Abstract
Background/Objectives:
High dietary fibre intakes may protect against obesity by influencing colonic fermentation and the colonic microbiota. Though, recent studies suggest that increased colonic fermentation contributes to adiposity. Diet influences the composition of the gut microbiota. Previous research has not evaluated dietary intakes, body mass index (BMI), faecal microbiota and short chain fatty acid (SCFA) in the same cohort. Our objectives were to compare dietary intakes, faecal SCFA concentrations and gut microbial profiles in healthy lean (LN, BMI⩽25) and overweight or obese (OWOB, BMI>25) participants.
Design:
We collected demographic information, 3-day diet records, physical activity questionnaires and breath and faecal samples from 94 participants of whom 52 were LN and 42 OWOB.
Results:
Dietary intakes and physical activity levels did not differ significantly between groups. OWOB participants had higher faecal acetate (P=0.05), propionate (P=0.03), butyrate (P=0.05), valerate (P=0.03) and total short chain fatty acid (SCFA; P=0.02) concentrations than LN. No significant differences in Firmicutes to Bacteroides/Prevotella (F:B) ratio was observed between groups. However, in the entire cohort, Bacteroides/Prevotella counts were negatively correlated with faecal total SCFA (r=−0.32, P=0.002) and F:B ratio was positively correlated with faecal total SCFA (r=0.42, P<0.0001). Principal component analysis identified distinct gut microbiota and SCFA–F:B ratio components, which together accounted for 59% of the variation. F:B ratio loaded with the SCFA and not with the microbiota suggesting that SCFA and F:B ratio vary together and may be interrelated.
Conclusions:
The results support the hypothesis that colonic fermentation patterns may be altered, leading to different faecal SCFA concentrations in OWOB compared with LN humans. More in-depth studies looking at the metabolic fate of SCFA produced in LN and OWOB participants are needed in order to determine the role of SCFA in obesity.
Similar content being viewed by others
Introduction
Colonic fermentation is a complex process that occurs through the interactions of many microbial species and involves the anaerobic breakdown of dietary fibre, protein and peptides.1, 2, 3 The principal end products of colonic fermentation are the SCFA, acetate, propionate and butyrate, the gases hydrogen, carbon dioxide and methane4, 5, 6 and energy, which is used by the microbiota for growth and maintenance of cellular functions.7 Small amounts of branched chain fatty acids (iso-butyrate, valerate and iso-valerate) are also formed from protein and amino acid degradation. The amount and type of dietary fibre are among the major determinants of gut microbial composition and SCFA production patterns.8 In humans, the SCFA produced account for 5–10% of total dietary energy.9
High intakes of dietary fibre are associated with a reduced risk of obesity.10, 11, 12 One of the many mechanisms by which the fibre may protect against obesity is via the SCFA-mediated modulation of the secretion of gut hormones involved in the regulation of food intake and energy balance.13, 14 However, recent work has suggested a causative role for the gut microbiota in obesity. Studies in animal models have shown that the obese microbiota differs from the lean microbiota15 and may produce more SCFA and, hence, extract more energy from a given diet than the lean microbiota.16 The same group also found that the relative proportion of Bacteroidetes was lower and Firmicutes was higher in obese than lean individuals;17 however, this has not been observed consistently in other studies.18, 19, 20 Recent studies have also shown that faecal SCFA concentrations were significantly higher in obese than lean participants.18, 21 It is not known if habitual dietary intakes, microbial profiles and faecal SCFA concentrations are interrelated.
Therefore, the objectives of this cross-sectional study were to compare dietary intakes, faecal SCFA concentrations, gut microbial profiles and physical activity levels in healthy lean (LN; body mass index (BMI)⩽25) versus overweight and obese (OWOB; BMI>25) participants. We hypothesised that LN and OWOB participants may have different dietary intakes and microbial profiles that may result in altered colonic fermentation patterns leading to different faecal SCFA concentrations.
Materials and methods
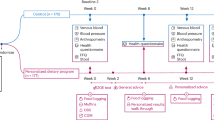
One hundred and two participants were recruited via advertisements posted around the University of Toronto campus and from a pool of participants previously involved in studies by our group. Ninety-four male or non-pregnant, non-lactating females over the age of 17 years completed the study. Participants were excluded for any of the following reasons: regular use of antibiotics (⩾1 course per year over the last 5 years), any use of antibiotics, laxatives or other drugs known to influence gastrointestinal function in the 3 months before the study, presence of inflammatory bowel disease, malabsorption, gastrointestinal infection, short bowel or other conditions affecting gastrointestinal function or any recent (3 months before the study) illness or surgery requiring hospitalization. Participants were also excluded if they failed to provide a faecal sample within the study period. Participants in this study have been previously studied to examine the relationship between faecal Archaea, BMI, SCFA and fibre intake.22 This study was approved by the Research Ethics Board, University of Toronto. Written informed consent was obtained from all participants.
Participants were studied over two separate visits to the laboratory. At the first visit, they filled out questionnaires related to demographics, medical history, drug use and physical activity. Subject’s height and weight was measured and two breath samples were collected. Participants were given instructions on how to record their dietary intake and asked to keep a 3-day diet record, including one weekend day. Participants were also given a faecal collection kit, which consisted of the Fisherbrand commode specimen collection system (Fisher Scientific, Ottawa, ON, Canada), plastic bags and a styrofoam box containing dry ice. On the third day of the diet record or the day after, participants collected a faecal sample. The plastic bag containing the faecal sample was immediately placed on dry ice, brought to the lab within 24 h of being collected and then stored at −20 °C. At the second visit, participants returned the faecal sample and the dietary record and provided two more breath samples.
Breath samples were collected using the Easy Sampler with tube holder (Quintron Instrument Company, Milwaukee, WI, USA). Methane and hydrogen were measured by gas chromatography (Quintron Microlyzer, Model SC) in breath samples and simultaneously obtained room air. The concentrations reported were corrected by subtracting the hydrogen and methane of room air.
The faecal sample was weighed and homogenized in a 400 series masticator (IUL Instruments, S.A., Barcelona, Spain) for 1 min. Aliquots of faeces were then transferred to individual vials for determination of pH, SCFA and DNA extraction. Faecal pH was measured using a FE20 pH metre with a MT InLab Solids Pro electrode (Mettler-Toledo, Columbus, OH, USA). Faecal SCFA were analysed by gas chromatography as previously described.22
Faecal bacteria were enumerated in triplicate by quantitative real-time PCR using 50 ng of DNA as previously described.23 Primers and probes for Bacteroides/Prevotella (Bacteroidetes),24 Clostridium coccoides group, Clostridium leptum group, bifidobacteria, Escherichia coli and total bacteria were as in Furet et al.25 Enumeration of Archaea has been previously described.22 The F:B ratio was calculated by dividing the sum of C. coccoides and C. leptum cells by the number of Bacteroidetes cells.
Intake of nutrients was calculated from the 3-day diet records using ESHA Research’s Food Processor SQL, Version 10.9.0 (Salem, OR, USA). Dietary intakes are reported for 93 participants as one diet record was misplaced.
Physical activity levels were assessed using the Modifiable Activity Questionnaire.26 The total weekly physical activity was estimated by multiplying the intensity of each activity (defined in metabolic equivalents (MET)) by duration. The product for each activity was summed to give a total activity score in MET hours per week. MET levels were obtained from a compendium of physical activities and include moderate-intensity activities between three and six METs and vigorous-intensity activities greater than six METs.27, 28
Statistical analyses were carried out with IBM SPSS Statistics Version 21 (Armonk, NY, USA). Statistical analysis of the independent variables was done using the Student’s t-test (two-tailed) for unpaired data. The χ2-test was calculated using a computer spreadsheet (Microsoft Office Excel 2007, Microsoft Corp, Seattle, WA, USA). Pearson’s correlations were used to test associations between variables with normal distributions, for nonnormally distributed data Spearman’s rank test was used. Differences with P-values⩽0.05 (two-tailed) were considered to be statistically significant. The results are expressed as means±s.e.m.
A principal component analysis (PCA; IBM SPSS Statistics Version 21) was carried out in order to determine whether distinct patterns could be discerned from the data. The number of factors to be extracted was based on scree plot analysis, Kaiser–Meyer–Olkin measure of sampling adequacy (>0.6), communalities (for variable included in the analysis were >0.5), eigenvalues (factors with eigenvalues >1 were retained) and proportion of common variance explained (>10%). Variables with a complex structure (more than one loading) were excluded. Varimax (orthogonal) rotation was used to obtain a set of independent interpretable factors. The resulting factor pattern was interpreted using factor loadings of >0.4. These analyses were carried out with all participants pooled. Archaea 16 S rRNA gene copies and breath methane data were not included in the PCA analysis as they have been previously studied.22
Results
Participants were divided into two groups based on BMI; those with a BMI⩽25 were placed in the lean (LN, n=52 or 55.3% of all participants) group and those with a BMI>25 were placed in the overweight and obese group (OWOB, n=37 or 44.7% of all participants) (Table 1). Although the age range for LN, 18–67 years, was similar to that for OWOB, 21–60 years, OWOB participants were significantly older than LN (P=0.03, Table 1). Study participants were largely Caucasian (52%) and Asian (38%); there was no significant difference in the sex or ethnic makeup of the LN and OWOB groups (Table 1). Breath methane was significantly higher in LN compared with OWOB participants, no significant differences in breath hydrogen were observed and physical activity levels were similar between the two groups (Table 1).
Individual faecal SCFA concentrations were not significantly different between the LN and OWOB groups but faecal total SCFA was significantly higher in OWOB compared with LN participants (P=0.05). As the OWOB participants were significantly older, SCFA concentrations were adjusted for age. After age adjustment, the OWOB group had significantly higher acetate (P=0.05), propionate (P=0.03), butyrate (P=0.05), valerate (P=0.03) and total SCFA concentrations (P=0.02) than the lean group (Table 1).
Microbial profile of the LN and OWOB participants was not different except that the LN participants had higher numbers of Escherichia coli compared with OWOB (P=0.005, Table 2). The ratio of Firmicutes to Bacteroidetes was not significantly different between the groups (Table 2). Five participants (two LN & three OWOB) had F:B ratios less than 1, which resulted in the log F:B ratio being negative in these participants. No significant differences in the proportion of participants in the LN and OWOB group who were Archaea positive was observed (34.6 vs 45.2%, χ2=0.29).
When the groups were combined, BMI was inversely related to the number of Bacteroidetes (r=−0.21, P=0.04; Figure 1) and E. coli (r=−0.34, P=0.002) but no association with log F:B ratio was observed (Figure 1). One male had a BMI that was >4s.d. above the mean for all subjects, and his results were excluded. In the entire cohort, significant negative correlations between Bacteroidetes and acetate, propionate, butyrate and total SCFA (TSCFA) concentrations were observed (Figure 2) and the ratio of Firmicutes to Bacteroidetes was positively correlated with acetate, propionate, butyrate and TSCFA concentrations (Figure 2). Faecal pH was negatively correlated with acetate (r=−0.51, P<0.0001), propionate (r=−0.44, P<0.0001), butyrate (r=−0.58, P<0.0001) and TSCFA (r=−0.56, P<0.0001).
Relationship between Bacteroidetes and acetate, propionate, butyrate and TSCFA (a); Log F:B ratio and acetate, propionate and butyrate and TSCFA (b) in the entire cohort. Filled circles (●) represent the LN participants and open circles (○) represent the OWOB participants. The slopes of the lines for the LN and OWOB groups were not significantly different so only the regression line for all the data is shown.
The groups were well matched for mean nutrient intakes and no significant differences were observed between LN and OWOB for daily macronutrient (Table 3) and micronutrient intakes (Supplementary Table 1). Significant negative correlations were observed between total dietary fibre (TDF) per 1000 kcals and SCFA concentrations in the LN group; acetate (r=−0.28, P=0.05), propionate (r=−0.35, P=0.01), butyrate (r=−0.36, P=0.01) and TSCFA (r=−0.36, P=0.01), but these correlations were not significant in OWOB group. Similar negative correlations between TDF per 1000 kcals and SCFA concentrations were seen when the groups were combined; propionate (r=−0.24, P=0.02), butyrate (r=−0.27, P=0.01), TSCFA (r=−0.27, P<0.01) and AC:PR ratio (r=0.22, P=0.03). Intake of polyunsaturated fatty acid (PUFA) was significantly negatively correlated with Bacteroidetes (r=−0.21, P=0.05), all bacteria (r=−0.22, P=0.04) and Firmicutes (r=−0.25, P=0.02; Supplementary Table 2).
Table 4 displays the results of PCA of the various variables in the entire cohort. PCA is a variable reduction technique. It reduces a large set of variables into a smaller set of variables, called ‘principal components’, which account for most of the variance in the original variables. In this study, a three-factor solution, which was supported by the retention criteria described in the methods section, explained 76.2% of the total variance (Table 4). These factors were interpreted as (1) a ‘gut microbiota’ factor, which explained 31.2% of the variation, with positive loadings of Firmicutes, sum of quantified bacteria, C. leptum, all bacteria and C. coccoides; (2) a ‘SCFA–F:B ratio’ factor, which explained 27.6% of the variation, with positive loadings of TSCFA, acetate, butyrate, propionate and F:B ratio; (3) a ‘diet’ factor, which explained 17.2% of the variation, with positive loadings of available CHO, energy and TDF. Among the many variables that were tested in the model but had to be excluded were Bacteroidetes, which exhibited a complex structure with a positive loading on gut microbiota and negative loading on SCFA component. Age, BMI, E. coli and bifidobacteria counts had low communalities and so were excluded.
Discussion
The present study was more comprehensive than previous studies in that it compared dietary intakes, microbial profiles, faecal SCFA concentrations and physical activity levels in LN and OWOB participants. The significantly higher SCFA concentrations in OWOB participants when compared with LN suggest that colonic fermentation differs between the groups. An inverse association was also observed between Bacteroidetes counts and BMI, significant positive associations were observed between F:B ratio and SCFA concentrations and negative associations between Bacteroidetes counts and SCFA concentrations. These results show that F:B ratio and faecal SCFA concentrations are interrelated and may be linked to adiposity.
The Firmicutes and Bacteroidetes are the two most predominant phyla in the human colon and together comprise >90%29 of the total gut microbiota. An inverse association was observed in the entire cohort between BMI and the number of Bacteroidetes suggesting that host adiposity and gut microbiota may be associated. However, the log F:B ratio in LN and OWOB participants did not differ as has been previously reported.17 Other studies have either found a lower proportion of Bacteroidetes and no significant difference in Firmicutes, a lower F:B ratio in obese compared with lean or no change in F:B ratio in lean and obese participants.18, 19, 20, 30 It has been suggested that a higher F:B ratio may be associated with an increased energy harvest from colonic fermentation and increased production of SCFA as has been seen in animal studies.16 In humans, weight loss in obese participants resulted in a decrease in the F:B ratio from the obese to the lean pattern.17 Jumpertz et al.31 observed in lean individuals a 20% increase in Firmicutes, and that a corresponding decrease in Bacteroidetes was associated with an increased energy harvest of ∼150 kcal. However, from studies done so far in humans, no consensus has emerged and it is unclear if F:B ratio differs in lean and obese humans. The reasons for the contradictions in study results vary and may be related to low sample size, different methodologies (qPCR vs 16S rRNA gene-based sequencing vs denaturing gradient gel electrophoresis), study populations and unaccounted for confounding factors, for example, diet. In this study, a significant difference in F:B ratio was not observed between the groups, but BMI was associated with Bacteroidetes. This may occur because a continuum or gradient of species functionality exists in humans rather than a discontinuous variation with segregated types of gut bacteria.32 If the two major phyla exhibit a continuous gradient, F:B ratios will overlap between the LN and OWOB groups and this may mask differences between groups. A more accurate indicator of colonic fermentation differences may be to study SCFA metabolism in LN and OWOB participants.
Animal studies have shown that gut microbiota composition and concentrations of SCFA differ in lean and obese animals.15, 16 In this study, in adult humans, the OWOB group had significantly higher faecal acetate, propionate, butyrate, valerate and TSCFA concentrations compared with the LN group, a finding that has been reported earlier in similar studies.18, 21 Higher faecal SCFA concentrations could result from increased SCFA production and/or decreased SCFA absorption in the OWOB group or the obese microbiome may be associated with fewer microbial species that utilize SCFA as an energy source. Production of SCFA in humans is regulated by a number of host, environmental, dietary and microbial factors.33 The main factors that control SCFA production are interrelated and include type and amount of available substrate, composition of the gut microbiota and gut transit time.8, 34 In this study, dietary intakes in lean and obese participants were not significantly different and the groups did not have significantly different gut microbiota composition; transit times were not measured. The increased SCFA concentrations in the OWOB group were not explained by an increased efficiency of the fermentation process caused by the presence of Archaea because no significant difference in proportion of individuals who were Archaea positive was observed and the OWOB group had significantly lower concentrations of breath methane. In addition to the aforementioned factors, several other factors may indirectly affect gut microbiota composition and SCFA production including ageing, neuroendocrine system activity, stress and the gut environment.33, 35 The OWOB participants in this study were older and ageing could affect gut microbiota via the many effects associated with it, for example, its effect on colonic transit times, but, considering the mean age for the OWOB group was below 40 years, any effect of age on faecal SCFA concentrations in this group was minimal.
Alternatively, the higher faecal SCFA concentrations in the OWOB group may be due to decreased colonic absorption of SCFA. Absorption of SCFA from the colon is very efficient with less than 5% being excreted in faeces. Many studies measure faecal concentrations of SCFA as a surrogate to determine SCFA production but faecal SCFA concentrations reflects a balance between colonic production and absorption of SCFA. Previous work in healthy participants has shown that faecal SCFA concentrations may reflect SCFA absorption rather than its production.36 However, preliminary studies by our group have found no differences in SCFA absorption between lean and obese participants.37 Future studies need to look at an in vitro production of SCFA in LN and OWOB participants; specifically, how unabsorbable carbohydrate and gut microbiota from individuals with different F:B ratios influences SCFA production patterns in these groups.
It is unclear if the higher faecal SCFA concentrations in the OWOB group resulted from differences in F:B ratios because the groups had similar F:B ratios; nevertheless, our results show that in humans, F:B ratio and faecal SCFA are positively associated. The results of the PCA also show that F:B ratio and faecal SCFA are associated. The PCA identified distinct gut microbiota and SCFA–F:B ratio components which together account for 59% of the variation. Interestingly, F:B ratio loaded with the individual and total SCFA and not with the gut microbiota suggesting that SCFA and F:B ratio vary together and may be interrelated. Taken together, the results show that colonic fermentation may be altered by the presence of an obese microbiome leading to increased SCFA production. This is not consistent with the current knowledge that high fibre diets, which would also increase SCFA production, reduce risk for obesity. More studies are needed to determine the metabolic fate of the SCFA produced and to see whether the kinetics of SCFA production and metabolism differ in lean and obese humans.
Dietary intake is a key factor in the pathophysiology of obesity, and habitual dietary intakes also have a role in determining gut microbiota composition.38, 39, 40, 41, 42 No significant differences in dietary intakes between the two groups in this study were observed. Interestingly, TDF intakes were similar in the two groups even though the OWOB group had significantly higher faecal SCFA concentrations and this may imply that colonic fermentation in the obese microbiome is more efficient. It is also possible that there might have been some under reporting of dietary intakes in the OWOB group. Faecal SCFA were inversely related to the intake of TDF per 1000 kcals in LN but not in OWOB participants, which may suggest that the lean microbiome may ferment dietary fibre differently from the obese microbiome. The inverse association may also suggest that LN participants may absorb more SCFA or that there might be bacteria associated with the lean microbiome that may breakdown SCFA in order to utilize it as an energy source. Negative correlations were also observed between PUFA intakes and various microbial phyla. Most of the genes present in the gut microbiome do not generally engage in fatty acid breakdown.43 However, studies have shown that PUFA significantly alters colonic fermentation.44, 45 In vitro studies have shown a modulatory effect of PUFA concentrations on the growth and adhesion of different Lactobacillus strains.46 In rats, a decrease in the number of Bacteroides fragilis species and total anaerobes in the caecum was observed after fish oil consumption, which is rich in long chain n-3 PUFA.47 Dietary fatty acids may alter the fatty acid composition of the intestinal wall and modify the attachment site to promote or inhibit microbial colonization.
In conclusion, this human study shows that differences in colonic fermentation exist between LN and OWOB participants and that adiposity may be linked to gut microbiota and faecal SCFA concentrations in healthy humans. But, more studies need to be done in order to understand how this may affect energy homeostasis and the development of obesity. Measuring faecal SCFA concentrations to understand the role SCFA metabolism has in obesity is limited and future work needs to study SCFA kinetics in lean and obese humans.
References
Macfarlane GT, Cummings JH . The colonic flora, fermentation and large bowel digestive function. In: Phillips SF, Pemberton JH, Shorter RG, (eds). The Large Intestine: Physiology, Pathophysiology and Disease. Raven Press Ltd: New York, NY, USA, 1991. pp 51–92.
Cummings JH . Fermentation in the human large intestine: evidence and implications for health. Lancet 1983; 1: 1206–1209.
Smith CJ, Bryant MP . Introduction to metabolic activities of intestinal bacteria. Am J Clin Nutr 1979; 32: 149–157.
Topping DL, Clifton PM . Short-chain fatty acids and human colonic function:roles of resistant starch and nonstarch polysaccharides. Physiol Rev 2001; 81: 1031–1064.
Cummings JH . Short chain fatty acids in the human colon. Gut 1981; 22: 763–779.
Miller TL, Wolin MJ . Fermentations by saccharolytic intestinal bacteria. Am J Clin Nutr 1979; 32: 164–172.
Cummings JH, Macfarlane GT . The control and consequences of bacterial fermentation in the human colon. J Appl Bacteriol 1991; 70: 443–459.
Macfarlane GT, Gibson SR . Microbiological aspects of the production of short-chain fatty acids in the large bowel. In: Cummings JH, Rombeau JL, Sakata T, (eds). Physiological and Clinical Aspects of Short Chain Fatty Acid Metabolism. Cambridge University Press: Cambridge, 1995. pp 87–105.
Royall D, Wolever TMS, Jeejeebhoy KN . Clinical significance of colonic fermentation. Am J Gastroenterol 1990; 85: 1307–1312.
Du H, van der ADL, Boshuizen HC, Forouhi NG, Wareham NJ, Halkjaer J et al. Dietary fibre and subsequent changes in body weight and waist circumference in European men and women. Am J Clin Nutr 2010; 91: 329–336.
Anderson JW, Baird P, Davis RH Jr, Ferreri S, Knudtson M, Koraym A et al. Health benefits of dietary fibre. Nutr Rev 2009; 67: 88–205.
Liu S, Willett WC, Manson JE, Hu FB, Rosner B, Colditz G . Relation between changes in intakes of dietary fibre and grain products and changes in weight and development of obesity among middle-aged women. Am J Clin Nutr 2003; 78: 920–927.
Freeland KR, Wolever TM . Acute effects of intravenous and rectal acetate onglucagon-like peptide-1, peptide YY, ghrelin, adiponectin and tumour necrosis factor-alpha. Br J Nutr 2010; 103: 460–466.
Tarini J, Wolever TM . The fermentable fibre inulin increases postprandialserum short-chain fatty acids and reduces free-fatty acids and ghrelin in healthy subjects. Appl Physiol Nutr Metab 2010; 35: 9–16.
Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI . Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 2005; 102: 11070–11075.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI . An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006; 444: 1027–1031.
Ley RE, Turnbaugh PJ, Klein S, Gordon JI . Microbial ecology: human gut microbes associated with obesity. Nature 2006; 444: 1022–1023.
Schwiertz A, Taras D, Schäfer K, Beijer S, Bos NA, Donus C et al. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010; 18: 190–195.
Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE et al. A core gut microbiome in obese and lean twins. Nature 2009; 457: 480–484.
Duncan SH, Lobley GE, Holtrop G, Ince J, Johnstone AM, Louis P et al. Human colonic microbiota associated with diet, obesity and weight loss. Int J Obes 2008; 32: 1720–1724.
Teixeira TFS, Grześkowiak L, Franceschini SCC, Bressan J, CLLF Ferreira, Peluzio MCG . Higher level of faecal SCFA in women correlates with metabolic syndrome risk factors. Br J Nutr 2013; 109: 914–919.
Fernandes J, Wang A, Su W, Rozenbloom SR, Taibi A, Comelli EM et al. Age, dietary fibre, breath methane, and faecal short chain fatty acids are interrelated in Archaea-positive humans. J Nutr 2013; 143: 1269–1275.
Mouzaki M, Comelli EM, Arendt BM, Bonengel J, Fung SK, Fischer SE et al. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology 2013; 58: 120–127.
Mariat D, Firmesse O, Levenez F, Guimarăes V, Sokol H, Doré J et al. The Firmicutes/Bacteroidetes ratio of the human microbiota changes with age. BMC Microbiol 2009; 9: 123.
Furet JP, Firmesse O, Gourmelon M, Bridonneau C, Tap J, Mondot S et al. Comparative assessment of human and farm animal faecal microbiota using real-time quantitative PCR. FEMS Microbiol Ecol 2009; 68: 351–362.
Kriska AM, Caspersen CJ . Modifiable activity questionnaire. In: a Collection of Physical Activity Questionnaires for Health-Related Research Kriska AM, Caspersen CJ, Eds. Med Sci Sports Exer 1997; 29 (Suppl 6): S73–S78.
Ainsworth BE, Haskell WL, Whitt MC, Irwin ML, Swartz AM, Strath SJ et al. Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exer 2000; 32 (Suppl 9): S498–S504.
Ainsworth BE, Haskell WL, Leon AS, Jacobs DR Jr, Montoye HJ, Sallis JF et al. Compendium of physical activities: classification of energycosts of human physical activities. Med Sci Sports Exer 1993; 25: 71–80.
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M et al. Diversity of the human intestinal microbial flora. Science 2005; 308: 1635–1638.
Armougom F, Henry M, Vialettes B, Raccah D, Raoult D . Monitoring bacterialcommunity of human gut microbiota reveals an increase in Lactobacillus in obese patients and Methanogens in anorexic patients. PLoS One 2009; 4: e7125.
Jumpertz R, Le DS, Turnbaugh PJ, Trinidad C, Bogardus C, Gordon JI et al. Energy-balance studies reveal associations between gut microbes, caloric load, and nutrient absorption in humans. Am J Clin Nutr 2011; 94: 58–65.
Jeffery IB, Claesson MJ, O’Toole PW, Shanahan F . Categorization of the gut microbiota: enterotypes or gradients? Nat Rev Microbiol 2012; 10: 591–592.
Macfarlane S, Macfarlane GT . Regulation of short-chain fatty acid production. Proc Nutr Soc 2003; 62: 67–72.
Cummings JH . Diet and transit through the gut. J Plant Foods 1978; 3: 83–95.
Louis P, Scott KP, Duncan SH, Flint HJ . Understanding the effects of diet onbacterial metabolism in the large intestine. J Appl Microbiol 2007; 102: 1197–1208.
Vogt JA . Wolever TMS. Faecal acetate is inversely related to acetate absorption from the human rectum and distal colon. J Nutr 2003; 133: 3145–3148.
Rahat-Rozenbloom S, Fernandes J, Gloor GB, Wolever TM . Evidence for greater production of colonic short-chain fatty acids in overweight than lean humans. Int J Obes 2014. e-pub ahead of print 19 March 2014 doi:10.1038/ijo.2014.46.
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014; 505: 559–563.
McNulty NP, Wu M, Erickson AR, Pan C, Erickson BK, Martens EC et al. Effects of diet on resource utilization by a model human gut microbiota containing Bacteroides cellulosilyticus WH2, a symbiont with an extensive glycobiome. PLoS Biol 2013; 11: e1001637.
Muegge BD, Kuczynski J, Knights D, Clemente JC, González A, Fontana L et al. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 2011; 332: 970–974.
De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA 2010; 107: 14691–14696.
Mai V, McCrary QM, Sinha R, Glei M . Associations between dietary habits and body mass index with gut microbiota composition and faecal water genotoxicity: an observational study in African American and Caucasian American volunteers. Nutr J 2009; 8: 49.
Kurokawa K, Itoh T, Kuwahara T, Oshima K, Toh H, Toyoda A et al. Comparative metagenomics revealed commonly enriched gene sets in human gut microbiomes. DNA Res 2007; 14: 169–181.
Thompson L, Spiller RC . Impact of polyunsaturated fatty acids on human colonic bacterial metabolism: an in vitro and in vivo study. Br J Nutr 1995; 74: 733–741.
Treon SP, Fox ES, Broitman SA . Marine oils in the modulation of colonic flora and pH: considerations for colon cancer. Microb Ecol Health Dis 1989; 2: 115–122.
Kankaanpää PE, Salminen SJ, Isolauri E, Lee YK . The influence of polyunsaturated fatty acids on probiotic growth and adhesion. FEMS Microbiol Lett 2001; 194: 149–153.
Conlon MA, Bird AR . Interactive and individual effects of dietary non-digestible carbohydrates and oils on DNA damage, SCFA and bacteria in the large bowel of rats. Br J Nutr 2009; 101: 1171–1177.
Acknowledgements
This work was supported by grant no. 486906 (TMS Wolever, EM Comelli) from the Canadian Institutes for Health Research (CIHR), Institute of Nutrition, Metabolism and Diabetes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Nutrition & Diabetes website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Fernandes, J., Su, W., Rahat-Rozenbloom, S. et al. Adiposity, gut microbiota and faecal short chain fatty acids are linked in adult humans. Nutr & Diabetes 4, e121 (2014). https://doi.org/10.1038/nutd.2014.23
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nutd.2014.23
This article is cited by
-
Sex differences in a mouse model of diet-induced obesity: the role of the gut microbiome
Biology of Sex Differences (2024)
-
Evaluation of gut microbiota of iranian patients with celiac disease, non-celiac wheat sensitivity, and irritable bowel syndrome: are there any similarities?
BMC Gastroenterology (2023)
-
Resistant starches from dietary pulses modulate the gut metabolome in association with microbiome in a humanized murine model of ageing
Scientific Reports (2023)
-
Modulatory effect of PPAR-γ by acetate on cardiorenometabolic disturbance associated with high fat diet–fed insulin-resistant male Wistar rats
Comparative Clinical Pathology (2023)
-
Lemon fermented products prevent obesity in high-fat diet-fed rats by modulating lipid metabolism and gut microbiota
Journal of Food Science and Technology (2023)