Abstract
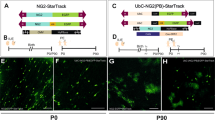
Low-grade glial neoplasms (astrocytomas) represent one of the most common brain tumors in the pediatric population. These tumors frequently form in the optic pathway (optic pathway gliomas, OPGs), especially in children with the neurofibromatosis type 1 (NF1)-inherited tumor predisposition syndrome. To model these tumors in mice, we have previously developed several Nf1 genetically-engineered mouse strains that form optic gliomas. However, there are three distinct macroglial cell populations in the optic nerve (astrocytes, NG2+ (nerve/glial antigen 2) cells and oligodendrocytes). The presence of NG2+ cells in the optic nerve raises the intriguing possibility that these cells could be the tumor-initiating cells, as has been suggested for adult glioma. In this report, we used a combination of complementary in vitro and novel genetically-engineered mouse strains in vivo to determine whether NG2+ cells could give rise to Nf1 optic glioma. First, we show that Nf1 inactivation results in a cell-autonomous increase in glial fibrillary acidic protein+ (GFAP+), but not in NG2+, cell proliferation in vitro. Second, similar to the GFAP-Cre transgenic strain that drives Nf1 optic gliomagenesis, NG2-expressing cells also give rise to all three macroglial lineages in vivo. Third, in contrast to the GFAP-Cre strain, Nf1 gene inactivation in NG2+ cells is not sufficient for optic gliomagenesis in vivo. Collectively, these data demonstrate that NG2+ cells are not the cell of origin for mouse optic glioma, and support a model in which gliomagenesis requires Nf1 loss in specific neuroglial progenitors during embryogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Blazo MA, Lewis RA, Chintagumpala MM, Frazier M, McCluggage C, Plon SE . Outcomes of systematic screening for optic pathway tumors in children with neurofibromatosis type 1. Am J Med Genet A 2004; 127A: 224–229.
Lund AM, Skovby F . Optic gliomas in children with neurofibromatosis type 1. Eur J Pediatr 1991; 150: 835–838.
Listernick R, Darling C, Greenwald M, Strauss L, Charrow J . Optic pathway tumors in children: the effect of neurofibromatosis type 1 on clinical manifestations and natural history. J Pediatr 1995; 127: 718–722.
Czyzyk E, Jozwiak S, Roszkowski M, Schwartz RA . Optic pathway gliomas in children with and without neurofibromatosis 1. J Child Neurol 2003; 18: 471–478.
Listernick R, Louis DN, Packer RJ, Gutmann DH . Optic pathway gliomas in children with neurofibromatosis 1: consensus statement from the NF1 optic pathway glioma task force. Ann Neurol 1997; 41: 143–149.
Bajenaru ML, Hernandez MR, Perry A, Zhu Y, Parada LF, Garbow JR et al. Optic nerve glioma in mice requires astrocyte Nf1 gene inactivation and Nf1 brain heterozygosity. Cancer Res 2003; 63: 8573–8577.
Zhu Y, Harada T, Liu L, Lush ME, Guignard F, Harada C et al. Inactivation of NF1 in CNS causes increased glial progenitor proliferation and optic glioma formation. Development 2005; 132: 5577–5588.
Bajenaru ML, Zhu Y, Hedrick NM, Donahoe J, Parada LF, Gutmann DH . Astrocyte-specific inactivation of the neurofibromatosis 1 gene (NF1) is insufficient for astrocytoma formation. Mol Cell Biol 2002; 22: 5100–5113.
Dahiya S, Lee da Y, Gutmann DH . Comparative characterization of the human and mouse third ventricle germinal zones. J Neuropathol Exp Neurol 2011; 70: 622–633.
Doetsch F, Garcia-Verdugo JM, Alvarez-Buylla A . Regeneration of a germinal layer in the adult mammalian brain. Proc Natl Acad Sci USA 1999; 96: 11619–11624.
Yeh TH, Lee da Y, Gianino SM, Gutmann DH . Microarray analyses reveal regional astrocyte heterogeneity with implications for neurofibromatosis type 1 (NF1)-regulated glial proliferation. Glia 2009; 57: 1239–1249.
Assanah M, Lochhead R, Ogden A, Bruce J, Goldman J, Canoll P . Glial progenitors in adult white matter are driven to form malignant gliomas by platelet-derived growth factor-expressing retroviruses. J Neurosci 2006; 26: 6781–6790.
Masui K, Suzuki SO, Torisu R, Goldman JE, Canoll P, Iwaki T . Glial progenitors in the brainstem give rise to malignant gliomas by platelet-derived growth factor stimulation. Glia 2010; 58: 1050–1065.
Neymeyer V, Tephly TR, Miller MW . Folate and 10-formyltetrahydrofolate dehydrogenase (FDH) expression in the central nervous system of the mature rat. Brain Res 1997; 766: 195–204.
Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB . NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn 2001; 222: 218–227.
Levine JM, Reynolds R, Fawcett JW . The oligodendrocyte precursor cell in health and disease. Trends Neurosci 2001; 24: 39–47.
Zhu X, Bergles DE, Nishiyama A . NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development 2008; 135: 145–157.
Lee da Y, Gianino SM, Gutmann DH . Innate neural stem cell heterogeneity determines the patterning of glioma formation in children. Cancer Cell 2012; 22: 131–138.
Dawson MR, Polito A, Levine JM, Reynolds R . NG2-expressing glial progenitor cells: an abundant and widespread population of cycling cells in the adult rat CNS. Mol Cell Neurosci 2003; 24: 476–488.
Raff MC, Abney ER, Cohen J, Lindsay R, Noble M . Two types of astrocytes in cultures of developing rat white matter: differences in morphology, surface gangliosides, and growth characteristics. J Neurosci 1983; 3: 1289–1300.
Raff MC, Abney ER, Miller RH . Two glial cell lineages diverge prenatally in rat optic nerve. Dev Biol 1984; 106: 53–60.
Miller RH, David S, Patel R, Abney ER, Raff MC . A quantitative immunohistochemical study of macroglial cell development in the rat optic nerve: in vivo evidence for two distinct astrocyte lineages. Dev Biol 1985; 111: 35–41.
Stallcup WB, Beasley L . Bipotential glial precursor cells of the optic nerve express the NG2 proteoglycan. J Neurosci 1987; 7: 2737–2744.
Baracskay KL, Kidd GJ, Miller RH, Trapp BD . NG2-positive cells generate A2B5-positive oligodendrocyte precursor cells. Glia 2007; 55: 1001–1010.
Yoo S, Wrathall JR . Mixed primary culture and clonal analysis provide evidence that NG2 proteoglycan-expressing cells after spinal cord injury are glial progenitors. Dev Neurobiol 2007; 67: 860–874.
Ju PJ, Liu R, Yang HJ, Xia YY, Feng ZW . Clonal analysis for elucidating the lineage potential of embryonic NG2(+) cells. Cytotherapy 2012; 14: 608–620.
Mokry J, Karbanova J, Filip S, Cizkova D, Pazour J, English D . Phenotypic and morphological characterization of in vitro oligodendrogliogenesis. Stem Cells Dev 2008; 17: 333–341.
Winkler EA, Bell RD, Zlokovic BV . Central nervous system pericytes in health and disease. Nat Neurosci 2011; 14: 1398–1405.
Wiranowska M, Ladd S, Smith SR, Gottschall PE . CD44 adhesion molecule and neuro-glial proteoglycan NG2 as invasive markers of glioma. Brain Cell Biol 2006; 35: 159–172.
Al-Mayhani MT, Grenfell R, Narita M, Piccirillo S, Kenney-Herbert E, Fawcett JW et al. NG2 expression in glioblastoma identifies an actively proliferating population with an aggressive molecular signature. Neuro Oncol 2011; 13: 830–845.
Schrappe M, Klier FG, Spiro RC, Waltz TA, Reisfeld RA, Gladson CL . Correlation of chondroitin sulfate proteoglycan expression on proliferating brain capillary endothelial cells with the malignant phenotype of astroglial cells. Cancer Res 1991; 51: 4986–4993.
Ellis JA, Castelli M, Bruce JN, Canoll P, Ogden AT . Retroviral delivery of platelet-derived growth factor to spinal cord progenitor cells drives the formation of intramedullary gliomas. Neurosurgery 2012; 70: 198–204.
Wang J, Svendsen A, Kmiecik J, Immervoll H, Skaftnesmo KO, Planaguma J et al. Targeting the NG2/CSPG4 proteoglycan retards tumour growth and angiogenesis in preclinical models of GBM and melanoma. PLoS One 2011; 6: e23062.
Ogden AT, Waziri AE, Lochhead RA, Fusco D, Lopez K, Ellis JA et al. Identification of A2B5+CD133- tumor-initiating cells in adult human gliomas. Neurosurgery 2008; 62: 505–514.
Tchoghandjian A, Baeza N, Colin C, Cayre M, Metellus P, Beclin C et al. A2B5 cells from human glioblastoma have cancer stem cell properties. Brain Pathol 2010; 20: 211–221.
Liu C, Sage JC, Miller MR, Verhaak RG, Hippenmeyer S, Vogel H et al. Mosaic analysis with double markers reveals tumor cell of origin in glioma. Cell 2011; 146: 209–221.
Alcantara Llaguno S, Chen J, Kwon CH, Jackson EL, Li Y, Burns DK et al. Malignant astrocytomas originate from neural stem/progenitor cells in a somatic tumor suppressor mouse model. Cancer Cell 2009; 15: 45–56.
Ono K, Yasui Y, Rutishauser U, Miller RH . Focal ventricular origin and migration of oligodendrocyte precursors into the chick optic nerve. Neuron 1997; 19: 283–292.
Gao L, Miller RH . Specification of optic nerve oligodendrocyte precursors by retinal ganglion cell axons. J Neurosci 2006; 26: 7619–7628.
Tchoghandjian A, Fernandez C, Colin C, El Ayachi I, Voutsinos-Porche B, Fina F et al. Pilocytic astrocytoma of the optic pathway: a tumour deriving from radial glia cells with a specific gene signature. Brain 2009; 132 (Part 6): 1523–1535.
Silber J, Lim DA, Petritsch C, Persson AI, Maunakea AK, Yu M et al. miR-124 and miR-137 inhibit proliferation of glioblastoma multiforme cells and induce differentiation of brain tumor stem cells. BMC Med 2008; 6: 14.
Kwon CH, Zhao D, Chen J, Alcantara S, Li Y, Burns DK et al. Pten haploinsufficiency accelerates formation of high-grade astrocytomas. Cancer Res 2008; 68: 3286–3294.
Abel TW, Clark C, Bierie B, Chytil A, Aakre M, Gorska A et al. GFAP-Cre-mediated activation of oncogenic K-ras results in expansion of the subventricular zone and infiltrating glioma. Mol Cancer Res 2009; 7: 645–653.
Zhu Y, Romero MI, Ghosh P, Ye Z, Charnay P, Rushing EJ et al. Ablation of NF1 function in neurons induces abnormal development of cerebral cortex and reactive gliosis in the brain. Genes Dev 2001; 15: 859–876.
Madisen L, Zwingman TA, Sunkin SM, Oh SW, Zariwala HA, Gu H et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci 2010; 13: 133–140.
Brannan CI, Perkins AS, Vogel KS, Ratner N, Nordlund ML, Reid SW et al. Targeted disruption of the neurofibromatosis type-1 gene leads to developmental abnormalities in heart and various neural crest-derived tissues. Genes Dev 1994; 8: 1019–1029.
Hegedus B, Banerjee D, Yeh TH, Rothermich S, Perry A, Rubin JB et al. Preclinical cancer therapy in a mouse model of neurofibromatosis-1 optic glioma. Cancer Res 2008; 68: 1520–1528.
Acknowledgements
We appreciate the excellent technical assistance of Angela Petti and Belinda McMahan in the Ophthalmology Core Facility for preparing the optic nerve sections. This work was funded by a grant from the National Cancer Institute (U01-CA141549 to DHG), whereas the Ophthalmology Core Facility is funded by a grant from the National Eye Institute (EY02687).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Solga, A., Gianino, S. & Gutmann, D. NG2-cells are not the cell of origin for murine neurofibromatosis-1 (Nf1) optic glioma. Oncogene 33, 289–299 (2014). https://doi.org/10.1038/onc.2012.580
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.580
Keywords
This article is cited by
-
Changes in the proliferative capacity of NG2 cell subpopulations during postnatal development of the mouse hippocampus
Brain Structure and Function (2017)
-
The molecular and cell biology of pediatric low-grade gliomas
Oncogene (2014)