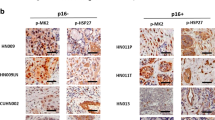
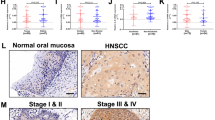
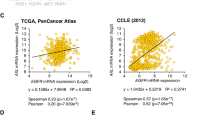
Abstract
The inhibitor-κB kinase-nuclear factor-κB (IKK-NF-κB) and epidermal growth factor receptor-activator protein-1 (EGFR-AP1) pathways are often co-activated and promote malignant behavior, but the underlying basis for this relationship is unclear. Resistance to inhibitors of IKKβ or EGFR is observed in head and neck squamous cell carcinomas (HNSCC). Here, we reveal that both IKKα and β contribute to nuclear activation of canonical and alternate NF-κB/REL family transcription factors, and overexpression of signal components that enhance co-activation of the EGFR-AP1 pathway. We observed that IKKα and IKKβ exhibit increased protein expression, nuclear localization, and phosphorylation in HNSCC tissues and cell lines. Individually, IKK activity varied among different cell lines, but overexpression of both IKKs induced the strongest NF-κB activation. Conversely, siRNA knock down of both IKKs significantly decreased nuclear localization and phosphorylation of canonical RELA and IκBα and alternative p52 and RELB subunits. Knock down of both IKKs more effectively inhibited NF-κB activation, broadly modulated gene expression and suppressed cell proliferation and migration. Global expression profiling revealed that NF-κB, cytokine, inflammatory response and growth factor signaling are among the top pathways and networks regulated by IKKs. Importantly, IKKα and IKKβ together promoted the expression and activity of transforming growth factor α, EGFR and AP1 transcription factors cJun, JunB and Fra1. Knock down of AP1 subunits individually decreased 8/15 (53%) of IKK-targeted genes sampled and similarly inhibited cell proliferation and migration. Mutations of NF-κB and AP1-binding sites abolished or decreased IKK-induced interleukin-8 (IL-8) promoter activity. Compounds such as wedelactone with dual IKK inhibitory activity and geldanomycins that block IKKα/β and EGFR pathways were more active than IKKβ-specific inhibitors in suppressing NF-κB activation and proliferation and inducing cell death. We conclude that IKKα and IKKβ cooperatively activate NF-κB and EGFR/AP1 networks of signaling pathways and contribute to the malignant phenotype and the intrinsic or acquired therapeutic resistance of HNSCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hacker H, Karin M . Regulation and function of IKK and IKK-related kinases. Sci STKE 2006; 2006: 13.
Scheidereit C . IkappaB kinase complexes: gateways to NF-kappaB activation and transcription. Oncogene 2006; 25: 6685–6705.
Karin M, Greten FR . NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 2005; 5: 749–759.
Van Waes C . Nuclear factor-kappaB in development, prevention, and therapy of cancer. Clin Cancer Res 2007; 13: 1076–1082.
Duffey DC, Chen Z, Dong G, Ondrey FG, Wolf JS, Brown K et al. Expression of a dominant-negative mutant inhibitor-kappaBalpha of nuclear factor-kappaB in human head and neck squamous cell carcinoma inhibits survival, proinflammatory cytokine expression, and tumor growth in vivo. Cancer Res 1999; 59: 3468–3474.
Sunwoo JB, Chen Z, Dong G, Yeh N, Crowl Bancroft C, Sausville E et al. Novel proteasome inhibitor PS-341 inhibits activation of nuclear factor-kappa B, cell survival, tumor growth, and angiogenesis in squamous cell carcinoma. Clin Cancer Res 2001; 7: 1419–1428.
Delhase M, Hayakawa M, Chen Y, Karin M . Positive and negative regulation of IkappaB kinase activity through IKKbeta subunit phosphorylation. Science 1999; 284: 309–313.
Adams J . The proteasome: a suitable antineoplastic target. Nat Rev Cancer 2004; 4: 349–360.
Nagashima K, Sasseville VG, Wen D, Bielecki A, Yang H, Simpson C et al. Rapid TNFR1-dependent lymphocyte depletion in vivo with a selective chemical inhibitor of IKKbeta. Blood 2006; 107: 4266–4273.
Allen C, Saigal K, Nottingham L, Arun P, Chen Z, Van Waes C . Bortezomib-induced apoptosis with limited clinical response is accompanied by inhibition of canonical but not alternative nuclear factor-{kappa}B subunits in head and neck cancer. Clin Cancer Res 2008; 14: 4175–4185.
Chen Z, Ricker JL, Malhotra PS, Nottingham L, Bagain L, Lee TL et al. Differential bortezomib sensitivity in head and neck cancer lines corresponds to proteasome, nuclear factor-kappaB and activator protein-1 related mechanisms. Mol Cancer Ther 2008; 7: 1949–1960.
Argiris A, Duffy A, Kummar S, Simone NL, Arai Y, Kim S et al. Early tumor progression associated with enhanced EGFR signaling with bortezomib, cetuximab, and radiotherapy for head and neck cancer. Clin Cancer Res 2011; 17: 5755–5764.
Liu B, Park E, Zhu F, Bustos T, Liu J, Shen J et al. A critical role for I kappaB kinase alpha in the development of human and mouse squamous cell carcinomas. Proc Natl Acad Sci USA 2006; 103: 17202–17207.
Marinari B, Moretti F, Botti E, Giustizieri ML, Descargues P, Giunta A et al. The tumor suppressor activity of IKKalpha in stratified epithelia is exerted in part via the TGF-beta antiproliferative pathway. Proc Natl Acad Sci USA 2008; 105: 17091–17096.
Lee TL, Yeh J, Van Waes C, Chen Z . Epigenetic modification of SOCS-1 differentially regulates STAT3 activation in response to interleukin-6 receptor and epidermal growth factor receptor signaling through JAK and/or MEK in head and neck squamous cell carcinomas. Mol Cancer Ther 2006; 5: 8–19.
Pernas FG, Allen CT, Winters ME, Yan B, Friedman J, Dabir B et al. Proteomic signatures of epidermal growth factor receptor and survival signal pathways correspond to gefitinib sensitivity in head and neck cancer. Clin Cancer Res 2009; 15: 2361–2372.
Sharafinski ME, Ferris RL, Ferrone S, Grandis JR . Epidermal growth factor receptor targeted therapy of squamous cell carcinoma of the head and neck. Head Neck 2010; 32: 1412–1421.
Grandis JR, Tweardy DJ . Elevated levels of transforming growth factor alpha and epidermal growth factor receptor messenger RNA are early markers of carcinogenesis in head and neck cancer. Cancer Res 1993; 53: 3579–3584.
Shaulian E, Karin M . AP-1 as a regulator of cell life and death. Nat Cell Biol 2002; 4: E131–E136.
Ondrey FG, Dong G, Sunwoo J, Chen Z, Wolf JS, Crowl-Bancroft CV et al. Constitutive activation of transcription factors NF-(kappa)B, AP-1, and NF-IL6 in human head and neck squamous cell carcinoma cell lines that express pro-inflammatory and pro-angiogenic cytokines. Mol Carcinog 1999; 26: 119–129.
Mishra A, Bharti AC, Saluja D, Das BC . Transactivation and expression patterns of Jun and Fos/AP-1 super-family proteins in human oral cancer. Int J Cancer 2010; 126: 819–829.
Park E, Liu B, Xia X, Zhu F, Jami WB, Hu Y . Role of IKKalpha in skin squamous cell carcinomas. Future Oncol 2011; 7: 123–134.
Ndlovu MN, Van Lint C, Van Wesemael K, Callebert P, Chalbos D, Haegeman G et al. Hyperactivated NF-{kappa}B and AP-1 transcription factors promote highly accessible chromatin and constitutive transcription across the interleukin-6 gene promoter in metastatic breast cancer cells. Mol Cell Biol 2009; 29: 5488–5504.
Liu B, Xia X, Zhu F, Park E, Carbajal S, Kiguchi K et al. IKKalpha is required to maintain skin homeostasis and prevent skin cancer. Cancer Cell 2008; 14: 212–225.
Yang X, Lu H, Yan B, Romano RA, Bian Y, Friedman J et al. DeltaNp63 versatilely regulates a Broad NF-kappaB gene program and promotes squamous epithelial proliferation, migration, and inflammation. Cancer Res 2011; 71: 3688–3700.
Yamamoto Y, Yin MJ, Gaynor RB . IkappaB kinase alpha (IKKalpha) regulation of IKKbeta kinase activity. Mol Cell Biol 2000; 20: 3655–3666.
Zandi E, Rothwarf DM, Delhase M, Hayakawa M, Karin M . The IkappaB kinase complex (IKK) contains two kinase subunits, IKKalpha and IKKbeta, necessary for IkappaB phosphorylation and NF-kappaB activation. Cell 1997; 91: 243–252.
Younes F, Quartey EL, Kiguwa S, Partridge M . Expression of TNF and the 55-kDa TNF receptor in epidermis, oral mucosa, lichen planus and squamous cell carcinoma. Oral Dis 1996; 2: 25–31.
Madge LA, May MJ . Classical NF-kappaB activation negatively regulates noncanonical NF-kappaB-dependent CXCL12 expression. J Biol Chem 2010; 285: 38069–38077.
Basak S, Hoffmann A . Crosstalk via the NF-kappaB signaling system. Cytokine Growth Factor Rev 2008; 19: 187–197.
Barre B, Perkins ND . The Skp2 promoter integrates signaling through the NF-kappaB, p53, and Akt/GSK3beta pathways to regulate autophagy and apoptosis. Mol Cell 2010; 38: 524–538.
Barre B, Perkins ND . Phosphorylation of the p52 NF-kappaB subunit. Cell Cycle 2010; 9: 4774–4775.
Wolf MJ, Seleznik GM, Zeller N, Heikenwalder M . The unexpected role of lymphotoxin beta receptor signaling in carcinogenesis: from lymphoid tissue formation to liver and prostate cancer development. Oncogene 2010; 29: 5006–5018.
Madge LA, Kluger MS, Orange JS, May MJ . Lymphotoxin-alpha 1 beta 2 and LIGHT induce classical and noncanonical NF-kappa B-dependent proinflammatory gene expression in vascular endothelial cells. J Immunol 2008; 180: 3467–3477.
Savinova OV, Hoffmann A, Ghosh G . The Nfkb1 and Nfkb2 proteins p105 and p100 function as the core of high-molecular-weight heterogeneous complexes. Mol Cell 2009; 34: 591–602.
Scheidereit C . NF-kappaB and DNA Damage Keystone Symposia, NF-kappaB Signaling and Biology: From Bench to Bedside 2012. Whistler, British Columbia, Canada.
Lam LT, Davis RE, Pierce J, Hepperle M, Xu Y, Hottelet M et al. Small molecule inhibitors of IkappaB kinase are selectively toxic for subgroups of diffuse large B-cell lymphoma defined by gene expression profiling. Clin Cancer Res 2005; 11: 28–40.
Kobori M, Yang Z, Gong D, Heissmeyer V, Zhu H, Jung YK et al. Wedelolactone suppresses LPS-induced caspase-11 expression by directly inhibiting the IKK complex. Cell Death Differ 2004; 11: 123–130.
Hertlein E, Wagner AJ, Jones J, Lin TS, Maddocks KJ, Towns WH et al. 17-DMAG targets the nuclear factor-kappaB family of proteins to induce apoptosis in chronic lymphocytic leukemia: clinical implications of HSP90 inhibition. Blood 2010; 116: 45–53.
Hu Y, Baud V, Delhase M, Zhang P, Deerinck T, Ellisman M et al. Abnormal morphogenesis but intact IKK activation in mice lacking the IKKalpha subunit of IkappaB kinase. Science 1999; 284: 316–320.
Ammirante M, Luo JL, Grivennikov S, Nedospasov S, Karin M . B-cell-derived lymphotoxin promotes castration-resistant prostate cancer. Nature 2010; 464: 302–305.
Lam LT, Davis RE, Ngo VN, Lenz G, Wright G, Xu W et al. Compensatory IKKalpha activation of classical NF-kappaB signaling during IKKbeta inhibition identified by an RNA interference sensitization screen. Proc Natl Acad Sci USA 2008; 105: 20798–20803.
Basak S, Shih VF, Hoffmann A . Generation and activation of multiple dimeric transcription factors within the NF-kappaB signaling system. Mol Cell Biol 2008; 28: 3139–3150.
Fusco AJ, Savinova OV, Talwar R, Kearns JD, Hoffmann A, Ghosh G . Stabilization of RelB requires multidomain interactions with p100/p52. J Biol Chem 2008; 283: 12324–12332.
Hinz M, Arslan SC, Scheidereit C . It takes two to tango: IkappaBs, the multifunctional partners of NF-kappaB. Immunol Rev 2012; 246: 59–76.
Anest V, Cogswell PC, Baldwin AS . IkappaB kinase alpha and p65/RelA contribute to optimal epidermal growth factor-induced c-fos gene expression independent of IkappaBalpha degradation. J Biol Chem 2004; 279: 31183–31189.
Cohen J, Chen Z, Lu SL, Yang XP, Arun P, Ehsanian R et al. Attenuated transforming growth factor beta signaling promotes nuclear factor-kappaB activation in head and neck cancer. Cancer Res 2009; 69: 3415–3424.
Bian Y, Hall B, Sun ZJ, Molinolo A, Chen W, Gutkind JS et al. Loss of TGF-beta signaling and PTEN promotes head and neck squamous cell carcinoma through cellular senescence evasion and cancer-related inflammation. Oncogene 2012; 31: 3322–3332.
Freudlsperger C, Bian Y, Contag Wise S, Burnett J, Coupar J, Yang X et al. TGF-β and NF-κB signal pathway cross-talk is mediated through TAK1 and SMAD7 in a subset of head and neck cancers. Oncogene 2013; 32: 1549–1559.
Krappmann D, Wegener E, Sunami Y, Esen M, Thiel A, Mordmuller B et al. The IkappaB kinase complex and NF-kappaB act as master regulators of lipopolysaccharide-induced gene expression and control subordinate activation of AP-1. Mol Cell Biol 2004; 24: 6488–6500.
Gomard T, Michaud HA, Tempe D, Thiolon K, Pelegrin M, Piechaczyk M . An NF-kappaB-dependent role for JunB in the induction of proinflammatory cytokines in LPS-activated bone marrow-derived dendritic cells. PloS one 2010; 5: e9585.
Carayol N, Chen J, Yang F, Jin T, Jin L . States D, et al. A dominant function of IKK/NF-kappaB signaling in global lipopolysaccharide-induced gene expression. J Biol Chem 2006; 281: 31142–31151.
Anest V, Hanson JL, Cogswell PC, Steinbrecher KA, Strahl BD, Baldwin AS . A nucleosomal function for IkappaB kinase-alpha in NF-kappaB-dependent gene expression. Nature 2003; 423: 659–663.
Zerbini LF, Wang Y, Cho JY, Libermann TA . Constitutive activation of nuclear factor kappaB p50/p65 and Fra-1 and JunD is essential for deregulated interleukin 6 expression in prostate cancer. Cancer Res 2003; 63: 2206–2215.
Debinski W, Gibo DM . Fos-related antigen 1 (Fra-1) pairing with and transactivation of JunB in GBM cells. Cancer Biol Ther 2011; 11: 254–262.
Bivona TG, Hieronymus H, Parker J, Chang K, Taron M, Rosell R et al. FAS and NF-kappaB signalling modulate dependence of lung cancers on mutant EGFR. Nature 2011; 471: 523–526.
Yemelyanov A, Gasparian A, Lindholm P, Dang L, Pierce JW, Kisseljov F et al. Effects of IKK inhibitor PS1145 on NF-kappaB function, proliferation, apoptosis and invasion activity in prostate carcinoma cells. Oncogene 2006; 25: 387–398.
Bednarski BK, Ding X, Coombe K, Baldwin AS, Kim HJ . Active roles for inhibitory kappaB kinases alpha and beta in nuclear factor-kappaB-mediated chemoresistance to doxorubicin. Mol Cancer Ther 2008; 7: 1827–1835.
Egorin MJ, Lagattuta TF, Hamburger DR, Covey JM, White KD, Musser SM et al. Pharmacokinetics, tissue distribution, and metabolism of 17-(dimethylaminoethylamino)-17-demethoxygeldanamycin (NSC 707545) in CD2F1 mice and Fischer 344 rats. Cancer Chemother Pharmacol 2002; 49: 7–19.
Acknowledgements
This project is supported by NIDCD Intramural projects Z01-DC-00016, Z01-DC-00073 and Z01-DC-00074. We thank Dr Michael Karin (UC San Diego) and Dr Ulrich Siebenlist (NIH/NIAID) for kindly providing the IKK plasmids; Dr Neil Perkins (Newcastle University, UK) for kindly providing anti-p-p52 antibody; Dr Jialing Zhang for technical assistance; and Dr Michael Karin, Dr Christophe Cataisson (NCI/NIH) and Dr Claire Sauvageot (Millennium Pharmaceuticals, Inc) for critique of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
LD was an employee at Millennium Pharmaceuticals, Inc. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Nottingham, L., Yan, C., Yang, X. et al. Aberrant IKKα and IKKβ cooperatively activate NF-κB and induce EGFR/AP1 signaling to promote survival and migration of head and neck cancer. Oncogene 33, 1135–1147 (2014). https://doi.org/10.1038/onc.2013.49
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.49
Keywords
This article is cited by
-
Myeloid-derived growth factor alleviates non-alcoholic fatty liver disease alleviates in a manner involving IKKβ/NF-κB signaling
Cell Death & Disease (2023)
-
The chaperone system in cancer therapies: Hsp90
Journal of Molecular Histology (2023)
-
CRSP8 promotes thyroid cancer progression by antagonizing IKKα-induced cell differentiation
Cell Death & Differentiation (2021)
-
Inhibition of kinase IKKβ suppresses cellular abnormalities induced by the human papillomavirus oncoprotein HPV 18E6
Scientific Reports (2021)
-
Combinatorial treatment with Gefitinib and Bay11-7085 sensitizes primary Gefitinib-resistant OSCC cells by influencing the EGFR- NFκB signaling axis
Medical Oncology (2021)