Abstract
The presence of a characteristic chimeric fusion as the initiating genomic event is one defining feature of Spitz neoplasms. Characterization of specific subtypes of Spitz neoplasms allows for better recognition facilitating diagnosis. Data on clinical outcomes of the specific tumor types may help in predicting behavior. In this study we present the largest series to date on ROS1 fusion Spitz neoplasms. We present the clinical, morphologic, and genomic features of 17 cases. We compared the morphologic features of these 17 cases to a cohort of 99 other non-ROS1 Spitz neoplasms to assess for features that may have high specificity for ROS1 fusions. These tumors consisted of ten Spitz nevi and seven Spitz tumors. None of the cases met criteria for a diagnosis of Spitz melanoma. Morphologically, the ROS1 fusion tumors of this series were characterized by a plaque-like or nodular silhouette, often densely cellular intraepidermal melanocyte proliferation, frequent pagetosis, tendency toward spindle cell cytomorphology, low grade nuclear atypia, and floating nests with occasional transepidermal elimination. However, there was a significant range in microscopic appearances, including two cases with morphologic features of a desmoplastic Spitz nevus. Different binding partners to ROS1 were identified with PWWP2A and TPM3 being the most common. No case had a recurrence or metastasis. Our findings document that most ROS1 fusion Spitz neoplasms have some typical characteristic microscopic features, while a small proportion will have features overlapping with other genomic subtypes of Spitz neoplasms. Preliminary evidence suggests that they tend to be indolent or low grade neoplasms.
Similar content being viewed by others
Introduction
The family of Spitz neoplasms is defined in the most recent edition of the World Health Organization Classification of Skin Tumors (4th edition) as a melanocytic neoplasm with a characteristic Spitz fusion or a mutation in HRAS with Spitzoid morphologic features. Recent studies have attempted to correlate specific clinical and morphologic findings in the various fusion subgroups such as ALK, NTRK1, NTRK3, MAPK, BRAF, and ROS1 [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16]. Genomic fusions involving the ROS1 oncogene are seen in 7–17% of Spitz neoplasms [17, 18]. However, thus far only one study of six cases has described the morphologic features of ROS1 Spitz neoplasms [13].
In this study, we report the clinical, histologic, and molecular findings in 17 ROS1 fusion Spitz neoplasms in order to better characterize this subset of Spitz neoplasms. We compared a number of morphologic features in this set of ROS1 fusions to a control set of 99 non-ROS1 Spitz melanocytic neoplasms which have also been assessed by next generation sequencing (NGS). We describe characteristic morphologic features and report those morphologic features statistically more frequent in ROS1 Spitz compared to other subtypes of Spitz neoplasms. We also report for the first time the occurrence of ROS1 fusions in two cases of desmoplastic Spitz nevi (SN).
Materials and methods
Case selection and genomic sequencing
Study approval and waiver of consent for use of archived tissue were obtained through the Northwestern Institutional Review Board. The dermatopathology database at Northwestern was searched for SN, atypical Spitz tumor (AST), and Spitz melanomas (SM) in which a ROS1 fusion was identified by NGS. We identified eight cases matching the above criteria. The paired normal tissue were identified for Case #1, #2, #5, and #6. Additionally, nine cases were contributed from the personal consultation files of KJ Busam at Memorial Sloan Kettering Cancer Center in New York. We also identified 99 cases consisting of 20 SN, 53 ST, and 26 SM. Each diagnosis was made at the time of clinical presentation based on morphology with incorporation of FISH or array CGH in select cases. The control group included 59 fusions consisting of the following genes: ALK (n = 14), MAP3K8 (n = 12), BRAF (n = 6), NTRK1 (n = 10), NTRK3 (n = 6), RET (n = 4), MET (n = 1), RASGRF (n = 1), RAF1 (n = 1), MAP3K3 (n = 1), FGFR (n = 1), ERBB4 (n = 1), and PRKDC (n = 1). Additionally, there were five MAP3K8 truncations. Lastly there were mutations in 19 cases in the following genes: BRAF (n = 8), NRAS (n = 4), HRAS (n = 5), GNAQ (n = 1), and ROS1 (n = 1). In 16 cases no known fusions or mutations were identified.
“Spitzoid” morphology was identified according to the World Health Organization Classification of Skin Tumors (4th edition) and other relevant literature [19,20,21,22]. NGS with a 1171 cancer related gene panel for DNA and a whole transcriptome sequencing on each case was performed with using the Tempus xO platform and variant-calling [23, 24]. The 1711-gene assay is validated and designed to target therapeutically actionable genes.
Tumor classification and clinicopathologic features
In total there were 17 cases with ROS1 fusions. The clinical features including age, sex, and site of the tumors were summarized from the medical record. Morphologic features were assessed by two board certified dermatopathologist experienced in the assessment of melanocytic tumors. The following morphologic features were evaluated: silhouette (plaque, wedge, or nodular), cytology (epithelioid, spindled, or both), nuclear atypia (mild, moderate, or severe), pigmentation (absent, focal, or extensive), host inflammatory reaction (absent, non brisk, or brisk), cell size (small, intermediate, large), mitotic figures per mm2, and for the absence or presence of Kamino body, maturation, ulceration, epidermal hyperplasia, plexiform growth, epithelioid sheets, pagetosis, nesting in the adnexa, and desmoplasia.
Mild nuclear atypia was defined as a slightly larger nucleus than conventional nevomelanocytes. Moderate atypia was defined as a nuclear size similar to the size of keratinocytes with a hyperchromatic nuclear membrane, visible nucleolus, and variable chromatin quality. Severe nuclear atypia was defined as a nuclear size larger than keratinocytes with a hyperchromatic nuclear membrane, prominent and/or multiple nucleoli, and coarse chromatin. For host inflammatory reaction, a brisk response was defined as a diffuse infiltration of lymphocytes across the entire base of the tumor; a non-brisk response was defined as a focal infiltration of lymphocytes that does not cover the entire base [25]. For cell size, the size of melanocytes was compared to the basal keratinocytes [26]. Cells about the size of basal keratinocytes were considered small, those moderately larger than basal keratinocytes were intermediate in size and cells nearly twice the size of basal keratinocytes were considered large. Clinical information including age, gender, and site of tumor was also included for analysis.
Statistical analysis
All statistical analyses were performed in R Studio v1.2.5001 to compare morphologic features across the groups Spitz neoplasms. Fisher’s exact test or Chi square test was used to compare associations in categorical variables. Student’s t test was used to compare mean values. A p value of <0.05 was considered statistically significant. All tests were two sided.
Results
Clinical findings in ROS1 fusion Spitz neoplasms
The final diagnosis from the time of clinical care in the set of 17 ROS1 Spitz neoplasms was Spitz nevus in ten cases and Spitz tumor in seven cases. In none of the cases was a diagnosis of Spitz melanoma favored. The patient ages ranged from 3 to 58 with a mean age of 19 years old. There were ten female and seven male patients. The body site of involvement was highly variable with four in the head/neck region, three on the upper extremities, three on the trunk, and seven on the lower extremities. Grossly, all cases were pink to red papules. In 14 cases the clinical impression was available. In seven cases the clinician suspected an atypical Spitz nevus and in one of these cases a dermoscopic description of radial streaming was provided. In two cases the clinical impression was dermatofibroma, in two cases it was benign nevus, in two cases it was pyogenic granuloma and in one case it was cyst.
Follow-up was available for 13 of 17 cases (Table 1). The average follow-up time was 23 months and ranged from 4 to 95 months. In 12 cases the lesions were re-excised with clear margins with no evidence of recurrence. One of these 12 cases also had a sentinel lymph node biopsy (SLNB) which was negative. In one case the original biopsy was incisional and no further re-excision was performed. There was persistent tumor at a follow exam 4 months later.
Morphologic and immunohistochemical findings in ROS1 fusions Spitz neoplasms
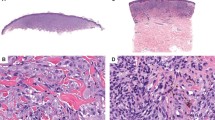
The low power silhouette on the 16 ROS1 cases was mostly that of either a plaque like (n = 7) or nodular pattern (n = 7). Two cases had a wedge shaped silhouette and one was polypoid. In 12 cases the cytomorphology was a mixed pattern of epithelioid and spindle cells while in four cases there was a predominance of spindle cells. In all cases the atypia was mild or moderate with none of the cases having high grade nuclear atypia (P = 0.006) (Fig. 1). This was statistically significant with ROS1 cases being less likely to have high grade nuclear atypia than the group of non-ROS1 Spitz neoplasms. The cell sizes were also all small to intermediate with none of the cases having large cells and this was also statistically significant (P = 0.001). Maturation was present in all cases and this was also statistically significant (P = 0.044). There was also a tendency for lower mitotic rate 1.3/mm2 (P = 0.001) (Table 2). Kamino bodies were also more common in this type of Spitz (8/17) than non-ROS1 Spitz (P = 0.025).
a At 40× one can appreciate the plaque-like silhouette of this ROS1 fusion Atypical Spitz Tumor. b At 100× the epidermal hyperplasia with a predominance of nests with spindle-shaped melanocytes can be seen in a back-to-back pattern crowding the epidermis. c At 200× one can appreciate the transepidermal elimination of small nests into the stratum corneum. d 400× demonstrates the Spitzoid cytomorphology with relatively low-grade nuclear atypia.
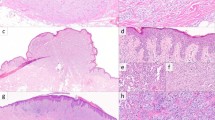
Thirteen of 17 cases had overlying epidermal hyperplasia. Fourteen of 17 cases were completely amelanotic. Lobulated nests were seen in two cases and nesting in the adnexa in five cases. Five cases had notable pagetosis in the epidermis. None of these features were statistically significant compared to non-ROS1 Spitz neoplasms. Nine of 17 cases had floating nests defined as nests situated above the basal layer and in three cases there was transepidermal elimination of nests (Figs. 1, 2). Myxoid changes were not identified in any of the cases. Two cases were characterized by prominent stromal desmoplasia, and were morphologically best characterized as a desmoplastic Spitz nevus (Fig. 3).
Immunohistochemical staining for ROS1 was performed in 16 cases. Fifteen of the 16 cases showed strong positive staining (Fig. 4). In one case only a blush staining was seen which was not convincingly positive.
Genomic findings in ROS1 fusion Spitz neoplasms
The fusion partner was identified in 16 of the 17 cases in the study. The most common genomic fusions among the 16 ROS1 cases were a PWWP2A–ROS1 fusion seen in six cases and a TPM3–ROS1 fusion also seen in five cases. Other recurrent fusion partners included a PPFIBP1–ROS1 fusion seen in two cases, and fusions partners involving MYH9–ROS1, CAPRINI1–ROS1, and MYO5A–ROS1 were each seen in one case (Table 3).
Three cases had copy number aberrations identified by NGS and SNP arrays. Two cases had copy number aberrations identified by NGS and one case had a copy number aberration identified by SNP array. Copy number loss of BCL11B, FGF3, CARD11, FBXO11, FLT4, GRIN2A, HGF, MGMT, MYCN, MYOD1, NPM1, NTRK3, PLAG1, PTPRT, RET, TERT, and TLX1 were identified in case 2. This case was negative for copy number alterations when tested by a SNP array platform. Copy number gains of HOXA9, JUN, and MDM2 were identified in case 6. Case 10 had an isolated loss at 6q.
Discussion
Among two studies sequencing a large number of Spitz neoplasms the frequency of ROS1 fusions varied from 7 to 17% [17, 18]. The vast majority of these cases were diagnosed as either Spitz nevus or Spitz tumor. In this study ten were diagnosed as Spitz nevus and seven as Spitz tumor. We did not identify any cases that met the criteria of a Spitz melanoma. In the study from Wiesner et al. where kinase fusions in Spitz neoplasms were first described [17], 3 of 24 ROS1 fusions were designated as Spitz melanoma, but no adverse clinical outcome was reported. This study from Wiesner et al. is the larger series on ROS1 fusions but does not discuss morphologic features. Thus far there is only one study involving 6 cases of ROS1 fusions which were all designated as Spitz tumors by Donati et al. which discusses morphologic features [13].
While there is limited clinical outcomes information available on Spitz tumors with ROS1 fusions, among the 13 cases with follow-up in this study and the six cases from Donati et al. there are no reported recurrences or metastases after complete excision of the primary tumors. One case in our series had a SLNB which was also negative. Thus, preliminary evidence suggests that most Spitz tumors with ROS1 fusions are likely indolent or at least in a much lower risk category compared to Spitz neoplasms with BRAF or MAP3K8 fusions which seem to constitute much of the more aggressive variants of Spitz neoplasms [9,10,11, 15, 27,28,29].
We did not identify morphologic features which could allow for a definitive diagnosis of a ROS1 fusion by microscopic review alone but there were some characteristic features. This included a tendency for plaque-like or nodular silhouette without a deeply infiltrative component with a combination of epithelioid and spindle cell cytomorphology. Statistically significant features included lack of high grade cytologic atypia in all cases, lack of larger cell type, presence of maturation, frequent Kamino bodies and lower mitotic rate. These findings are consistent with the fact that all cases were diagnosed as Spitz nevus or Spitz tumor and none were thought to be Spitz melanoma.
In our cases, 13/17 had epidermal hyperplasia and 5/17 had notable epidermal pagetosis. Two cases had lobulated nests and four had nesting in the adnexa. None of these features were statistically significant as they can be seen in a broad spectrum of Spitz subtypes. In particular many of these features can overlap with NTRK1 fusion Spitz neoplasms. Donati et al. reported transepidermal elimination of nests and myxoid changes as being present in all six cases. Another highly characteristic feature was floating nests seen in 9 of 17 cases with transepidermal elimination of nests in three cases. We did not identify significant mucinous changes though a colloidal iron was not performed. Although none of these features are totally specific, one might anticipate a ROS1 fusion in compound plaque-like Spitz neoplasm with prominent intraepidermal component, Kamino bodies, with small to intermediate sized cells with low grade cytology, pagetosis, and floating nests within the epidermis.
An interesting and novel observation is the detection of a ROS1 fusion in two desmoplastic SN. This illustrates the wide spectrum of microscopic features associated with ROS1 fusions, but it also documents that the desmoplastic phenotype among SN is not limited to HRAS aberrations. Gains of 11p (location of HRAS) and/or HRAS mutations have previously been thought to be typical of desmoplastic SN. While they likely represent the most common aberration associated with a desmoplastic Spitz nevus, we hereby document two cases with a ROS1 kinase fusion associated with a desmoplastic phenotype.
In the 17 cases in this series, six different fusion partners were identified. This included PWWP2A (n = 6), TPM3 (n = 5), PPFIBP1 (n = 2), MYO5A (n = 1), CAPRINI1 (n = 1), and MYH9 (n = 1). PWWP2A was also the most frequent fusion partner in the series from Donati et al. A figure showing the chimeric protein model and the breakpoint of the fusions can be found in Fig. 5. Previous in vivo studies show rising levels of phosphorylation produced by this fusion protein indicating that the ROS1 kinase is being constitutively activated [17].
Functional domains are displayed. Breakpoints were indicated by the black arrows above the each schematic. Recurrent ROS1 fusion breakapart were identified in intron 34 and 35. Positive and negative strands of DNA sequence were marked with “+” and “–”, respectively. Transcribed DNA strand was highlighted in red. Arrows at the end of sequence indicate the direction of transcription.
ROS1 fusions have been identified in 9% SM and 1.3% in melanomas from previous studies [17, 30]. There are no cases of ROS1 fusion melanoma in the TCGA database. ROS1 fusions are also seen in a subset of 1–2% non-small cell lung cancers. More recently ROS1 fusions were identified in 9 of 130 gliomas from an infant population [31]. Also, rare cases of ROS1 fusions in angiosarcoma, thyroid, and breast cancer have been reported [32,33,34]. Interestingly in melanocytic neoplasms with ROS1 fusions the tumors seem to have an indolent clinical behavior.
In conclusion, this study describes the largest series to date on ROS1 fusion Spitz neoplasms. They seem to represent a lower grade group of tumors with generally indolent behavior. We could not find specific morphologic aberrations that were predictive of the molecular aberration but identified a number of features that were enriched in the group of ROS1 fusion tumors. They included a plaque or nodular silhouette with a cellular intraepidermal component, frequent Kamino bodies, a slight predisposition toward spindle cytology, a lower grade of cytologic atypia, and floating nests/transepidermal elimination of nests. We also report for the first time the association of a desmoplastic phenotype with ROS1 fusions.
Data availability
Processed sequencing data (vcf files and count files) can be found through GEO Series accession number GSE142443.
References
Busam KJ, Kutzner H, Cerroni L, Wiesner T. Clinical and pathologic findings of Spitz nevi and atypical Spitz tumors with ALK fusions. Am J Surg Pathol. 2014;38:925–33.
Yeh I, de la Fouchardiere A, Pissaloux D, Mully TW, Garrido MC, Vemula SS, et al. Clinical, histopathologic, and genomic features of Spitz tumors with ALK fusions. Am J Surg Pathol. 2015;39:581–91.
Saraggi D, Salmaso R, Zamuner C, Munari G, Lanza C, Alaibac M, et al. Prevalence of ALK gene alterations among the spectrum of plexiform spitzoid lesions. J Am Acad Dermatol. 2018;79:728–35.
Lee CY, Sholl LM, Zhang B, Merkel EA, Amin SM, Guitart J, et al. Atypical Spitzoid neoplasms in childhood: a molecular and outcome study. Am J Dermatopathol. 2017;39:181–6.
Yeh I, Busam KJ, McCalmont TH, LeBoit PE, Pissaloux D, Alberti L, et al. Filigree-like rete ridges, lobulated nests, rosette-like structures, and exaggerated maturation characterize Spitz tumors with NTRK1 fusion. Am J Surg Pathol. 2019;43:737–46.
Kiuru M, Jungbluth A, Kutzner H, Wiesner T, Busam KJ. Spitz tumors: comparison of histological features in relationship to immunohistochemical staining for ALK and NTRK1. Int J Surg Pathol. 2016;24:200–6.
Yeh I, Tee MK, Botton T, Shain AH, Sparatta AJ, Gagnon A, et al. NTRK3 kinase fusions in Spitz tumours. J Pathol. 2016;240:282–90.
Quan VL, Panah E, Zhang B, Shi K, Mohan LS, Gerami P. The role of gene fusions in melanocytic neoplasms. J Cutan Pathol. 2019;46:878–87.
Quan VL, Zhang B, Mohan LS, Shi K, Isales M, Panah E, et al. Activating structural alterations in MAPK genes are distinct genetic drivers in a unique subgroup of Spitzoid neoplasms. Am J Surg Pathol. 2019;43:538–48.
Houlier A, Pissaloux D, Masse I, Tirode F, Karanian M, Pincus L, et al. Melanocytic tumors with MAP3K8 fusions: report of 33 cases with morphological-genetic correlations. Mod Pathol. 2020;33:846–57.
Newman S, Fan L, Pribnow A, Silkov A, Rice SV, Lee S, et al. Clinical genome sequencing uncovers potentially targetable truncations and fusions of MAP3K8 in Spitzoid and other melanomas. Nat Med. 2019;25:597–602.
Perron E, Pissaloux D, Neub A, Hohl D, Tartar M, Mortier L, et al. Unclassified sclerosing malignant melanomas with AKAP9-BRAF gene fusion: a report of two cases and review of BRAF fusions in melanocytic tumors. Virchows Arch. 2018;472:469–76.
Donati M, Kastnerova L, Martinek P, Gossmann P, Sticová E, Hadravský L, et al. Spitz tumors with ROS1 fusions: a clinicopathological study of 6 cases, including FISH for chromosomal copy number alterations and mutation analysis using next-generation sequencing. Am J Dermatopathol. 2020;42:92–102.
Donati M, Kastnerova L, Ptakova N, Michal M, Kazakov DV. Polypoid atypical Spitz tumor with a fibrosclerotic stroma, CLIP2-BRAF fusion, and homozygous loss of 9p21. Am J Dermatopathol. 2020;42:204–7.
Ross JS, Wang K, Chmielecki J, Gay L, Johnson A, Chudnovsky J, et al. The distribution of BRAF gene fusions in solid tumors and response to targeted therapy. Int J Cancer. 2016;138:881–90.
Yeh I, Botton T, Talevich E, Shain AH, Sparatta AJ, de la Fouchardiere A, et al. Activating MET kinase rearrangements in melanoma and Spitz tumours. Nat Commun. 2015;6:7174.
Wiesner T, He J, Yelensky R, Esteve-Puig R, Botton T, Yeh I, et al. Kinase fusions are frequent in Spitz tumours and spitzoid melanomas. Nat Commun 2014;5:3116.
Quan VL, Zhang B, Zhang Y, Mohan L, Shi K, Wagner A, et al. Integrating next-generation sequencing with morphology improves prognostic and biologic classification of Spitz neoplasms. J Investig Dermatol. 2020;140:1599–608.
Busam KJ, Gerami P, Scolyer RA. Pathology of melanocytic tumors. Philadelphia, PA: Elsevier; 2018.
Crowson AN, Magro CM, Mihm MC. The melanocytic proliferations: a comprehensive textbook of pigmented lesions. Hoboken, NJ: John Wiley & Sons Inc; 2013.
Mooi W, Krausz T. Pathology of melanocytic disorders. BocaRaton, FL: CRC Press; 2007.
Barnhill RL, Piepkorn M, Busam KJ. Pathology of malignant melanoma. New York, NY: Springer; 2004.
Beaubier N, Tell R, Huether R, Bontrager M, Bush S, Parsons J, et al. Clinical validation of the Tempus xO assay. Oncotarget. 2018;9:25826–32.
Beaubier N, Tell R, Lau D, Parsons JR, Bush S, Perera J, et al. Clinical validation of the tempus xT next-generation targeted oncology sequencing assay. Oncotarget. 2019;10:2384–96.
Busam KJ, Antonescu CR, Marghoob AA, Marghoob M, Nehal KS, Sachs DL, et al. Histologic classification of tumor-infiltrating lymphocytes in primary cutaneous malignant melanoma. A study of interobserver agreement. Am J Clin Pathol. 2001;115:856–60.
Cichorek M, Wachulska M, Stasiewicz A, Tymińska A. Skin melanocytes: biology and development. Postepy Dermatol Alergol. 2013;30:30–41.
Wu G, Barnhill RL, Lee S, Li Y, Shao Y, Easton J, et al. The landscape of fusion transcripts in spitzoid melanoma and biologically indeterminate spitzoid tumors by RNA sequencing. Mod Pathol. 2016;29:359–69.
Lee S, Barnhill RL, Dummer R, Dalton J, Wu J, Pappo A, et al. TERT promoter mutations are predictive of aggressive clinical behavior in patients with Spitzoid melanocytic neoplasms. Sci Rep. 2015;5:11200.
Botton T, Yeh I, Nelson T, Vemula S, Sparatta A, Garrido M, et al. Recurrent BRAF kinase fusions in melanocytic tumors offer an opportunity for targeted therapy. Pigment Cell Melanoma Res. 2013;26:845–51.
Turner J, Couts K, Sheren J, Saichaemchan S, Ariyawutyakorn W, Avolio I, et al. Kinase gene fusions in defined subsets of melanoma. Pigment Cell Melanoma Res. 2017;30:53–62.
Clarke M, Mackay A, Ismer B, Pickles J, Tatevossian R, Newman S, et al. Infant high grade gliomas comprise multiple subgroups characterized by novel targetable gene fusions and favorable outcomes. Cancer Discov. 2020;10:942–63.
Ritterhouse LL, Wirth LJ, Randolph GW, Sadow PM, Ross DS, Liddy W, et al. ROS1 rearrangement in thyroid cancer. Thyroid. 2016;26:794–7.
Marks EI, Pamarthy S, Dizon D, Birnbaum A, Yakirevich E, Safran H, et al. ROS1-GOPC/FIG: a novel gene fusion in hepatic angiosarcoma. Oncotarget. 2019;10:245–51.
Wu X, Wang H, Fang M, Li C, Zeng Y, Wang K. ALK or ROS1-rearranged breast metastasis from lung adenocarcinoma: a report of 2 cases. Tumori. 2019;105:NP67–71.
Funding
This work was supported by the IDP Foundation and in part by the Cancer Center Support Grant of the National Institutes of Health/National Cancer Institute under award number P30CA008748.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
PG has served as a consultant for Myriad Genomics, DermTech Int., Merck and Castle Biosciences and has received honoraria for this. All other authors report no relevant conflicts of interest. This work is original and has not been previously published.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gerami, P., Kim, D., Compres, E.V. et al. Clinical, morphologic, and genomic findings in ROS1 fusion Spitz neoplasms. Mod Pathol 34, 348–357 (2021). https://doi.org/10.1038/s41379-020-00658-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-020-00658-w
This article is cited by
-
Novel gene fusion discovery in Spitz tumours and its relevance in diagnostics
Virchows Archiv (2023)
-
Morphologic features in a series of 352 Spitz melanocytic proliferations help predict their oncogenic drivers
Virchows Archiv (2022)
-
Genome-wide copy number variations as molecular diagnostic tool for cutaneous intermediate melanocytic lesions: a systematic review and individual patient data meta-analysis
Virchows Archiv (2021)