Abstract
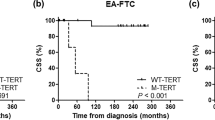
Telomerase activation through induction of its catalytic component telomerase reverse transcriptase (TERT) expression is essential for malignant transformation. TERT promoter mutations namely C228T and C250T that stimulate TERT transcription and telomerase activation have recently been identified in many human malignancies. We thus determined these mutations and their biological and clinical implications in thyroid carcinomas in the present study. The TERT promoter was sequenced in 10 thyroid cancer cell lines and 144 tumors from 20 patients with anaplastic thyroid carcinoma (ATC), 51 with papillary thyroid carcinoma (PTC), 36 with follicular thyroid carcinoma (FTC), and 37 with medullary thyroid carcinoma (MTC). We identified C228T or C250T mutation in 6/8 of ATC cell lines, as well as in tumor tissue from 10/20, 13/51, 8/36 and 0/37 patients with ATC, PTC, FTC and MTC, respectively. In PTC patients, these mutations were exclusively present in the group with age >45 years (P<0.0001), and highly correlated shorter telomeres (P<0.0001) and distant metastasis (P=0.028). The previous radioactivity exposure did not induce the mutation. The presence of C228T or C250T was an independent predictor associated with shorter disease-related survival (DRS) in the entire cohort (P<0.0001), as well as among patients >45 years (P=0.021). ATC patients carrying the mutation survived shorter than those without mutations, although not statistically significant (P=0.129). The TERT promoter mutation was associated with overall survival (P=0.038) and DRS (P=0.058) of FTC patients. Taken together, age- and shorter telomere-dependent TERT promoter mutations occur frequently in follicular cell-derived thyroid carcinoma (ATC, PTC and FTC) but not in parafollicular cell-originated MTC, and may serve as a marker for aggressive disease and poor outcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Xing M . Molecular pathogenesis and mechanisms of thyroid cancer. Nat Rev Cancer 2013; 13: 184–199.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Pacini F, Cantara S, Capezzone M, Marchisotta S . Telomerase and the endocrine system. Nat Rev Endocrinol 2011; 7: 420–430.
Blackburn EH, Greider CW, Szostak JW . Telomeres and telomerase: the path from maize, Tetrahymena and yeast to human cancer and aging. Nat Med 2006; 12: 1133–1138.
Cong YS, Wright WE, Shay JW . Human telomerase and its regulation. Microbiol Mol Biol Rev 2002; 66: 407–425.
Shay JW, Bacchetti S . A survey of telomerase activity in human cancer. Eur J Cancer 1997; 33: 787–791.
Daniel M, Peek GW, Tollefsbol TO . Regulation of the human catalytic subunit of telomerase (hTERT). Gene 2012; 498: 135–146.
Asaad NY, Abd El-Wahed MM, Mohammed AG . Human telomerase reverse transcriptase (hTERT) gene expression in thyroid carcinoma: diagnostic and prognostic role. J Egypt Natl Canc Inst 2006; 18: 8–16.
Saji M, Xydas S, Westra WH, Liang CK, Clark DP, Udelsman R et al. Human telomerase reverse transcriptase (hTERT) gene expression in thyroid neoplasms. Clin Cancer Res 1999; 5: 1483–1489.
Straight AM, Patel A, Fenton C, Dinauer C, Tuttle RM, Francis GL . Thyroid carcinomas that express telomerase follow a more aggressive clinical course in children and adolescents. J Endocrinol Invest 2002; 25: 302–308.
Wang Y, Kowalski J, Tsai HL, Marik R, Prasad N, Somervell H et al. Differentiating alternative splice variant patterns of human telomerase reverse transcriptase in thyroid neoplasms. Thyroid 2008; 18: 1055–1063.
Umbricht CB, Saji M, Westra WH, Udelsman R, Zeiger MA, Sukumar S . Telomerase activity: a marker to distinguish follicular thyroid adenoma from carcinoma. Cancer Res 1997; 57: 2144–2147.
Lerma E, Mora J . Telomerase activity in ‘suspicious’ thyroid cytology. Cancer 2005; 105: 492–497.
Kammori M, Nakamura K, Hashimoto M, Ogawa T, Kaminishi M, Takubo K . Clinical application of human telomerase reverse transcriptase gene expression in thyroid follicular tumors by fine-needle aspirations using in situ hybridization. Int J Oncol 2003; 22: 985–991.
Guerra LN, Miler EA, Moiguer S, Karner M, Orlandi AM, Fideleff H et al. Telomerase activity in fine needle aspiration biopsy samples: application to diagnosis of human thyroid carcinoma. Clin Chim Acta 2006; 370: 180–184.
Aogi K, Kitahara K, Buley I, Backdahl M, Tahara H, Sugino T et al. Telomerase activity in lesions of the thyroid: application to diagnosis of clinical samples including fine-needle aspirates. Clin Cancer Res 1998; 4: 1965–1970.
Bornstein-Quevedo L, Garcia-Hernandez ML, Camacho-Arroyo I, Herrera MF, Angeles AA, Trevino OG et al. Telomerase activity in well-differentiated papillary thyroid carcinoma correlates with advanced clinical stage of the disease. Endocr Pathol 2003; 14: 213–219.
Cheng AJ, Lin JD, Chang T, Wang TC . Telomerase activity in benign and malignant human thyroid tissues. Br J Cancer 1998; 77: 2177–2180.
Hoang-Vu C, Boltze C, Gimm O, Poremba C, Dockhorn-Dworniczak B, Kohrle J et al. Expression of telomerase genes in thyroid carcinoma. Int J Oncol 2002; 21: 265–272.
Brousset P, Chaouche N, Leprat F, Branet-Brousset F, Trouette H, Zenou RC et al. Telomerase activity in human thyroid carcinomas originating from the follicular cells. J Clin Endocrinol Metab 1997; 82: 4214–4216.
Foukakis T, Gusnanto A, Au AY, Hoog A, Lui WO, Larsson C et al. A PCR-based expression signature of malignancy in follicular thyroid tumors. Endocr Relat Cancer 2007; 14: 381–391.
Horn S, Figl A, Rachakonda PS, Fischer C, Sucker A, Gast A et al. TERT promoter mutations in familial and sporadic melanoma. Science 2013; 339: 959–961.
Huang FW, Hodis E, Xu MJ, Kryukov GV, Chin L, Garraway LA . Highly recurrent TERT promoter mutations in human melanoma. Science 2013; 339: 957–959.
Killela PJ, Reitman ZJ, Jiao Y, Bettegowda C, Agrawal N, Diaz LA Jr. et al. TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc Natl Acad Sci USA 2013; 110: 6021–6026.
Tallet A, Nault J-C, Renier A, Hysi I, Galateau-Sallé F, Cazes A et al. Overexpression and promoter mutation of the TERT gene in malignant pleural mesothelioma. Oncogene 2014; 33: 3748–3752.
Dinets A, Hulchiy M, Sofiadis A, Ghaderi M, Hoog A, Larsson C et al. Clinical, genetic, and immunohistochemical characterization of 70 Ukrainian adult cases with post-Chornobyl papillary thyroid carcinoma. Eur J Endocrinol 2012; 166: 1049–1060.
Liu X, Bishop J, Shan Y, Pai S, Liu D, Murugan AK et al. Highly prevalent TERT promoter mutations in aggressive thyroid cancers. Endocr Relat Cancer 2013; 20: 603–610.
Liu Z, Li Q, Li K, Chen L, Li W, Hou M et al. Telomerase reverse transcriptase promotes epithelial-mesenchymal transition and stem cell-like traits in cancer cells. Oncogene 2013; 32: 4203–4213.
Park JI, Venteicher AS, Hong JY, Choi J, Jun S, Shkreli M et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 2009; 460: 66–72.
Cao Y, Li H, Deb S, Liu JP . TERT regulates cell survival independent of telomerase enzymatic activity. Oncogene 2002; 21: 3130–3138.
Stewart SA, Hahn WC, O'Connor BF, Banner EN, Lundberg AS, Modha P et al. Telomerase contributes to tumorigenesis by a telomere length-independent mechanism. Proc Natl Acad Sci USA 2002; 99: 12606–12611.
Gertler R, Rosenberg R, Stricker D, Friederichs J, Hoos A, Werner M et al. Telomere length and human telomerase reverse transcriptase expression as markers for progression and prognosis of colorectal carcinoma. J Clin Oncol 2004; 22: 1807–1814.
Domont J, Pawlik TM, Boige V, Rose M, Weber JC, Hoff PM et al. Catalytic subunit of human telomerase reverse transcriptase is an independent predictor of survival in patients undergoing curative resection of hepatic colorectal metastases: a multicenter analysis. J Clin Oncol 2005; 23: 3086–3093.
Sanders RP, Drissi R, Billups CA, Daw NC, Valentine MB, Dome JS . Telomerase expression predicts unfavorable outcome in osteosarcoma. J Clin Oncol 2004; 22: 3790–3797.
Sputova K, Garbe JC, Pelissier FA, Chang E, Stampfer MR, Labarge MA . Aging phenotypes in cultured normal human mammary epithelial cells are correlated with decreased telomerase activity independent of telomere length. Genome Integr 2013; 4: 4.
Cawthon RM . Telomere measurement by quantitative PCR. Nucleic Acids Res 2002; 30: e47.
Acknowledgements
We thank Drs N-E Heldin (Uppsala University, Sweden), and BG Robinson and Mr. E Wennerberg (University of Sydney, Australia) for cell lines. The study was funded by grants from the Swedish Cancer Society, the Swedish Research Council, Cancer Society in Stockholm, the Stockholm County Council and Karolinska Institutet. Dr M Hulchiy (Kyiv CIty Teaching Endocrinological Center, Ukraine) for help with retrieval of Ukraine PTC tissue samples.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, T., Wang, N., Cao, J. et al. The age- and shorter telomere-dependent TERT promoter mutation in follicular thyroid cell-derived carcinomas. Oncogene 33, 4978–4984 (2014). https://doi.org/10.1038/onc.2013.446
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.446