Abstract
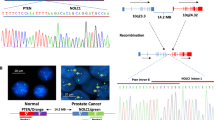
Fusion genes act as potent oncogenes, resulting from chromosomal rearrangements or abnormal transcription in many human cancers. Although multiple gastric cancer genomes have been sequenced, the driving recurrent gene fusions have not been well characterized. Here, we used paired-end transcriptome sequencing to identify novel gene fusions in 18 human gastric cancer cell lines and 18 pairs of primary human gastric cancer tissues and their adjacent normal tissues. Multiple samples revealed expression of PPP1R1B-STARD3 fusion transcript. The presence of PPP1R1B-STARD3 correlated with elevated levels of PPP1R1B mRNA. PPP1R1B-STARD3 fusion transcript was detected in 21.3% of primary human gastric cancers but not in adjacent matched normal gastric tissues. Based on reverse transcription PCR analysis of DNA, unlike other fusions described in gastric cancer, the PPP1R1B-STARD3 appears to be generated by RNA processing without chromosomal rearrangement. Overexpression of PPP1R1B-STARD3 in MKN-28 significantly increased cell proliferation and colony formation. This increased proliferation was mediated by activation of phosphatidylinositol-3-kinase (PI3K)/AKT signaling. Furthermore, expression of PPP1R1B-STARD3 fusion transcript enhanced the tumor growth of MKN-28 cells in athymic nude mice. These findings show that PPP1R1B-STARD3 fusion transcript has a key role in subsets of gastric cancers through the activation of PI3K/AKT signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D . Global cancer statistics. CA Cancer J Clin 2011; 61: 69–90.
Guggenheim DE, Shah MA . Gastric cancer epidemiology and risk factors. J Surg Oncol 2013; 107: 230–236.
Guilford P, Hopkins J, Harraway J, McLeod M, McLeod N, Harawira P et al. E-cadherin germline mutations in familial gastric cancer. Nature 1998; 392: 402–405.
Deng N, Goh LK, Wang H, Das K, Tao J, Tan IB et al. A comprehensive survey of genomic alterations in gastric cancer reveals systematic patterns of molecular exclusivity and co-occurrence among distinct therapeutic targets. Gut 2012; 61: 673–684.
Hold GL, Rabkin CS, Chow WH, Smith MG, Gammon MD, Risch HA et al. A functional polymorphism of toll-like receptor 4 gene increases risk of gastric carcinoma and its precursors. Gastroenterology 2007; 132: 905–912.
Wei J, Noto J, Zaika E, Romero-Gallo J, Correa P, El-Rifai W et al. Pathogenic bacterium Helicobacter pylori alters the expression profile of p53 protein isoforms and p53 response to cellular stresses. Proc Natl Acad Sci USA 2012; 109: E2543–E2550.
Ito K . [Tumor suppressive functions of RUNX3 in gastric carcinogenesis]. Seikagaku 2012; 84: 278–282.
Wang XS, Prensner JR, Chen G, Cao Q, Han B, Dhanasekaran SM et al. An integrative approach to reveal driver gene fusions from paired-end sequencing data in cancer. Nat Biotechnol 2009; 27: 1005–1011.
Prensner JR, Chinnaiyan AM . Oncogenic gene fusions in epithelial carcinomas. Curr Opin Genet Dev 2009; 19: 82–91.
Mitelman F, Johansson B, Mertens F . The impact of translocations and gene fusions on cancer causation. Nat Rev Cancer 2007; 7: 233–245.
Akiva P, Toporik A, Edelheit S, Peretz Y, Diber A, Shemesh R et al. Transcription-mediated gene fusion in the human genome. Genome Res 2006; 16: 30–36.
Zayed A, Couban S, Hayne O, Sparavalo N, Shawwa A, Sadek I et al. Acute promyelocytic leukemia: a novel PML/RARalpha fusion that generates a frameshift in the RARalpha transcript and ATRA resistance. Leuk Lymphoma 2007; 48: 489–496.
Poulin F, Brueschke A, Sonenberg N . Gene fusion and overlapping reading frames in the mammalian genes for 4E-BP3 and MASK. J Biol Chem 2003; 278: 52290–52297.
Li H, Wang J, Mor G, Sklar J . A neoplastic gene fusion mimics trans-splicing of RNAs in normal human cells. Science 2008; 321: 1357–1361.
Kannan K, Wang L, Wang J, Ittmann MM, Li W, Yen L . Recurrent chimeric RNAs enriched in human prostate cancer identified by deep sequencing. Proc Natl Acad Sci USA 2011; 108: 9172–9177.
Persson M, Andren Y, Mark J, Horlings HM, Persson F, Stenman G . Recurrent fusion of MYB and NFIB transcription factor genes in carcinomas of the breast and head and neck. Proc Natl Acad Sci USA 2009; 106: 18740–18744.
Brene S, Lindefors N, Ehrlich M, Taubes T, Horiuchi A, Kopp J et al. Expression of mRNAs encoding ARPP-16/19, ARPP-21, and DARPP-32 in human brain tissue. J Neurosci 1994; 14: 985–998.
Greengard P, Allen PB, Nairn AC . Beyond the dopamine receptor: the DARPP-32/protein phosphatase-1 cascade. Neuron 1999; 23: 435–447.
Fienberg AA, Hiroi N, Mermelstein PG, Song W, Snyder GL, Nishi A et al. DARPP-32: regulator of the efficacy of dopaminergic neurotransmission. Science 1998; 281: 838–842.
Beckler A, Moskaluk CA, Zaika A, Hampton GM, Powell SM, Frierson HF Jr et al. Overexpression of the 32-kilodalton dopamine and cyclic adenosine 3′,5′-monophosphate-regulated phosphoprotein in common adenocarcinomas. Cancer 2003; 98: 1547–1551.
Belkhiri A, Zaika A, Pidkovka N, Knuutila S, Moskaluk C, El-Rifai W . Darpp-32: a novel antiapoptotic gene in upper gastrointestinal carcinomas. Cancer Res 2005; 65: 6583–6592.
Mukherjee K, Peng D, Brifkani Z, Belkhiri A, Pera M, Koyama T et al. Dopamine and cAMP regulated phosphoprotein MW 32 kDa is overexpressed in early stages of gastric tumorigenesis. Surgery 2010; 148: 354–363.
Robinson DR, Kalyana-Sundaram S, Wu YM, Shankar S, Cao X, Ateeq B et al. Functionally recurrent rearrangements of the MAST kinase and Notch gene families in breast cancer. Nat Med 2011; 17: 1646–1651.
Katoh M . Evolutionary recombination hotspot around GSDML-GSDM locus is closely linked to the oncogenomic recombination hotspot around the PPP1R1B-ERBB2-GRB7 amplicon. Int J Oncol 2004; 24: 757–763.
Yoon K, Lee S, Han TS, Moon SY, Yun SM, Kong SH et al. Comprehensive genome- and transcriptome-wide analyses of mutations associated with microsatellite instability in Korean gastric cancers. Genome Res 2013; 23: 1109–1117.
Zhu S, Belkhiri A, El-Rifai W . DARPP-32 increases interactions between epidermal growth factor receptor and ERBB3 to promote tumor resistance to gefitinib. Gastroenterology 2011; 141: 1738–1748.
Hennessy BT, Smith DL, Ram PT, Lu Y, Mills GB . Exploiting the PI3K/AKT pathway for cancer drug discovery. Nat Rev Drug Discov 2005; 4: 988–1004.
Courtney KD, Corcoran RB, Engelman JA . The PI3K pathway as drug target in human cancer. J Clin Oncol 2010; 28: 1075–1083.
El-Rifai W, Smith MF Jr, Li G, Beckler A, Carl VS, Montgomery E et al. Gastric cancers overexpress DARPP-32 and a novel isoform, t-DARPP. Cancer Res 2002; 62: 4061–4064.
Wang K, Ubriaco G, Sutherland LC . RBM6-RBM5 transcription-induced chimeras are differentially expressed in tumours. BMC Genomics 2007; 8: 348.
Palanisamy N, Ateeq B, Kalyana-Sundaram S, Pflueger D, Ramnarayanan K, Shankar S et al. Rearrangements of the RAF kinase pathway in prostate cancer, gastric cancer and melanoma. Nat Med 2010; 16: 793–798.
Tao J, Deng NT, Ramnarayanan K, Huang B, Oh HK, Leong SH et al. CD44-SLC1A2 gene fusions in gastric cancer. Sci Transl Med 2011; 3: 77ra30.
Trapnell C, Pachter L, Salzberg SL . TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 2009; 25: 1105–1111.
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 2010; 28: 511–515.
McPherson A, Hormozdiari F, Zayed A, Giuliany R, Ha G, Sun MG et al. deFuse: an algorithm for gene fusion discovery in tumor RNA-Seq data. PLoS Comput Biol 2011; 7: e1001138.
Acknowledgements
This work was supported in part by the National Research Foundation of Korea (NRF) grants (2009-0081756 and 2012M3A9C4048736 to S-JK). JB and SL were supported by TheragenEtex and Genome Research Foundation internal funds. JB was supported by the Industrial Strategic Technology Development Program (10040231) funded by the Ministry of Knowledge Economy (MKE, Korea).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare competing financial interests. Some authors are present employees of TheragenEtex, and some of them have personal financial interests as shareholders in TheragenEtex.
Additional information
Author Contributions
SMY and KY performed research, analyzed data and wrote the manuscript; SL analyzed and interpreted sequencing data; EK and PK performed research and collected data; JMK performed an animal study; JC, YC and HY contributed to the manuscript; T-SH, S-HK and H-KY collected clinical information and prepared clinical samples; SJ and JB analyzed and collected sequencing data; S-JK designed research, interpreted data and drafted the manuscript.
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yun, S., Yoon, K., Lee, S. et al. PPP1R1B-STARD3 chimeric fusion transcript in human gastric cancer promotes tumorigenesis through activation of PI3K/AKT signaling. Oncogene 33, 5341–5347 (2014). https://doi.org/10.1038/onc.2013.472
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.472
Keywords
This article is cited by
-
Dopamine and cAMP-regulated phosphoprotein 32 kDa (DARPP-32) and survival in breast cancer: a retrospective analysis of protein and mRNA expression
Scientific Reports (2019)
-
RhoGAP domain-containing fusions and PPAPDC1A fusions are recurrent and prognostic in diffuse gastric cancer
Nature Communications (2018)
-
A unified model of the hierarchical and stochastic theories of gastric cancer
British Journal of Cancer (2017)
-
miR-3188 regulates nasopharyngeal carcinoma proliferation and chemosensitivity through a FOXO1-modulated positive feedback loop with mTOR–p-PI3K/AKT-c-JUN
Nature Communications (2016)