Abstract
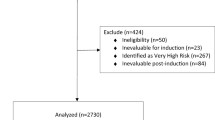
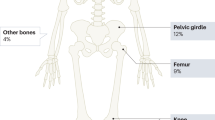
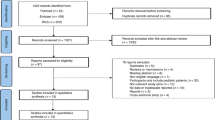
The majority of patients with Hodgkin Lymphoma (HL) can be cured with stage and risk adapted treatment today. Therefore, current research focuses on reducing long-term sequelae of treatment. Osteonecrosis (ON) is a severe long-term complication of HL treatment which has so far not been systematically evaluated. Hence, we investigated incidence, risk factors and timing of symptomatic ON in HL patients. Further endpoints included localization, intervention and outcome of ON. We included all qualified HL patients of the randomized German Hodgkin Study Group trials HD10-15 and HD18, recruited between 05/1998 and 07/2014 and aged from 16 to 60 years. Among 11 330 patients, 66 developed symptomatic ON after first-line treatment, 83.3% within three years. The incidence of symptomatic ON was 0.2% in early-stage HL and 1.0% in advanced-stage HL. Logistic regression revealed the total cumulative corticosteroid dose to be a strong risk factor interacting with younger age. Male sex additionally increased the risk of symptomatic ON. The prognostic value of the corresponding logistic regression model was rather high (AUC = 0.78). Other tested potential risk factors including obesity, IPS and radiotherapy did not further increase the risk of ON. Further development of current treatment protocols should aim to reduce the cumulative corticosteroid dose.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Skoetz N, Trelle S, Rancea M, Haverkamp H, Diehl V, Engert A, et al. Effect of initial treatment strategy on survival of patients with advanced-stage Hodgkin’s lymphoma: a systematic review and network meta-analysis. Lancet Oncol. 2013;14:943–52.
Borchmann S, von Tresckow B, Engert A. Current developments in the treatment of early-stage classical Hodgkin lymphoma. Curr Opin Oncol. 2016;28:377–83.
Behringer K, Mueller H, Goergen H, Thielen I, Eibl AD, Stumpf V, et al. Gonadal function and fertility in survivors after Hodgkin lymphoma treatment within the German Hodgkin study group HD13 to HD15 Trials. J Clin Oncol. 2013;31:231–9.
Eichenauer DA, Thielen I, Haverkamp H, Franklin J, Behringer K, Halbsguth T, et al. Therapy-related acute myeloid leukemia and myelodysplastic syndromes in patients with hodgkin lymphoma: a report from the German Hodgkin Study Group. Blood. 2014;123:1658–64.
Schaapveld M, Aleman BMP, van Eggermond AM, Janus CPM, Krol ADG, van der Maazen RWM, et al. Second cancer risk up to 40 years after treatment for Hodgkin’s lymphoma. N Engl J Med. 2015;373:2499–511.
Behringer K, Goergen H, Müller H, Thielen I, Brillant C, Kreissl S, et al. Cancer-related fatigue in patients with and survivors of Hodgkin lymphoma: the impact on treatment outcome and social reintegration. J Clin Oncol. 2016;34:4329–37.
Kreissl S, Mueller H, Goergen H, Mayer A, Brillant C, Behringer K, et al. Cancer-related fatigue in patients with and survivors of Hodgkin’s lymphoma: a longitudinal study of the German Hodgkin Study Group. Lancet Oncol. 2016;17:1453–62.
te Winkel ML, Pieters R, Hop WCJ, de Groot-Kruseman HA, Lequin MH, van der Sluis IM, et al. Prospective study on incidence, risk factors, and long-term outcome of osteonecrosis in pediatric acute lymphoblastic leukemia. J Clin Oncol. 2011;29:4143–50.
Prosnitz LR, Lawson JP, Friedlaender GE, Farber LR, Pezzimenti JF. Avascular necrosis of bone in Hodgkin’s disease patients treated with combined modality therapy. Cancer. 1981;47:2793–7.
Ellis J, MacLeod U, Sammon D, Lindsay E, Erskine JG. Osteonecrosis following treatment for Hodgkin’s disease. Clin Lab Haematol. 1994;16:3–8.
Enrici RM, Anselmo AP, Donato V, Santoro M, Tombolini V. Avascular osteonecrosis in patients treated for Hodgkin’s disease. Eur J Haematol. 1998;61:204–9.
Fosså A, Fiskvik IH, Kolstad A, Lauritzsen GF, Aurlien E, Blystad AK, et al. Two escalated followed by six standard BEACOPP in advanced-stage high-risk classical Hodgkin lymphoma: high cure rates but increased risk of aseptic osteonecrosis. Ann Oncol. 2012;23:1254–9.
Renedo RJG, Sousa MM, Pérez SF, Zabalbeascoa JR, Carro LP. Avascular necrosis of the femoral head in patients with Hodgkin’s disease. Hip Int. 2010;20:473–81.
Engert A, Plütschow A, Eich HT, Lohri A, Dörken B, Borchmann P, et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N Engl J Med. 2010;363:640–52.
Eich HT, Diehl V, Görgen H, Pabst T, Markova J, Debus J, et al. Intensified chemotherapy and dose-reduced involved-field radiotherapy in patients with early unfavorable Hodgkin’s lymphoma: final analysis of the German Hodgkin Study Group HD11 trial. J Clin Oncol. 2010;28:4199–206.
Borchmann P, Haverkamp H, Diehl V, Cerny T, Markova J, Ho AD, et al. Eight cycles of escalated-dose BEACOPP compared with four cycles of escalated-dose BEACOPP followed by four cycles of baseline-dose BEACOPP with or without radiotherapy in patients with advanced-stage Hodgkin’s lymphoma: final analysis of the HD12 trial of the German Hodgkin Study Group. J Clin Oncol. 2011;29:4234–42.
Behringer K, Goergen H, Hitz F, Zijlstra JM, Greil R, Markova J, et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): an open-label, randomised, non-inferiority trial. Lancet. 2015;385:1418–27.
von Tresckow B, Plutschow A, Fuchs M, Klimm B, Markova J, Lohri A, et al. Dose-intensification in early unfavorable Hodgkin’s lymphoma: final analysis of the German Hodgkin Study Group HD14 Trial. J Clin Oncol. 2012;30:907–13.
Borchmann P, Goergen H, Kobe C, Lohri A, Greil R, Eichenauer DA, et al. PET-guided treatment in patients with advanced-stage Hodgkin’s lymphoma (HD18): final results of an open-label, international, randomised phase 3 trial by the German Hodgkin Study Group. Lancet. 2017. https://doi.org/10.1016/S0140-6736(17)32134-7.
Engert A, Haverkamp H, Kobe C, Markova J, Renner C, Ho A, et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin’s lymphoma (HD15 trial): a randomised, open-label, phase 3 non-inferiority trial. Lancet. 2012;379:1791–9.
NCI, NIH, DHHS, National Cancer Institute. Common terminology criteria for adverse events v4.0. NIH publication; 2009.
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–55.
Cooper C, Steinbuch M, Stevenson R, Miday R, Watts NB. The epidemiology of osteonecrosis: findings from the GPRD and THIN databases in the UK. Osteoporos Int. 2010;21:569–77.
Mattano LAJ, Sather HN, Trigg ME, Nachman JB. Osteonecrosis as a complication of treating acute lymphoblastic leukemia in children: a report from the Children’s Cancer Group. J Clin Oncol. 2000;18:3262–72.
Sharma S, Yang S, Rochester R, Britton L, Leung WH, Yang J, et al. Prevalence of osteonecrosis and associated risk factors in children before allogeneic BMT. Bone Marrow Transplant. 2011;46:813–9.
Kaste SC, Karimova EJ, Neel MD. Osteonecrosis in children after therapy for malignancy. Am J Roentgenol. 2011;196:1011–8.
Mont MA, Baumgarten KM, Rifai A, Bluemke DA, Jones LC, Hungerford DS. Atraumatic osteonecrosis of the knee. J Bone Jt Surg Am. 2000;82:1279–90.
Moya-Angeler J, Gianakos AL, Villa JC, Ni A, Lane JM. Current concepts on osteonecrosis of the femoral head. World J Orthop. 2015;6:590–601.
Mont MA, Carbone JJ, Fairbank AC. Core decompression versus nonoperative management for osteonecrosis of the hip. Clin Orthop Relat Res. 1996:169–78.
Chan KL, Mok CC. Glucocorticoid-induced avascular bone necrosis: diagnosis and management. Open Orthop J. 2012;6:449–57.
Niinimäki RA, Harila-Saari AH, Jartti AE, Seuri RM, Riikonen PV, Pääkkö EL, et al. High body mass index increases the risk for osteonecrosis in children with acute lymphoblastic leukemia. J Clin Oncol. 2007;25:1498–504.
Pritchett JW. Statin therapy decreases the risk of osteonecrosis in patients receiving steroids. Clin Orthop Relat Res. 2001:173–8.
Jäger M, Zilkens C, Bittersohl B, Matheney T, Kozina G, Blondin D, et al. Efficiency of iloprost treatment for osseous malperfusion. Int Orthop. 2011;35:761–5.
Lai K.-A, Shen W.-J, Yang C.-Y, Shao C.-J, Hsu J.-T, Lin R.-M. The use of alendronate to prevent early collapse of the femoral head in patients with nontraumatic osteonecrosis. A randomized clinical study. J Bone Jt Surg Am. 2005;87:2155–9.
Kaste SC, Pei D, Cheng C, Neel MD, Bowman WP, Ribeiro RC, et al. Utility of early screening magnetic resonance imaging for extensive hip osteonecrosis in pediatric patients treated with glucocorticoids. J Clin Oncol. 2015;33:610–5.
Connors JM, Jurczak W, Straus DJ, Ansell SM, Kim WS, Gallamini A, et al. Brentuximab vedotin with chemotherapy for stage III or IV Hodgkin’s lymphoma. N Engl J Med. 2018;378:331–44.
Borchmann P, Eichenauer DA, Pluetschow A, Haverkamp H, Kreissl S, Fuchs M, et al. Targeted Beacopp variants in patients with newly diagnosed advanced stage classical Hodgkin lymphoma: final analysis of a randomized phase II study. Blood. 2015;126:580.
Acknowledgements
This study was presented in part at the 21st annual congress of the European Hematology Association (EHA) in Copenhagen, Denmark in June 2016 and at the 10th International Symposium on Hodgkin Lymphoma (ISHL) in October 2016 in Cologne, Germany. This study was supported by grants of the Deutsche Krebshilfe e.V. (German Cancer Aid) for the HD10-HD15 and HD18 trials.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Borchmann, S., Müller, H., Haverkamp, H. et al. Symptomatic osteonecrosis as a treatment complication in Hodgkin lymphoma: an analysis of the German Hodgkin Study Group (GHSG). Leukemia 33, 439–446 (2019). https://doi.org/10.1038/s41375-018-0240-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-018-0240-8