Abstract
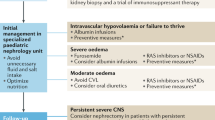
Congenital nephrotic syndrome (CNS), a challenging form of nephrotic syndrome, is characterized by massive proteinuria, hypoalbuminemia, and edema. Extensive leakage of plasma proteins is the main feature of CNS. Patients can be diagnosed in utero or during the first few weeks of life, usually before three months. The etiology of CNS can be related to either genetic or nongenetic etiologies. Pathogenic variants in NPHS1, NPHS2, LAMB2, WT1, and PLCE1 genes have been implicated in this disease. The clinical course is complicated by significant edema, infections, thrombosis, hypothyroidism, failure to thrive, and others. Obtaining vascular access, frequent intravenous albumin infusions, diuretic use, infection prevention, and nutritional support are the mainstay management during their first month of life. The best therapy for these patients is kidney transplantation. CNS diagnosis and treatment continue to be a challenge for clinicians. This review increases the awareness about the pathogenesis, diagnosis, and management of CNS patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hinkes BG, Mucha B, Vlangos CN, Gbadegesin R, Liu J, Hasselbacher K, et al. Nephrotic syndrome in the first year of life: two thirds of cases are caused by mutations in 4 genes (NPHS1, NPHS2, WT1, and LAMB2). Pediatrics. 2007;119:e907–19.
Jalanko H. Congenital nephrotic syndrome. Pediatr Nephrol. 2009;24:2121–8.
Lenkkeri U, Männikkö M, McCready P, Lamerdin J, Gribouval O, Niaudet PM, et al. Structure of the gene for congenital nephrotic syndrome of the finnish type (NPHS1) and characterization of mutations. Am J Hum Genet. 1999;64:51–61.
Kestilä M, Lenkkeri U, Männikkö M, Lamerdin J, McCready P, Putaala H, et al. Positionally cloned gene for a novel glomerular protein–nephrin–is mutated in congenital nephrotic syndrome. Mol Cell. 1998;1:575–82.
Boute N, Gribouval O, Roselli S, Benessy F, Lee H, Fuchshuber A, et al. NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resistant nephrotic syndrome. Nat Genet. 2000;24:349–54.
Suvanto M, Jahnukainen T, Kestilä M, Jalanko H. Podocyte proteins in congenital and minimal change nephrotic syndrome. Clin Exp Nephrol. 2015;19:481–8.
Shahin B, Papadopoulou ZL, Jenis EH. Congenital nephrotic syndrome associated with congenital toxoplasmosis. J Pediatr. 1974;85:366–70.
Basker M, Agarwal I, Bendon KS. Congenital nephrotic syndrome-a treatable cause. Ann Trop Paediatr. 2007;27:87–90.
Besbas N, Bayrakci US, Kale G, Cengiz AB, Akcoren Z, Akinci D, et al. Cytomegalovirus-related congenital nephrotic syndrome with diffuse mesangial sclerosis. Pediatr Nephrol. 2006;21:740–2.
Chen Y, Zhang Y, Wang F, Zhang H, Zhong X, Xiao H, et al. Analysis of 14 Patients With Congenital Nephrotic Syndrome. Front Pediatr. 2019;7:341.
Evans DG, Lyon AJ. Fatal congenital cytomegalovirus infection acquired by an intra-uterine transfusion. Eur J Pediatr. 1991;150:780–1.
Batisky DL, Roy S, Gaber LW. Congenital nephrosis and neonatal cytomegalovirus infection: a clinical association. Pediatr Nephrol. 1993;7:741–3.
Kim YH, Song JH, Kim CJ, Yang EM. Congenital syphilis presenting with only nephrotic syndrome: reemergence of a forgotten disease. J Korean Med Sci. 2017;32:1374–6.
Massengill SF, Richard GA, Donnelly WH. Infantile systemic lupus erythematosus with onset simulating congenital nephrotic syndrome. J Pediatr. 1994;124:27–31.
Dudley J, Fenton T, Unsworth J, Chambers T, MacIver A, Tizard J. Systemic lupus erythematosus presenting as congenital nephrotic syndrome. Pediatr Nephrol. 1996;10:752–5.
Cooper JM, Sánchez PJ. Congenital syphilis. Semin Perinatol. 2018;42:176–84.
Debiec H, Nauta J, Coulet F, van der Burg M, Guigonis V, Schurmans T, et al. Role of truncating mutations in MME gene in fetomaternal alloimmunisation and antenatal glomerulopathies. Lancet. 2004;364:1252–9.
Debiec H, Guigonis V, Mougenot B, Decobert F, Haymann JP, Bensman A, et al. Antenatal membranous glomerulonephritis due to anti-neutral endopeptidase antibodies. N. Engl J Med. 2002;346:2053–60.
Nortier JL, Debiec H, Tournay Y, Mougenot B, Nöel JC, Deschodt-Lanckman MM, et al. Neonatal disease in neutral endopeptidase alloimmunization: lessons for immunological monitoring. Pediatr Nephrol. 2006;21:1399–405.
Gigante M, Piemontese M, Gesualdo L, Iolascon A, Aucella F. Molecular and genetic basis of inherited nephrotic syndrome. Int J Nephrol. 2011;2011:792195.
Heeringa SF, Vlangos CN, Chernin G, Hinkes B, Gbadegesin R, Liu J, et al. Thirteen novel NPHS1 mutations in a large cohort of children with congenital nephrotic syndrome. Nephrol Dialysis Transplant: Off Publ Eur Dialysis Transpl Assoc - Eur Ren Assoc. 2008;23:3527–33.
Levy M, Feingold J. Estimating prevalence in single-gene kidney diseases progressing to renal failure. Kidney Int. 2000;58:925–43.
Ismaili K, Pawtowski A, Boyer O, Wissing KM, Janssen F, Hall M, et al. Genetic forms of nephrotic syndrome: a single-center experience in Brussels. Pediatr Nephrol. 2009;24:287–94.
Patrakka J, Kestilä M, Wartiovaara J, Ruotsalainen V, Tissari P, Lenkkeri U, et al. Congenital nephrotic syndrome (NPHS1): features resulting from different mutations in Finnish patients. Kidney Int. 2000;58:972–80.
Huttunen NP, Rapola J, Vilska J, Hallman N. Renal pathology in congenital nephrotic syndrome of Finnish type: a quantitative light microscopic study on 50 patients. Int J Pediatr Nephrol. 1980;1:10–6.
Machuca E, Benoit G, Nevo F, Tête MJ, Gribouval O, Pawtowski A. Genotype-phenotype correlations in non-Finnish congenital nephrotic syndrome. J Am Soc Nephrol. 2010;21:1209–17.
Guaragna M, Lutaif A, Piveta C, Souza M, De Souza S, Henriques T, et al. NPHS2 mutations account for only 15% of nephrotic syndrome cases. BMC Med Genet. 2015;16:88.
Lipska-Ziętkiewicz BS, Ozaltin F, Hölttä T, Bockenhauer D, Bérody S, Levtchenko E, et al. Genetic aspects of congenital nephrotic syndrome: a consensus statement from the ERKNet-ESPN inherited glomerulopathy working group. Eur J Hum Genet. 2020;28:1368–78.
Koziell A, Grech V, Hussain S, Lee G, Lenkkeri U, Tryggvason K, et al. Genotype/phenotype correlations of NPHS1 and NPHS2 mutations in nephrotic syndrome advocate a functional inter-relationship in glomerular filtration. Hum Mol Genet. 2002;11:379–88.
Mikó Á, Menyhárd D, Kaposi A, Antignac C, Tory K. The mutation-dependent pathogenicity of NPHS2 p.R229Q: a guide for clinical assessment. Hum Mutat. 2018;39:1854–60.
Pritchard-Jones K, Fleming S, Davidson D, Bickmore W, Porteous D, Gosden C, et al. The candidate Wilms’ tumour gene is involved in genitourinary development. Nature. 1990;346:194–7.
Mrowka C, Schedl A. Wilms’ tumor suppressor gene WT1: from structure to renal pathophysiologic features. J Am Soc Nephrol. 2000;11:S106–15.
Nishi K, Inoguchi T, Kamei K, Hamada R, Hataya H, Ogura M, et al. Detailed clinical manifestations at onset and prognosis of neonatal-onset Denys-Drash syndrome and congenital nephrotic syndrome of the Finnish type. Clin Exp Nephrol. 2019;23:1058–65.
Drash A, Sherman F, Hartmann WH, Blizzard RM. A syndrome of pseudohermaphroditism, Wilms’ tumor, hypertension, and degenerative renal disease. J Pediatr. 1970;76:585–93.
Urbach J, Drukker A, Rosenmann E. Diffuse mesangial sclerosis–light, immunofluorescent and electronmicroscopy findings. Int J Pediatr Nephrol. 1985;6:101–4.
Coppes M, Huff V, Pelletier J. Denys-Drash syndrome: Relating a clinical disorder to genetic alterations in the tumor suppressor gene WT1. J Pediatrics. 1993;123:673–78.
Habib R, Loirat C, Gubler MC, Niaudet P, Bensman A, Levy M, et al. The nephropathy associated with male pseudohermaphroditism and Wilms’ tumor (Drash syndrome): a distinctive glomerular lesion–report of 10 cases. Clin Nephrol. 1985;24:269–78.
Barbaux S, Niaudet P, Gubler MC, Grünfeld JP, Jaubert F, Kuttenn F, et al. Donor splice-site mutations in WT1 are responsible for Frasier syndrome. Nat Genet. 1997;17:467–70.
Fujita S, Sugimoto K, Miyazawa T, Yanagida H, Tabata N, Okada M, et al. A female infant with Frasier syndrome showing splice site mutation in Wilms’ tumor gene (WT1) intron 9. Clin Nephrol. 2010;73:487–91.
Zenker M, Aigner T, Wendler O, Tralau T, Müntefering H, Fenski R, et al. Human laminin beta2 deficiency causes congenital nephrosis with mesangial sclerosis and distinct eye abnormalities. Hum Mol Genet. 2004;13:2625–32.
Zenker M, Tralau T, Lennert T, Pitz S, Mark K, Madlon H, et al. Congenital nephrosis, mesangial sclerosis, and distinct eye abnormalities with microcoria: an autosomal recessive syndrome. Am J Med Genet A. 2004;130a:138–45.
Hasselbacher K, Wiggins RC, Matejas V, Hinkes BG, Mucha B, Hoskins BE, et al. Recessive missense mutations in LAMB2 expand the clinical spectrum of LAMB2-associated disorders. Kidney Int. 2006;70:1008–12.
Lin PY, Tseng MH, Zenker M, Rao J, Hildebrandt F, Lin SH, et al. Galloway-Mowat syndrome in Taiwan: OSGEP mutation and unique clinical phenotype. Orphanet J Rare Dis. 2018;13:226.
Colin E, Cong E, Mollet G, Guichet A, Gribouval O, Arrondel C, et al. Loss-of-function mutations in WDR73 are responsible for microcephaly and steroid-resistant nephrotic syndrome: Galloway-Mowat syndrome. Am J Hum Genet. 2014;95:637–48.
Gosens I, den Hollander AI, Cremers FP, Roepman R. Composition and function of the Crumbs protein complex in the mammalian retina. Exp Eye Res. 2008;86:713–26.
van den Hurk JA, Rashbass P, Roepman R, Davis J, Voesenek KE, Arends ML, et al. Characterization of the Crumbs homolog 2 (CRB2) gene and analysis of its role in retinitis pigmentosa and Leber congenital amaurosis. Mol Vis. 2005;11:263–73.
Slavotinek A, Kaylor J, Pierce H, Cahr M, DeWard SJ, Schneidman-Duhovny D, et al. CRB2 mutations produce a phenotype resembling congenital nephrosis, Finnish type, with cerebral ventriculomegaly and raised alpha-fetoprotein. Am J Hum Genet. 2015;96:162–9.
Has C, Spartà G, Kiritsi D, Weibel L, Moeller A, Vega-Warner V, et al. Integrin α3 mutations with kidney, lung, and skin disease. N. Engl J Med. 2012;366:1508–14.
López LC, Schuelke M, Quinzii CM, Kanki T, Rodenburg RJ, Naini A, et al. Leigh syndrome with nephropathy and CoQ10 deficiency due to decaprenyl diphosphate synthase subunit 2 (PDSS2) mutations. Am J Hum Genet. 2006;79:1125–9.
Hata D, Miyazaki M, Seto S, Kadota E, Muso E, Takasu K, et al. Nephrotic syndrome and aberrant expression of laminin isoforms in glomerular basement membranes for an infant with Herlitz junctional epidermolysis bullosa. Pediatrics. 2005;116:e601–7.
Patrakka J, Martin P, Salonen R, Kestilä M, Ruotsalainen V, Männikkö M, et al. Proteinuria and prenatal diagnosis of congenital nephrosis in fetal carriers of nephrin gene mutations. Lancet. 2002;359:1575–7.
Heinonen S, Ryynänen M, Kirkinen P, Penttilä I, Syrjänen K, Seppälä M, et al. Prenatal screening for congenital nephrosis in east Finland: results and impact on the birth prevalence of the disease. Prenat Diagn. 1996;16:207–13.
Boyer O, Schaefer F, Haffner D, Bockenhauer D, Hölttä T, Bérody S, et al. Management of congenital nephrotic syndrome: consensus recommendations of the ERKNet-ESPN Working Group. Nat Rev Nephrol. 2021;17:277–89.
Bérody S, Heidet L, Gribouval O, Harambat J, Niaudet P, Baudouin V, et al. Treatment and outcome of congenital nephrotic syndrome. Nephrol Dial Transpl. 2019;34:458–67.
Dufek S, Holtta T, Trautmann A, Ylinen E, Alpay H, Ariceta G, et al. Management of children with congenital nephrotic syndrome: challenging treatment paradigms. Nephrol Dial Transpl. 2019;34:1369–77.
Hölttä T, Jalanko H. Congenital nephrotic syndrome: is early aggressive treatment needed? Yes. Pediatr Nephrol. 2020;35:1985–90.
Boyer O, Bérody S. Congenital nephrotic syndrome: is early aggressive treatment needed?-No. Pediatr Nephrol. 2020;35:1991–6.
Dufek S, Ylinen E, Trautmann A, Alpay H, Ariceta G, Aufricht C, et al. Infants with congenital nephrotic syndrome have comparable outcomes to infants with other renal diseases. Pediatr Nephrol. 2019;34:649–55.
Mattoo TK. Hypothyroidism in infants with nephrotic syndrome. Pediatr Nephrol. 1994;8:657–9.
Lau KK, Chan HH, Massicotte P, Chan AK. Thrombotic complications of neonates and children with congenital nephrotic syndrome. Curr Pediatr Rev. 2014;10:169–76.
Holmberg C, Antikainen M, Rönnholm K, Ala Houhala M, Jalanko H. Management of congenital nephrotic syndrome of the Finnish type. Pediatr Nephrol. 1995;9:87–93.
Takasato M, Er PX, Chiu HS, Maier B, Baillie GJ, Ferguson C, et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature. 2015;526:564–8.
Hölttä T, Bonthuis M, Van Stralen KJ, Bjerre A, Topaloglu R, Ozaltin F, et al. Timing of renal replacement therapy does not influence survival and growth in children with congenital nephrotic syndrome caused by mutations in NPHS1: data from the ESPN/ERA-EDTA Registry. Pediatr Nephrol. 2016;31:2317–25.
Acknowledgements
We thank Dr. William Day of the University of Arizona Microscopy Core for assistance with electron microscopy, Dr. Johnathan Zuckerman at the University of California Los Angles for providing NPHS1 pathogenic variant micrograph, and Dr. Koichiro Ichimura at Juntendo University, Japan for providing colored podocytes micrograph (Fig. 3).
Author information
Authors and Affiliations
Contributions
ASA, RA, SB wrote the manuscript. ASA provided the figures, wrote, and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
AbuMaziad, A.S., Abusaleh, R. & Bhati, S. Congenital nephrotic syndrome. J Perinatol 41, 2704–2712 (2021). https://doi.org/10.1038/s41372-021-01279-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-021-01279-0
This article is cited by
-
Water-soluble vitamins and trace elements in children with chronic kidney disease stage 5d
Pediatric Nephrology (2024)
-
Effects of a novel ANLN E841K mutation associated with SRNS on podocytes and its mechanism
Cell Communication and Signaling (2023)
-
Hypogammaglobulinemia in neonates: illustrative cases and review of the literature
Journal of Perinatology (2023)