Abstract
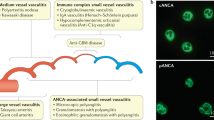
Polyarteritis nodosa (PAN) is a vasculitic disease characterized primarily by necrotizing vasculitis — inflammatory lesions in blood vessels that lead to vessel wall necrosis. Our understanding of PAN and necrotizing vasculitis has evolved over time. In addition to PAN, necrotizing vasculitis is now a recognized feature of a broad range of diseases with different aetiopathogenesis. For example, necrotizing vasculitis associated with hepatitis B virus infection has a different aetiopathogeneis to PAN and is now classified as a separate disease. Additionally, although 'classic' PAN is not an inherited disease, mutations in specific genes, such as ADA2 (also known as CECR1), can result in a necrotizing vasculopathy similar to PAN. The literature also suggests that the course of PAN differs in childhood-onset disease and in cases confined to the skin (so-called cutaneous PAN). Dissecting PAN and other autoinflammatory diseases with PAN-like features has enabled more-specific therapies and might also help us better understand the pathogenesis of these devastating conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hoffman, G. S. & Calabrese, L. H. Vasculitis: determinants of disease patterns. Nat. Rev. Rheumatol. 10, 454–462 (2014).
Jennette, J. C. & Falk, R. J. Pathogenesis of antineutrophil cytoplasmic autoantibody-mediated disease. Nat. Rev. Rheumatol. 10, 463–473 (2014).
Kussmaul, A. & Maier, R. Über eine bisher nicht beschriebene eigentümliche Arterienerkrankung (periarteritis nodosa) [German]. Deutsch. Arch. Klin. Med. 1, 484–518 (1866).
Ferrari, E. Über polyarteritis acuta nodosa (sogenannte periarteritis nodosa) und ihre beziehungen zur polymyositis und polyneuritis acuta [German]. Beitr. Pathol. Anat. 34, 350–386 (1903).
Zeek, P. M. Periarteritis nodosa; a critical review. Am. J. Clin. Pathol. 22, 777–790 (1952).
Godman, G. C. & Churg, J. Wegener's granulomatosis: pathology and review of the literature. AMA Arch. Pathol. 58, 533–553 (1954).
Jennette, J. C. et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum. 37, 187–192 (1994).
Wegener, F. Periarteritis nodosa und ihre bedeutung bei einem fall von chronischer ruhr [German]. Dtsch. Z. Gerichtl. Med. 25, 93–101 (1935).
Davies, D. J., Moran, J. E., Niall, J. F. & Ryan, G. B. Segmental necrotising glomerulonephritis with antineutrophil antibody: possible arbovirus aetiology? Br. Med. J. (Clin. Res. Ed.) 285, 606 (1982).
van der Woude, F. J. et al. Autoantibodies against neutrophils and monocytes: tool for diagnosis and marker of disease activity in Wegener's granulomatosis. Lancet 1, 425–429 (1985).
Eleftheriou, D. et al. Systemic polyarteritis nodosa in the young: a single-center experience over thirty-two years. Arthritis Rheum. 65, 2476–2485 (2013).
Iudici, M. et al. Brief report: childhood-onset systemic necrotizing vasculitides: long-term data from the French Vasculitis Study Group Registry. Arthritis Rheumatol. 67, 1959–1965 (2015).
Maeda, M. et al. Clinical observation of 14 cases of childhood polyarteritis nodosa in Japan. Acta Paediatr. Jpn 39, 277–279 (1997).
Merlin, E. et al. Long-term outcome of children with pediatric-onset cutaneous and visceral polyarteritis nodosa. Joint Bone Spine 82, 251–257 (2015).
Mondal, R. et al. Childhood polyarteritis nodosa: a prospective multicentre study from eastern India. Indian J. Pediatr. 81, 371–374 (2014).
Ozen, S. Juvenile polyarteritis: is it a different disease? J. Rheumatol. 31, 831–832 (2004).
Ozen, S., Besbas, N., Saatci, U. & Bakkaloglu, A. Diagnostic criteria for polyarteritis nodosa in childhood. J. Pediatr. 120, 206–209 (1992).
Lightfoot, R. W. et al. The American College of Rheumatology 1990 criteria for the classification of polyarteritis nodosa. Arthritis Rheum. 33, 1088–1093 (1990).
Jennette, J. C. et al. 2012 revised International Chapel Hill Consensus Conference nomenclature of vasculitides. Arthritis Rheum. 65, 1–11 (2013).
Ozen, S. & Bilginer, Y. A clinical guide to autoinflammatory diseases: familial Mediterranean fever and next-of-kin. Nat. Rev. Rheumatol. 10, 135–147 (2014).
Ozen, S. et al. Juvenile polyarteritis: results of a multicenter survey of 110 children. The Journal of pediatrics. 145, 517–522 (2004).
Ozen, S. et al. EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part II: final classification criteria. Ann. Rheum. Dis. 69, 798–806 (2010).
Alibaz-Oner, F. et al. The clinical spectrum of medium-sized vessel vasculitis. Arthritis Care Res. http://dx.doi.org/10.1002/acr.23007 (2017).
Shimojima, Y., Ishii, W., Kishida, D., Fukushima, K. & Ikeda, S. I. Imbalanced expression of dysfunctional regulatory T cells and T-helper cells relates to immunopathogenesis in polyarteritis nodosa. Mod. Rheumatol. 27, 102–109 (2017).
Wigley, R. D. & Couchman, K. G. Polyarteritis nodosa-like disease in outbred mice. Nature 211, 319–320 (1966).
Jennette, J. C. & Falk, R. J. Small-vessel vasculitis. N. Engl. J. Med. 337, 1512–1523 (1997).
Gocke, D. J. et al. Association between polyarteritis and Australia antigen. Lancet 2, 1149–1153 (1970).
Trepo, C. & Thivolet, J. Hepatitis associated antigen and periarteritis nodosa (PAN). Vox Sang. 19, 410–411 (1970).
Guillevin, L. et al. Polyarteritis nodosa related to hepatitis B virus. A prospective study with long-term observation of 41 patients. Medicine (Baltimore) 74, 238–253 (1995).
Zhou, Q. et al. Loss-of-function mutations in TNFAIP3 leading to A20 haploinsufficiency cause an early-onset autoinflammatory disease. Nat. Genet. 48, 67–73 (2016).
Watts, R. A. & Scott, D. G. I. in Vasculitis 2nd edn (eds Ball, G. V. & Bridges, S. L.) 7–21 (Oxford Univ. Press, 2008).
Ben-Zvi, I. & Livneh, A. Chronic inflammation in FMF: markers, risk factors, outcomes and therapy. Nat. Rev. Rheumatol. 7, 105–112 (2011).
Balbir-Gurman, A., Nahir, A. M. & Braun-Moscovici, Y. Vasculitis in siblings with familial Mediterranean fever: a report of three cases and review of the literature. Clin. Rheumatol. 26, 1183–1185 (2007).
Ozen, S. et al. Polyarteritis nodosa in patients with familial Mediterranean fever (FMF): a concomitant disease or a feature of FMF? Semin. Arthritis Rheum. 30, 281–287 (2001).
Tunca, M. et al. Familial Mediterranean fever (FMF) in Turkey: results of a nationwide multicenter study. Medicine (Baltimore) 84, 1–11 (2005).
Yalcinkaya, F. et al. Prevalence of the MEFV gene mutations in childhood polyarteritis nodosa. J. Pediatr. 151, 675–678 (2007).
Hernandez-Rodriguez, J., Alba, M. A., Prieto-Gonzalez, S. & Cid, M. C. Diagnosis and classification of polyarteritis nodosa. J. Autoimmun. 48–49, 84–89 (2014).
Kalyoncu, M. et al. Are carriers for MEFV mutations “healthy”? Clin. Exp. Rheumatol. 24 (5 Suppl. 42), S120–S122 (2006).
Akse-Onal, V. et al. Decrease in the rate of secondary amyloidosis in Turkish children with FMF: are we doing better? Eur. J. Pediatr. 169, 971–974 (2010).
Navon Elkan, P. et al. Mutant adenosine deaminase 2 in a polyarteritis nodosa vasculopathy. N. Engl. J. Med. 370, 921–931 (2014).
Zhou, Q. et al. Early-onset stroke and vasculopathy associated with mutations in ADA2. N. Engl. J. Med. 370, 911–920 (2014).
Batu, E. D. et al. A case series of adenosine deaminase 2-deficient patients emphasizing treatment and genotype-phenotype correlations. J. Rheumatol. 42, 1532–1534 (2015).
Nanthapisal, S. et al. Deficiency of adenosine deaminase type 2: a description of phenotype and genotype in fifteen cases. Arthritis Rheumatol. 68, 2314–2322 (2016).
Stoffels, M. Z. Q., Chen, C., Aksentijevich, I., Kastner, D. & Merkel, P. A. Update on CECR1 molecular diagnostics: new mutations in the deficiency of ADA2 (DADA2) and the North American polyarteritis nodosa (PAN) cohort. Pediatr. Rheumatol. 13 (Suppl. 1), O20 (2015).
Liu, Y. et al. Activated STING in a vascular and pulmonary syndrome. N. Engl. J. Med. 371, 507–518 (2014).
Henderson, L. A. et al. Expanding the spectrum of recombination-activating gene 1 deficiency: a family with early-onset autoimmunity. J. Allergy Clin. Immunol. 132, 969–971.e2 (2013).
Gadola, S. D., Moins-Teisserenc, H. T., Trowsdale, J., Gross, W. L. & Cerundolo, V. TAP deficiency syndrome. Clin. Exp. Immunol. 121, 173–178 (2000).
Mukhtyar, C. et al. EULAR recommendations for the management of primary small and medium vessel vasculitis. Ann. Rheum. Dis. 68, 310–317 (2009).
Yates, M. et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann. Rheum. Dis. 75, 1583–1594 (2016).
Dolezalova, P. et al. Disease activity assessment in childhood vasculitis: development and preliminary validation of the Paediatric Vasculitis Activity Score (PVAS). Ann. Rheum. Dis. 72, 1628–1633 (2013).
Beresford, M. W. Share recommendations on connective tissue diseases [abstract SP0062]. Ann. Rheum. Dis. 75 (Suppl. 2), 16 (2016).
Xiao, H. et al. Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulonephritis and vasculitis in mice. J. Clin. Invest. 110, 955–963 (2002).
Jennette, J. C., Falk, R. J., Hu, P. & Xiao, H. Pathogenesis of antineutrophil cytoplasmic autoantibody-associated small-vessel vasculitis. Annu. Rev. Pathol. 8, 139–160 (2013).
Jennette, J. C. & Falk, R. J. L1. Pathogenesis of ANCA-associated vasculitis: observations, theories and speculations. Presse Med. 42, 493–498 (2013).
Jennette, J. C. L17. What can we expect from the revised Chapel Hill Consensus Conference nomenclature of vasculitis? Presse Med. 42, 550–555 (2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
S.O. declares that she has acted as a consultant for Novartis and R-Pharm, and has received honoraria from Sobi.
Rights and permissions
About this article
Cite this article
Ozen, S. The changing face of polyarteritis nodosa and necrotizing vasculitis. Nat Rev Rheumatol 13, 381–386 (2017). https://doi.org/10.1038/nrrheum.2017.68
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2017.68
This article is cited by
-
Inflammatory comorbidities ın the largest pediatric Familial Mediterranean fever cohort: a multicenter retrospective study of Pediatric Rheumatology Academy (PeRA)-Research Group (RG)
Clinical Rheumatology (2024)
-
Differentiation of idiopathic pulmonary hemosiderosis from rheumatologic and autoimmune diseases causing diffuse alveolar hemorrhage: establishing a diagnostic approach
Clinical Rheumatology (2022)
-
A young girl with severe polyarteritis nodosa successfully treated with tocilizumab: a case report
Pediatric Rheumatology (2021)
-
Thoracic mass lesion in a 14-year-old girl: Answers
Pediatric Nephrology (2021)