Abstract
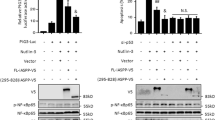
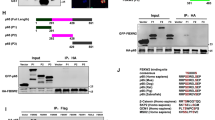
The alternative nuclear factor-kappaB (NF-κB) -activation pathway proceeds via inducible p100 processing, leading to the activation of RelB-containing dimers. This pathway is aberrantly activated in several types of tumors; however, a direct role for RelB in the control of cell proliferation is still largely unexplored. Here, we demonstrate that RelB provides cell proliferation-inhibitory signals in murine fibroblasts. In agreement with these results, RelB ectopic expression inhibits xenograft tumor growth in vivo, whereas RelB knockdown enhances it. Significantly, we show that RelB inhibits cell proliferation and tumor growth in a p53-dependent manner. Mechanistic studies indicate that RelB regulates the transcription of the p53 tumor-suppressor gene through direct recruitment to the p53 promoter, thus increasing both p53 protein levels and expression of p53 target genes such as p21. Our findings define a novel link between NF-κB and growth-inhibitory pathways involving the RelB-dependent transcriptional upregulation of p53. Furthermore, they suggest that inhibition of RelB in some tumor types that retain wild-type p53 may diminish rather than improve therapeutic responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Oeckinghaus A, Ghosh S . The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol 2009; 1: a000034.
Hayden MS, Ghosh S . Shared principles in NF-kappaB signaling. Cell 2008; 132: 344–362.
Karin M, Lin A . NF-kappaB at the crossroads of life and death. Nat Immunol 2002; 3: 221–227.
Fan Y, Dutta J, Gupta N, Fan G, Gelinas C . Regulation of programmed cell death by NF-kappaB and its role in tumorigenesis and therapy. Adv Exp Med Biol 2008; 615: 223–250.
Naugler WE, Karin M . NF-kappaB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev 2008; 18: 19–26.
Basseres DS, Baldwin AS . Nuclear factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic initiation and progression. Oncogene 2006; 25: 6817–6830.
Baud V, Karin M . Is NF-kappaB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov 2009; 8: 33–40.
Chen F, Castranova V . Nuclear factor-kappaB, an unappreciated tumor suppressor. Cancer Res 2007; 67: 11093–11098.
Perkins ND . NF-kappaB: tumor promoter or suppressor? Trends Cell Biol 2004; 14: 64–69.
Dajee M, Lazarov M, Zhang JY, Cai T, Green CL, Russell AJ et al. NF-kappaB blockade and oncogenic Ras trigger invasive human epidermal neoplasia. Nature 2003; 421: 639–643.
Chen F, Lu Y, Castranova V, Li Z, Karin M . Loss of Ikkbeta promotes migration and proliferation of mouse embryo fibroblast cells. J Biol Chem 2006; 281: 37142–37149.
Maeda S, Kamata H, Luo JL, Leffert H, Karin M . IKKbeta couples hepatocyte death to cytokine-driven compensatory proliferation that promotes chemical hepatocarcinogenesis. Cell 2005; 121: 977–990.
Gapuzan ME, Yufit PV, Gilmore TD . Immortalized embryonic mouse fibroblasts lacking the RelA subunit of transcription factor NF-kappaB have a malignantly transformed phenotype. Oncogene 2002; 21: 2484–2492.
Saccani S, Pantano S, Natoli G . Modulation of NF-kappaB activity by exchange of dimers. Mol Cell 2003; 11: 1563–1574.
Perkins ND . Integrating cell-signalling pathways with NF-kappaB and IKK function. Nat Rev Mol Cell Biol 2007; 8: 49–62.
Ak P, Levine AJ . p53 and NF-kappaB: different strategies for responding to stress lead to a functional antagonism. FASEB J. 2010; 24: 3643–3652.
Ryan KM, Ernst MK, Rice NR, Vousden KH . Role of NF-kappaB in p53-mediated programmed cell death. Nature 2000; 404: 892–897.
Wu L, D'Amico A, Winkel KD, Suter M, Lo D, Shortman K . RelB is essential for the development of myeloid-related CD8alpha- dendritic cells but not of lymphoid-related CD8alpha+ dendritic cells. Immunity 1998; 9: 839–847.
Weih F, Carrasco D, Durham SK, Barton DS, Rizzo CA, Ryseck RP et al. Multiorgan inflammation and hematopoietic abnormalities in mice with a targeted disruption of RelB, a member of the NF-kappa B/Rel family. Cell 1995; 80: 331–340.
Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell 2007; 12: 115–130.
Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng WJ et al. Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma. Cancer Cell 2007; 12: 131–144.
Wang X, Belguise K, Kersual N, Kirsch KH, Mineva ND, Galtier F et al. Oestrogen signalling inhibits invasive phenotype by repressing RelB and its target BCL2. Nat Cell Biol 2007; 9: 470–478.
Xu Y, Josson S, Fang F, Oberley TD, Clair DK, Wan XS et al. RelB enhances prostate cancer growth: implications for the role of the nuclear factor-kappaB alternative pathway in tumorigenicity. Cancer Res 2009; 69: 3267–3271.
Vousden KH, Prives C . Blinded by the light: the growing complexity of p53. Cell 2009; 137: 413–431.
Levine AJ, Oren M . The first 30 years of p53: growing ever more complex. Nat Rev Cancer 2009; 9: 749–758.
Tergaonkar V, Pando M, Vafa O, Wahl G, Verma I . p53 stabilization is decreased upon NFkappaB activation: a role for NFkappaB in acquisition of resistance to chemotherapy. Cancer Cell 2002; 1: 493–503.
Xia Y, Padre RC, De Mendoza TH, Bottero V, Tergaonkar VB, Verma IM . Phosphorylation of p53 by IkappaB kinase 2 promotes its degradation by beta-TrCP. Proc Natl Acad Sci USA 2009; 106: 2629–2634.
Jacque E, Tchenio T, Piton G, Romeo PH, Baud V . RelA repression of RelB activity induces selective gene activation downstream of TNF receptors. Proc Natl Acad Sci USA 2005; 102: 14635–14640.
Dejardin E . The alternative NF-kappaB pathway from biochemistry to biology: pitfalls and promises for future drug development. Biochem Pharmacol 2006; 72: 1161–1179.
Mise N, Drosten M, Racek T, Tannapfel A, Putzer BM . Evaluation of potential mechanisms underlying genotype-phenotype correlations in multiple endocrine neoplasia type 2. Oncogene 2006; 25: 6637–6647.
Espinosa JM, Verdun RE, Emerson BM . p53 functions through stress- and promoter-specific recruitment of transcription initiation components before and after DNA damage. Mol Cell 2003; 12: 1015–1027.
An W, Kim J, Roeder RG . Ordered cooperative functions of PRMT1, p300, and CARM1 in transcriptional activation by p53. Cell 2004; 117: 735–748.
Roy B, Reisman D . Positive and negative regulatory elements in the murine p53 promoter. Oncogene 1996; 13: 2359–2366.
Hellin AC, Calmant P, Gielen J, Bours V, Merville MP . Nuclear factor - kappaB-dependent regulation of p53 gene expression induced by daunomycin genotoxic drug. Oncogene 1998; 16: 1187–1195.
Mauro C, Leow SC, Anso E, Rocha S, Thotakura AK, Tornatore L et al. NF-kappaB controls energy homeostasis and metabolic adaptation by upregulating mitochondrial respiration. Nat Cell Biol 2011; 13: 1272–1279.
Huang WC, Ju TK, Hung MC, Chen CC . Phosphorylation of CBP by IKKalpha promotes cell growth by switching the binding preference of CBP from p53 to NF-kappaB. Mol Cell 2007; 26: 75–87.
Schumm K, Rocha S, Caamano J, Perkins ND . Regulation of p53 tumour suppressor target gene expression by the p52 NF-kappaB subunit. EMBO J. 2006; 25: 4820–4832.
Barre B, Perkins ND . The Skp2 promoter integrates signaling through the NF-kappaB, p53, and Akt/GSK3beta pathways to regulate autophagy and apoptosis. Mol Cell 2010; 38: 524–538.
Hailfinger S, Nogai H, Pelzer C, Jaworski M, Cabalzar K, Charton JE et al. Malt1-dependent RelB cleavage promotes canonical NF-kappaB activation in lymphocytes and lymphoma cell lines. Proc Natl Acad Sci USA 2011; 108: 14596–14601.
Zhang JY, Green CL, Tao S, Khavari PA . NF-kappaB RelA opposes epidermal proliferation driven by TNFR1 and JNK. Genes Dev 2004; 18: 17–22.
Hu Y, Baud V, Oga T, Kim KI, Yoshida K, Karin M . IKKalpha controls formation of the epidermis independently of NF-kappaB. Nature 2001; 410: 710–714.
Park E, Zhu F, Liu B, Xia X, Shen J, Bustos T et al. Reduction in IkappaB kinase alpha expression promotes the development of skin papillomas and carcinomas. Cancer Res 2007; 67: 9158–9168.
Liu B, Park E, Zhu F, Bustos T, Liu J, Shen J et al. A critical role for I kappaB kinase alpha in the development of human and mouse squamous cell carcinomas. Proc Natl Acad Sci USA 2006; 103: 17202–17207.
Maeda G, Chiba T, Kawashiri S, Satoh T, Imai K . Epigenetic inactivation of IkappaB Kinase-alpha in oral carcinomas and tumor progression. Clin Cancer Res 2007; 13: 5041–5047.
Kieusseian A, Chagraoui J, Kerdudo C, Mangeot PE, Gage PJ, Navarro N et al. Expression of Pitx2 in stromal cells is required for normal hematopoiesis. Blood 2006; 107: 492–500.
Acknowledgements
We thank F Weih, U Siebenlist, K Vousden, B Vogelstein, T Jacks, J Cleveland, M Oren, D Reisman, G Kroemer, JL Perfettini, M Körner, LA Martinez, T Tchenio, J Browning and I Verma for valuable material; G Piton for technical assistance; N Boggetto for cell sorting (ImagoSeine, Institut Monod, France, Région Ile-de-France grant no. E539); and LL Pritchard for critically reading the manuscript. This work was supported by grants to VB from Agence Nationale pour la Recherche (ANR), Association pour la Recherche sur le Cancer, Belgian InterUniversity Attraction Pole, Cancéropole Ile-de-France and Université Paris Descartes, post-doctoral funding from ANR (HA and KB), doctoral fellowships from Ministère de la Recherche et des Technologies, Ligue Nationale contre le Cancer, and Société Française du Cancer (EJ), and an award from the Bettencourt-Schueller Fundation (EJ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website .
Rights and permissions
About this article
Cite this article
Jacque, E., Billot, K., Authier, H. et al. RelB inhibits cell proliferation and tumor growth through p53 transcriptional activation. Oncogene 32, 2661–2669 (2013). https://doi.org/10.1038/onc.2012.282
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.282
Keywords
This article is cited by
-
Alternative NF-κB signaling promotes colorectal tumorigenesis through transcriptionally upregulating Bcl-3
Oncogene (2018)
-
Evaluation of NF-κB subunit expression and signaling pathway activation demonstrates that p52 expression confers better outcome in germinal center B-cell-like diffuse large B-cell lymphoma in association with CD30 and BCL2 functions
Modern Pathology (2015)
-
NF-κB pathways in hematological malignancies
Cellular and Molecular Life Sciences (2014)