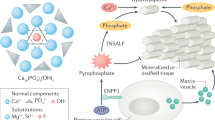
Abstract
Calcium pyrophosphate (CPP) crystal deposition (CPPD) is associated with ageing and osteoarthritis, and with uncommon disorders such as hyperparathyroidism, hypomagnesemia, hemochromatosis and hypophosphatasia. Elevated levels of synovial fluid pyrophosphate promote CPP crystal formation. This extracellular pyrophosphate originates either from the breakdown of nucleotide triphosphates by plasma-cell membrane glycoprotein 1 (PC-1) or from pyrophosphate transport by the transmembrane protein progressive ankylosis protein homolog (ANK). Although the etiology of apparent sporadic CPPD is not well-established, mutations in the ANK human gene (ANKH) have been shown to cause familial CPPD. In this Review, the key regulators of pyrophosphate metabolism and factors that lead to high extracellular pyrophosphate levels are described. Particular emphasis is placed on the mechanisms by which mutations in ANKH cause CPPD and the clinical phenotype of these mutations is discussed. Cartilage factors predisposing to CPPD and CPP-crystal-induced inflammation and current treatment options for the management of CPPD are also described.
Key Points
-
Calcium pyrophosphate (CPP) crystal deposition (CPPD) is associated with ageing, osteoarthritis, hemochromatosis, hyperparathyroidism and hypomagnesemia
-
High levels of extracellular inorganic pyrophosphate (PPi) facilitates CPP crystal formation
-
Progressive ankylosis protein homolog (ANK), a transmembrane PPi transporter, and plasma-cell membrane glycoprotein 1 (PC-1) generate extracellular PPi
-
Mutations in ANKH lead to familial CPPD
-
Methotrexate and/or hydroxychloroquine, and interleukin-1 antagonists might be treatment options for chronic CPP crystal inflammatory arthritis and resistant acute CPP crystal arthritis, respectively
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Neame, R. & Doherty, M. in Rheumatology 4th edn (eds Hochberg, M. C. et al.) 1845–1858 (Mosby, Edinburgh, 2008).
Viriyavejkul, P., Wilairatana, V., Tanavalee, A. & Jaovisidha, K. Comparison of characteristics of patients with and without calcium pyrophosphate dihydrate crystal deposition disease who underwent total knee replacement surgery for osteoarthritis. Osteoarthritis Cartilage 15, 232–235 (2007).
Derfus, B. A. et al. The high prevalence of pathologic calcium crystals in pre-operative knees. J. Rheumatol. 29, 570–574 (2002).
McCarthy, G. M. in Rheumatology 4th edn (eds Hochberg, M. C. et al.) 1859–1868 (Mosby, Edinburgh, 2008).
McCarty, D. J., Kohn, N. H. & Faires, J. S. The significance of calcium phosphate crystals in the synovial fluid of arthritic patients: the 'pseudogout syndrome'. I. Clinical aspects. Ann. Intern. Med. 56, 711–737 (1962).
McCarty, D. J. Calcium pyrophosphate dihydrate crystal deposition disease—1975. Arthritis Rheum. 19 (Suppl. 3), 275–285 (1976).
Zhang, W. et al. EULAR evidence-based recommendations for calcium pyrophosphate deposition (CPPD). Part I: terminology and diagnosis. Ann. Rheum. Dis. 69 (Suppl. 3), 93 (2010).
Felson, D. T., Anderson, J. J., Naimark, A., Kannel, W. & Meenan, R. F. The prevalence of chondrocalcinosis in the elderly and its association with knee osteoarthritis: the Framingham Study. J. Rheumatol. 16, 1241–1245 (1989).
Zhang, W., Neame, R., Doherty, S. & Doherty, M. Relative risk of knee chondrocalcinosis in siblings of index cases with pyrophosphate arthropathy. Ann. Rheum. Dis. 63, 969–973 (2004).
Ramonda, R. et al. Prevalence of chondrocalcinosis in Italian subjects from northeastern Italy. The Pro. V. A. (PROgetto Veneto Anziani) study. Clin. Exp. Rheumatol. 27, 981–984 (2009).
Neame, R. L., Carr, A. J., Muir, K. & Doherty, M. UK community prevalence of knee chondrocalcinosis: evidence that correlation with osteoarthritis is through a shared association with osteophyte. Ann. Rheum. Dis. 62, 513–518 (2003).
Sanmartí, R. et al. Prevalence of articular chondrocalcinosis in elderly subjects in a rural area of Catalonia. Ann. Rheum. Dis. 52, 418–422 (1993).
Zhang, Y. et al. Lower prevalence of chondrocalcinosis in Chinese subjects in Beijing than in white subjects in the United States: the Beijing Osteoarthritis Study. Arthritis Rheum. 54, 3508–3512 (2006).
Malaviya, A. N. et al. Calcium pyrophosphatase dihydrate (CPPD) crystal deposition disease in a teaching hospital in Kuwait. Ann. Rheum. Dis. 60, 416–419 (2001).
Doherty, M., Watt, I. & Dieppe, P. A. Localised chondrocalcinosis in post-meniscectomy knees. Lancet 1, 1207–1210 (1982).
Fuerst, M. et al. Articular cartilage mineralization in osteoarthritis of the hip. BMC Musculoskelet. Disord. 10, 166 (2009).
Fuerst, M. et al. Calcification of articular cartilage in human osteoarthritis. Arthritis Rheum. 60, 2694–2703 (2009).
Richette, P., Bardin, T. & Doherty, M. An update on the epidemiology of calcium pyrophosphate dihydrate crystal deposition disease. Rheumatology (Oxford) 48, 711–715 (2009).
Calò, L., Punzi, L. & Semplicini, A. Hypomagnesemia and chondrocalcinosis in Bartter's and Gitelman's syndrome: review of the pathogenetic mechanisms. Am. J. Nephrol. 20, 347–350 (2000).
Richette, P. et al. Hypomagnesemia associated with chondrocalcinosis: a cross-sectional study. Arthritis Rheum. 57, 1496–1501 (2007).
Hollingworth, P., Williams, P. L. & Scott, J. T. Frequency of chondrocalcinosis of the knees in asymptomatic hyperuricaemia and rheumatoid arthritis: a controlled study. Ann. Rheum. Dis. 41, 344–346 (1982).
Doherty, M., Dieppe, P. & Watt, I. Low incidence of calcium pyrophosphate dihydrate crystal deposition in rheumatoid arthritis, with modification of radiographic features in coexistent disease. Arthritis Rheum. 27, 1002–1009 (1984).
Rothschild, B. CPPD complicating other forms of inflammatory arthritis. Clin. Rheumatol. 26, 1130–1131 (2007).
Gerster, J. C., Varisco, P. A., Kern, J., Dudler, J. & So, A. K. CPPD crystal deposition disease in patients with rheumatoid arthritis. Clin. Rheumatol. 25, 468–469 (2006).
Hughes, A. E., McGibbon, D., Woodward, E., Dixey, J. & Doherty, M. Localisation of a gene for chondrocalcinosis to chromosome 5p. Hum. Mol. Genet. 4, 1225–1228 (1995).
Williams, C. J. et al. Mutations in the amino terminus of ANKH in two US families with calcium pyrophosphate dihydrate crystal deposition disease. Arthritis Rheum. 48, 2627–2631 (2003).
Andrew, L. J. et al. Refinement of the chromosome 5p locus for familial calcium pyrophosphate dihydrate deposition disease. Am. J. Hum. Genet. 64, 136–145 (1999).
Doherty, M., Hamilton, E., Henderson, J., Misra, H. & Dixey, J. Familial chondrocalcinosis due to calcium pyrophosphate dihydrate crystal deposition in English families. Br. J. Rheumatol. 30, 10–15 (1991).
Hamza, M. et al. HLA-antigens in a Tunisian familial chondrocalcinosis. Dis. Markers 8, 109–112 (1990).
Gaucher, A. et al. Hereditary diffuse articular chondrocalcinosis. Dominant manifestation without close linkage with the HLA system in a large pedigree. Scand. J. Rheumatol. 6, 217–221 (1977).
Hamza, M., Meddeb, N. & Bardin, T. Hereditary chondrocalcinosis in a Tunisian family. Clin. Exp. Rheumatol. 10, 43–49 (1992).
Baldwin, C. T. et al. Linkage of early-onset osteoarthritis and chondrocalcinosis to human chromosome 8q. Am. J. Hum. Genet. 56, 692–697 (1995).
Pendleton, A. et al. Mutations in ANKH cause chondrocalcinosis. Am. J. Hum. Genet. 71, 933–940 (2002).
Béjia, I. et al. Familial calcium pyrophosphate dihydrate deposition disease. A Tunisian kindred. Joint Bone Spine 71, 401–408 (2004).
Gaudreau, A. et al. Familial articular chondrocalcinosis in Quebec. Arthritis Rheum. 24, 611–615 (1981).
Fernandez Dapica, M. P. & Gómez-Reino, J. J. Familial chondrocalcinosis in the Spanish population. J. Rheumatol. 13, 631–633 (1986).
Balsa, A. et al. Familial articular chondrocalcinosis in Spain. Ann. Rheum. Dis. 49, 531–535 (1990).
Zhang, Y. et al. Association of sporadic chondrocalcinosis with a 4-basepair G-to-A transition in the 5′-untranslated region of ANKH that promotes enhanced expression of ANKH protein and excess generation of extracellular inorganic pyrophosphate. Arthritis Rheum. 52, 1110–1117 (2005).
Sahinbegovic, E. et al. Musculoskeletal disease burden of hereditary hemochromatosis. Arthritis Rheum. doi:10.1002/art.27712.
Derfus, B. A. et al. Articular cartilage vesicles generate calcium pyrophosphate dihydrate-like crystals in vitro. Arthritis Rheum. 35, 231–240 (1992).
Pritzker, K. P., Cheng, P. T. & Renlund, R. C. Calcium pyrophosphate crystal deposition in hyaline cartilage. Ultrastructural analysis and implications for pathogenesis. J. Rheumatol. 15, 828–835 (1988).
Ryan, L. M. & McCarty, D. J. Understanding inorganic pyrophosphate metabolism: toward prevention of calcium pyrophosphate dihydrate crystal deposition. Ann. Rheum. Dis. 54, 939–941 (1995).
Harmey, D. et al. Concerted regulation of inorganic pyrophosphate and osteopontin by Akp2, Enpp1, and Ank: an integrated model of the pathogenesis of mineralization disorders. Am. J. Pathol. 164, 1199–1209 (2004).
Derfus, B. et al. Characterization of an additional articular cartilage vesicle fraction that generates calcium pyrophosphate dihydrate crystals in vitro. J. Rheumatol. 22, 1514–1519 (1995).
Derfus, B. et al. Human osteoarthritic cartilage matrix vesicles generate both calcium pyrophosphate dihydrate and apatite in vitro. Calcif. Tissue Int. 63, 258–262 (1998).
Mitton, E., Gohr, C. M., McNally, M. T. & Rosenthal, A. K. Articular cartilage vesicles contain RNA. Biochem. Biophys. Res. Commun. 388, 533–538 (2009).
Grassi, W., Meenagh, G., Pascual, E. & Filippucci, E. “Crystal clear”-sonographic assessment of gout and calcium pyrophosphate deposition disease. Semin. Arthritis Rheum. 36, 197–202 (2006).
Muehleman, C. et al. Association between crystals and cartilage degeneration in the ankle. J. Rheumatol. 35, 1108–1117 (2008).
Beutler, A., Rothfuss, S., Clayburne, G., Sieck, M. & Schumacher, H. R. Jr . Calcium pyrophosphate dihydrate crystal deposition in synovium. Relationship to collagen fibers and chondrometaplasia. Arthritis Rheum. 36, 704–715 (1993).
Ishikawa, K., Masuda, I., Ohira, T. & Yokoyama, M. A histological study of calcium pyrophosphate dihydrate crystal-deposition disease. J. Bone Joint Surg. Am. 71, 875–886 (1989).
Mandel, N. S., Mandel, G. S., Carroll, D. J. & Halverson, P. B. Calcium pyrophosphate crystal deposition. An in vitro study using a gelatin matrix model. Arthritis Rheum. 27, 789–796 (1984).
Terkeltaub, R. A. Inorganic pyrophosphate generation and disposition in pathophysiology. Am. J. Physiol. Cell Physiol. 281, C1–C11 (2001).
Russell, R. G. Metabolism of inorganic pyrophosphate (PPi). Arthritis Rheum. 19 (Suppl. 3), 465–478 (1976).
Murray, R. K., Granner, D. K., Mayes, P. A. & Rodwell, V. W. (eds) Harper's Biochemistry 24th edn, 112 (Appleton & Lange, Stamford, 1996).
Ryan, L. M., Cheung, H. S. & McCarty, D. J. Release of pyrophosphate by normal mammalian articular hyaline and fibrocartilage in organ culture. Arthritis Rheum. 24, 1522–1527 (1981).
Rosenthal, A. K., McCarty, B. A., Cheung, H. S. & Ryan, L. M. A comparison of the effect of transforming growth factor β1 on pyrophosphate elaboration from various articular tissues. Arthritis Rheum. 36, 539–542 (1993).
Altman, R. D., Muniz, O. E., Pita, J. C. & Howell, D. S. Articular chondrocalcinosis. Microanalysis of pyrophosphate (PPi) in synovial fluid and plasma. Arthritis Rheum. 16, 171–178 (1973).
Doherty, M., Belcher, C., Regan, M., Jones, A. & Ledingham, J. Association between synovial fluid levels of inorganic pyrophosphate and short term radiographic outcome of knee osteoarthritis. Ann. Rheum. Dis. 55, 432–436 (1996).
Thouverey, C., Bechkoff, G., Pikula, S. & Buchet, R. Inorganic pyrophosphate as a regulator of hydroxyapatite or calcium pyrophosphate dihydrate mineral deposition by matrix vesicles. Osteoarthritis Cartilage 17, 64–72 (2009).
Cheng, P. T. & Pritzker, K. P. Pyrophosphate, phosphate ion interaction: effects on calcium pyrophosphate and calcium hydroxyapatite crystal formation in aqueous solutions. J. Rheumatol. 10, 769–777 (1983).
Johnson, K., Pritzker, K., Goding, J. & Terkeltaub, R. The nucleoside triphosphate pyrophosphohydrolase isozyme PC-1 directly promotes cartilage calcification through chondrocyte apoptosis and increased calcium precipitation by mineralizing vesicles. J. Rheumatol. 28, 2681–2691 (2001).
Gurley, K. A., Reimer, R. J. & Kingsley, D. M. Biochemical and genetic analysis of ANK in arthritis and bone disease. Am. J. Hum. Genet. 79, 1017–1029 (2006).
Hessle, L. et al. Tissue-nonspecific alkaline phosphatase and plasma cell membrane glycoprotein-1 are central antagonistic regulators of bone mineralization. Proc. Natl Acad. Sci. USA 99, 9445–9449 (2002).
Graff, R. D., Lazarowski, E. R., Banes, A. J. & Lee, G. M. ATP release by mechanically loaded porcine chondrons in pellet culture. Arthritis Rheum. 43, 1571–1579 (2000).
Ryan, L. M., Kurup, I. V., Derfus, B. A. & Kushnaryov, V. M. ATP-induced chondrocalcinosis. Arthritis Rheum. 35, 1520–1525 (1992).
Lotz, M. et al. Interleukin 1β suppresses transforming growth factor-induced inorganic pyrophosphate (PPi) production and expression of the PPi-generating enzyme PC-1 in human chondrocytes. Proc. Natl Acad. Sci. USA 92, 10364–10368 (1995).
Johnson, K. et al. Up-regulated expression of the phosphodiesterase nucleotide pyrophosphatase family member PC-1 is a marker and pathogenic factor for knee meniscal cartilage matrix calcification. Arthritis Rheum. 44, 1071–1081 (2001).
Johnson, K. et al. Differential mechanisms of inorganic pyrophosphate production by plasma cell membrane glycoprotein-1 and B10 in chondrocytes. Arthritis Rheum. 42, 1986–1997 (1999).
Hirose, J., Masuda, I. & Ryan, L. M. Expression of cartilage intermediate layer protein/nucleotide pyrophosphohydrolase parallels the production of extracellular inorganic pyrophosphate in response to growth factors and with aging. Arthritis Rheum. 43, 2703–2711 (2000).
Johnson, K., Farley, D., Hu, S. I. & Terkeltaub, R. One of two chondrocyte-expressed isoforms of cartilage intermediate-layer protein functions as an insulin-like growth factor 1 antagonist. Arthritis Rheum. 48, 1302–1314 (2003).
Garimella, R., Bi, X., Anderson, H. C. & Camacho, N. P. Nature of phosphate substrate as a major determinant of mineral type formed in matrix vesicle-mediated in vitro mineralization: an FTIR imaging study. Bone 38, 811–817 (2006).
Rosenthal, A. K., Gohr, C. M., Uzuki, M. & Masuda, I. Osteopontin promotes pathologic mineralization in articular cartilage. Matrix Biol. 26, 96–105 (2007).
Zaka, R. et al. P5L mutation in Ank results in an increase in extracellular inorganic pyrophosphate during proliferation and nonmineralizing hypertrophy in stably transduced ATDC5 cells. Arthritis Res. Ther. 8, R164 (2006).
Ho, A. M., Johnson, M. D. & Kingsley, D. M. Role of the mouse Ank gene in control of tissue calcification and arthritis. Science 289, 265–270 (2000).
Caswell, A., Guilland-Cumming, D. F., Hearn, P. R., McGuire, M. K. & Russell, R. G. Pathogenesis of chondrocalcinosis and pseudogout. Metabolism of inorganic pyrophosphate and production of calcium pyrophosphate dihydrate crystals. Ann. Rheum. Dis. 42 (Suppl. 1), 27–37 (1983).
Hamilton, E. B. Diseases associated with CPPD deposition disease. Arthritis Rheum. 19 (Suppl. 3), 353–357 (1976).
Zhang, L. et al. Phosphodiesterase activity of alkaline phosphatase in ATP-initiated Ca2+ and phosphate deposition in isolated chicken matrix vesicles. J. Biol. Chem. 280, 37289–37296 (2005).
Wang, W., Xu, J., Du, B. & Kirsch, T. Role of the progressive ankylosis gene (Ank) in cartilage mineralization. Mol. Cell. Biol. 25, 312–323 (2005).
Rosen, F. et al. Differential effects of aging on human chondrocyte responses to transforming growth factor β: increased pyrophosphate production and decreased cell proliferation. Arthritis Rheum. 40, 1275–1281 (1997).
Hakim, F. T. et al. Hereditary joint disorder in progressive ankylosis (Ank/Ank) mice. I. Association of calcium hydroxyapatite deposition with inflammatory arthropathy. Arthritis Rheum. 27, 1411–1420 (1984).
Netter, P., Bardin, T., Bianchi, A., Richette, P. & Loeuille, D. The ANKH gene and familial calcium pyrophosphate dihydrate deposition disease. Joint Bone Spine 71, 365–368 (2004).
Jubeck, B. et al. Promotion of articular cartilage matrix vesicle mineralization by type I collagen. Arthritis Rheum. 58, 2809–2817 (2008).
Kalya, S. & Rosenthal, A. K. Extracellular matrix changes regulate calcium crystal formation in articular cartilage. Curr. Opin. Rheumatol. 17, 325–329 (2005).
Ishikawa, K. Chondrocytes that accumulate proteoglycans and inorganic pyrophosphate in the pathogenesis of chondrocalcinosis. Arthritis Rheum. 28, 118–120 (1985).
Rosenthal, A. K., Masuda, I., Gohr, C. M., Derfus, B. A. & Le, M. The transglutaminase, Factor XIIIA, is present in articular chondrocytes. Osteoarthritis Cartilage 9, 578–581 (2001).
Heinkel, D., Gohr, C. M., Uzuki, M. & Rosenthal, A. K. Transglutaminase contributes to CPPD crystal formation in osteoarthritis. Front. Biosci. 9, 3257–3261 (2004).
Rosenthal, A. K., Gohr, C. M., Henry, L. A. & Le, M. Participation of transglutaminase in the activation of latent transforming growth factor beta1 in aging articular cartilage. Arthritis Rheum. 43, 1729–1733 (2000).
Rosenthal, A. K. & Henry, L. A. Thyroid hormones induce features of the hypertrophic phenotype and stimulate correlates of CPPD crystal formation in articular chondrocytes. J. Rheumatol. 26, 395–401 (1999).
Masuda, I., Ishikawa, K. & Usuku, G. A histologic and immunohistochemical study of calcium pyrophosphate dihydrate crystal deposition disease. Clin. Orthop. Relat. Res. 263, 272–287 (1991).
Pasquetti, P. et al. Joint lavage and pseudogout. Ann. Rheum. Dis. 63, 1529–1530 (2004).
Liote, F. & Ea, H. K. Recent developments in crystal-induced inflammation pathogenesis and management. Curr. Rheumatol. Rep. 9, 243–250 (2007).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Higo, T., Duronio, V., Tudan, C., Burt, H. M. & Jackson, J. K. Calcium pyrophosphate dihydrate crystal-induced inhibition of neutrophil apoptosis: involvement of Bcl-2 family members. Inflamm. Res. 59, 71–81 (2010).
Liu-Bryan, R. & Liote, F. Monosodium urate and calcium pyrophosphate dihydrate (CPPD) crystals, inflammation, and cellular signaling. Joint Bone Spine 72, 295–302 (2005).
Tudan, C., Jackson, J. K., Blanis, L., Pelech, S. L. & Burt, H. M. Inhibition of TNF-α-induced neutrophil apoptosis by crystals of calcium pyrophosphate dihydrate is mediated by the extracellular signal-regulated kinase and phosphatidylinositol 3-kinase/Akt pathways up-stream of caspase 3. J. Immunol. 165, 5798–5806 (2000).
Liu, Y. Z., Jackson, A. P. & Cosgrove, S. D. Contribution of calcium-containing crystals to cartilage degradation and synovial inflammation in osteoarthritis. Osteoarthritis Cartilage 17, 1333–1340 (2009).
Fam, A. G. et al. Acceleration of experimental lapine osteoarthritis by calcium pyrophosphate microcrystalline synovitis. Arthritis Rheum. 38, 201–210 (1995).
Martinez Sanchis, A. & Pascual, E. Intracellular and extracellular CPPD crystals are a regular feature in synovial fluid from uninflamed joints of patients with CPPD related arthropathy. Ann. Rheum. Dis. 64, 1769–1772 (2005).
Schumacher, H. R., Fishbein, P., Phelps, P., Tse, R. & Krauser, R. Comparison of sodium urate and calcium pyrophosphate crystal phagocytosis by polymorphonuclear leukocytes. Effects of crystal size and other factors. Arthritis Rheum. 18, 783–792 (1975).
Swan, A., Heywood, B., Chapman, B., Seward, H. & Dieppe, P. Evidence for a causal relationship between the structure, size, and load of calcium pyrophosphate dihydrate crystals, and attacks of pseudogout. Ann. Rheum. Dis. 54, 825–830 (1995).
NICE clinical guideline 59. Osteoarthritis: the care and management of osteoarthritis in adults (National Institute for Health and Clinical Excellence London, 2008).
Hilliquin, P., Le Devic, P. & Menkes, C. J. Comparison of the efficacy of nonsurgical synovectomy (synoviorthesis) and joint lavage in knee osteoarthritis with effusions. Rev. Rhum. Engl. Ed. 63, 93–102 (1996).
Doherty, M. & Dieppe, P. A. Effect of intra-articular yttrium-90 on chronic pyrophosphate arthropathy of the knee. Lancet 2, 1243–1246 (1981).
Pascual, E. Management of crystal arthritis. Rheumatology (Oxford) 38, 912–916 (1999).
Roane, D. W. et al. Prospective use of intramuscular triamcinolone acetonide in pseudogout. J. Rheumatol. 24, 1168–1170 (1997).
Announ, N. & Guerne, P. A. Treating difficult crystal pyrophosphate dihydrate deposition disease. Curr. Rheumatol. Rep. 10, 228–234 (2008).
Tabatabai, M. R. & Cummings, N. A. Intravenous colchicine in the treatment of acute pseudogout. Arthritis Rheum. 23, 370–374 (1980).
Alvarellos, A. & Spilberg, I. Colchicine prophylaxis in pseudogout. J. Rheumatol. 13, 804–805 (1986).
McGonagle, D., Tan, A. L., Madden, J., Emery, P. & McDermott, M. F. Successful treatment of resistant pseudogout with anakinra. Arthritis Rheum. 58, 631–633 (2008).
Terkeltaub, R. et al. The interleukin 1 inhibitor rilonacept in treatment of chronic gouty arthritis: results of a placebo controlled monosequence crossover, non-randomised, single blind-pilot study. Ann. Rheum. Dis. 68, 1613–1617 (2009).
Chollet-Janin, A., Finckh, A., Dudler, J. & Guerne, P. A. Methotrexate as an alternative therapy for chronic calcium pyrophosphate deposition disease: an exploratory analysis. Arthritis Rheum. 56, 688–692 (2007).
Doan, T. H. T., Chevalier, X., Leparc, J. M., Richette, P., Bardin, T. & Forestier, R. French Society for Rheumatology Osteoarthritis Section. Premature enthusiasm for the use of methotrexate for refractory chondrocalcinosis: Comment on the article by Chollet-Janin et al. Arthritis Rheum. 58, 2 (2008).
Rothschild, B. & Yakubov, L. E. Prospective 6-month, double-blind trial of hydroxychloroquine treatment of CPDD. Compr. Ther. 23, 327–331 (1997).
Doherty, M. & Dieppe, P. Double blind, placebo controlled trial of magnesium carbonate in chronic pyrophosphate arthropathy. Ann. Rheum. Dis. 42, 2 (1983).
Hunter, G. K., Grynpas, M. D., Cheng, P. T. & Pritzker, K. P. Effect of glycosaminoglycans on calcium pyrophosphate crystal formation in collagen gels. Calcif. Tissue Int. 41, 164–170 (1987).
Rosenthal, A. K. & Ryan, L. M. Probenecid inhibits transforming growth factor-β1 induced pyrophosphate elaboration by chondrocytes. J. Rheumatol. 21, 896–900 (1994).
Kannampuzha, J. V., Tupy, J. H. & Pritzker, K. P. Mercaptopyruvate inhibits tissue-nonspecific alkaline phosphatase and calcium pyrophosphate dihydrate crystal dissolution. J. Rheumatol. 36, 8 (2009).
Shinozaki, T. & Pritzker, K. P. Regulation of alkaline phosphatase: implications for calcium pyrophosphate dihydrate crystal dissolution and other alkaline phosphatase functions. J. Rheumatol. 23, 677–683 (1996).
Cheung, H. S. Phosphocitrate as a potential therapeutic strategy for crystal deposition disease. Curr. Rheumatol. Rep. 3, 24–28 (2001).
Derfus, B. et al. Transforming growth factor β-1 stimulates articular chondrocyte elaboration of matrix vesicles capable of greater calcium pyrophosphate precipitation. Osteoarthritis Cartilage 9, 189–194 (2001).
Masuda, I. et al. Variations in site and levels of expression of chondrocyte nucleotide pyrophosphohydrolase with aging. J. Bone Miner. Res. 16, 868–875 (2001).
Rosenthal, A. K. & Henry, L. A. Retinoic acid stimulates pyrophosphate elaboration by cartilage and chondrocytes. Calcif. Tissue Int. 59, 128–133 (1996).
Kojima, S., Nara, K. & Rifkin, D. B. Requirement for transglutaminase in the activation of latent transforming growth factor-β in bovine endothelial cell. J. Cell Biol. 121, 439–448 (1993).
Hirose, J., Ryan, L. M. & Masuda, I. Up-regulated expression of cartilage intermediate-layer protein and ANK in articular hyaline cartilage from patients with calcium pyrophosphate dihydrate crystal deposition disease. Arthritis Rheum. 46, 3218–3229 (2002).
Sohn, P., Crowley, M., Slattery, E. & Serra, R. Developmental and TGF β mediated regulation of Ank mRNA expression in cartilage and bone. Osteoarthirtis Cartilage 10, 482–490 (2002).
Rosenthal, A. K., Derfus, B. A. & Henry, L. A. Transglutaminase activity in aging articular chondrocytes and articular cartilage vesicles. Arthritis Rheum. 40, 966–970 (1997).
Olmez, U., Ryan, L. M., Kurup, I. V. & Rosenthal, A. K. Insulin-like growth factor-1 suppresses pyrophosphate elaboration by transforming growth factor β1-stimulated chondrocytes and cartilage. Osteoarthritis Cartilage 2, 149–154 (1994).
Cailotto, F. et al. Inorganic pyrophosphate generation by transforming growth factor β1 is mainly dependent on ANK induction by Ras/Raf-1/extracellular signal-regulated kinase pathways in chondrocytes. Arthritis Res. Ther. 9, R122 (2007).
Ryan, L. M., Wortmann, R. L., Karas, B., Lynch, M. P. & McCarty, D. J. Pyrophosphohydrolase activity and inorganic pyrophosphate content of cultured human skin fibroblasts. Elevated levels in some patients with calcium pyrophosphate dihydrate deposition disease. J. Clin. Invest. 77, 1689–1693 (1986).
Williams, C. J. et al. Autosomal dominant familial calcium pyrophosphate dihydrate deposition disease is caused by mutation in the transmembrane protein ANKH. Am. J. Hum. Genet. 71, 985–991 (2002).
Lust, G., Faure, G., Netter, P., Gaucher, A. & Seegmiller, J. E. Evidence of a generalized metabolic defect in patients with hereditary chondrocalcinosis. Increased inorganic pyrophosphate in cultured fibroblasts and lymphoblasts. Arthritis Rheum. 24, 1517–1521 (1981).
Wang, J., Tsui, H. W., Beier, F. & Tsui, F. W. The CPPDD-associated ANKH M48T mutation interrupts the interaction of ANKH with the sodium/phosphate cotransporter PiT-1. J. Rheumatol. 36, 1265–1272 (2009).
Wang, J., Tsui, H. W., Beier, F., Pritzker, K. P., Inman, R. D. & Tsui, F. W. The ANKH DeltaE490 mutation in calcium pyrophosphate dihydrate crystal deposition disease (CPPDD) affects tissue non-specific alkaline phosphatase (TNAP) activities. Open Rheumatol. J. 2, 23–30 (2008).
Author information
Authors and Affiliations
Contributions
A. Abhishek and M. Doherty contributed equally to researching data for the article, providing a substantial contribution to discussions of the content, writing the article, and to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Abhishek, A., Doherty, M. Pathophysiology of articular chondrocalcinosis—role of ANKH. Nat Rev Rheumatol 7, 96–104 (2011). https://doi.org/10.1038/nrrheum.2010.182
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2010.182
This article is cited by
-
Cartilage calcification in osteoarthritis: mechanisms and clinical relevance
Nature Reviews Rheumatology (2023)
-
Tensin-3 is involved in osteogenic versus adipogenic fate of human bone marrow stromal cells
Cellular and Molecular Life Sciences (2023)
-
Management of temporomandibular joint diseases: a rare case report of coexisting calcium pyrophosphate crystal deposition and synovial chondromatosis
BMC Oral Health (2022)
-
Association between serum zinc and copper concentrations and copper/zinc ratio with the prevalence of knee chondrocalcinosis: a cross-sectional study
BMC Musculoskeletal Disorders (2020)
-
Differences in intracellular localisation of ANKH mutants that relate to mechanisms of calcium pyrophosphate deposition disease and craniometaphyseal dysplasia
Scientific Reports (2020)