Abstract
Aceruloplasminemia is an autosomal recessive disorder of iron metabolism characterized by diabetes, retinal degeneration, and neurologic symptoms. Affected patients evidence marked parenchymal iron accumulation in conjunction with an absence of circulating serum ceruloplasmin and molecular genetic analysis reveals inherited mutations in the ceruloplasmin gene. Taken together with earlier studies that characterized ceruloplasmin as a ferroxidase and recent work indicating an essential role for a homologous multicopper oxidase in iron metabolism in Saccharomyces cerevisiae, these findings reveal an essential role for ceruloplasmin in human iron metabolism. The presence of neurologic symptoms in patients with aceruloplasminemia is unique among the characterized disorders of iron metabolism, and recent findings indicate that astrocyte-specific ceruloplasmin gene expression is critical for iron metabolism and neuronal survival in the retina and basal ganglia. The discovery of this disease provides new insights into the pathways of CNS iron metabolism of direct relevance to a variety of nutritional and genetic disorders of childhood.
Similar content being viewed by others
Main
The introduction of electrophoresis for the separation of biologic molecules earned Arne Tiselius the Nobel Prize in chemistry and ushered in a new era of investigation resulting in the characterization of the proteins of human plasma. In the course of such studies, Holmberg and Laurell(1) used ammonium sulfate to obtain a bluish green fraction of plasma from which they isolated a protein containing most of the copper found in serum. Owing to the sky blue color of the purified protein they named their discovery caeruloplasmin(1). In this same year, Wilson's disease was found to result from hepatic copper accumulation and soon thereafter Scheinberg and Gitlin(2) demonstrated a deficiency of ceruloplasmin in the sera of patients with this disorder. Despite intense investigation the biologic role of ceruloplasmin has only recently been revealed by identification of patients with aceruloplasminemia.
PHYSIOLOGIC FUNCTION
Ceruloplasmin is a multicopper oxidase, using the facile electron chemistry of copper to oxidize selected substrates via a single-step, four-electron reduction of oxygen. These enzymes have been highly conserved in evolution and are characterized by the presence of three types of spectroscopically distinct copper ions(3). In ceruloplasmin, three of these copper ions form a trinuclear cluster responsible for oxygen binding and activation during enzymatic catalysis(4). Recent determination of the crystal structure of ceruloplasmin provides a model for the process of electron transfer from the oxidized substrate to molecular oxygen and permits delineation of the amino acids which directly contribute to the trinuclear copper center(5).
Ceruloplasmin is an abundant serum protein that contains greater than 95% of the copper present in the blood plasma(6). This protein is synthesized in hepatocytes and secreted into the serum with six atoms of copper incorporated during biosynthesis(7). Ceruloplasmin is an acute-phase reactant, and the serum concentration increases during infection, inflammation, and trauma largely due to effects on hepatic ceruloplasmin gene expression(8). The intracellular copper concentration does not affect the rate of synthesis or secretion of apoceruloplasmin; however, a failure to incorporate copper during biosynthesis results in the secretion into the plasma of an unstable apoprotein which is devoid of oxidase activity and rapidly turned over(9,10). In patients with Wilson's disease an inability to transfer copper into the secretory pathway for holoceruloplasmin biosynthesis results in a decrease in serum ceruloplasmin concentration secondary to the rapid degradation of the secreted apoprotein. Consistent with this observation, the Wilson's disease gene encodes a copper-transporting ATPase which has been shown to reside in the trans-Golgi network of hepatocytes(11–13) (Fig. 1).
Localization of the Wilson's disease protein in HepG2 cells. Immunofluorescence using a polyclonal antibody reveals abundant Wilson's disease protein in the trans-Golgi network of the secretory pathway. This protein transports copper into this pathway for incorporation into apoceruloplasmin before secretion of the holoprotein into the plasma.
Although ceruloplasmin can oxidize several substrates in vitro, Freiden and colleagues demonstrated that this protein plays a direct role in the mobilization of iron from parenchymal tissues and the subsequent oxidation and incorporation of ferric iron into circulating apotransferrin(14,15). In a series of elegant metabolic studies, Cartwright and colleagues demonstrated that pigs made copper deficient by dietary restriction developed iron overload in conjunction with a diminution of circulating ceruloplasmin. These copper-deficient animals had impairment in tissue iron release into the plasma that was restored after administration of ceruloplasmin(16,17). Support for the role of ceruloplasmin as a ferroxidase also comes from genetic experiments in Saccharomyces cerevisiae, which reveal that high affinity iron uptake in this organism is dependent upon the presence of the homologous multicopper oxidase Fet3(18). Fet3 functions as a ferroxidase at the plasma membrane, oxidizing iron in the extracellular milieu and acting in concert with a membrane permease to promote iron uptake(19). The Wilson's disease protein homolog has also been identified in this organism and shown to be essential for delivery of copper to Fet3 in the yeast secretory pathway, revealing a remarkable evolutionary conservation of the function of these copper proteins in iron homeostasis(20,21).
The complete amino acid sequence of ceruloplasmin was determined by Putnam and colleagues using protein chemistry(22). These studies demonstrated the single chain structure of this protein and revealed an internal triplicate homology within the sequence. Subsequently, human ceruloplasmin cDNA clones were isolated and characterized which confirmed the amino acid sequence determined by earlier protein analysis(23–25). Studies using these clones localized the ceruloplasmin gene to chromosome 3q23-25 and revealed abundant, tissue-specific expression of this gene in the liver. The human ceruloplasmin gene consists of 19 exons spanning greater than 45 kb(26). A processed pseudogene for ceruloplasmin has been detected on chromosome 8 by in situ hybridization(27). Although not expressed, the occurrence of this sequence in the human genome has methodologic implications in the detection of ceruloplasmin gene mutations in patients with aceruloplasminemia(vide infra).
METABOLISM
Evidence of ceruloplasmin synthesis can be detected in the human fetal liver and yolk sac as early as the 5th wk of gestation(28). The serum concentration rises steadily throughout gestation, and half-life analysis at birth indicates that nearly all the ceruloplasmin present in newborn serum is derived from endogenous synthesis(28). The serum concentration of ceruloplasmin remains low for the first several postnatal months, and adult levels are not achieved until the 1st y of life. This rise in serum ceruloplasmin concentration parallels the onset of biliary copper excretion and represents the slow postnatal increase in holoceruloplasmin synthesis. Metabolic studies using131 I indicate a t1/2 of 5.5 d for human ceruloplasmin(29,30). Kinetic studies using radioactive copper yield a similar half-life, indicating little or no exchange of ceruloplasmin copper in tissues(31). This observation suggests that, although ceruloplasmin contains almost all the copper found in the serum, this protein plays no direct role in copper transport.
About 10% of circulating ceruloplasmin is apoprotein, which is synthesized and secreted from the liver without copper and turns over rapidly in the circulation with a t1/2 of 5 h. Biosynthetic studies reveal that copper availability serves only to determine the ratio of apo- and holoceruloplasmin secreted from the cell and thus at steady state the serum ceruloplasmin concentration represents the sum of the these two forms, determined by intracellular copper availability and the observed differences in their turnover(32,33). Given the differences in half-lives, these data indicate that under normal circumstances the rate of synthesis and secretion of the apo-and holoprotein is roughly equivalent. As anticipated from such a model, an increase in the hepatic copper pool of normal individuals results in a sustained increase in the concentration of holoceruloplasmin in the serum(34).
ACERULOPLASMINEMIA
In addition to the decreased serum ceruloplasmin in patients with Wilson's disease, earlier studies revealed that approximately 15% of obligate heterozygotes have a slightly decreased serum ceruloplasmin and a 3-fold increase in hepatic copper concentration(35,36). In the course of these investigations, individuals were identified with inherited differences in serum ceruloplasmin concentration where the incidence and inheritance pattern suggested this was not due to the heterozygote state of Wilson's disease(37,38). Analysis of one such family led to the designation of hereditary hypoceruloplasminemia and suggested that a decrease in serum ceruloplasmin concentration to 50% of normal is not associated with any clinical abnormality(39). Despite these earlier findings, two reports appeared in the literature which described individuals with dementia, diabetes, retinal degeneration, and basal ganglia symptoms in association with a complete absence of circulating serum ceruloplasmin(40,41). Analysis of the serum ceruloplasmin concentrations in families of affected patients revealed an autosomal recessive inheritance pattern, and molecular genetic studies have demonstrated the presence of mutations in the ceruloplasmin gene(42–48).
A typical pedigree in one such family from Belfast, Ireland, is shown in Figure 2 along with the DNA sequence from exon 13, indicating a single nucleotide deletion in affected family members. As would be anticipated from the incidence and inheritance pattern of this disease, consanguinity has been observed in most cases. Thus far, six distinct mutations have been characterized in the ceruloplasmin gene, each of which would result in an alteration of the open reading frame such that the amino acid ligands in the carboxyl-terminus determinating the essential trinuclear copper cluster would be eliminated (Table 1). As ceruloplasmin synthesized without this region would be incapable of binding copper, these results are consistent with the observed lack of detectable ceruloplasmin oxidase activity in the serum of affected patients.
Pedigree and nucleotide sequence analysis in a kindred with aceruloplasminemia. (A) A pedigree of an affected family is shown with serum ceruloplasmin concentrations as indicated(mg/dL). Solid squares indicate proband and his younger brother.(B) Sequence ladder of normal and mutant allele in heterozygote V-6 as well as the affected individuals. (Reproduced from Harris et al.(46): Quarterly Journal of Medicine, 89:355-359, 1996, with permission from Oxford University Press.)
PATHOLOGY
Patients with aceruloplasminemia have normal liver histology on biopsy with no evidence of hepatic copper accumulation but a marked increase in iron in both hepatocytes and reticuloendothelial cells(49). The serum ferritin concentration is elevated, the serum iron concentration is decreased, and most patients have a mild anemia. Autopsy findings reveal iron in the pancreas with accumulation in the islets of Langerhans in association with a selective loss of insulin-producing cells(49). All patients evidence abnormal glucose metabolism and most develop insulin-dependent diabetes.
The disruption of iron homeostasis in aceruloplasminemia is understood by considering the role of ceruloplasmin as a plasma ferroxidase. Within the iron cycle ceruloplasmin functions to oxidize ferrous iron for subsequent transfer to plasma apotransferrin. This transferrin-bound iron is then predominantly used for Hb biosynthesis during erythropoiesis. Once in the circulation the Hb-bound iron remains within the erythrocyte until these cells are turned over in the reticuloendothelial system, and the iron is then subsequently released and recycled to transferrin (Fig. 3). Ceruloplasmin is essential for the release of iron from these reticuloendothelial storage sites. The low serum iron in patients with aceruloplasminemia reflects this impairment in iron release from the reticuloendothelial system; however, the mild anemia indicates that alternative mechanisms of iron oxidation are available to provide sufficient transferrin-bound ferric iron for erythropoiesis. In addition, the absence of ceruloplasmin leads to an accumulation of ferrous iron in the plasma, which is rapidly removed from the circulation by the liver and other tissues, analogous to what is observed in atransferrinemia, hereditary hemochromatosis, and transfusion-dependent iron overload(50). In the healthy individual 5% of the normal serum ceruloplasmin concentration is sufficient to sustain plasma iron turnover rates, explaining why abnormalities of iron homeostasis are not observed in patients with Wilson's disease(17). Although all patients with aceruloplasminemia have low plasma copper owing to the absence of this protein in the serum, copper metabolism in such patients is entirely normal, indicating that there is no essential role for ceruloplasmin in copper homeostasis.
Role of ceruloplasmin as a plasma ferroxidase. The systemic iron cycle is shown illustrating the function of ceruloplasmin (Cp) in mediating iron release from the reticuloendothelial system and subsequent oxidation and incorporation into transferrin (Tf). (Reproduced with permission from Harris et al(6): American Journal of Clinical Nutrition 67:972S-977S.)
NEUROPATHOLOGY
The predominant clinical features in aceruloplasminemia are neurologic and result from progressive degeneration of the basal ganglia. T2-weighted magnetic resonance brain imaging reveals decreased signal intensity in the basal ganglia, consistent with iron deposition in these regions. Furthermore, examination of brain tissue at autopsy demonstrates abundant iron deposition in neurons and glia within the basal ganglia and thalamus with clear evidence of neuronal loss in these same tissues(49). Patients exhibit clinical symptoms consistent with these findings, including subcortical dementia, dystonia, dysarthria, and movement disorders. Although visual symptoms are not prominent clinically, ophthalmologic examination does reveal iron deposition and photoreceptor loss in the peripheral regions of the retina. Involvement of the CNS distinguishes aceruloplasminemia from other known disorders of iron homeostasis and indicates that ceruloplasmin plays an essential role in brain iron metabolism. Interestingly, these pathologic findings of iron accumulation are similar to those observed in Hallervorden-Spatz disease, but no information is currently available on the mechanisms of iron accumulation in this rare disease(51).
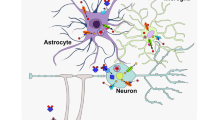
Ceruloplasmin does not cross the blood-brain barrier, and this implies that any direct role for this protein in CNS iron metabolism must involve expression within this site. Indeed examination of ceruloplasmin gene expression within the CNS reveals astrocyte-specific gene expression throughout the cerebral microvasculature(52,53). Ceruloplasmin is also expressed by astrocytes surrounding specific neurons in the substantia nigra and other basal ganglia as well as by astrocytic Müller glia cells in the retina(52–54) (Fig. 4). Biosynthetic studies using primary cultures of neurons and glia confirm this cell-specific expression and indicate that ceruloplasmin is synthesized and secreted by astrocytes with kinetics identical to that observed in hepatocytes(52). Although a glycophosphotidylinositol-linked membrane form of ceruloplasmin is detected in an astrocyte cell line, pulse-chase studies indicate that all of the ceruloplasmin synthesized by primary astrocyte cultures is secreted into the media, making the relevance of this anchored form unclear at this time(55).
Ceruloplasmin gene expression in the substantia nigra. (A and B) In situ hybridization of human substantia nigra pars compacta reveals ceruloplasmin mRNA in astrocytes (arrows) surrounding dopaminergic neurons(mdn). Original magnification, ×1000 (A),×2000 (B). (Reproduced from Klomp and Gitlin(53): Human Molecular Genetics, 5:1898-1996, 1996, with permission from Oxford University Press.)
The observation that ceruloplasmin is synthesized in the CNS suggests that the absence of this protein at this site in affected patients is directly responsible for the iron accumulation and neuronal degeneration. As such it is envisioned that astrocyte-secreted ceruloplasmin may function in an analogous manner as in the plasma, oxidizing ferrous iron after the release of this metal from the cerebral microvasculature (Fig. 5). In aceruloplasminemia, the inappropriate accumulation of ferrous iron by glial cells may then lead to cell-specific injury with subsequent loss of glia-derived trophic factors essential for neuronal survival. Alternatively it is also possible that the accumulation of ferrous iron may directly result in oxidant-mediated injury to the CNS. This model would be consistent with observations in patients with aceruloplasminemia indicative of free-radical mediated tissue injury including an increase in plasma lipid peroxidation and an interruption of peroxisomal β-oxidation of fatty acids(56,57). It is also possible that neuronal degeneration results from impaired iron delivery to neurons owing to the diminution of transferrin-bound ferric iron. Elucidation of the precise function of ceruloplasmin in the CNS as well as mechanisms of neuronal loss in affected patients must await the development of a suitable animal model in which to examine this process directly.
Model of ceruloplasmin function in CNS. Ferrous iron (Fe2+) which accumulates in the absence of ceruloplasmin ferroxidase activity is shown as resulting in tissue injury via free radical generation. (Reproduced with permission from Harris et al.(6): American Journal of Clinical Nutrition 67:972S-977S.)
Although aceruloplasminemia is a fatal neurodegenerative disease, clinical and laboratory studies indicate that iron accumulation precedes the onset of neurologic symptoms by several years. This observation suggests a potential therapeutic approach, and a preliminary study of the iron-chelator deferoxamine did indicate a reduction in body iron stores as well as amelioration of neurologic symptoms in a severely affected patient(58). If these preliminary observations are supported by clinical trials, this approach may prove useful as a preventative therapy in asymptomatic individuals. As the complete absence of serum ceruloplasmin in diagnostic of this disorder, the development of an effective therapy would make it feasible to consider population screening.
CONCLUSION
The presence of mutations in the ceruloplasmin gene in patients with CNS iron accumulation and neuronal degeneration indicates an essential role for this protein in brain iron homeostasis and neuronal survival. The pathophysiology of aceruloplasminemia suggests the presence of an iron cycle in the CNS analogous to that previously characterized in the systemic circulation. Presumably the presence of such a cycle serves to minimize the effects of systemic iron deficiency on CNS function. This observation has important implications for the cognitive defects observed in children with sustained iron deficiency in the perinatal period where previous studies have suggested a critical window for iron repletion within the CNS(59). In addition, recent studies suggest that iron-mediated free-radical injury is a central mechanism underlying hypoxicischemic brain damage in the newborn infant(60). As holoceruloplasmin biosynthesis is diminished in the fetus and newborn infant it will be worth pursuing the role of this protein in iron homeostasis in the newborn brain. Thus the discovery of aceruloplasminemia provides a starting point for elucidation of the mechanisms of CNS iron homeostasis during development and may permit the creation of novel therapeutic approaches to prevent or ameliorate neurodegeneration in a variety of childhood diseases where abnormalities in brain iron metabolism have been implicated.
References
Holmberg CG, Laurell CB 1948 Investigations in serum copper. II. Isolation of the copper containing protein, and a description of some of its properties. Acta Chem Scand 2: 550–556.
Scheinberg IH, Gitlin D 1952 Deficiency of ceruloplasmin in patients with hepatolenticular degeneration (Wilson's disease). Science 116: 484–485.
Adman ET 1991 Copper protein structures. Adv Protein Chem 42: 145–197.
Calabrese L, Carbonaro M, Giovanni M 1993 Presence of coupled trinuclear copper cluster in mammalian ceruloplasmin is essential for efficient electron transfer to oxygen. J Biol Chem 264: 6183–6187.
Zaitseva I, Zaitsev V, Cara G, Mojhkov K, Bar B, Lindley PF 1996 The x-ray structure of human ceruloplasmin at 3.1 D resolution. Nature of the copper centers. J Biol Inorgan Chem 1: 15–23.
Harris ZL, Klomp LW, Gitlin JD 1998 Aceruloplasminemia: an inherited neurodegenerative disease with impaired iron homeostasis. Am J Clin Nutr 67: 972S–977S.
Ortel TL, Takahashi N, Putnam FW 1984 Structural model of human ceruloplasmin based on internal triplication, hydrophilic/hydrophobic character, and secondary structure of domains. Proc Natl Acad Sci USA 81: 4761–4765.
Gitlin JD 1988 Transcriptional regulation of ceruloplasmin gene expression during inflammation. J Biol Chem 263: 6281–6287.
Holtzman NA, Guamnitz BM 1970 Identification of an apoceruloplasmin-like substance in the plasma of copper-deficient rats. J Biol Chem 245: 2350–2353.
Holtzman NA, Guamnitz BM 1970 Studies of the rate of release and turnover of ceruloplasmin and apoceruloplasmin in rat plasma. J Biol Chem 245: 2354–2358.
Hung IH, Suzuki M, Yamaguchi Y, Yuan DS, Klausner RD, Gitlin JD 1997 Biochemical characterization of the Wilson disease protein and functional expression in the yeast Saccharomyces cerevisiae. J Biol Chem 272: 21461–21466.
Yang X-L, Miura N, Kawarada Y, Terada K, Petrukhin K, Conrad GT, Sugiyama T 1997 Two forms of Wilson disease protein produced by alternative splicing are localized in distinct cellular compartments. Biochem J 326: 897–902.
Terada K, Nakako T, Yang X-L, Iida M, Aiba N, Minamiya T, Nakai M, Sakaki T, Miura N, Sugiyama T 1998 Restoration of holoceruloplasmin synthesis in LEC rat after infusion of recombinant adenovirus bearing WND cDNA. J Biol Chem 273: 1815–1820.
Osaki S, Johnson DA, Frieden E 1966 The possible significance of the ferrous oxidase activity of ceruloplasmin in normal human serum. J Biol Chem 241: 2746–2751.
Osaki S, Johnson DA, Frieden E 1971 The mobilization of iron from the perfused mammalian liver by a serum copper enzyme, ferroxidase I. J Biol Chem 246: 3018–3023.
Lee GR, Nacht S, Lukens JN, Cartwright GE 1968 Iron metabolism in copper-deficient swine. J Clin Invest 47: 2058–2069.
Roeser HP, Lee GR, Nacht S, Cartwright GE 1970 The role of ceruloplasmin in iron metabolism. J Clin Invest 49: 2408–2417.
Askwith C, Eide D, VanHo A, Bernard PS, Li L, Davis-Kaplan S, Sipe DM, Kaplan J 1994 The FET3 gene of S. cerevisiae encodes a multicopper oxidase required for ferrous iron uptake. Cell 76: 403–410.
Stearman R, Yuan DS, Yamaguchi-Iwa Y, Klausner RD, Dancis A 1996 A permease-oxidase complex involved in high-affinity iron uptake in yeast. Science 271: 1552–1557.
Yuan DS, Stearman R, Dancis A, Dunn T, Beeler T, Klausner RD 1995 The Menkes/Wilson disease gene homologue in yeast provides copper to a ceruroloplasmin-like oxidase required for iron uptake. Proc Natl Acad Sci USA 92: 2632–2636.
Yuan DS, Dancis A, Klausner RD 1997 Restriction of copper export in Saccharomyces cerevisiae to a late Golgi or post-Golgi compartment in the secretory pathway. J Biol Chem 272: 25787–25793.
Takahashi N, Ortel TL, Putnam FW 1984 Single-chain structure of human ceruloplasmin: the complete amino acid sequence of the whole molecule. Proc Natl Acad Sci USA 81: 390–394.
Koschinsky ML, Funk WD, VanOost BA, MacGillivray RT 1986 Complete cDNA sequence of human preceruloplasmin. Proc Natl Acad Sci USA 83: 5086–5090.
Yang F, Naylor SL, Lum JB, Cutshaw S, McCombs JL, Naberhaus KH, McGill JR, Adrian GS, Moore CM, Barnett DR, Bowman BH 1986 Characterization, mapping, and expression of the human ceruloplasmin gene. Proc Natl Acad Sci USA 83: 3257–3261.
Mercer JF, Grimes A 1986 Isolation of a human ceruloplasmin cDNA clone that includes the N-terminal leader sequence. FEBS Lett 203: 185–190.
Daimon M, Yamatani K, Igarashi M, Fukase N, Kawanami T, Kato T, Tominaga M, Sasaki H 1995 Fine structure of the human ceruloplasmin gene. Biochem Biophys Res Commun 208: 1028–1035.
Koschinsky ML, Chow BK, Schwartz J, Hamerton JL, MacGillivray RT 1987 Isolation and characterization of a processed gene for human ceruloplasmin. Biochemistry 26: 7760–7767.
Gitlin D, Biasucci A 1969 Development of γG,γA, γM, βIC-βIA, C1 esterase inhibitor, ceruloplasmin, transferrin, hemopexin, haptoglobin, fibrinogen, plasminogen,α1-antitrypsin, orosomucoid, β-lipoprotein,α2-macroglobulin, and prealbumin in the human conceptus. J Clin Invest 48: 1433–1446.
Sternlieb I, Morell AG, Tucker WD, Greene MW, Scheinberg IH 1961 The incorporation of copper into ceruloplasmin in vivo: studies with copper-64 and copper-67. J Clin Invest 40: 1834–1840.
Kekki M, Koskelo P, Nikkila EA 1966 Turnover of iodine-131-labelled ceruloplasmin in human beings. Nature 209: 1252–1253.
Gitlin D, Janeway CA 1960 Turnover of the copper and protein moieties of ceruloplasmin. Nature 185: 693
Sato M, Gitlin JD 1991 Mechanisms of copper incorporation during the biosynthesis of human ceruloplasmin. J Biol Chem 266: 5128–5134.
Gitlin JD, Schroeder JJ, Lee-Ambrose LM, Cousins RJ 1992 Mechanisms of ceruloplasmin biosynthesis in normal and copper-deficient rats. Biochem J 282: 835–839.
Holtzman NA, Elliott DA 1966 Copper intoxication. Report of a case with observations on ceruloplasmin. N Engl J Med 275: 347–352.
Sternlieb I, Scheinberg IH 1968 Prevention of Wilson's disease in asymptomatic patients. N Engl J Med 278: 352–359.
Gibbs K, Walshe JM 1979 A study of the caeruloplasmin concentrations found in 75 patients with Wilson's disease, their kinships and various control groups. Q J Med 48: 447–463.
Cartwright GR, Markowitz H, Shields S, Wintrobe MM 1960 Studies on copper metabolism. XXIX. Am J Med 28: 555–563.
Cox DW 1966 Factors influencing serum ceruloplasmin levels in normal individuals. J Lab Clin Med 68: 893–904.
Edwards CQ, Williams DM, Cartwright GE 1979 Hereditary hypoceruloplasminemia. Clin Genet 15: 311–316.
Miyajima H, Nishimura Y, Mizoguchi K, Sakanoto M, Shimizu T, Honda N 1987 Familial apoceruloplasmin deficiency associated with blepharospasm and retinal degeneration. Neurology 37: 761–767.
Logan JI, Harveyson KB, Wisdom GB, Hughes AE, Archbold GP 1994 Hereditary caeruloplasmin deficiency, dementia and diabetes mellitus. Q J Med 87: 663–670.
Harris ZL, Takahashi Y, Miyajima H, Serizawa M, MacGillivray RTA, Gitlin JD 1995 Aceruloplasminemia: Molecular characterization of this disorder of iron metabolism. Proc Natl Acad Sci USA 92: 2539–2543.
Yoshida K, Furihata K, Takeda S, Nakamura A, Yamamoto K, Hiyamuta S, Ikeda S, Shimizu N, Yanagisawa N 1995 A mutation in the ceruloplasmin gene is associated with systemic hemosiderosis humans. Nat Genet 9: 267–272.
Takahashi Y, Miyajima H, Shirabe S, Nagataki S, Suenaga A, Gitlin JD 1996 Characterization of a nonsense mutation in the ceruloplasmin gene resulting in diabetes and neurodegenerative disease. Hum Mol Genet 5: 81–84.
Daimon M, Kato T, Kawanami T, Tominaga M, Igarashi M, Yamatani K, Sasaki H 1995 A nonsense mutation of the ceruloplasmin deficiency with diabetes mellitus. Biochem Biophys Res Commun 217: 89–95.
Harris ZL, Migas MD, Hughes AE, Logan JI, Gitlin JD 1995 Familial dementia due to a frameshift mutation in the caeruloplasmin gene. Q J Med 89: 355–359.
Okamoto N, Wada S, Oga T, Kawabata Y, Baba Y, Habu D, Takeda Z, Wada Y 1996 Hereditary ceruloplasmin deficiency with hemosiderosis. Hum Genet 97: 755–758.
Yazaki M, Yoshida K, Nakamura A, Furihata K, Yonekawa M, Okabe T, Yamashita N, Ohta M, Ikeda S 1998 A novel splicing mutation in the ceruloplasmin gene responsible for hereditary ceruloplasmin deficiency with hemosiderosis. J Neurol Sci 156: 30–34.
Morita H, Ikeda S, Yamamoto K, Morita S, Yoshida K, Nomoto S, Kato M, Yanagisawa N 1995 Hereditary ceruloplasmin deficiency with hemosiderosis: a clinicopathological study of a Japanese family. Ann Neurol 37: 646–656.
Craven CM, Alexander J, Eldridge M, Kushner JP, Bernstein S, Kaplan J 1987 Tissue distribution and clearance kinetics of non-transferrin-found iron in the hypotransferrinemic mouse: a rodent model for hemochromatosis. Proc Natl Acad Sci USA 84: 3457–3461.
Swaiman KF 1991 Hallervorden-Spatz syndrome and brain iron metabolism. Arch Neurol 48: 1285–1293.
Klomp LWJ, Farhangrazi ZS, Dugan LL, Gitlin JD 1996 Ceruloplasmin gene expression in the murine central nervous system. J Clin Invest 98: 207–215.
Klomp LWJ, Gitlin JD 1996 Expression of the ceruloplasmin gene in the human retina and brain: implications for a pathogenic model in aceruloplasminemia. Hum Mol Genet 5: 1989–1996.
Levin LA, Geszvain KM 1998 Expression of ceruloplasmin in the retina: induction after optic nerve crush. Invest Ophthalmol Vis Sci 39: 157–163.
Patel BN, David S 1997 A novel glycosylphosphatidylinositol-anchored form of ceruloplasmin is expressed by mammalian astrocytes. J Biol Chem 272: 20185–20190.
Miyajima H, Yoshitoma T, Masahiro S, Kaneko E, Gitlin JD 1996 Increased plasma lipid peroxidation in patients with aceruloplasminemia. Free Radic Biol Med 20: 757–760.
Miyajima H, Adachi J, Tatsuno Y, Takahashi Y, Fujimoto M, Kaneko E, Gitlin JD 1998 Increased very long-chain fatty acids in erythrocyte membranes of patients with aceruloplasminemia. Neurology 50: 130–136.
Miyajima H, Takahashi Y, Kamata T, Shimizu H, Sakai N, Gitlin JD 1997 Use of desferrioxamine in the treatment of aceruloplasminemia. Ann Neurol 41: 404–407.
Lozoff B, Jimenez E, Wolf AW 1991 Long-term developmental outcome of infants with iron deficiency. N Engl J Med 325: 687–694.
Dorrepaal CA, Berger HM, Benders MJ, van Zoeren-Grobben D, Van de Bor M, Van Bel F 1996 Nonprotein-bound iron in postasphyxial reperfusion injury of the newborn. Pediatrics 98: 883–889.
Acknowledgements
I gratefully acknowledge the collaboration of my colleagues Z. Leah Harris, Leo W.J. Klomp, Hiroaki Miyajima, Yoshitomo Takahashi, Hiroshi Morita, Ross MacGillivray, John Logan, and Anne Hughes.
Author information
Authors and Affiliations
Additional information
Supported in part by funds from National Institutes of Health Grants DK44464 and HL41536. J.D.G. is the recipient of a Burroughs Wellcome Fund Scholar Award in Experimental Therapeutics.Recipient of the Society for Pediatric Research 1998 E. Mead Johnson Award for Research in Pediatrics and presented at the 1998 Annual Meeting of the Pediatric Academic Societies, New Orleans, LA.
Rights and permissions
About this article
Cite this article
Gitlin, J. Aceruloplasminemia. Pediatr Res 44, 271–276 (1998). https://doi.org/10.1203/00006450-199809000-00001
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199809000-00001
This article is cited by
-
Lysosomal iron modulates NMDA receptor-mediated excitation via small GTPase, Dexras1
Molecular Brain (2016)
-
Role of hepcidin and its downstream proteins in early brain injury after experimental subarachnoid hemorrhage in rats
Molecular and Cellular Biochemistry (2016)
-
Iron transport across the blood–brain barrier: development, neurovascular regulation and cerebral amyloid angiopathy
Cellular and Molecular Life Sciences (2015)
-
Ceruloplasmin and what it might do
Journal of Neural Transmission (2007)
-
Disturbance of iron metabolism in Parkinson’s disease — ultrasonography as a biomarker
Neurotoxicity Research (2006)