Abstract
Mismatch repair (MMR) deficiency is an indicator of good prognosis in localized colon cancer but also associated with lack of expression of caudal-type homeobox transcription factor 2 (CDX2) and high tumor grade; markers that in isolation indicate a poor prognosis. Our study aims to identify clinically relevant prognostic subgroups by combining information about tumor grade, MMR phenotype, and CDX2 expression. Immunohistochemistry for MMR proteins and CDX2 was performed in 544 patients with colon cancer stage II–III, including a cohort from a randomized trial. In patients with proficient MMR (pMMR) and CDX2 negativity, hazard ratio (HR) for cancer death was 2.93 (95% CI 1.23–6.99, p = 0.015). Cancer-specific survival for pMMR/CDX2-negative cases was 35.8 months (95% CI 23.4–48.3) versus 52.1–53.5 months (95% CI 45.6–58.6, p = 0.001) for the remaining cases (CDX2-positive tumors or deficient MMR (dMMR)/CDX2-negative tumors). In our randomized cohort, high tumor grade was predictive of response to adjuvant fluorouracil–levamisole in pMMR patients, with a significant interaction between tumor grade and treatment (p = 0.036). For pMMR patients, high tumor grade was a significant marker of poor prognosis in the surgery-only group (HR 4.60 (95% CI 1.68–12.61), p = 0.003) but not in the group receiving chemotherapy (HR 0.66 (95% CI 0.15–3.00), p = 0.587). To conclude, patients with pMMR and CDX2 negativity have a very poor prognosis. Patients with pMMR and high-graded tumors have a poor prognosis but respond well to adjuvant chemotherapy. CDX2 expression and tumor grade did not impact prognosis in patients with dMMR.
Similar content being viewed by others
Introduction
Colorectal cancer is the second most common cause of cancer death worldwide [1]. To reduce the risk of relapse after colon cancer surgery, adjuvant chemotherapy is offered to patients with stage III disease and to high-risk patients with stage II disease [2]. Still, the definition of high-risk colon cancer remains controversial [3, 4]. Biomarkers identifying patients with a high probability of relapse or patients with tumors sensitive to adjuvant chemotherapy could significantly improve treatment stratification across cancer stage [5, 6].
Microsatellite instability (MSI) is a manifestation of defective DNA mismatch repair (dMMR) and is found in 15–20% of patients with localized colon cancer [7,8,9]. It is caused by a germline mutation in one or more of the MMR genes, double somatic MMR gene inactivation or a somatic hypermethylation of the MLH-1 gene promoter [8,9,10,11]. The high number of mutations associated with dMMR leads to expression of cell membrane neo-antigens. This often facilitates a strong anti-tumor immune response [6, 9], contributing to the low risk of relapse for dMMR patients with localized disease [7, 9]. In addition, treatment with fluorouracil (5-FU) yields no survival benefit in patients with stage II colon cancer and dMMR [12, 13]. Therefore, stage II colon cancer patients with dMMR are usually not eligible for adjuvant chemotherapy [2]. In metastatic colorectal cancer, dMMR predicts response to anti PD-1 immunotherapy and an studies suggest an improved prognosis for cases with concurrent BRAF-wild type [11].
However, dMMR is also associated with markers that in isolation have a negative impact on prognosis, such as high tumor grade and lack of expression of cell maturation marker caudal-type homeobox transcription factor 2 (CDX2) [14,15,16,17]. CDX2 is an emerging biomarker in colon cancer [16, 18, 19]. This nuclear transcription factor regulates proliferation and differentiation of intestinal epithelial cells and is a diagnostic biomarker for mature colon epithelial tissue [20, 21]. CDX2 immunohistochemistry (IHC) is integrated in the clinical diagnostics of gastrointestinal adenocarcinomas in cancers of unknown origin [8, 22]. In the pivotal study by Dalerba et al., lack of CDX2 expression identified a high-risk subgroup of localized colon cancer patients. The authors also proposed that CDX2-negative stage II patients benefit from adjuvant chemotherapy [16].
Histological grading is based on the degree of tumor glandular formation. High tumor grade is found in ~20% of colon adenocarcinomas and is considered a high-risk trait in stage II colon cancer and a marker of poor prognosis [2, 8, 15]. The predictive role of high tumor grade on chemotherapy effect in colon cancer is not established. Tumors with dMMR are detected in several morphological subtypes but overrepresented in mucinous adenocarcinomas, medullary carcinomas, and colon adenocarcinoma NOS (not otherwise specified) with high-grade morphology [7, 11, 17]. Central studies challenge the role of histological grading in the definition of high-risk localized colon cancer [23, 24], but the prognostic effect of high tumor grade may be confounded when not incorporating MMR phenotype in the assessment [25]. Rosty et al. propose that for colon adenocarcinoma NOS, the positive prognostic impact of dMMR might outweigh the negative impact of high tumor grade on survival [25].
Prognostic assessments for patients with tumors featuring dMMR, CDX2 negativity and high-grade morphology are conflicting, despite the large overlap between these traits. To choose the optimal treatment for these patients, the impact of MMR phenotype on the prognosis of CDX2-negative and on high-grade tumors needs to be established.
Our study aims to assess the prognostic value of MMR phenotype in CDX2-negative and/or high-grade colon cancer. We hypothesize that by combining information about tumor grading, CDX2 expression and MMR phenotype, we can identify clinically relevant prognostic subgroups. This report also assesses the impact of CDX2 negativity, MMR phenotype, and tumor grading on adjuvant chemotherapy effect in our randomized controlled cohort.
Material and methods
Study cohort
The study cohort originates from two different patient materials. For material 1 (the Norwegian Gastrointestinal Cancer Group (NGICG) cohort), tissue from primary tumors was obtained from patients in a randomized controlled trial previously described by Dahl et al. [26]. In brief, 425 patients with colon or rectal cancer stage II and III were included between 1993 and 1996 and randomized to treatment with 5-FU and levamisole after surgery or to surgery only. Material 2 is a population-based cohort consisting of 374 patients undergoing complete (D3) mesocolic excision for stage I–IV colon cancer at Haraldsplass Deaconess Hospital (HDH) [27, 28]. Patients were included between January 2007 and December 2011. Thorough 5-year follow-up data and clinicopathological data were available for both materials [26, 27]. For the current study, we included cases from both materials with colon cancer stage II or III, 544 patients in total (276 patients from the NGICG cohort and 268 from the HDH cohort).
Immunohistochemistry
Tissue microarrays (TMAs) with 1 mm core diameter were constructed from formalin fixed paraffin embedded tissue in sections of 3–5 μm. CDX2 staining was performed using the diagnostic protocol on a Ventana BenchMark Ultra. After deparaffinization and rehydration, target retrieval was done with CC1 reagent (Ventana Medical systems) for 48 min. For the primary antibody (CDX2 clone EPR2764Y, Cell Marque, anti-rabbit monoclonal, 1:200 dilution), incubation time was 32 min. OptiView amplifier was used (4 min), slides were counterstained with hematoxylin, dehydrated, and mounted. Positive controls included tonsil, appendix, and pancreas. MMR protein staining for MLH-1 (1:60, DAKO, M3640, clone ES05), MSH2 (1:300, Biocare Medical, CM219B, clone FE11), MSH6 (1:50, DAKO, M3646, clone EP49), and PMS2 (1:50, DAKO, M3647, clone EP51) was performed as previously described [28]. Epitope retrieval (HIER) was done in pH9 TE-buffer and the results visualized using the MACH3 HRP-Polymer (Biocare Medical) detection kit.
Analysis of tissue microarrays
Two of the authors (KEH and KAA), blinded from other patient data, scored each tissue core for percentage of CDX2-positive tumor cells. Staining intensity was also recorded. Scoring details are included in Supplementary Material 3. The TMAs included 1–3 cores for each patient. For cases with intratumoral heterogeneity, we estimated the average CDX2 expression. In accordance with Dalerba et al., cases were divided into four groups: a: no staining (0–5% positive cells), b: weak/scattered staining in a minority of cells (5–49% positive cells), c: moderate/strong staining in a majority of cells (50–95% positive cells), d: strong staining in all cells (95–100% positive cells). Tumors in category A and B were defined as CDX2 negative. As weak staining is a known diagnostic pitfall in CDX2 IHC [29] cases with weak staining in the majority of cells were regarded CDX2-positive (category c). This applied to one case only.
MMR protein expression was assessed by KEH, YM, NBR, and MPM. Scoring details are found in Supplementary Material 3. Loss of expression was defined as less than 5% positive tumor cells in the presence of retained expression in internal control cells (stromal cells and/or normal colon epithelium of the same tissue core), in ordinance with other studies [30]. Cases with negative staining in tumor cells and negative staining of internal control cells were omitted from analysis. There is no consensus on how to interpret equivocal MMR IHC staining patterns as this group includes cases both with and without MMR germline mutation [31]. We therefore decided to exclude cases with equivocal MMR-staining patterns. We categorized patients as MMR deficient if staining was absent for MLH-1 + PMS2, PMS2 alone, MSH2 + MSH6, or MSH6 alone.
Validation in whole tissue sections
Whole tissue sections from the HDH cohort were used to validate the CDX2 and MMR IHC (Supplementary Material 3) and to assess heterogeneity in MMR protein expression [32, 33].
Tumor grading
We recorded information about tumor grading from the original patient pathology reports. We used a two-tiered grading system (low versus high grade) in order to reduce interobserver variation [8].
Statistics
Differences in CDX2 expression and tumor grade between subgroups were tested using Pearson’s chi square test and Student’s t test. We used Cancer-specific survival (CSS) as outcome variable, defined as time from randomization until death of colon cancer or death caused by treatment complications. The effect of CDX2 expression and tumor grade on CSS in different subgroups was assessed using Kaplan–Meier curves with log rank test and the multivariate Cox proportional hazards model. We adjusted for clinicopathological variables known to be relevant in colon cancer using the enter method. All p values were two-sided and regarded statistically significant if <0.05. As we consider our study exploratory, we made no adjustments for multiple comparisons. Statistical analyses were performed in IBM SPSS Statistics for Windows (v25.0) and R [34].
Results
Patient characteristics, tumor grade, CDX2 expression, and MMR phenotype
The study cohort consists of 544 patients with stage II–III colon cancer (Table 1). CDX2 staining was successful for 443 patients: 23 (5.2%) had no staining, 18 (4.1%) had weak/scattered staining in a minority of cells, and 56 (12.6%) had moderate/strong staining in a majority of cells (Fig. 1). Strong staining in all cells was registered for 346 tumors (78.1%) and for the 308 (100%) corresponding samples with normal intestinal epithelium. Interobserver agreement between the four categories were 89, and 99% between positive and negative cases. CDX2 negativity (41 patients, 9.3%) was significantly associated with right sided colon cancer, dMMR and high tumor grade (Supplementary Table 1).
Validation in whole tissue sections (Supplementary Material 3) showed a satisfactory compliance between TMA and whole tissue CDX2 IHC. Discrepant results represented cases with scoring values close to the chosen cut-off of 50% positive cells.
Proficient mismatch repair (pMMR) was detected in 377 patients (69.3%) and dMMR in 105 (19.3%) patients. Out of the 479 patients (88.1%) with colon adenocarcinoma NOS, 406 patients (84.8%) had low-grade tumors and 71 patients (14.8%) had high-grade tumors. High tumor grade was associated with dMMR, CDX2 negativity and right sided colon cancer (Supplementary Table 1). The MMR phenotyping based on the whole tissue sections IHC complied with the MMR phenotyping based on the TMA IHC. However, we found two cases with different staining results; one case had heterogenous MLH-1-staining pattern in the whole tissue section and retained MLH-1 expression in the TMA. It was regarded as having retained MMR expression due to retained PMS2. Another case was regarded as retained MSH2 expression in TMA but lost expression in whole tissue section. This did not result in changes in MMR phenotype as this case had loss of expression for MSH6 in both TMA and the whole tissue section.
To illustrate the large overlap between dMMR, CDX2 negativity and high tumor grade, an Euler diagram was made for patients with colon adenocarcinoma NOS and valid staining for both MMR and CDX2 (Fig. 2).
CDX2 expression status could not be determined in 101 (18.6%) cases due to technical issues, e.g., few tumor cells in tissue cores, tissue core detached from slide during staining or necrosis in tissue core. MMR phenotype could not be determined in 62 cases (11.4%) due to the mentioned technical issues, equivocal staining patterns or absence of staining in normal tissue used as internal positive control. Our goodness of fit analyses showed no significantly different distribution of clinicopathological variables between cases with successful CDX2- or MMR staining and the original study cohort (Supplementary Table 2).
Patients with pMMR and CDX2 negativity have a very poor prognosis
In Kaplan–Meier analyses with log rank test, there was no statistically significant difference in CSS between patients with dMMR and pMMR (p = 0.333, Supplementary Fig. 1A). Patients with CDX2-negative tumors had a significantly shorter 5-year CSS than patients with CDX2-positive tumors (47.8 (95% confidence interval (CI) 41.7–53.9) versus 53.6 (95% CI 52.1–55.1) months, p = 0.024) (Supplementary Fig. 1B). CDX2 negativity was not an independent adverse prognostic marker in multivariate Cox regression analyses when analyzing the whole study cohort (hazard ratio (HR) 1.70, 95% CI 0.83–3.49, p = 0.145) (Table 2).
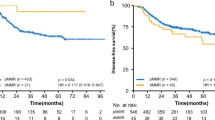
When considering MMR phenotype and histology in addition to CDX2 status, we identified subgroups with high risk of death from colon cancer. CDX2 negativity was a strong marker of poor prognosis in our multivariate models in patients with pMMR only, (HR 2.93, 95% CI 1.23–6.99, p = 0.015), patients with colon adenocarcinoma NOS histology (HR 3.03, 95% CI 1.34–6.88, p = 0.008) and patients with colon adenocarcinoma NOS + pMMR (HR 5.18, 95% CI 2.25–11.90, p < 0.001) (Table 2), with HRs surpassing UJCC stage III versus II in the latter group. Patients with pMMR and CDX2 negativity had a 5-year mean CSS of 35.8 months (95% CI 23.4–48.3) compared to 52.1–53.5 months (95% CI 45.6–58.6) (p = 0.001) for patients with CDX2 positive tumors or dMMR/CDX2-negative tumors (Fig. 3). CDX2 expression did not affect CSS in the dMMR subgroup (p = 0.501). The predictive value of CDX2 expression was analyzed in our randomized cohort (NGICG). CDX2 negativity did not predict effect of adjuvant chemotherapy in our uni- or multivariate models, but due to lack of statistical power of the CDX2-negative subgroup this analysis is considered explorative.
MMR phenotype directs the influence of high tumor grade on prognosis and effect of adjuvant chemotherapy for patients with adenocarcinoma NOS
In this section, the predictive and prognostic impact of tumor grade will be discussed for patients with adenocarcinoma NOS only to eliminate bias introduced by cases with other colon cancer histology.
Overall, patients with high tumor grade had a worse 5-year CSS than patients with low tumor grade (48.6 months (95% CI 44.4–52.8) versus 54.6 months (95% CI 53.2–56.0) p = 0.002). (Supplementary Fig. 1C). When separating patients into categories based on grade and MMR phenotype, the group with pMMR and high-grade tumors had a poor prognosis compared to the other groups (CSS 43.7 months (95% CI 37.3–50.0) versus 54.2–55.4 months (95% CI 50.3–60.0), p < 0.001) (Fig. 4).
In multivariate Cox regression analyses, high tumor grade was an independent negative prognostic factor in colon adenocarcinoma NOS (pMMR and dMMR) but with a higher HR for the colon adenocarcinoma NOS and pMMR subgroup (Table 2).
To avoid patient selection bias, the predictive effect of tumor grade was assessed in our randomized cohort only. In the group randomized to receive surgery only, there was a large difference in mean 5-year CSS between pMMR patients with high-grade tumors versus pMMR patients with low-grade tumors (CSS 30.2 months (95% CI 19.5–41.0) versus 54.6 months (95% CI 51.6–57.6)). In contrast, in the group randomized to adjuvant chemotherapy, there was no statistically significant difference in CSS between these groups (Fig. 5). As tumor grade did not affect benefit of chemotherapy in the dMMR group (p = 0.947), a split multivariate Cox regression model with treatment × tumor grade interaction was used as a predictive model for pMMR patients (Table 3). There was a significant interaction between tumor grade and treatment (p = 0.036). High tumor grade was a significant marker of poor prognosis in the surgery-only group (HR 4.60 (95% CI 1.68–12.6), p = 0.003) but not in the group receiving chemotherapy (HR 0.66 (95% CI 0.15–3.00), p = 0.587). The results from our Kaplan–Meier curves and multivariate models indicate that although high tumor grade is a marker of poor prognosis in pMMR patients, these tumors respond well to adjuvant 5-FU-based chemotherapy.
Discussion
In this study, we have assessed the prognostic and predictive impact of CDX2 expression and tumor grade in the context of MMR phenotype. We observe that patients with tumors that are both pMMR and CDX2 negative have a particularly poor prognosis. Our study also shows that pMMR combined with high-grade morphology indicates a particularly poor prognosis in colon adenocarcinoma NOS treated with surgery alone. Still, this group of patients respond very well to adjuvant 5-FU-based chemotherapy.
The frequency of CDX2 negativity in our material (9.3%) is in line with the reported rate (4–15%) in localized colon cancer [14, 16, 17, 35,36,37,38,39]. Loss of CDX2 expression may be a result of epigenetic silencing by CpG island promoter hypermethylation [19, 38, 40, 41], a mechanism also known to cause dMMR by silencing of MLH-1 [9, 40]. This might explain the large overlap between CDX2 negativity and dMMR. This observation is supported by previous studies: 45–81% of CDX2-negative cases have dMMR, and 25–41% of dMMR cases are CDX2 negative [14, 17, 36, 37, 39, 42].
In our study, CDX2 negativity was an adverse prognostic marker for patients with pMMR tumors. Few other studies have differentiated between pMMR and dMMR tumors when assessing the effect of CDX2 negativity on prognosis and we propose that not stratifying may understate the prognostic impact of CDX2 negativity in previous studies [14, 43]. Pilati et al. studied the relation between CDX2 negativity and the consensus molecular subtype (CMS) classification. CDX2 negativity was an adverse prognostic marker in the CMS4 group and for cases with MSS but not in the CMS1 group or for cases with MSI [35]. Other studies also support our findings; Lugli et al. demonstrated an association between CDX2 negativity and advanced tumor stage in cases with pMMR but not for cases with dMMR. Yet, they were unable to demonstrate any prognostic value for CDX2 negativity in multivariate analyses, this may be caused by not including MMR phenotype in their multivariate models [22]. Ryan et al. studied dMMR tumors only and conclude that CDX2 negativity is not a prognostic marker in this group [44]. Slik et al. demonstrate a higher frequency of death from CRC for patients with CDX2 negativity and MSS compared to patients with CDX2 negativity and MSI.
In contrast to our results, Ma et al. report a negative prognostic impact of SATB2 and CDX2 in patients with dMMR but not pMMR [42]. However, independent prognostic value was only reported for the combined SATB2/CDX2 marker, not for CDX2 negativity alone. Kim et al. studied 109 patients with MSI-H colorectal cancer. The combination of CDX2 negativity and CK 20 loss indicated a poor prognosis, but CDX2 negativity alone was not an independent prognostic marker in multivariate analyses [40].
In our study, the prognostic value of CDX2 expression was analyzed in the joined patient material (NGICG and HDH). The two groups have similar clinicopathological data including fraction of dMMR, high tumor grade and CDX2 negativity. The indications for adjuvant chemotherapy changed between the time of including patients in these two different studies and this probably explains the statistically significant difference in survival between the two hospitals. We have adjusted for differences in survival between the cohorts in the multivariate model (variable “hospital”, Table 2). The number of CDX2-negative cases is in line with other studies but the number of cases with aberrant CDX2 expression is low. This calls for caution when interpreting our findings and validation in larger patient cohorts.
Despite the high incidence of dMMR tumors in localized colon cancer with high-grade morphology [7, 17], high tumor grade is regarded a negative prognostic factor [2]. Rosty et. al found that the negative prognostic impact of high tumor grade was restricted to MSS tumors and proposed that the grading of colon adenocarcinoma should be made in accordance with MSI status [25]. In our study, patients with pMMR and high-grade morphology had a very poor prognosis, but high-grade morphology did not influence the prognosis of dMMR patients, supporting these findings. The eUpdate of the ESMO guidelines also emphasizes the importance of assessing established high-risk factors in the context of MMR/MSI phenotype. Here, MSS, and either T4 or more than one validated risk factor (number of examined lymph nodes <12, primary tumor perforation or occlusion, high tumor grade), constitute the newly defined “very high risk” group in colon cancer stage II [4].
Liu et al. reported that stage IIA colon cancer patients with poorly differentiated or undifferentiated tumors benefit from adjuvant chemotherapy [45], in accordance with our results for stage II–III pMMR patients. However, they assessed high-grade adenocarcinomas together with tumors with other histology (undifferentiated tumors) and did not stratify by MSI/MMR phenotype.
The randomized design of the NGICG study offers unique opportunities to study markers that predict effect of adjuvant chemotherapy. We therefore assessed predictive markers in this material only. In our study, a predictive effect of high tumor grade was demonstrated when analyzing stage II and III colon cancer together, but due to the limited number of events in stage II colon cancer in the randomized cohort, our study was underpowered to show this effect in the colon cancer II subgroup alone. The adjuvant chemotherapy in the randomized study consisted of 5-FU-levamisol. Today, 5-FU or capecitabine in combination with oxaliplatin is the standard regimen for adjuvant chemotherapy in stage III CRC in Europe, but as most of the survival effect of the XELOX/FLOX/FOLFOX treatment can be attributed to the fluorouracil component [46], we expect our results to be applicable to these combinations as well.
This study emphasizes the importance of assessing CDX2 expression and tumor grade in the context of MMR phenotype. We propose that CDX2, together with MSI or MMR phenotype, should be implemented in the routine diagnostics of colon cancer stage II–III. Patients with pMMR and CDX2 negativity represent a high-risk group irrespective of cancer stage. We therefore suggest that adjuvant chemotherapy is considered for pMMR CDX2-negative stage II patients.
Our study is, to our knowledge, the first study to assess the predictive impact of high tumor grade on effect of adjuvant chemotherapy in pMMR patients in a randomized trial. We demonstrate that although patients with pMMR and high tumor grade have an inferior prognosis after surgery alone, they seem to respond very well to adjuvant chemotherapy.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Labianca R, Nordlinger B, Beretta GD, Mosconi S, Mandala M, Cervantes A, et al. Early colon cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24 Suppl 6:vi64–72.
Babcock BD, Aljehani MA, Jabo B, Choi AH, Morgan JW, Selleck MJ, et al. High-risk stage II colon cancer: not all risks are created equal. Ann Surg Oncol. 2018;25:1980–5.
eUpdate- Early Colon Cancer Treatment Recommendations, [Internet], ESMO Guidelines Committee, Lugano, Switzerland, 2019 [cited 2 January 2020]. Available from https://www.esmo.org/guidelines/gastrointestinal-cancers/localised-colon-cancer/eupdate-early-colon-cancer-treatment-recommendations.
Gray R, Barnwell J, McConkey C, Hills RK, Williams NS, Kerr DJ. Adjuvant chemotherapy versus observation in patients with colorectal cancer: a randomised study. Lancet. 2007;370:2020–9.
Guinney J, Dienstmann R, Wang X, de Reynies A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015;21:1350–6.
Benatti P, Gafa R, Barana D, Marino M, Scarselli A, Pedroni M, et al. Microsatellite instability and colorectal cancer prognosis. Clin Cancer Res. 2005;11:8332–40.
Fleming M, Ravula S, Tatishchev SF, Wang HL. Colorectal carcinoma: pathologic aspects. J Gastrointest Oncol. 2012;3:153–73.
Boland CR, Goel A. Microsatellite instability in colorectal cancer. Gastroenterology. 2010;138:2073–87.e3.
Wang T, Lee LH, Vyas M, Zhang L, Ganesh K, Firat C, et al. Colorectal carcinoma with double somatic mismatch repair gene inactivation: clinical and pathological characteristics and response to immune checkpoint blockade. Mod Pathol. 2019;32:1551–62.
WHO Classification of Tumors Editorial Board. Digestive system tumors. 5th ed. Lyon: International Agency for Research on Cancer; 2019.
Sargent DJ, Marsoni S, Monges G, Thibodeau SN, Labianca R, Hamilton SR, et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J Clin Oncol. 2010;28:3219–26.
Koenig JL, Toesca DAS, Harris JP, Tsai CJ, Haraldsdottir S, Lin AY, et al. Microsatellite instability and adjuvant chemotherapy in stage II colon cancer. Am J Clin Oncol. 2019;42:573–80.
Hansen TF, Kjaer-Frifeldt S, Eriksen AC, Lindebjerg J, Jensen LH, Sorensen FB, et al. Prognostic impact of CDX2 in stage II colon cancer: results from two nationwide cohorts. Br J Cancer. 2018;119:1367–73.
Greene FL, Stewart AK, Norton HJ. A new TNM staging strategy for node-positive (stage III) colon cancer: an analysis of 50,042 patients. Ann Surg. 2002;236:416–21.
Dalerba P, Sahoo D, Paik S, Guo X, Yothers G, Song N, et al. CDX2 as a prognostic biomarker in stage II and stage III colon cancer. N Engl J Med. 2016;374:211–22.
Romiti A, Roberto M, Marchetti P, Di Cerbo A, Falcone R, Campisi G, et al. Study of histopathologic parameters to define the prognosis of stage II colon cancer. Int J Colorectal Dis. 2019;34:905–13.
Bruun J, Sveen A, Barros R, Eide PW, Eilertsen I, Kolberg M, et al. Prognostic, predictive, and pharmacogenomic assessments of CDX2 refine stratification of colorectal cancer. Mol Oncol. 2018;12:1639–55.
Graule J, Uth K, Fischer E, Centeno I, Galvan JA, Eichmann M, et al. CDX2 in colorectal cancer is an independent prognostic factor and regulated by promoter methylation and histone deacetylation in tumors of the serrated pathway. Clin Epigenetics. 2018;10:120.
Suh E, Traber PG. An intestine-specific homeobox gene regulates proliferation and differentiation. Mol Cell Biol. 1996;16:619–25.
Werling RW, Yaziji H, Bacchi CE, Gown AM. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. Am J Surg Pathol. 2003;27:303–10.
Lugli A, Tzankov A, Zlobec I, Terracciano LM. Differential diagnostic and functional role of the multi-marker phenotype CDX2/CK20/CK7 in colorectal cancer stratified by mismatch repair status. Mod Pathol. 2008;21:1403–12.
Gill S, Loprinzi CL, Sargent DJ, Thome SD, Alberts SR, Haller DG, et al. Pooled analysis of fluorouracil-based adjuvant therapy for stage II and III colon cancer: who benefits and by how much? J Clin Oncol. 2004;22:1797–806.
Jessup JM, Stewart A, Greene FL, Minsky BD. Adjuvant chemotherapy for stage III colon cancer: implications of race/ethnicity, age, and differentiation. JAMA. 2005;294:2703–11.
Rosty C, Williamson EJ, Clendenning M, Walters RJ, Win AK, Jenkins MA, et al. Should the grading of colorectal adenocarcinoma include microsatellite instability status? Hum Pathol. 2014;45:2077–84.
Dahl O, Fluge O, Carlsen E, Wiig JN, Myrvold HE, Vonen B, et al. Final results of a randomised phase III study on adjuvant chemotherapy with 5 FU and levamisol in colon and rectum cancer stage II and III by the Norwegian Gastrointestinal Cancer Group. Acta Oncol. 2009;48:368–76.
Stanisavljevic L, Assmus J, Storli KE, Leh SM, Dahl O, Myklebust MP. CXCR4, CXCL12 and the relative CXCL12-CXCR4 expression as prognostic factors in colon cancer. Tumour Biol. 2016;37:7441–52.
Jacob H, Stanisavljevic L, Storli KE, Hestetun KE, Dahl O, Myklebust MP. Identification of a sixteen-microRNA signature as prognostic biomarker for stage II and III colon cancer. Oncotarget. 2017;8:87837–47.
NordiQC: assessments and recommended protocols, [Internet], Nordic immunohistochemical Quality Control (NordiQC), Aalborg, Denmark 2016 [cited 13 June 2020]. Available from https://www.nordiqc.org/downloads/assessments/83_39.pdf.
Chen W, Frankel WL. A practical guide to biomarkers for the evaluation of colorectal cancer. Mod Pathol. 2019;32 Suppl 1:1–15.
Sarode VR, Robinson L. Screening for Lynch syndrome by immunohistochemistry of mismatch repair proteins: significance of indeterminate result and correlation with mutational studies. Arch Pathol Lab Med. 2019;143:1225–33.
Joost P, Veurink N, Holck S, Klarskov L, Bojesen A, Harbo M, et al. Heterogenous mismatch-repair status in colorectal cancer. Diagn Pathol. 2014;9:126.
McCarthy AJ, Capo-Chichi JM, Spence T, Grenier S, Stockley T, Kamel-Reid S, et al. Heterogenous loss of mismatch repair (MMR) protein expression: a challenge for immunohistochemical interpretation and microsatellite instability (MSI) evaluation. J Pathol Clin Res. 2019;5:115–29.
Team RDC. R: a language and environment for statistical computing. Vienna: Team RDC; 2017.
Pilati C, Taieb J, Balogoun R, Marisa L, de Reynies A, Laurent-Puig P. CDX2 prognostic value in stage II/III resected colon cancer is related to CMS classification. Ann Oncol. 2017;28:1032–5.
Olsen J, Eiholm S, Kirkeby LT, Espersen ML, Jess P, Gogenur I, et al. CDX2 downregulation is associated with poor differentiation and MMR deficiency in colon cancer. Exp Mol Pathol. 2016;100:59–66.
Lundberg IV, Edin S, Eklof V, Oberg A, Palmqvist R, Wikberg ML. SOX2 expression is associated with a cancer stem cell state and down-regulation of CDX2 in colorectal cancer. BMC Cancer. 2016;16:471.
Bae JM, Lee TH, Cho NY, Kim TY, Kang GH. Loss of CDX2 expression is associated with poor prognosis in colorectal cancer patients. World J Gastroenterol. 2015;21:1457–67.
Slik K, Turkki R, Carpen O, Kurki S, Korkeila E, Sundstrom J, et al. CDX2 loss with microsatellite stable phenotype predicts poor clinical outcome in stage II colorectal carcinoma. Am J Surg Pathol. 2019;43:1473–82.
Kim JH, Rhee YY, Bae JM, Cho NY, Kang GH. Loss of CDX2/CK20 expression is associated with poorly differentiated carcinoma, the CpG island methylator phenotype, and adverse prognosis in microsatellite-unstable colorectal cancer. Am J Surg Pathol. 2013;37:1532–41.
Baba Y, Nosho K, Shima K, Freed E, Irahara N, Philips J, et al. Relationship of CDX2 loss with molecular features and prognosis in colorectal cancer. Clin Cancer Res. 2009;15:4665–73.
Ma C, Olevian D, Miller C, Herbst C, Jayachandran P, Kozak MM, et al. SATB2 and CDX2 are prognostic biomarkers in DNA mismatch repair protein deficient colon cancer. Mod Pathol. 2019;32:1217–31.
Knosel T, Chen Y, Hotovy S, Settmacher U, Altendorf-Hofmann A, Petersen I. Loss of desmocollin 1-3 and homeobox genes PITX1 and CDX2 are associated with tumor progression and survival in colorectal carcinoma. Int J Colorectal Dis. 2012;27:1391–9.
Ryan EJ, Creavin B, Khaw YL, Kelly ME, Mohan HM, Geraghty R, et al. Effects of CDX2 on prognosis and chemotherapy responsiveness in mismatch repair-deficient colorectal cancer. BJS Open. 2018;2:456–63.
Liu Q, Luo D, An H, Zhang S, Cai S, Li Q, et al. Survival benefit of adjuvant chemotherapy for patients with poorly differentiated stage IIA colon cancer. J Cancer. 2019;10:1209–15.
Andre T, Boni C, Navarro M, Tabernero J, Hickish T, Topham C, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009;27:3109–16.
Fluge Ø, Gravdal K, Carlsen E, Vonen B, Kjellevold K, Refsum S, et al. Expression of EZH2 and Ki-67 in colorectal cancer and associations with treatment response and prognosis. Br J Cancer. 2009;101:1282–9.
Acknowledgements
We thank the Department of Pathology Ålesund for performing MMR protein staining, the Department of Pathology, Haukeland University Hospital for performing CDX2 staining and the Mohn Laboratory for Cancer Research for excellent facilities. We want to give a special acknowledgement to Ole Johnny Steffensen for invaluable technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics
The Regional Committee for Medical Research Ethics of Western Norway and the Data Inspectorate for National Registries approved the study protocols. All patients signed informed consents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Hestetun, K.E., Aasebø, K., Rosenlund, N.B. et al. Mismatch repair phenotype determines the implications of tumor grade and CDX2 expression in stage II–III colon cancer. Mod Pathol 34, 161–170 (2021). https://doi.org/10.1038/s41379-020-0634-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-020-0634-9
This article is cited by
-
The Prognostic Role of Mismatch Repair Status and CDX-2 Expression with Inflammatory Markers and Pathological Risk Factors in Stage II and III Colon Cancer: Multicenter Real-Life Data
Journal of Gastrointestinal Cancer (2023)
-
Effects of deficient mismatch repair on the prognosis of patients with stage II and stage III colon cancer during different postoperative periods
BMC Cancer (2022)
-
Loss of CDX2 in colorectal cancer is associated with histopathologic subtypes and microsatellite instability but is prognostically inferior to hematoxylin–eosin-based morphologic parameters from the WHO classification
British Journal of Cancer (2021)