Key Points
-
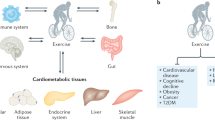
Persistent systemic inflammation is a typical feature of inflammatory rheumatic diseases such as rheumatoid arthritis and systemic lupus erythematosus
-
Chronic inflammation predisposes to insulin resistance, dyslipidaemia, endothelial dysfunction, accelerated atherosclerosis and neurodegeneration, and thereby to a network of chronic diseases such as type 2 diabetes mellitus, cardiovascular disease and dementia
-
Disease-specific symptoms and comorbidities might negatively affect mobility, physical activity and physical capacity of patients with inflammatory rheumatic diseases
-
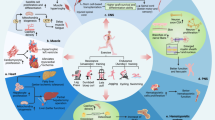
Physical inactivity can cause the accumulation of visceral fat, which, along with comorbidities, might further enhance the development of chronic diseases in a 'vicious cycle' of chronic inflammation
-
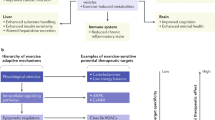
During exercise, skeletal muscle produces myokines, which might mediate either a direct anti-inflammatory response with each bout of exercise or improvements in comorbidities, thereby indirectly having anti-inflammatory effects
-
Exercise is no longer thought to aggravate inflammation; rather, physical activity is now advocated as an anti-inflammatory therapy for patients with rheumatic diseases
Abstract
Persistent systemic inflammation, a typical feature of inflammatory rheumatic diseases, is associated with a high cardiovascular risk and predisposes to metabolic disorders and muscle wasting. These disorders can lead to disability and decreased physical activity, exacerbating inflammation and the development of a network of chronic diseases, thus establishing a 'vicious cycle' of chronic inflammation. During the past two decades, advances in research have shed light on the role of exercise as a therapy for rheumatic diseases. One of the most important of these advances is the discovery that skeletal muscle communicates with other organs by secreting proteins called myokines. Some myokines are thought to induce anti-inflammatory responses with each bout of exercise and mediate long-term exercise-induced improvements in cardiovascular risk factors, having an indirect anti-inflammatory effect. Therefore, contrary to fears that physical activity might aggravate inflammatory pathways, exercise is now believed to be a potential treatment for patients with rheumatic diseases. In this Review, we discuss how exercise disrupts the vicious cycle of chronic inflammation directly, after each bout of exercise, and indirectly, by improving comorbidities and cardiovascular risk factors. We also discuss the mechanisms by which some myokines have anti-inflammatory functions in inflammatory rheumatic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McInnes, I. B. & Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat. Rev. Immunol. 7, 429–442 (2007).
Sitia, S. et al. Cardiovascular involvement in systemic autoimmune diseases. Autoimmun. Rev. 8, 281–286 (2009).
Weiss, G. & Schett, G. Anaemia in inflammatory rheumatic diseases. Nat. Rev. Rheumatol. 9, 205–215 (2013).
Van, G. H. & Charles-Schoeman, C. The heart in inflammatory myopathies. Rheum. Dis. Clin. North Am. 40, 1–10 (2014).
Roman, M. J. et al. Prevalence and correlates of accelerated atherosclerosis in systemic lupus erythematosus. N. Engl. J. Med. 349, 2399–2406 (2003).
Del Rincón, I. D., Williams, K., Stern, M. P., Freeman, G. L. & Escalante, A. High incidence of cardiovascular events in a rheumatoid arthritis cohort not explained by traditional cardiac risk factors. Arthritis Rheum. 44, 2737–2745 (2001).
Esdaile, J. M. et al. Traditional Framingham risk factors fail to fully account for accelerated atherosclerosis in systemic lupus erythematosus. Arthritis Rheum. 44, 2331–2337 (2001).
Han, C. et al. Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J. Rheumatol. 33, 2167–2172 (2006).
Mancuso, C. A., Perna, M., Sargent, A. B. & Salmon, J. E. Perceptions and measurements of physical activity in patients with systemic lupus erythematosus. Lupus 20, 231–242 (2011).
Sokka, T. et al. Physical inactivity in patients with rheumatoid arthritis: data from twenty-one countries in a cross-sectional, international study. Arthritis Rheum. 59, 42–50 (2008).
Prioreschi, A., Hodkinson, B., Avidon, I., Tikly, M. & McVeigh, J. A. The clinical utility of accelerometry in patients with rheumatoid arthritis. Rheumatology (Oxford) 52, 1721–1727 (2013).
Petersen, A. M. & Pedersen, B. K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 98, 1154–1162 (2005).
Pedersen, B. K. Muscle as a secretory organ. Compr. Physiol. 3, 1337–1362 (2013).
Pedersen, B. K. & Febbraio, M. A. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 8, 457–465 (2012).
Steensberg, A., Fischer, C. P., Keller, C., Møller, K. & Pedersen, B. K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Endocrinol. Metab. 285, E433–E437 (2003).
Sutej, P. G. & Hadler, N. M. Current principles of rehabilitation for patients with rheumatoid arthritis. Clin. Orthop. Relat. Res. 265, 116–124 (1991).
Lundberg, I. E. & Nader, G. A. Molecular effects of exercise in patients with inflammatory rheumatic disease. Nat. Clin. Pract. Rheumatol. 4, 597–604 (2008).
Pearson, T. A. et al. Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 107, 499–511 (2003).
Wallin, K. et al. Midlife rheumatoid arthritis increases the risk of cognitive impairment two decades later: a population-based study. J. Alzheimers Dis. 31, 669–676 (2012).
Matter, C. M. & Handschin, C. RANTES (regulated on activation, normal T cell expressed and secreted), inflammation, obesity, and the metabolic syndrome. Circulation 115, 946–948 (2007).
Khovidhunkit, W., Memon, R. A., Feingold, K. R. & Grunfeld, C. Infection and inflammation-induced proatherogenic changes of lipoproteins. J. Infect. Dis. 181 (Suppl. 3), S462–S472 (2000).
Sattar, N., McCarey, D. W., Capell, H. & McInnes, I. B. Explaining how “high-grade” systemic inflammation accelerates vascular risk in rheumatoid arthritis. Circulation 108, 2957–2963 (2003).
Takeda, S., Sato, N. & Morishita, R. Systemic inflammation, blood-brain barrier vulnerability and cognitive/non-cognitive symptoms in Alzheimer disease: relevance to pathogenesis and therapy. Front. Aging Neurosci. 6, 171 (2014).
Collins, L. M., Toulouse, A., Connor, T. J. & Nolan, Y. M. Contributions of central and systemic inflammation to the pathophysiology of Parkinson's disease. Neuropharmacology 62, 2154–2168 (2012).
Plomgaard, P. et al. Tumor necrosis factor-α induces skeletal muscle insulin resistance in healthy human subjects via inhibition of Akt substrate 160 phosphorylation. Diabetes 54, 2939–2945 (2005).
Nguyen, M. T. et al. JNK and tumor necrosis factor-α mediate free fatty acid-induced insulin resistance in 3T3-L1 adipocytes. J. Biol. Chem. 280, 35361–35371 (2005).
Plomgaard, P., Fischer, C. P., Ibfelt, T., Pedersen, B. K. & van Hall, G. Tumor necrosis factor-α modulates human in vivo lipolysis. J. Clin. Endocrinol. Metab. 93, 543–549 (2008).
Vallance, P., Collier, J. & Bhagat, K. Infection, inflammation, and infarction: does acute endothelial dysfunction provide a link? Lancet 349, 1391–1392 (1997).
Yudkin, J. S., Eringa, E. & Stehouwer, C. D. “Vasocrine” signalling from perivascular fat: a mechanism linking insulin resistance to vascular disease. Lancet 365, 1817–1820 (2005).
Plomgaard, P., Keller, P., Keller, C. & Pedersen, B. K. TNF-α, but not IL-6, stimulates plasminogen activator inhibitor-1 expression in human subcutaneous adipose tissue. J. Appl. Physiol. 98, 2019–2023 (2005).
Murakami, M. & Nishimoto, N. The value of blocking IL-6 outside of rheumatoid arthritis: current perspective. Curr. Opin. Rheumatol. 23, 273–277 (2011).
Calabrese, L. H. & Rose-John, S. IL-6 biology: implications for clinical targeting in rheumatic disease. Nat. Rev. Rheumatol. http://dx.doi.org/10.1038/nrrheum.2014.127.
Pedersen, B. K. & Febbraio, M. A. Muscle as an endocrine organ: focus on muscle-derived Interleukin-6. Physiol. Rev. 88, 1379–1406 (2008).
Muñoz-Cánoves, P., Scheele, C., Pedersen, B. K. & Serrano, A. L. Interleukin-6 myokine signaling in skeletal muscle: a double-edged sword? FEBS J. 280, 4131–4148 (2013).
Pal, M., Febbraio, M. A. & Whitham, M. From cytokine to myokine: the emerging role of interleukin-6 in metabolic regulation. Immunol. Cell Biol. 92, 331–339 (2014).
Steensberg, A. et al. Acute interleukin-6 administration does not impair muscle glucose uptake or whole body glucose disposal in healthy humans. J. Physiol. 548, 631–638 (2003).
Febbraio, M. A., Hiscock, N., Sacchetti, M., Fischer, C. P. & Pedersen, B. K. Interleukin-6 is a novel factor mediating glucose homeostasis during skeletal muscle contraction. Diabetes 53, 1643–1648 (2004).
Wallenius, V. et al. Interleukin-6-deficient mice develop mature-onset obesity. Nat. Med. 8, 75–79 (2002).
Fosgerau, K. et al. Interleukin-6 autoantibodies are involved in the pathogenesis of a subset of type 2 diabetes. J. Endocrinol. 204, 265–273 (2010).
Kubaszek, A. et al. Promoter polymorphisms of the TNF-α (G.–308A) and IL-6 (C–174G) genes predict the conversion from impaired glucose tolerance to type 2 diabetes: the Finnish Diabetes Prevention Study. Diabetes 52, 1872–1876 (2003).
Chugai Pharmaceutical. Atlizumab: anti-IL-6 receptor antibody-Chugai, anti-interleukin-6 receptor antibody-Chugai, MRA-Chugai. BioDrugs 17, 369–372 (2003).
Choy, E. H. et al. Therapeutic benefit of blocking interleukin-6 activity with an anti-interleukin-6 receptor monoclonal antibody in rheumatoid arthritis: a randomized, double-blind, placebo-controlled, dose-escalation trial. Arthritis Rheum. 46, 3143–3150 (2002).
Nishimoto, N. et al. Treatment of rheumatoid arthritis with humanized anti-interleukin-6 receptor antibody: a multicenter, double-blind, placebo-controlled trial. Arthritis Rheum. 50, 1761–1769 (2004).
Van Hall, G. et al. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 88, 3005–3010 (2003).
Rosenvinge, A., Krogh-Madsen, R., Baslund, B. & Pedersen, B. K. Insulin resistance in patients with rheumatoid arthritis: effect of anti-TNFalpha therapy. Scand. J. Rheumatol. 36, 91–96 (2007).
Hansson, G. K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 352, 1685–1695 (2005).
Yudkin, J. S. Inflammation, obesity, and the metabolic syndrome. Horm. Metab. Res. 39, 707–709 (2007).
Krogh-Madsen, R., Plomgaard, P., Keller, P., Keller, C. & Pedersen, B. K. Insulin stimulates interleukin-6 and tumor necrosis factor-α gene expression in human subcutaneous adipose tissue. Am. J. Physiol. Endocrinol. Metab. 286, E234–E238 (2004).
Krogh-Madsen, R. et al. Effect of short-term intralipid infusion on the immune response during low-dose endotoxemia in humans. Am. J. Physiol. Endocrinol. Metab. 294, E371–E379 (2008).
Dandona, P. et al. Insulin inhibits intranuclear nuclear factor κB and stimulates IκB in mononuclear cells in obese subjects: evidence for an anti-inflammatory effect? J. Clin. Endocrinol. Metab. 86, 3257–3265 (2001).
Roubenoff, R. Physical activity, inflammation, and muscle loss. Nutr. Rev. 65, S208–S212 (2007).
Walsmith, J. & Roubenoff, R. Cachexia in rheumatoid arthritis. Int. J. Cardiol. 85, 89–99 (2002).
Lilleby, V. et al. Body composition, lipid and lipoprotein levels in childhood-onset systemic lupus erythematosus. Scand. J. Rheumatol. 36, 40–47 (2007).
Marighela, T. F., Genaro, P. S., Pinheiro, M. M., Szejnfeld, V. L. & Kayser, C. Risk factors for body composition abnormalities in systemic sclerosis. Clin. Rheumatol. 32, 1037–1044 (2013).
Needham, M. & Mastaglia, F. L. Inclusion body myositis: current pathogenetic concepts and diagnostic and therapeutic approaches. Lancet Neurol. 6, 620–631 (2007).
Nordemar, R., Ekblom, B., Zachrisson, L. & Lundqvist, K. Physical training in rheumatoid arthritis: a controlled long-term study. I. Scand. J. Rheumatol. 10, 17–23 (1981).
Marcora, S. et al. Preliminary evidence for cachexia in patients with well-established ankylosing spondylitis. Rheumatology (Oxford) 45, 1385–1388 (2006).
Wiesinger, G. F. et al. Aerobic capacity in adult dermatomyositis/polymyositis patients and healthy controls. Arch. Phys. Med. Rehabil. 81, 1–5 (2000).
Do Prado, D. L. et al. Abnormal chronotropic reserve and heart rate recovery in patients with SLE: a case–control study. Lupus 20, 717–720 (2011).
Ekdahl, C. & Broman, G. Muscle strength, endurance, and aerobic capacity in rheumatoid arthritis: a comparative study with healthy subjects. Ann. Rheum. Dis. 51, 35–40 (1992).
Halvorsen, S. et al. Physical fitness in patients with ankylosing spondylitis: comparison with population controls. Phys. Ther. 92, 298–309 (2012).
De Oliveira, N. C. et al. Reduced exercise capacity in systemic sclerosis patients without pulmonary involvement. Scand. J. Rheumatol. 36, 458–461 (2007).
Munsterman, T., Takken, T. & Wittink, H. Are persons with rheumatoid arthritis deconditioned? A review of physical activity and aerobic capacity. BMC Musculoskelet. Disord. 13, 202–213 (2012).
Pedersen, B. K. The diseasome of physical inactivity—and the role of myokines in muscle–fat cross talk. J. Physiol. 587, 5559–5568 (2009).
Pischon, T. et al. General and abdominal adiposity and risk of death in Europe. N. Engl. J. Med. 359, 2105–2120 (2008).
Laye, M. J., Thyfault, J. P., Stump, C. S. & Booth, F. W. Inactivity induces increases in abdominal fat. J. Appl. Physiol. (1985) 102, 1341–1347 (2007).
Olsen, R. H., Krogh-Madsen, R., Thomsen, C., Booth, F. W. & Pedersen, B. K. Metabolic responses to reduced daily steps in healthy nonexercising men. JAMA 299, 1261–1263 (2008).
Krogh-Madsen, R. et al. Normal physical activity obliterates the deleterious effects of a high-caloric intake. J. Appl. Physiol. (1985) 116, 231–239 (2014).
Santos, M. J., Vinagre, F., Canas da Silva, J., Gil, V. & Fonseca, J. E. Body composition phenotypes in systemic lupus erythematosus and rheumatoid arthritis: a comparative study of Caucasian female patients. Clin. Exp. Rheumatol. 29, 470–476 (2011).
Giles, J. T. et al. Abdominal adiposity in rheumatoid arthritis: association with cardiometabolic risk factors and disease characteristics. Arthritis Rheum. 62, 3173–3182 (2010).
Booth, F. W. & Laye, M. J. Lack of adequate appreciation of physical exercise's complexities can pre-empt appropriate design and interpretation in scientific discovery. J. Physiol. 587, 5527–5539 (2009).
Baslund, B. et al. Effect of 8 wk of bicycle training on the immune system of patients with rheumatoid arthritis. J. Appl. Physiol. 75, 1691–1695 (1993).
Häkkinen, A., Hannonen, P., Nyman, K., Lyyski, T. & Häkkinen, K. Effects of concurrent strength and endurance training in women with early or longstanding rheumatoid arthritis: comparison with healthy subjects. Arthritis Rheum. 49, 789–797 (2003).
Strasser, B. et al. The effects of strength and endurance training in patients with rheumatoid arthritis. Clin. Rheumatol. 30, 623–632 (2011).
Stavropoulos-Kalinoglou, A. et al. Individualised aerobic and resistance exercise training improves cardiorespiratory fitness and reduces cardiovascular risk in patients with rheumatoid arthritis. Ann. Rheum. Dis. 72, 1819–1825 (2013).
Lemmey, A. B. et al. Effects of high-intensity resistance training in patients with rheumatoid arthritis: a randomized controlled trial. Arthritis Rheum. 61, 1726–1734 (2009).
Baillet, A. et al. Efficacy of cardiorespiratory aerobic exercise in rheumatoid arthritis: meta-analysis of randomized controlled trials. Arthritis Care Res. (Hoboken) 62, 984–992 (2010).
Ramsey-Goldman, R. et al. A pilot study on the effects of exercise in patients with systemic lupus erythematosus. Arthritis Care Res. 13, 262–269 (2000).
Tench, C. M., McCarthy, J., McCurdie, I., White, P. D. & D'Cruz, D. P. Fatigue in systemic lupus erythematosus: a randomized controlled trial of exercise. Rheumatology (Oxford) 42, 1050–1054 (2003).
Dos Reis-Neto, E. T., da Silva, A. E., Monteiro, C. M., de Camargo, L. M. & Sato, E. I. Supervised physical exercise improves endothelial function in patients with systemic lupus erythematosus. Rheumatology (Oxford) 52, 2187–2195 (2013).
Perandini, L. A. et al. Exercise training can attenuate the inflammatory milieu in woman with systemic lupus erythematosus. J. Appl. Physiol. (1985) 117, 639–647 (2014).
Prado, D. M. et al. Exercise training in childhood-onset systemic lupus erythematosus: a controlled randomized trial. Arthritis Res. Ther. 15, R46 (2013).
Niedermann, K. et al. Effect of cardiovascular training on fitness and perceived disease activity in people with ankylosing spondylitis. Arthritis Care Res. (Hoboken) 65, 1844–1852 (2013).
Analay, Y., Ozcan, E., Karan, A., Diracoglu, D. & Aydin, R. The effectiveness of intensive group exercise on patients with ankylosing spondylitis. Clin. Rehabil. 17, 631–636 (2003).
Hidding, A., van der Linden, S. & de Witte, L. Therapeutic effects of individual physical therapy in ankylosing spondylitis related to duration of disease. Clin. Rheumatol. 12, 334–340 (1993).
Rosu, M. O., Topa, I., Chirieac, R. & Ancuta, C. Effects of pilates, McKenzie and Heckscher training on disease activity, spinal motility and pulmonary function in patients with ankylosing spondylitis: a randomized controlled trial. Rheumatol. Int. 34, 367–372 (2014).
Oliveira, N. C., dos Santos Sabbag, L. M., de Sá Pinto, A. L., Borges, C. L. & Lima, F. R. Aerobic exercise is safe and effective in systemic sclerosis. Int. J. Sports Med. 30, 728–732 (2009).
Antonioli, C. M. et al. An individualized rehabilitation program in patients with systemic sclerosis may improve quality of life and hand mobility. Clin. Rheumatol. 28, 159–165 (2009).
Pinto, A. L. et al. Efficacy and safety of concurrent training in systemic sclerosis. J. Strength. Cond. Res. 25, 1423–1428 (2011).
Alexanderson, H., Bergegård, J., Björnådal, L. & Nordin, A. Intensive aerobic and muscle endurance exercise in patients with systemic sclerosis: a pilot study. BMC Res. Notes 7, 86 (2014).
Alexanderson, H. & Lundberg, I. E. Exercise as a therapeutic modality in patients with idiopathic inflammatory myopathies. Curr. Opin. Rheumatol. 24, 201–207 (2012).
De Salles Painelli, V. et al. The possible role of physical exercise on the treatment of idiopathic inflammatory myopathies. Autoimmun. Rev. 8, 355–359 (2009).
Alexanderson, H., Dastmalchi, M., Esbjörnsson-Liljedahl, M., Opava, C. H. & Lundberg, I. E. Benefits of intensive resistance training in patients with chronic polymyositis or dermatomyositis. Arthritis Rheum. 57, 768–777 (2007).
Nader, G. A. et al. A longitudinal, integrated, clinical, histological and mRNA profiling study of resistance exercise in myositis. Mol. Med. 16, 455–464 (2010).
Spector, S. A. et al. Safety and efficacy of strength training in patients with sporadic inclusion body myositis. Muscle Nerve. 20, 1242–1248 (1997).
Johnson, L. G. et al. Improvement in aerobic capacity after an exercise program in sporadic inclusion body myositis. J. Clin. Neuromuscul. Dis. 10, 178–184 (2009).
Gualano, B. et al. Resistance training with vascular occlusion in inclusion body myositis: a case study. Med. Sci. Sports Exerc. 42, 250–254 (2010).
Dastmalchi, M. et al. Effect of physical training on the proportion of slow-twitch type I muscle fibers, a novel nonimmune-mediated mechanism for muscle impairment in polymyositis or dermatomyositis. Arthritis Rheum. 57, 1303–1310 (2007).
Alemo Munters, L. et al. Improved exercise performance and increased aerobic capacity after endurance training of patients with stable polymyositis and dermatomyositis. Arthritis Res. Ther. 15, R83 (2013).
Neuberger, G. B. et al. Effects of exercise on fatigue, aerobic fitness, and disease activity measures in persons with rheumatoid arthritis. Res. Nurs. Health. 20, 195–204 (1997).
Pedersen, B. K. & Saltin, B. Evidence for prescribing exercise as therapy in chronic disease. Scand. J. Med. Sci. Sports 16, 3–63 (2006).
Metsios, G. S. et al. Association of physical inactivity with increased cardiovascular risk in patients with rheumatoid arthritis. Eur. J. Cardiovasc. Prev. Rehabil. 16, 188–194 (2009).
Barnes, J. N. et al. Arterial stiffening, wave reflection, and inflammation in habitually exercising systemic lupus erythematosus patients. Am. J. Hypertens. 24, 1194–1200 (2011).
Volkmann, E. R. et al. Low physical activity is associated with proinflammatory high-density lipoprotein and increased subclinical atherosclerosis in women with systemic lupus erythematosus. Arthritis Care Res. (Hoboken) 62, 258–265 (2010).
Metsios, G. S. et al. Individualised exercise improves endothelial function in patients with rheumatoid arthritis. Ann. Rheum. Dis. 73, 748–751 (2014).
Benatti, F. B. et al. The effects of exercise on lipid profile in systemic lupus erythematosus and healthy individuals: a randomized trial. Rheumatol. Int. http://dx.doi.org/10.1007/s00296-014-3081-4.
Janse van Rensburg, D. C., Ker, J. A., Grant, C. C. & Fletcher, L. Effect of exercise on cardiac autonomic function in females with rheumatoid arthritis. Clin. Rheumatol. 31, 1155–1162 (2012).
Miossi, R. et al. Using exercise training to counterbalance chronotropic incompetence and delayed heart rate recovery in systemic lupus erythematosus: a randomized trial. Arthritis Care Res. (Hoboken) 64, 1159–1166 (2012).
Votruba, S. B., Horvitz, M. A. & Schoeller, D. A. The role of exercise in the treatment of obesity. Nutrition 16, 179–188 (2000).
Wong, S. L. et al. Cardiorespiratory fitness is associated with lower abdominal fat independent of body mass index. Med. Sci. Sports Exerc. 36, 286–291 (2004).
Plasqui, G. et al. Physical activity and body composition in patients with ankylosing spondylitis. Arthritis Care Res. (Hoboken) 64, 101–107 (2012).
Kipen, Y., Briganti, E. M., Strauss, B. J., Littlejohn, G. O. & Morand, E. F. Three year follow-up of body composition changes in pre-menopausal women with systemic lupus erythematosus. Rheumatology (Oxford) 38, 59–65 (1999).
Nordemar, R., Edström, L. & Ekblom, B. Changes in muscle fibre size and physical performance in patients with rheumatoid arthritis after short-term physical training. Scand. J. Rheumatol. 5, 70–76 (1976).
Sharif, S. et al. Resistance exercise reduces skeletal muscle cachexia and improves muscle function in rheumatoid arthritis. Case. Rep. Med. 2011, 205691 (2011).
Häkkinen, A. et al. Effects of prolonged combined strength and endurance training on physical fitness, body composition and serum hormones in women with rheumatoid arthritis and in healthy controls. Clin. Exp. Rheumatol. 23, 505–512 (2005).
Marcora, S. M., Lemmey, A. B. & Maddison, P. J. Can progressive resistance training reverse cachexia in patients with rheumatoid arthritis? Results of a pilot study. J. Rheumatol. 32, 1031–1039 (2005).
Omori, C. H. et al. Exercise training in juvenile dermatomyositis. Arthritis Care Res. (Hoboken) 64, 1186–1194 (2012).
Lyngberg, K., Danneskiold-Samsøe, B. & Halskov, O. The effect of physical training on patients with rheumatoid arthritis: changes in disease activity, muscle strength and aerobic capacity. A clinically controlled minimized cross-over study. Clin. Exp. Rheumatol. 6, 253–260 (1988).
De Jong, Z. et al. Long term high intensity exercise and damage of small joints in rheumatoid arthritis. Ann. Rheum. Dis. 63, 1399–1405 (2004).
Van den Ende, C. H. et al. Effect of intensive exercise on patients with active rheumatoid arthritis: a randomised clinical trial. Ann. Rheum. Dis. 59, 615–621 (2000).
De Jong, Z. et al. Is a long-term high-intensity exercise program effective and safe in patients with rheumatoid arthritis? Results of a randomized controlled trial. Arthritis Rheum. 48, 2415–2424 (2003).
Rall, L. C. et al. Effects of progressive resistance training on immune response in aging and chronic inflammation. Med. Sci. Sports Exerc. 28, 1356–1365 (1996).
Perandini, L. A. et al. Inflammatory cytokine kinetics to single bouts of acute moderate and intense aerobic exercise in women with active and inactive systemic lupus erythematosus. Exercise Immun. Rev. (in press) (2014).
Bazzoni, F. & Beutler, B. The tumor necrosis factor ligand and receptor families. N. Engl. J. Med. 334, 1717–1725 (1996).
Da Silva, A. E., dos Reis-Neto, E. T., da Silva, N. P. & Sato, E. I. The effect of acute physical exercise on cytokine levels in patients with systemic lupus erythematosus. Lupus. 22, 1479–1483 (2013).
Bearne, L. M., Scott, D. L. & Hurley, M. V. Exercise can reverse quadriceps sensorimotor dysfunction that is associated with rheumatoid arthritis without exacerbating disease activity. Rheumatology (Oxford) 41, 157–166 (2002).
Dessein, P. H., Joffe, B. I. & Singh, S. Biomarkers of endothelial dysfunction, cardiovascular risk factors and atherosclerosis in rheumatoid arthritis. Arthritis Res. Ther. 7, R634–R643 (2005).
Pedersen, B. K. et al. Searching for the exercise factor: is IL-6 a candidate? J. Muscle Res. Cell. Motil. 24, 113–119 (2003).
Bortoluzzi, S., Scannapieco, P., Cestaro, A., Danieli, G. A. & Schiaffino, S. Computational reconstruction of the human skeletal muscle secretome. Proteins 62, 776–792 (2006).
Yoon, J. H. et al. Comparative proteomic analysis of the insulin-induced L6 myotube secretome. Proteomics 9, 51–60 (2009).
Henningsen, J., Rigbolt, K. T., Blagoev, B., Pedersen, B. K. & Kratchmarova, I. Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol. Cell Proteomics. 9, 2482–2496 (2010).
Pedersen, B. K. Muscular IL-6 and its role as an energy sensor. Med. Sci. Sports Exerc. 44, 392–396 (2012).
Bruunsgaard, H. et al. Exercise-induced increase in interleukin-6 is related to muscle damage. J. Physiol. 499, 833–841 (1997).
Peake, J., Nosaka, K., & Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 11, 64–85 (2005).
Toft, A. D. et al. Cytokine response to eccentric exercise in young and elderly humans. Am. J. Physiol. Cell Physiol. 283, C289–C295 (2002).
Starkie, R., Ostrowski, S. R., Jauffred, S., Febbraio, M. & Pedersen, B. K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-α production in humans. FASEB J. 17, 884–886 (2003).
Mauer, J. et al. Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nat. Immunol. 15, 423–430 (2014).
Yang, H. et al. Treadmill exercise promotes interleukin 15 expression in skeletal muscle and interleukin 15 receptor α expression in adipose tissue of high-fat diet rats. Endocrine 43, 579–585 (2013).
Rinnov, A. et al. Endurance training enhances skeletal muscle interleukin-15 in human male subjects. Endocrine 45, 271–278 (2014).
Grabstein, K. H. et al. Cloning of a T cell growth factor that interacts with the β chain of the interleukin-2 receptor. Science 264, 965–968 (1994).
Furmanczyk, P. S. & Quinn, L. S. Interleukin-15 increases myosin accretion in human skeletal myogenic cultures. Cell Biol. Int. 27, 845–851 (2003).
Quinn, L. S., Haugk, K. L. & Damon, S. E. Interleukin-15 stimulates C2 skeletal myoblast differentiation. Biochem. Biophys. Res. Commun. 239, 6–10 (1997).
Quinn, L. S., Haugk, K. L. & Grabstein, K. H. Interleukin-15: a novel anabolic cytokine for skeletal muscle. Endocrinology 136, 3669–3672 (1995).
Carbo, N. et al. Interleukin-15 mediates reciprocal regulation of adipose and muscle mass: a potential role in body weight control. Biochim. Biophys. Acta 1526, 17–24 (2001).
Quinn, L. S., Strait-Bodey, L., Anderson, B. G., Argilés, J. M. & Havel, P. J. Interleukin-15 stimulates adiponectin secretion by 3T3-L1 adipocytes: evidence for a skeletal muscle-to-fat signaling pathway. Cell Biol. Int. 29, 449–457 (2005).
Nielsen, A. R. et al. Association between IL-15 and obesity: IL-15 as a potential regulator of fat mass. J. Clin. Endocrinol. Metab. 93, 4486–4493 (2008).
Raschke, S. & Eckel, J. Adipo-myokines: two sides of the same coin—mediators of inflammation and mediators of exercise. Mediators Inflamm. 2013, 320724 (2013).
Pedersen, B. K. et al. Role of exercise-induced brain-derived neurotrophic factor production in the regulation of energy homeostasis in mammals. Exp. Physiol. 94, 1153–1160 (2009).
Hojman, P. et al. Erythropoietin over-expression protects against diet-induced obesity in mice through increased fat oxidation in muscles. PLoS ONE 4, e5894 (2009).
Rao, R. R. et al. Meteorin-like Is a hormone that regulates immune-adipose Interactions to increase beige fat thermogenesis. Cell 157, 1279–1291 (2014).
Varjú, C., Pethö, E., Kutas, R. & Czirják, L. The effect of physical exercise following acute disease exacerbation in patients with dermato/polymyositis. Clin. Rehabil. 17, 83–87 (2003).
Escalante, A., Miller, L. & Beardmore, T. D. Resistive exercise in the rehabilitation of polymyositis/dermatomyositis. J. Rheumatol. 20, 1340–1344 (1993).
Acknowledgements
The authors are grateful to CAPES (process 12824-13-5) and FAPESP (process 2011-24093-2) for financial support. The Centre of Inflammation and Metabolism (CIM) is supported by a grant from the Danish National Research Foundation (DNRF55). The Centre for Physical Activity Research is supported by a grant from Trygfonden. CIM is part of the UNIK Project: Food, Fitness & Pharma for Health and Disease, supported by the Danish Ministry of Science, Technology, and Innovation. CIM is a member of DD2, the Danish Center for Strategic Research in Type 2 Diabetes (the Danish Council for Strategic Research grant numbers 09-067009 and 09-075724). The Copenhagen Muscle Research Centre is supported by a grant from the Capital Region of Denmark.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to researching data for the article, providing a substantial contribution to discussions of the content, writing the article, and to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Benatti, F., Pedersen, B. Exercise as an anti-inflammatory therapy for rheumatic diseases—myokine regulation. Nat Rev Rheumatol 11, 86–97 (2015). https://doi.org/10.1038/nrrheum.2014.193
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.193
This article is cited by
-
Diabetes, Sport und Bewegung
Die Diabetologie (2024)
-
Muscle-to-organ cross-talk mediated by interleukin 6 during exercise: a review
Sport Sciences for Health (2024)
-
Exercise suppresses neuroinflammation for alleviating Alzheimer’s disease
Journal of Neuroinflammation (2023)
-
Diabetes, Sport und Bewegung
Die Diabetologie (2023)
-
Übersetzung der 2018 EULAR Empfehlungen zu körperlicher Aktivität von Menschen mit entzündlich-rheumatischen und degenerativen Erkrankungen ins Deutsche und sprachliche Validierung im deutschsprachigen Raum mit medizinischen Fachpersonen
Zeitschrift für Rheumatologie (2023)