Abstract
Background:
To determine whether the presence of intra-amniotic infection and elevated proinflammatory cytokine levels in amniotic fluid (AF) are associated with failure in the newborn hearing screen (NHS) test in very preterm neonates.
Methods:
This is a retrospective cohort study of 112 premature singleton neonates born to women with preterm labor or preterm premature rupture of membranes at ≤32 wk. AF obtained through amniocentesis was cultured, and interleukin-6 (IL-6) and IL-8 levels were determined.
Results:
Fourteen (12.5%) neonates failed the NHS test. The prevalence of a positive AF culture was 40% (45/112). Multiple logistic regression analyses indicated that intra-amniotic infection was significantly associated with failure in the NHS test after adjusting for baseline covariates such as maternal white blood cell count (WBC) and periventricular leukomalacia. However, the IL-6 and IL-8 levels in AF were not significantly associated with hearing screen failure. Moreover, neither gestational age at birth nor birth weight was associated with NHS failure.
Conclusion:
The presence of intra-amniotic infection, but not elevated levels of AF IL-6 and IL-8, may contribute to the risk for failure in the NHS test in very preterm neonates. This finding suggests that intra-amniotic infection in utero might contribute to the development of congenital sensorineural hearing loss.
Similar content being viewed by others
Main
Preterm birth is the leading cause of neonatal morbidity, especially neurological sequelae, and mortality (1).One of the most devastating neurodevelopmental impairmentsexperienced by preterm neonates during infancy is sensorineural hearing loss (SNHL), which leads to functional disability if not detected early. The prevalence of SNHL was estimated to be 1.8–12.8%in very-low-birth-weight neonates (2). Despite the clinical relevance and higher prevalence of SNHL in preterm neonates, little is known about the risk factors and preventive measure, especially antenatally, for the development of SNHL.
Intra-amniotic infection and/or inflammation (defined as elevated proinflammatory cytokine levels in amniotic fluid (AF)), are strongly associated with preterm birth and is detected in up to 42% of women with preterm labor and preterm premature rupture of membranes (PROM) (3,4,5,6). In addition, it has also been well established that intra-amniotic infection and/or inflammation are involved in the pathogenesis of adverse long-term neonatal neurologic and pulmonary sequelae, particularly in the immature organs (7,8,9). However, no rigorous clinical investigation addressing the relation between intra-amniotic infection and/or inflammation and the development of SNHL has been conducted yet. Recently, Leung et al. demonstrated that the fetal inflammatory response syndrome, defined as the presence of funisitis and/or cord serum interelukin-6 (IL-6) > 18.2 pg/ml, was significantly associated with an increased risk of hearing screen failure (10). Interestingly, funisitis or elevated IL-6 levels in cord blood have been reported to be closely related to infection in AF (11,12). These findings suggest the possible association of intra-amniotic infection with the pathogenesis of SNHL. It is important to clarify this issue because it can potentially lead to the development and advancement of strategies, particularly starting antenatally, for preventing and treating SNHL. Indeed, pharmacological treatments to enhance anti-inflammatory properties reduced cochlear spiral ganglion neuronal deaths in infant rat models of bacterial meningitis, thereby attenuating SNHL (13). Given this result, we aimed to determine whether the presence of intra-amniotic infection and elevated pro-inflammatory cytokine levels in AF are associated with hearing screen failure in very preterm neonates. We selected AF IL-6 and IL-8 as markers of intra-amniotic inflammation because these cytokines have been reported to be the strongest predictors of adverse pregnancy outcomes, including intra-amniotic infection, preterm birth, and long-term neurologic and pulmonary sequelae (4,7,8,14,15).
Methods
This retrospective cohort study was approved by the local ethics committee at Seoul National University Bundang Hospital (IRB no. B-1105/128-102). The study population consisted of all consecutive women with preterm labor or preterm PROM, and their infants born between 23 + 0 and 32 + 0 wk gestation who were admitted to a neonatal intensive care unit (NICU) between July 2004 and September 2012. The inclusion criteria were as follows: (i) singleton gestation; (ii) infants whose mothers underwent trans-abdominal amniocentesis for the evaluation of the microbiologic and inflammatory status of the amniotic cavity on admission; (iii) infants who survived for at least 90 d after birth; and (iv) infants who underwent hearing screening examinations. The exclusion criteria were twin or higher-order multiple births, major congenital anomalies, and transfer to another hospital after amniocentesis. Gestational age was calculated from the first day of the last menstrual period, and confirmed with first- or second-trimester ultrasonography. Written informed consent was obtained from all patients for the amniocentesis procedure, and for the collection and use of AF samples.
Trans-abdominal amniocentesis was performed with ultrasound guidance, and the aseptically obtained AF was cultured for aerobic and anaerobic bacteria and genital mycoplasma, and analyzed for white blood cell count (WBC) according to previously detailed methods (16). The remaining AF was centrifuged at 1,500g at 4 °C for 10 min, and the supernatant was aliquoted and immediately stored at −70 °C until assayed. IL-6 and IL-8 in stored AF were measured by using an enzyme-linked immunosorbent assay human DuoSet Kit (R&D Systems, Minneapolis, MN). The range of the IL-6 and IL-8 standard curves was 7.8–600 and 31.2–2,000 pg/ml, respectively. All samples were assayed in duplicate. The intra- and inter-assay coefficients of variation for two different proteins were <10% and <15%, respectively. Amniocentesis has been routinely offered at our institution to patients who were admitted with the diagnosis of preterm labor and intact membranes or preterm PROM less than 34.0 wk to assess the microbial and inflammatory status in amniotic cavity or fetal lung maturation. However, the decision for completing an amniocentesis was at the discretion of the attending obstetrician. The results of AF culture and WBC, but not AF IL-6 and IL-8, were available to the managing obstetricians and neonatologists. The C-reactive protein level and WBC were measured immediately after amniocentesis, and details about these measurements have been previously described (16).
Electronic medical records on hearing screen failure in one or both ears of the included preterm singleton neonates were reviewed twice by two audiologists (J.H.R. and J.H.P.), who were blinded to the results of AF analysis and to the maternal and neonatal details. The automated auditory brainstem response (AABR) was mostly performed as a newborn hearing screen (NHS) test; the otoacoustic emission test was done in 14 cases for which AABR was not available. If the infant failed either the AABR or the otoacoustic emission test, the same test was repeated. Neonates who failed two consecutive screenings of either AABR or otoacoustic emission, in one or both ears, were classified as having a hearing screen failure. The results were recorded as either “refer” (further confirmation tests, such as an auditory brainstem response threshold test, needed) or “pass” (normal). Therefore, the primary outcome measure was a refer result from one or both ears before hospital discharge.
Tissue samples of the placenta were obtained from the placental membranes, umbilical cord, and chorionic plate. They were fixed in 10% neutral-buffered formalin and embedded in paraffin. Sections of the tissue blocks were stained with hemotoxylin and eosin. Histopathologic examination was performed by experienced board-certified pathologists who were blinded to the clinical information. The presence of acute inflammation was noted and classified as grade 1 or 2 ( Table 1 ) according to the criteria previously published (17). Acute histologic chorioamnionitis was defined as the presence of acute inflammatory change in any tissue sample (amnion, chorion-decidua, umbilical cord, or chorionic plate). Funisitis was diagnosed in the presence of neutrophil infiltration into the umbilical vessel walls or Wharton’s jelly. For each placenta, the total grade of histologic chorioamnionitis was calculated as the sum of histologic grades in the amnion (0–2), chorion-decidua (0–2), umbilical cord (0–2), and chorionic plate (0–2). Grade 0 (noninflamed placenta) was defined as a total grade of histologic chorioamnionitis of zero (0), grade 1 (mild chorioamnionitis) as a total grade of histologic chorioamnionitis less than or equal to the median value (2) in placentas with leukocyte infiltration, and grade 2 (severe chorioamnionitis) as a total grade of histologic chorioamnionitis above the median value (2).
Intra-amniotic infection was defined as a positive AF culture. Diagnostic criteria and management of preterm labor and preterm PROM have been previously described in detail elsewhere (16,18). Decisions regarding the use and type of prophylactic antibiotics were left to the discretion of the attending obstetrician and treatments with tocolytics, corticosteroids, and antibiotics were started after amniocentesis. Diagnostic criteria of clinical chorioamnionitis have been described in detail elsewhere (18). Proven sepsis was identified when causative organisms of systemic inflammation were identified on at least two sets of blood cultures. Respiratory distress syndrome, bronchopulmonary dysplasia, intraventricular hemorrhage, periventricular leukomalacia (PVL), and necrotizing enterocolitis were also diagnosed according to the definitions previously described in detail (4).
A previous study reported the incidence of failure in the NHS test for very preterm infants to be 16% (10). We assumed that the SD for AF IL-6 would be 15.0 ng/ml based on our previously published study (19). Therefore, by using an α of 0.05 and a β of 0.2 (power = 80%), and assuming a ratio of 1:5 for infants who failed vs. those who passed the hearing screen, we retrospectively calculated that a total sample size of 108 was needed to detect a 11 ng/ml difference in AF IL-6 between the two groups. In our previous study (20), the SD of AF IL-8 was shown to be 4.62 ng/ml, and thus, a sample size of 108 is expected to be large enough for the AF IL-8 to yield a significant difference.
Bivariate analysis was conducted by using Student’s t-test, Mann-Whitney U-test, Fisher’s exact test, or χ2 test, as appropriate. The normality for continuous variables in groups was determined by using the Shapiro-Wilk test. Multivariable logistic regression was then performed to determine the independent relation between positive AF cultures and cytokine levels in AF and failure in the NHS test after adjusting for baseline variables. Variables showing a statistically significant correlation or a tendency of correlation with refer on the NHS test in univariate analysis (P < 0.15) were included in the logistic analysis. All statistical analyses were performed with SPSS for Windows version 21.0 (IBM SPSS, Chicago, IL). All reported P values are two-sided, and P values <0.05 were considered statistically significant.
Results
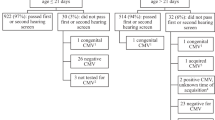
During the study period, 112 women with preterm labor (n = 66) or preterm PROM (n = 46) and their neonates met the eligibility criteria and were included in the final analysis. The mean gestational age at birth of the study cohort was 29.2 wk (SD, 1.9 wk; range, 24.0–32.0 wk), and the mean birth weight was 1,369 g (SD, 347 g; range, 680–2,275 g). Microorganisms were isolated from 45 of 112 AF samples (40.2%), including 20 from women with preterm labor and intact membranes and 25 from women with preterm PROM. The microorganisms isolated from the amniotic cavity included Ureaplasmaurealyticum (n = 38), Mycoplasma hominis (n = 31), Escherichia coli (n = 1), Staphylococcus aureus (n = 1), Streptococcus agalactiae (n = 1), Streptococcus mitis (n = 1), Lactobacillus sp. (n = 1), unidentified Gram-negative rods (n = 1), and unidentified Gram-positive cocci (n = 2). Polymicrobial invasion was present in 31 of 45 cases (68.8%). The NHS tests were conducted at mean corrected gestational age of 36.5 ± 5.3 wk. Ninety-eight neonates (87.5%) passed the NHS test in both ears, whereas 14 (12.5%) failed the NHS test. Among 14 neonates with failure in the NHS test, 5 (4.2%) had a unilateral failure and 9 (7.6%) had a bilateral failure.
Table 2 shows the maternal and obstetric characteristics of the study population according to the NHS results. Mothers delivering infants who showed refer on the NHS test had a significantly higher prevalence of preterm PROM, funisitis, and clinical chorioamnionitis, and a lower median maternal WBC than mothers who delivered infants that passed the NHS tests. However, there were no significant differences in the maternal demographics, antenatal medications, antibiotic-to-delivery interval, duration of antibiotic use, and histologic chorioamnionitis between the two groups. In addition, the mean maternal WBC counts were not significantly different between women with preterm labor and intact membranes and women with preterm PROM (preterm labor, mean ± SD, 13,039 ± 4,019 cells/mm3 vs. preterm PROM, mean ± SD, 12,309 ± 3,714 cells/mm3; P = 0.332). There were no correlations between failure in the NHS test and the severity of inflammation in the amnion, chorion-decidua, and chorionic plate, and histologic chorioamnionitis as reflected by the total grade points. However, we found a correlation between failure in the NHS test and the grade of inflammation in the umbilical cord ( Table 3 ).
Table 4 shows the AF culture results and the AF WBC, IL-6 level, and IL-8 level according to the NHS results. Mothers delivering infants who failed the NHS test had a higher rate of positive AF cultures than mothers who delivered infants that passed NHS. However, AF WBC and the levels of AF IL-6 and IL-8 were not significantly different between the two groups.
Table 5 shows the neonatal characteristics according to the NHS results. In bivariate analyses, neither gestational age at birth nor birth weight was associated with failure in the NHS test. Moreover, the median peak serum bilirubin level during NICU admission and the rates of low Apgar scores and major treatments (such as mechanical ventilation; continuous positive airway pressure; and use of surfactants, diuretics, and aminoglycoside antibiotics) were comparable between infants who passed and those who failed the NHS test. However, PVL had a statistically significant association with refer on the NHS test (P = 0.005).
Multivariable logistic regression analysis was performed to estimate the association between positive AF cultures or elevated levels of IL-6 and IL-8 in AF and failure in the NHS test, with adjustment for baseline variables. The following parameters, at a P value of <0.15 based on the univariate analyses, were included in the multivariate logistic regression analysis:pretermPROM, funisitis, clinical chorioamnionitis, positive AF cultures, maternal WBC, and PVL. Before performing logistic regression analysis, we checked for multicollinearity among the variables by using a χ2 test. Significant correlations were found among preterm PROM, funisitis, clinical chorioamnionitis, and positive AF cultures in bivariable analyses (P = 0.044 to <0.001), and then they were analyzed in separate models ( Table 6 ). After adjustments for maternal WBC and PVL, a positive AF culture was significantly associated with failure in the NHS test ( Table 6 ). Moreover, in the logistic regression models, preterm PROM, funisitis, and clinical chorioamnionitis were significantly associated with failure in the NHS test after adjustments for maternal WBC and PVL. When these six variables were simultaneously entered into logistic regression analysis, PVL was independently associated with failure in the NHS test, whereas elevated maternal WBC was statistically significantly associated with a reduced incidence of refer on the NHS test.
Discussion
This is the first report to document the relation between intra-amniotic infection and hearing screen failure that places a neonate at risk for SNHL. Although we did not include follow-up confirmatory test results in our analyses, two consecutive failures in AABR screening tests at or before discharge from the NICU strongly indicated a high-risk group for permanent SNHL (21). Furthermore, auditory brainstem response (ABR) is a very sensitive indicator of alteration of brain or neural maturation. Earlier measurement of AABR as a screening test at NICU discharge might be superior to confirmatory ABRdone at 2–3 mo after birth in elucidating subtly but potentially contributing factors to SNHL that were possibly tolerated before the confirmatory ABR tests (22).
Our data clearly demonstrated that the presence of intra-amniotic infection, but not elevated levels of proinflammatory cytokines in AF, may contribute to the risk for hearing screen failure in very preterm infants. We also noted that funisitis, which is well known to occur more frequently among patients with positive AF culture results (3,5,11,16), was significantly associated with hearing screen failure. Collectively, these observations suggest that fetal exposure to infection in AF might be involved in the pathophysiological mechanism of SNHL, and that antenatal use of antimicrobial therapy for the treatment and prevention of infection in AF might be helpful in reducing the likelihood of SNHL in preterm neonates.
In the context of prenatal exposure to infection/inflammation, the current study showed that the presence of intra-amniotic infection remained associated with hearing screen failure even after adjustments for confounders. In addition, we observed that the presence of funisitis significantly increased the probability of hearing screen failure, which is consistent with the recent study by Leung et al. (10). However, in the present study, in contrast to intra-amniotic infection and funisitis, elevated IL-6 and IL-8 levels in AF did not show any association with hearing screen failure. This was an unexpected result, as IL-6 and IL-8 levels in AF are one of the most sensitive markers for predicting intra-amniotic infection (4,14,19). Although we cannot explicitly elucidate the discrepancy between intra-amniotic infection and elevated cytokine levels in AF in terms of their association with the hearing screening failure, this discrepancy can be interpreted as follows: a large inoculum of bacteria, but not proinflammatory cytokine per se, in the amniotic cavity, which can gain directly access to the fetus, may contribute significantly to the development of hearing loss. In fact, infection in the AF may be primarily an infection of the fetus rather than of the mother (23), and is the most advanced and serious stage of ascending intrauterine infection during which fetal infection, as manifested by funisitis/elevated IL-6 level in cord blood, occurs (24). On the contrary, elevated cytokine levels in AF (15,25) are found (i) during the early stage when infection is limited to the chorion-decidua (histologic chorioamnionitis), and thus, microorganisms may not gain access to the amniotic cavity and fetus; (ii) in patients with negative AF culture results caused by the small size of the inocula, in whom microbial fingerprints are detected in AF onpolymerase chain reaction (26,27); and (iii) in cases that are caused by inflammatory mechanisms other than infection, which may not be involved in the etiology of hearing screen failure. In support of our view, Forner et al. found that the risk of hearing deficit increased with a viral load in blood at birth of ≥17,000 copies/ml in 34 newborns with congenital asymptomatic cytomegalovirus infection born to women with primary cytomegalovirus infection during pregnancy (28). Similar explanations can be applied to our findings of a lack of association between failure in the NHS test and histologic chorioamnionitis, which is mainly a maternal host response (24).
How intra-amniotic infection may predispose premature neonates to the subsequent development of SNHL can only be speculated at present. Direct spreading of microorganisms from the infected AF into the fetal circulation could cause fetal bacteremia. The resulting sepsis may cause ischemic damage to the inner ear due to septic emboli, as previously documented with meningitis (29). Alternatively, a similar phenomenon as the matrix metalloproteinase 9-mediated opening of the blood–brain barrier stimulated by infection/inflammation (30,31) could also occur for the blood–labyrinth barrier, allowing the direct spread of microorganisms into the labyrinth. It may be possible that infection/inflammation in the cerebrospinal fluid may reach the labyrinth through the cochlear aqueduct (32).
Our finding that preterm PROM, funisitis, clinical chorioamnionitis, and positive AF cultures were significantly correlated with each other is in accordance with the results of many previous studies (3,5,11,16). From the clinical point of view, our observation that these parameters were significantly associated with hearing screen failure is important because it allows clinicians to identify infants, before the time of birth, who are at a higher risk for abnormal findings on the NHS test and the subsequent development of SNHL. In the present study, the rate of a refer result on the NHS test in infants born at ≤32.0 wk gestation is similar to those of recent large-scale studies (10,33). On the contrary, the prevalence of intra-amniotic infection in the current study was 40%, which is significantly higher than the 26.4% (P < 0.001) that Yoon et al. previously reported in infants born at ≤35.0 wk gestation (11). However, the discrepancy between these two studies is natural, given that the prevalence of positive AF cultures and elevated proinflammatory cytokine levels in AF have been inversely related to gestational age at amniocentesis and at delivery (3,4,5,6,18).
In the literature, bronchopulmonary dysplasia, intraventricular hemorrhage, and PVL have been reported as risk factors for SNHL and neurosensory impairment in very-low-birth-weight infants (34,35,36,37). In accordance with the previous reports, our results also showed that PVL is independently associated with hearing screen failure. This relation cannot be explained clearly; however, several plausible explanations can be postulated about the primary mechanism that plays a role in the co-occurrence of these diseases, including the following: (i) shared common risk factors for these diseases, such as mechanical ventilation and prolonged supplemental oxygen use, and (ii) the complications/events (e.g., cardiorespiratory instability and infection) after or during receiving a common procedure (e.g., patent ductus arteriosus ligation).
A unique finding of this study is that elevated maternal WBC was independently significantly associated with a reduced probability of a refer result on the NHS test. This is an unexpected result, disagreeing with our other observation that demonstrated that maternal inflammation as shown by a maternal C-reactive protein levels at amniocentesis was not related to the abnormal hearing screen results. We cannot explain the underlying mechanism for the beneficial effect of elevated maternal WBC on the auditory system in neonates; however, it is presumably because hematologic change in peripheral blood (e.g., WBC elevation) reflecting maternal immunity might affect the production of maternal IgG and then the transfer of passive immunity during the perinatal and postnatal periods (38,39), contributing to vulnerability to infections associated with the development of hearing loss (1).
The current study has several limitations. First, the results of AF culture and WBC were routinely reported to the managing obstetricians, which might have affected the timing of delivery and starting of antibiotic therapy. However, it is unlikely that this bias would have influenced our finding because the culture results in AF were not usually available until at least 48–72 h after the amniocentesis, and gestational age at birth and use of antibiotics were considered in our statistical analyses. Second, we could not determine whether intra-amniotic infection and IL-6 or IL-8 levels in AF correlated with SNHL, because of the low prevalence of SNHL and the limited number of patients. Further studies are needed to examine whether permanent hearing loss is initially mediated by infection or inflammatory process in AF. Third, the current study did not include cultures for viruses (i.e., TORCH) that have been known to be involved in the pathophysiological mechanism of hearing loss development (1,40). Fourth, we did not perform a full characterization of pro- and anti-inflammatory cytokines in AF, but only measured the most relevant inflammatory markers (i.e., IL-6 and IL-8) consistently reported to be the best predictors of intra-amniotic infection, preterm birth, and long-term neurologic sequelae (4,6,7,14,15).
In conclusion, the presence of intra-amniotic infection significantly increased the probability of abnormal NHS results, whereas elevated proinflammatory cytokine levels in AF (i.e., intra-amniotic inflammation) did not. Future studies are needed to investigate the impact of early administration of antibiotics, by which therapeutic levels can be achieved in the fetal compartment, immediately after the diagnosis of intra-amniotic infection on NHS outcomes and hearing impairment in neonates.
Statement of Financial Support
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant No. HI 14C1798).
Disclosure
The authors report no conflict of interest.
References
Mwaniki MK, Atieno M, Lawn JE, Newton CR. Long-term neurodevelopmental outcomes after intrauterine and neonatal insults: a systematic review. Lancet 2012;379:445–52.
Cristobal R, Oghalai JS. Hearing loss in children with very low birth weight: current review of epidemiology and pathophysiology. Arch Dis Child Fetal Neonatal Ed 2008;93:F462–8.
Shim SS, Romero R, Hong JS, et al. Clinical significance of intra-amniotic inflammation in patients with preterm premature rupture of membranes. Am J Obstet Gynecol 2004;191:1339–45.
Park KH, Kim SN, Oh KJ, Lee SY, Jeong EH, Ryu A. Noninvasive prediction of intra-amniotic infection and/or inflammation in preterm premature rupture of membranes. Reprod Sci 2012;19:658–65.
Yoon BH, Romero R, Moon JB, et al. Clinical significance of intra-amniotic inflammation in patients with preterm labor and intact membranes. Am J Obstet Gynecol 2001;185:1130–6.
Jung HJ, Park KH, Kim SN, et al. Non-invasive prediction of intra-amniotic inflammation in women with preterm labor. Ultrasound Obstet Gynecol 2011;37:82–7.
Yoon BH, Romero R, Park JS, et al. Fetal exposure to an intra-amniotic inflammation and the development of cerebral palsy at the age of three years. Am J Obstet Gynecol 2000;182:675–81.
Yoon BH, Romero R, Jun JK, et al. Amniotic fluid cytokines (interleukin-6, tumor necrosis factor-alpha, interleukin-1 beta, and interleukin-8) and the risk for the development of bronchopulmonary dysplasia. Am J Obstet Gynecol 1997;177:825–30.
Hagberg H, Gressens P, Mallard C. Inflammation during fetal and neonatal life: implications for neurologic and neuropsychiatric disease in children and adults. Ann Neurol 2012;71:444–57.
Leung JC, Cifra CL, Agthe AG, Sun CJ, Viscardi RM. Antenatal factors modulate hearing screen failure risk in preterm infants. Arch Dis Child Fetal Neonatal Ed 2016;101:56–61.
Yoon BH, Romero R, Park JS, et al. The relationship among inflammatory lesions of the umbilical cord (funisitis), umbilical cord plasma interleukin 6 concentration, amniotic fluid infection, and neonatal sepsis. Am J Obstet Gynecol 2000;183:1124–9.
Romero R, Gomez R, Ghezzi F, et al. A fetal systemic inflammatory response is followed by the spontaneous onset of preterm parturition. Am J Obstet Gynecol 1998;179:186–93.
Meli DN, Coimbra RS, Erhart DG, et al. Doxycycline reduces mortality and injury to the brain and cochlea in experimental pneumococcal meningitis. Infect Immun 2006;74:3890–6.
Jacobsson B, Mattsby-Baltzer I, Andersch B, et al. Microbial invasion and cytokine response in amniotic fluid in a Swedish population of women with preterm prelabor rupture of membranes. Acta Obstet Gynecol Scand 2003;82:423–31.
Jacobsson B, Mattsby-Baltzer I, Hagberg H. Interleukin-6 and interleukin-8 in cervical and amniotic fluid: relationship to microbial invasion of the chorioamniotic membranes. BJOG 2005;112:719–24.
Lee SY, Park KH, Jeong EH, Oh KJ, Ryu A, Kim A. Intra-amniotic infection/inflammation as a risk factor for subsequent ruptured membranes after clinically indicated amniocentesis in preterm labor. J Korean Med Sci 2013;28:1226–32.
Yoon BH, Romero R, Kim CJ, et al. Amniotic fluid interleukin-6: a sensitive test for antenatal diagnosis of acute inflammatory lesions of preterm placenta and prediction of perinatal morbidity. Am J Obstet Gynecol 1995;172:960–70.
Ryu A, Park KH, Oh KJ, Lee SY, Jeong EH, Park JW. Predictive value of combined cervicovaginal cytokines and gestational age at sampling for intra-amniotic infection in preterm premature rupture of membranes. Acta Obstet Gynecol Scand 2013;92:517–24.
Oh KJ, Park KH, Kim SN, Jeong EH, Lee SY, Yoon HY. Predictive value of intra-amniotic and serum markers for inflammatory lesions of preterm placenta. Placenta 2011;32:732–6.
Jung EY, Park KH, Lee SY, Ryu A, Joo JK, Park JW. Predicting outcomes of emergency cerclage in women with cervical insufficiency using inflammatory markers in maternal blood and amniotic fluid. Int J Gynaecol Obstet 2016;132:165–9.
Benito-Orejas JI, Ramírez B, Morais D, Almaraz A, Fernández-Calvo JL. Comparison of two-step transient evoked otoacoustic emissions (TEOAE) and automated auditory brainstem response (AABR) for universal newborn hearing screening programs. Int J Pediatr Otorhinolaryngol 2008;72:1193–201.
Ptok M. Early detection of hearing impairment in newborns and infants. Dtsch Arztebl Int 2011;108:426–31.
Eschenbach DA. Amniotic fluid infection and cerebral palsy. Focus on the fetus. JAMA 1997;278:247–8.
Romero R, Chaiworapongsa T, Espinoza J. Micronutrients and intrauterine infection, preterm birth and the fetal inflammatory response syndrome. J Nutr 2003;133(5 Suppl 2):1668S–73S.
McNamara MF, Wallis T, Qureshi F, Jacques SM, Gonik B. Determining the maternal and fetal cellular immunologic contributions in preterm deliveries with clinical or subclinical chorioamnionitis. Infect Dis Obstet Gynecol 1997;5:273–9.
Romero R, Miranda J, Chaiworapongsa T, et al. A novel molecular microbiologic technique for the rapid diagnosis of microbial invasion of the amniotic cavity and intra-amniotic infection in preterm labor with intact membranes. Am J Reprod Immunol 2014;71:330–58.
Hitti J, Riley DE, Krohn MA, et al. Broad-spectrum bacterial rDNA polymerase chain reaction assay for detecting amniotic fluid infection among women in premature labor. Clin Infect Dis 1997;24:1228–32.
Forner G, Abate D, Mengoli C, Palù G, Gussetti N. High cytomegalovirus (CMV) DNAemia predicts CMV sequelae in asymptomatic congenitally infected newborns born to women with primary infection during pregnancy. J Infect Dis 2015;212:67–71.
Harburg TD, Leonard HA, Kimbrough RC 3rd, Jones SR. Group B streptococcal meningitis appearing as acute deafness in an adult. Arch Neurol 1984;41:214–6.
Leib SL, Leppert D, Clements J, Täuber MG. Matrix metalloproteinases contribute to brain damage in experimental pneumococcal meningitis. Infect Immun 2000;68:615–20.
Leppert D, Leib SL, Grygar C, Miller KM, Schaad UB, Holländer GA. Matrix metalloproteinase (MMP)-8 and MMP-9 in cerebrospinal fluid during bacterial meningitis: association with blood-brain barrier damage and neurological sequelae. Clin Infect Dis 2000;31:80–4.
Bhatt SM, Lauretano A, Cabellos C, et al. Progression of hearing loss in experimental pneumococcal meningitis: correlation with cerebrospinal fluid cytochemistry. J Infect Dis 1993;167:675–83.
Lieu JE, Champion G. Prediction of auditory brainstem reflex screening referrals in high-risk infants. Laryngoscope 2006;116:261–7.
Martínez-Cruz CF, Poblano A, Fernández-Carrocera LA. Risk factors associated with sensorineural hearing loss in infants at the neonatal intensive care unit: 15-year experience at the National Institute of Perinatology (Mexico City). Arch Med Res 2008;39:686–94.
Meyer C, Witte J, Hildmann A, et al. Neonatal screening for hearing disorders in infants at risk: incidence, risk factors, and follow-up. Pediatrics 1999;104(4 Pt 1):900–4.
Vohr BR, Wright LL, Dusick AM, et al. Neurodevelopmental and functional outcomes of extremely low birth weight infants in the National Institute of Child Health and Human Development Neonatal Research Network, 1993-1994. Pediatrics 2000;105:1216–26.
Robertson CM, Howarth TM, Bork DL, Dinu IA. Permanent bilateral sensory and neural hearing loss of children after neonatal intensive care because of extreme prematurity: a thirty-year study. Pediatrics 2009;123:e797–807.
Kumar V, Sharma A. Neutrophils: Cinderella of innate immune system. Int Immunopharmacol 2010;10:1325–34.
Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M. IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol 2012;2012:985646.
Beswick R, Driscoll C, Kei J. Monitoring for postnatal hearing loss using risk factors: a systematic literature review. Ear Hear 2012;33:745–56.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, E., Choi, B., Rhee, J. et al. Relation between amniotic fluid infection or cytokine levels and hearing screen failure in infants at 32 wk gestation or less. Pediatr Res 81, 349–355 (2017). https://doi.org/10.1038/pr.2016.219
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2016.219
This article is cited by
-
Predictive potential of various plasma inflammation-, angiogenesis-, and extracellular matrix remodeling-associated mediators for intra-amniotic inflammation and/or microbial invasion of the amniotic cavity in preterm labor
Archives of Gynecology and Obstetrics (2024)
-
Proteomic analysis of plasma to identify novel biomarkers for intra-amniotic infection and/or inflammation in preterm premature rupture of membranes
Scientific Reports (2023)
-
Cord blood transforming growth factor-β-induced as predictive biomarker of retinopathy of prematurity in preterm infants
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
Identifying potential biomarkers related to pre-term delivery by proteomic analysis of amniotic fluid
Scientific Reports (2020)
-
A Protein Microarray Analysis of Plasma Proteins for the Prediction of Spontaneous Preterm Delivery in Women with Preterm Labor
Reproductive Sciences (2020)