-selective CRF antagonist, D-Phe-CRF (12-41; Bachem, Torrance, CA), was dissolved in 0.9% saline. Animals were transported to the testing room in their home cage and remained in the testing room until testing was initiated. ICV and bilateral intra-PFC infusions were performed 15-minutes prior to testing using 33 ga. needles at a rate of 250 nl/min for 2-minutes (500 nl total). Infusion needles were left in place for 2-minutes, after which the stylets were replaced. CNO (clozapine-N-oxide dihydrochloride, Tocris, Bristol, UK) was dissolved in 0.9% saline and administered subcutaneously 45-minutes before testing. To better assess the potential clinical utility of CRF antagonists, the small molecule CRF1 antagonist, NBI 35965 hydrochloride (Tocris, Bristol, UK), was dissolved in 0.9% saline and administered subcutaneously (1 ml/kg) 60-minutes before testing. Choice of antagonists and doses of all treatments are based on prior observations in males [6, 7] and limited pilot studies. The superficial cannula placement, combined with close attention to infusion procedures (cleanliness, needle straightness) avoids significant tissue damage/infection when the number of intra-tissue infusions is limited to 7-8, a limit that was followed in this study.
Histology and Immunohistochemistry
Needle placements were determined in 40-µm thick coronal sections stained with Neutral Red dye (Sigma-Aldrich, St. Louis, MO), as described previously [6]. Data from a given experiment were included only when histological analyses verified accurate placement of injector tip and minimal PFC damage. The location and extent of viral expression (as determined by mCherry) was determined as described previously [7].
In a subset of 3–5 animals per group, additional sections were labeled for either CRF-immunoreactivity (ir) or Fos-ir and mCherry viral expression as previously described [7]. 60X images were used to determine the number of CRF-ir or Fos-ir and mCherry per image.
Vaginal cytology
Vaginal cytology was used to determine distinct stages of the estrus cycle (metestrus, diestrus, estrus, proestrus) using criteria previously described [14, 15]. Briefly, 0.05–0.1 mL sterile 0.01 M phosphate-buffered saline (PBS) was applied with a sterile, disposable 1 mL syringe (BD, Franklin Lakes, NJ) inserted 3–5 mm into the vagina. Fluid was ejected and reabsorbed twice and then ejected onto a microscope slide. Wet samples were then evaluated for cycle phase using an Olympus BX51 microscope, and a representative portion was imaged at 10x magnification with an attached DP73 Olympus camera. Daily cycle phase identifications were tracked to monitor cycling and predict phase timing. Sample collection and evaluation occurred no more than one hour before testing. Dried samples were H&E stained according to Harris’ Hemotoxylin Protocol (National Diagnostics, 2011), then cover slipped using DPX mounting medium. Once dried, the samples were evaluated once more to confirm cycle phase.
Statistical analyses
When possible, each rat received each treatment dose in both proestrus and non-proestrus. However, with a limit on the number of infusions, this was not always possible. Thus, a linear mixed-effects model using JMP® Pro Version 12.2.0 (SAS Institute Inc.) was used to analyze behavioral measures, with treatment as a fixed effect factor and animal as a random effect factor. When statistical significance was indicated (P < 0.05), post-hoc comparisons between drug dose and vehicle were determined using Bonferroni-corrected t-tests. Data points greater than 2 standard deviations from the mean were considered outliers and excluded. This resulted in no more than 1 data point removed from any group.
Results
Working memory effects of CRF receptor activation and blockade in the dmPFC
CRF receptor activation
We first examined the working memory effects of caudal dmPFC CRF receptors. For this, bilateral intra-PFC infusions of varying doses of CRF (vehicle, 50 ng, 250 ng CRF) were made into the caudal dmPFC during proestrus, when ovarian steroids peak, vs. all other stages outside proestrus combined. Group sizes for the various treatments were as follows: proestrus, vehicle, n = 7; 50 ng, n = 11; 250 ng, n = 12; non-proestrus, vehicle, n = 14; 50 ng, n = 13; 250 ng, n = 15. As shown in Fig. 1A, activation of caudal dmPFC CRF receptors elicited a dose-dependent impairment in working memory performance outside of proestrus (F2,39.5 = 4.21, P = 0.022). This was not observed during proestrus (F2,32.8 = 1.03, P = 0.367). Additional studies examined the effects of CRF infusions into the rostral dmPFC (proestrus: vehicle, n = 7; 50 ng, n = 9; 250 ng, n = 8; non-proestrus: vehicle, n = 9; 50 ng, n = 7; 250 ng, n = 8). Rostral dmPFC infusions failed to affect working memory outside (Fig. 1B; F2,22.6 = 1.39, P = 0.270) or during proestrus (F2,14.1 = 0.24, P = 0.792).
A Representative photomicrograph depicting the main body of the ventral-most extent of the needle track within the caudal dmPFC. As this represents the main body of the track, it depicts the greatest extent of tissue damage. Schematics indicating location of infusion sites in all animals tested. Bar graphs represent percent change from baseline (±SEM) following bilateral intra-caudal dmPFC infusions of vehicle, 50 ng, or 250 ng CRF. CRF infusions impaired working memory outside (Non-Pro), but not during (Pro), proestrus. B CRF infusions into the rostral dmPFC did not significantly affect task performance. C Working memory effects of bilateral infusions of vehicle, 200 ng, or 500 ng of the CRF antagonist, D-Phe-CRF, into the caudal dmPFC outside of proestrus improved task performance. This antagonist significantly improved performance at the 500 ng dose. *P < 0.05 vs. vehicle. CC corpus callosum, dAcg dorsal anterior cingulate, IL infralimbic, PL prelimbic.
Caudal dmPFC CRF receptor blockade
Additional studies examined the working memory effects of endogenous CRF signaling in the caudal dmPFC outside of proestrus. Animals received bilateral infusions of vehicle (n = 9), or 200 ng (n = 11) or 500 ng (n = 13) of the non-selective CRF antagonist, D-Phe-CRF. CRF receptor blockade in the caudal dmPFC elicited a modest, yet significant improvement in working memory performance at the 500 ng dose of D-Phe-CRF (Fig. 1C; F2,18.8 = 3.92, P = 0.038).
Working memory effects of chemogenetic activation of caudal dmPFC CRF neurons
Chemogenetic activation
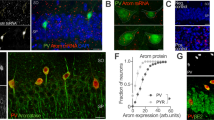
Additional studies examined the working memory effects of chemogenetic activation of dmPFC CRF neurons. Given observations described above, as well as our earlier observations [6, 7], these studies focused on the caudal dmPFC. For this, a viral cocktail encoding hM3Dq DREADDs (CRF-Cre + DIO-hM3Dq-mCherry) was infused into the caudal dmPFC (Fig. 2A). An additional group received a control virus cocktail that lacked the DREADD transgene (CRF-Cre + DIO-mCherry). For all viral treatments, robust reporter protein expression was observed within 3 weeks (Fig. 2A). Viral expression as measured by mCherry was limited to a radius of ~500 μm, filling the majority of the caudal dmPFC, with minimal spread into the ventromedial PFC or adjacent dmPFC hemifield (Figs. 2, 3). The viral cocktail transfected CRF neurons efficiently and selectively. Thus, 88.8 ± 0.03% of CRF-ir neurons displayed mCherry (n = 455 cells, 5 animals) while only 0.02% (n = 7) of mCherry-positive neurons failed to express CRF-ir. To assess whether the DREADD agonist, CNO, activates hM3Dq-expressing PFC CRF neurons, we examined Fos-ir in hM3Dq (n = 4) or viral control animals (n = 4) treated with 1 mg/kg CNO in the home-cage (highest dose used in subsequent studies). As was observed in males, CNO elicited robust colocalization (86.0 ± 0.01%) of Fos-ir and mCherry in hM3Dq-treated animals (n = 866 cells), but not viral control-treated animals (1.3 ± 0.01%, n = 839 cells).
A Schematic depicting dual viral system to activate PFC CRF neurons. One virus expresses Cre recombinase under the control of the CRF promoter (CRF-Cre) while a 2nd virus either expresses hM3Dq receptors and mCherry (hM3Dq) or mCherry alone (Control) in a Cre-dependent manner. 2x photomicrograph depicts robust mCherry expression bilaterally in the caudal dmPFC following infusion of the CRF-hM3Dq-viral cocktail; cc: corpus callosum. B Collapsed 2 µm z-stack demonstrating mCherry-positive cells (left panel, red) extensively colocalize with CRF-ir cells (middle, green) when the two images are merged (right panel); scale bar = 20 µm. C CNO treatment in hM3Dq animals elicits neuronal activation of mCherry-positive neurons, as measured by Fos-ir. Arrows indicate a subset of neurons displaying colocalized Fos-ir and mCherry. D Significant Fos-ir was not observed in control virus treated animals. Scale bars = 30 µm. CC corpus callosum.
A Schematics depict spread of control (left) and ‘excitatory’ (hM3Dq, right) virus in the caudal dmPFC from all animals tested. Bar graphs depict percent change from baseline (±SEM) following treatment with vehicle or varying doses of CNO outside of (Non-Pro) and during proestrus (Pro). Outside of proestrus, CNO dose-dependently impairs task performance in animals treated with the hM3Dq virus relative to vehicle and CNO-treated viral control animals. B In a separate group of hM3Dq tested outside of proestrus, animals received bilateral infusions of vehicle (VEH) or the CRF antagonist (Antag), D-Phe-CRF, into the caudal dmPFC after systemic vehicle or CNO. CNO-induced impairment observed in intra-PFC vehicle-treated animals was prevented with bilateral CRF antagonist infusions. **,***P < 0.01, 0.001. vs. vehicle; ++P < 0.01 vs. viral controls; ##P < 0.01 vs. intra-PFC vehicle + CNO. CC corpus callosum, dAcg, dorsal anterior cingulate, IL infralimbic PFC, PL prelimbic PFC.
Animals treated with the hM3Dq viral cocktail received either vehicle (non-proestrus: n = 10; proestrus: n = 8) or varying doses of CNO (non-proestrus: 0.3 mg/kg, n = 11; 1 mg/kg, n = 12; proestrus: 0.3 mg/kg, n = 8; 1 mg/kg, n = 7). Animals with control virus received the maximal CNO dose of 1 mg/kg (non-proestrus: n = 7; proestrus: n = 10). As shown in Fig. 3A, we observed dose-dependent effects of chemogenetic activation of caudal dmPFC CRF neurons on working memory accuracy outside of proestrus relative to both vehicle (F2,31 = 12.40, P < 0.001) and CNO-treated viral controls (F2,21.4 = 5.29, P = 0.014), with significant impairment seen with the highest dose of CNO. Within proestrus, chemogenetic activation of caudal dmPFC CRF neurons did not significantly affect working memory performance relative to vehicle (Fig. 3A; F2,17.4 = 0.17, P = 0.844) or CNO-treated viral controls (F2,17.3 = 0.13, P = 0.879).
Local receptor dependency?
Given caudal dmPFC CRF receptor activation impairs working memory, an additional study examined whether the working memory impairing effect of caudal dmPFC CRF neuronal activation (outside of proestrus) is dependent on local CRF receptors. Animals received intra-caudal dmPFC infusions of vehicle or the non-selective CRF antagonist, D-Phe-CRF (500 ng/hemisphere), 30-minutes after receiving systemic vehicle or the highest dose of CNO (1 mg/kg). In animals treated with intra-PFC vehicle, CNO elicited a robust working memory impairment that was prevented by local D-Phe-CRF infusions (Fig. 3B; n = 7, n = 8, F1,22.7 = 27.91, P < 0.001; CNO × antagonist interaction, F1,22 = 7.73, P = 0.011). While this dose of the antagonist significantly improved performance on its own in the study depicted in Fig. 1, the magnitude of this effect (~5%) is noticeably smaller than the ~14% reversal of the CNO-induced impairment in performance. Thus, additivity alone unlikely explains the ability of this antagonist to prevent the working memory impairing effect of chemogenetic activation of caudal dmPFC CRF neurons.
Global manipulations of brain CRF receptors
Additional studies examined the working memory effects of CRF and CRF antagonists when distributed more widely in the brain. We first examined the working memory effects of ICV CRF. Given observations described above as well as prior studies examining the sustained attention effects of ICV CRF [10], for these studies we only tested animals outside of proestrus. Animals were treated with either vehicle (n = 8) or varying doses of CRF (0.2 μg, n = 8; 1 µg, n = 8). As shown in Fig. 4A, ICV CRF dose-dependently impaired working memory performance outside of proestrus (F2,14 = 4.05; P = 0.041) with significant impairment observed at the 1 µg dose and a trend for impairment at the 250 ng dose (P = 0.052).
A ICV treatment with CRF outside of proestrus elicited a dose-dependent impairment in working memory. B Conversely, subcutaneous treatment with the CRF1-selective antagonist, NBI 35965, improved performance outside proestrus (Non-Pro) but not during proestrus (Pro). Bars represent mean (±SEM) percentage change in performance accuracy from baseline. *,**P < 0.05, 0.01 vs. vehicle.
Additional studies examined the working memory effects of systemic administration of the CRF1 receptor-selective antagonist, NBI 35965, in and outside proestrus. As shown in Fig. 4B, the highest dose of this antagonist significantly improved working memory outside of proestrus (F3,62.2 = 4.69; P = 0.005). During proestrus, this antagonist failed to significantly affect working memory performance (vehicle, n = 11; 5 mg/kg, n = 12; 10 mg/kg, n = 9; 20 mg/kg, n = 12, F3,37.7 = 0.38; P = 0.769). While there appears to be a trend towards improvement with the antagonist during proestrus, this in part reflects a modest and non-significant improvement in performance seen in vehicle-treated animals, a trend not observed in other components of this study.
Discussion
These results demonstrate that activation of both CRF receptors and neurons in the caudal dmPFC impairs working memory in female rats outside of proestrus. The working memory impairing effects of caudal dmPFC CRF neurons were dependent on the activation of local receptors in this region. Conversely, blocking CRF receptors within the caudal dmPFC or globally in the brain improved working memory, indicating endogenous CRF release under testing conditions. These actions are similar to those observed previously in males [6, 7]. In contrast, during the relatively short period of proestrus when circulating ovarian steroids reach peak levels, activation of caudal dmPFC CRF neurons or receptors, or blockade of CRF receptors globally in the brain, did not significantly affect working memory. In the current study, all animals completed all trials and rapidly consumed food rewards regardless of treatment, consistent with prior observations in males [6]. Thus, CRF-dependent changes in working memory performance do not appear to be associated with notable decreases in motivation for food rewards. Collectively, these observations add to a growing body of evidence for a prominent role of PFC CRF signaling in the regulation of PFC-dependent cognitive function [9], while identifying an important estrus stage-dependency in this action.
The working memory impairing effects of PFC CRF neurotransmission outside of proestrus are qualitatively similar to those observed previously in males. While this study does not permit making quantitative comparisons across studies, limited observations suggest there could be modest differences in sensitivity to CRF manipulations across males and females. Specifically, in the present studies 50 ng of intra-PFC CRF maximally impaired working memory in non-proestrus females, whereas in males a larger dose (100 ng) was necessary to elicit impairment [6]. Conversely, greater doses of intra-PFC (500 ng D-Phe-CRF) and systemic CRF antagonists (20 mg/kg NBI) were necessary to significantly improve working memory in non-proestrus females relative to males (200 ng intra-PFC D-Phe-CRF, 10 mg/kg NBI). Of potential relevance, a recent study in C57BL/6J mice demonstrated that females display greater CRF protein expression in the PFC across the lifespan compared to males [16]. Combined, these observations could suggest that females have greater levels of endogenous PFC CRF neurotransmission than males. Future studies will need to address this issue.
Anatomical and functional studies indicate the medial PFC of rodents can be divided into dorsal and ventral, as well as rostral and caudal, subfields [17,18,19]. For example, the dorsal aspect of the medial PFC, encompassing the dorsal anterior cingulate cortex and dorsal portion of the prelimbic PFC, more closely aligns with cognition-related circuitry and function while the ventromedial PFC (infralimbic PFC and ventral prelimbic PFC) is more closely associated with motivational and/or affective processes. Consistent with this, in females and/or males, we have observed working memory effects of CRF neurons and receptors in the caudal dmPFC, but not rostral or caudal ventromedial PFC [6, 7]. Of course, this oversimplifies the complexity of medial PFC structure and function, including the dorsomedial PFC [17, 20]. Future studies will need to better delineate the circuitry within the caudal dorsomedial PFC involved in CRF regulation of higher cognitive function.
Ovarian steroid regulation of CRF transmission and cognition
The protective effects of proestrus against the working memory-impairing effects of PFC CRF are similar to those observed with ICV CRF-induced impairment of sustained attention [10]. We recently demonstrated that caudal dmPFC CRF neurons also impair sustained attention in males [11]. However, unlike working memory, this action does not involve CRF receptors within the PFC [11]. Collectively, these observations suggest that the protective effects of proestrus against CRF-related impairment in PFC-dependent cognitive processes involves actions across multiple regions and circuits. PFC CRF neurons could target multiple regions beyond the PFC to influence PFC-dependent cognition, including the locus coeruleus [21] and the mediodorsal nucleus of the thalamus (MD) [22,23,24,25]. In terms of MD, recent observations in our lab demonstrate that this region receives an unusually dense projection from caudal dmPFC CRF neurons and that CRF infusions into the MD impair sustained attention. Ongoing studies are examining the role of the MD in the working memory and sustained attention actions of PFC CRF neurons. Future studies will need to definitively determine the broader projection targets of PFC CRF neurons and their roles in the regulation of PFC-dependent cognition.
Given estrogen and progesterone reach peak levels in proestrus [26], this argues for a role of one or both of these hormones in attenuating the cognition-impairing actions of CRF. Consistent with this, evidence indicates that both estrogen and progesterone can regulate the behavioral and synaptic effects of CRF neurotransmission. For example, treatment with progesterone or its metabolite, allopregnanolone, blunts CRF-induced increases in fear-potentiated startle [27]. Moreover, ovariectomized (OVX) rhesus macaques display elevated levels of CRF fibers and CRF1 receptors within the dorsal raphe relative to naturally-cycling females, and this effect is reversed with estradiol treatment [28]. It should be noted that although ovarian steroids can protect against some actions of CRF, they have also been observed to enhance CRF-induced grooming and elevate hypothalamic CRF levels [29, 30]. Thus, while ovarian steroids can modulate CRF action, the nature of this modulation is highly behavior- and neural circuit-dependent.
Currently, the mechanisms by which ovarian steroids exert protective effects against the working memory impairing actions of PFC CRF are unknown. However, the short duration of proestrus (12–18 h) may indicate they are not dependent on genomic actions at nuclear steroid receptors [31]. The membrane-associated estrogen receptors, Eα and Eβ, have been identified in the PFC of rats and non-human primates [32,33,34]. Upon activation, these receptors can initiate signal transduction by associating with metabotropic glutamate receptors (mGluRs) and regulating mGluR-dependent transmission independent of glutamate [35]. Furthermore, a third estrogen receptor, the G protein-coupled estrogen receptor 1 (also known as GPR30), is present in the rodent frontal cortex, and in vitro studies indicate this receptor can form heterodimers with CRF1 receptors at postsynaptic densities in the hippocampus [36,37,38]. Thus, ovarian steroids could modulate CRF action via direct action on CRF receptors, or indirectly via actions on CRF receptor associated 2nd messenger signaling pathways and/or on glutamatergic neurotransmission.
The current studies examined the working memory effects of PFC CRF neurotransmission in females and whether these actions are modulated across estrus cycle stages associated with low vs. high ovarian steroids. This is a critical step in better understanding both the neurobiology of PFC CRF and the potential utility of targeting CRF to treat PFC-dependent cognitive dysfunction. However, given these studies involved naturally cycling females, the degree to which the protective effects of proestrus are dependent on ovarian steroids remains unclear. For example, given the estrogen synthesizing enzyme, aromatase, is present in the PFC of adult males and females, it is possible that locally synthesized steroids could modulate the cognitive actions of CRF [39, 40]. However, whether PFC aromatase levels fluctuate across the estrus cycle is unknown. Future studies will need to definitively determine the role of ovarian steroids in proestrus-related protection against the cognition impairing effects of PFC CRF neurotransmission. While this was beyond the scope of the current studies, these observations provide a strong foundation for future research on this issue.
Translational relevance
When combined with earlier research, these studies demonstrate that systemic treatment with a CRF1 antagonist improves working memory and sustained attention in adult male and female rats outside of proestrus. The procognitive effects of these antagonists are similar to those seen with all approved treatments for ADHD, in both ADHD patients as well as healthy humans and animals [41]. Thus, CRF antagonists may be useful in the treatment of ADHD. In rodents, the CRF1 receptor predominates in the medial PFC. In contrast, CRF1 and CRF2 receptors are found within the PFC of non-human primates [42, 43]. To better understand the role of PFC CRF in higher cognitive function and to better assess the potential for CRF antagonists in treating PFC cognitive dysfunction in humans, it is important that this research be extended to non-human primates. Beyond providing guidance on the pharmacological treatment of cognitive dysfunction, these studies also suggest that dysregulated PFC CRF neurotransmission could contribute to PFC-dependent cognitive impairment. From this perspective, it is of relevance to note that there are sex differences in the incidence and phenotype of ADHD [3]. Ovarian steroid-dependent modulation of cognitive/behavioral actions of PFC CRF could contribute to these sex differences.
References
Fuster J, The prefrontal cortex. 5th ed. Academic Press: London, UK; 2015.
Piccinelli M, Wilkinson G. Gender differences in depression. Crit Rev Br J Psychiatry. 2000;177:486–92.
Ramtekkar UP, Reiersen AM, Todorov AA, Todd RD. Sex and age differences in attention-deficit/hyperactivity disorder symptoms and diagnoses: implications for DSM-V and ICD-11. J Am Acad Child Adolesc Psychiatry. 2010;49:217–28 e1-3.
Mendrek A, Mancini-Marie A. Sex/gender differences in the brain and cognition in schizophrenia. Neurosci Biobehav Rev. 2016;67:57–78.
Setlik J, Bond GR, Ho M. Adolescent prescription ADHD medication abuse is rising along with prescriptions for these medications. Pediatrics 2009;124:874–80.
Hupalo S, Berridge CW. Working memory impairing actions of Corticotropin-Releasing Factor (CRF) neurotransmission in the prefrontal cortex. Neuropsychopharmacology 2016;41:2733–40.
Hupalo S, Martin AJ, Green RK, Devilbiss DM, Berridge CW. Prefrontal Corticotropin-Releasing Factor (CRF) neurons act locally to modulate frontostriatal cognition and circuit function. J Neurosci. 2019;39:2080–90.
Bangasser DA, Eck SR, Ordones Sanchez E. Sex differences in stress reactivity in arousal and attention systems. Neuropsychopharmacology 2019;44:129–39.
Hupalo S, Bryce CA, Bangasser DA, Berridge CW, Valentino RJ, Floresco SB. Corticotropin-Releasing Factor (CRF) circuit modulation of cognition and motivation. Neurosci Biobehav Rev. 2019;103:50–50.
Cole RD, Kawasumi Y, Parikh V, Bangasser DA. Corticotropin releasing factor impairs sustained attention in male and female rats. Behav Brain Res. 2016;296:30–4.
Hupalo S, Spencer RC, Berridge CW. Prefrontal corticotropin-releasing factor neurons impair sustained attention via distal transmitter release. Eur J Neurosci. 2021;54:4182–96.
Becker JB, Arnold AP, Berkley KJ. Strategies and methods for research on sex differences in brain and behavior. Endocrinology 2006;146:1660–73.
Schmeichel B, Zemlan F, Berridge CW. A selective dopamine reuptake inhibitor imporves prefrontal cortex-dependent cognitive function: potential relevant to attention deficit hyperactivity disorder. Neuropharmacology 2012;64:321–28.
Hubscher CH, Brooks DL, Johnson JR. A quantitative method for assessing stages of the rat estrous cycle. Biotech Histochem. 2005;80:79–87.
Cora MC, Kooistra L, Travlos G. Vaginal cytology of the laboratory rat and mouse: review and criteria for the staging of the estrous cycle using stained vaginal smears. Toxicol Pathol. 2015;43:776–93.
Locci A, Yan Y, Rodriguez G, Dong H. Sex differences in CRF1, CRF, and CRFBP expression in C57BL/6J mouse brain across the lifespan and in response to acute stress. J Neurochem. 2021;158:943–59.
Gabbott PL, Warner TA, Jays PR, Salway P, Busby SJ. Prefrontal cortex in the rat: projections to subcortical autonomic, motor, and limbic centers. J Comp Neurol. 2005;492:145–77.
Voorn P, Vanderschuren LJ, Groenewegen HJ, Robbins TW, Pennartz CM. Putting a spin on the dorsal-ventral divide of the striatum. Trends Neurosci. 2004;27:468–74.
van Holstein M, Floresco SB. Dissociable roles for the ventral and dorsal medial prefrontal cortex in cue-guided risk/reward decision making. Neuropsychopharmacology 2020;45:683–93.
Bennett PJG, Maier E, Brecht M. Involvement of rat posterior prelimbic and cingulate area 2 in vocalization control. Eur J Neurosci. 2019;50:3164–80.
Spencer RC, Berridge CW. Receptor and circuit mechanisms underlying differential procognitive actions of psychostimulants. Neuropsychopharmacology 2019;44:1820–27.
Parnaudeau S, Bolkan SS, Kellendonk C. The mediodorsal thalamus: an essential partner of the prefrontal cortex for cognition. Biol Psychiatry. 2018;83:648–56.
Schmitt LI, Wimmer RD, Nakajima M, Happ M, Mofakham S, Halassa MM. Thalamic amplification of cortical connectivity sustains attentional control. Nature 2017;545:219–23.
Rikhye RV, Gilra A, Halassa MM. Thalamic regulation of switching between cortical representations enables cognitive flexibility. Nat Neurosci. 2018;21:1753–63.
Mantanona CP, Bozic T, Chudasama Y, Robbins TW, Dalley JW, Alsio J, et al. Dissociable contributions of mediodorsal and anterior thalamic nuclei in visual attentional performance: A comparison using nicotinic and muscarinic cholinergic receptor antagonists. J Psychopharm. 2020;34:1371–81.
Goldman JM, Murr AS, Cooper RL. The rodent estrous cycle: characterization of vaginal cytology and its utility in toxicological studies. Birth Defects Res B Dev Reprod Toxicol. 2007;80:84–97.
Toufexis DJ, Davis C, Hammond A, Davis M. Progesterone attenuates corticotropin-releasing factor-enhanced but not fear-potentiated startle via the activity of its neuroactive metabolite, allopregnanolone. J Neurosci. 2004;24:10280–7.
Sanchez RL, Reddy AP, Bethea CL. Ovarian steroid regulation of the midbrain corticotropin releasing factor and urocortin systems in macaques. Neuroscience 2010;171:893–909.
Viau V, Bingham B, Davis J, Lee P, Wong M. Gender and puberty interact on the stress-induced activation of parvocellular neurosecretory neurons and corticotropin-releasing hormone messenger ribonucleic acid expression in the rat. Endocrinology 2005;146:137–46.
Bangasser DA, Wiersielis KR. Sex differences in stress responses: a critical role for corticotropin-releasing factor. Hormones 2018;17:5–13.
Hazell GG, Yao ST, Roper JA, Prossnitz ER, O’Carroll AM, Lolait SJ. Localisation of GPR30, a novel G protein-coupled oestrogen receptor, suggests multiple functions in rodent brain and peripheral tissues. J Endocrinol. 2009;202:223–36.
Wang J, Cheng CM, Zhou J, Smith A, Weickert CS, Perlman WR, et al. Estradiol alters transcription factor gene expression in primate prefrontal cortex. J Neurosci Res. 2004;76:306–14.
Montague D, Weickert CS, Tomaskovic-Crook E, Rothmond DA, Kleinman JE, Rubinow DR. Oestrogen receptor alpha localisation in the prefrontal cortex of three mammalian species. J Neuroendocrinol. 2008;20:893–903.
Almey A, Cannell E, Bertram K, Filardo E, Milner TA, Brake WG. Medial prefrontal cortical estradiol rapidly alters memory system bias in female rats: ultrastructural analysis reveals membrane-associated estrogen receptors as potential mediators. Endocrinology 2014;155:4422–32.
Meitzen J, Mermelstein PG. Estrogen receptors stimulate brain region-specific metabotropic glutamate receptors to rapidly initiate signal transduction pathways. J Chem Neuroanat. 2011;42:236–41.
Prossnitz ER, Arterburn JB, Smith HO, Oprea TI, Sklar LA, Hathaway HJ. Estrogen signaling through the transmembrane G protein-coupled receptor GPR30. Annu Rev Physiol. 2008;70:165–90.
Hammond R, Nelson D, Gibbs RB. GPR30 co-localizes with cholinergic neurons in the basal forebrain and enhances potassium-stimulated acetylcholine release in the hippocampus. Psychoneuroendocrinology 2011;36:182–92.
Akama KT, Thompson LI, Milner TA, McEwen BS. Post-synaptic density-95 (PSD-95) binding capacity of G-protein-coupled receptor 30 (GPR30), an estrogen receptor that can be identified in hippocampal dendritic spines. J Biol Chem. 2013;288:6438–50.
Hojo Y, Hattori TA, Enami T, Furukawa A, Suzuki K, Ishii HT, et al. Adult male rat hippocampus synthesizes estradiol from pregnenolone by cytochromes P45017alpha and P450 aromatase localized in neurons. Proc Natl Acad Sci USA. 2004;101:865–70.
Barker JM, Galea LA. Sex and regional differences in estradiol content in the prefrontal cortex, amygdala, and hippocampus of adult male and female rats. Gen Comp Endocrinol. 2009;164:77–84.
Spencer RC, Devilbiss DM, Berridge CW. The cognition-enhancing effects of psychostimulants involve direct action in the prefrontal cortex. Biol Psychiatry. 2015;77:940–50.
Sanchez MM, Young LJ, Plotsky PM, Insel TR. Autoradiographic and in situ hybridization localization of corticotropin-releasing factor 1 and 2 receptors in nonhuman primate brain. J Comp Neurol. 1999;408:365–77.
Van Pett K, Viau V, Bittencourt JC, Chan RK, Li HY, Arias C, et al. Distribution of mRNAs encoding CRF receptors in brain and pituitary of rat and mouse. J Comp Neurol. 2000;428:191–212.
Funding
This work was supported by the National Institutes of Health grant MH116526 and the Office of the Vice Chancellor for Research and Graduate Education at the University of Wisconsin-Madison with funding from the Wisconsin Alumni Research Foundation.
Author information
Authors and Affiliations
Contributions
CWB assisted in study design, oversight of data collection, data analysis and, preparation and editing of the manuscript. AJM assisted in study design, data collection, data analysis, and, preparation and editing of the manuscript. SH assisted in preparation and editing of the manuscript. SN assisted in data collection and preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Berridge, C.W., Martin, A.J., Hupalo, S. et al. Estrus cycle-dependent working memory effects of prefrontal cortex corticotropin-releasing factor neurotransmission. Neuropsychopharmacol. 47, 2016–2023 (2022). https://doi.org/10.1038/s41386-022-01349-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-022-01349-7