Abstract
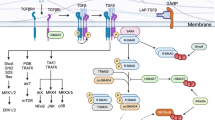
Type 17 T helper (TH17) cells are a population of CD4+ effector T cells that are distinct from TH1 and TH2 cells owing to their ability to produce interleukin (IL)-17. Although TH1 and TH2 cells are similar in mice and humans, TH17 cells differ in several ways. The differentiation of mouse TH17 cells requires transforming growth factor β and IL-6, whereas human naive T cells can develop into TH17 cells in the presence of IL-1β and IL-23 alone, transforming growth factor β having an indirect role in their development via the selective inhibition of TH1 cell expansion. In both mice and humans, a late developmental plasticity of TH17 cells towards the TH1 lineage has been shown. Mainly based on mouse gene knockout studies, TH17 lymphocytes have been found to have a pathogenic role in several autoimmune disorders; however, whether human autoimmune disorders, including rheumatoid arthritis (RA) and psoriasis, are prevalently TH1-mediated or TH17-mediated, is still unclear. Research suggests that both TH1 and TH17 cells are involved in RA pathogenesis, raising the possibility that interventions that target both the IL-23–IL-17 (TH17) and the IL-12–interferon γ (TH1) axes might be successful future therapeutic approaches for RA.
Key Points
-
It is our opinion that the origin and development of type 17 T helper (TH17) cells in humans is different than in mice
-
Human TH17 cells can be induced to produce interferon-γ in the presence of interleukin (IL)-12, thus becoming TH17/TH1 cells
-
TH17 cells are associated with several immune-mediated diseases, particularly psoriasis and rheumatoid arthritis, owing to the pleiotropic effects exerted by IL-17 and related-cytokines on osteoblasts, B cells and monocytes
-
The pathogenesis of rheumatoid arthritis is more complex than previously thought; both TH17 and TH1 cells are probably involved in the maintenance of inflammation that leads to joint destruction
-
Immune-mediated diseases could perhaps be treated more effectively by therapeutic interventions that target both the IL-23–IL-17 (TH17) and IL12–interferon-γ (TH1) axes, rather than just a single pathway
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mosmann, T. R., Cherwinski, C., Bond, M. W., Giedlin, M. A. & Coffman, R. L. Two types of murine helper T cell clones. I. Definition according to profiles of lymphokine activities and secreted proteins. J. Immunol. 136, 2348–2357 (1986).
Romagnani, S. Human TH1 and TH2 subsets: doubt no more. Immunol. Today 12, 256–257 (1991).
Romagnani, S. The TH1/TH2 paradigm. Immunol. Today 18, 263–266 (1997).
Gately, M. K. et al. The interleukin-12/interleukin-12-receptor system: role in normal and pathologic immune responses. Annu. Rev. Immunol. 16, 495–521 (1998).
Cua, D. J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
Murphy, C. A. et al. Divergent pro- and anti-inflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J. Exp. Med. 198, 1951–1958 (2003).
Yao, Z. et al. Herpesvirus Saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. Immunity 3, 811–821 (1995).
Dong, C. Regulation and pro-inflammatory function of interleukin-17 family cytokines. Immunol. Rev. 226, 80–86 (2008).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector Th17 and regulatory T cells. Nature 44, 235–238 (2006).
Mangan, P. R. et al. Transforming growth factor-beta induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Veldhoen, M., Hocking, R. J., Atkins, C. J., Locksley, R. M. & Stockinger, B. TGF-beta in the context of an inflammatory cytokine milieu supports differentiation of IL-17-producing T cells. Immunity 25, 179–189 (2006).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector Th17 and regulatory T cells. Nature 44, 235–238 (2006).
Huber, M. et al. IRF4 is essential for IL-21-mediated induction, amplification, and stabilization of the Th17 phenotype. Proc. Natl Acad. Sci. USA 105, 20846–20851 (2008).
Veldhoen, M., Hirota, K., Christensen, J., O'Garra, A. & Stockinger, B. Natural agonists for aryl hydrocarbon receptor in culture medium are essential for optimal differentiation of Th17 T cells. J. Exp. Med. 206, 43–49 (2009).
Ivanov, I. I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatopry IL-17+ T helper cells. Cell 126, 1121–1131 (2006).
Yang, X. O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors RORα and RORγ. Immunity 28, 29–39 (2008).
Chen, Z., Laurence, A. & O'Shea, J. J. Signal transduction pathways and transcriptional regulation in the control of Th17 differentiation. Semin. Immunol. 19, 400–408 (2007).
Ouyang, W., Koli, J. K. & Zheng, Y. The biological functions of T helper 17 cell effector cytokines in inflammation. Immunity 28, 454–467 (2008).
Dubin, P. J. & Kolls, J. K. Th17 cytokines and mucosal immunity. Immunol. Rev. 226, 160–171 (2008).
Takatori, H. et al. Lymphoid tissue inducer-like cells are an innate source of IL-17 and IL-22. J. Exp. Med. 206, 35–41 (2009).
Oukka, M. Th17 cells in immunity and autoimmunity. Ann. Rheum. Dis. 67 (Suppl. 3), 26–29 (2008).
Acosta-Rodriguez, E. V., Napolitani, G., Lanzavecchia, A. & Sallusto, F. Interleukins 1β and 6 but not transforming growth factor-β are essential for the differentiation of interleukin 17-producing human T helper cells. Nat. Immunol. 8, 942–949 (2007).
Wilson, N. J. et al. Development, cytokine profile and function of human interleukin-17-producing helper T cells. Nat. Immunol. 8, 950–957 (2007).
Evans, H. G., Suddason, T., Jackson, I., Taams, L. S. & Lord, G. M. Optimal induction of T helper 17 cells in humans requires T cell receptor ligation in the context of Toll-like receptor-activated monocytes. Proc. Natl Acad. Sci. USA 104, 17034–17039 (2007).
van Beelen, A. J. et al. Stimulation of the intracellular bacterial sensor NOD2 programs dendritic cells to promote interleukin-17 production in human memory T cells. Immunity 27, 660–669 (2007).
Manel, N., Unutmaz, D. & Littman, D. R. The differentiation of human Th17 cells requires transforming growth factor-β and induction of the nuclear receptor RORγt. Nat. Immunol. 9, 641–649 (2008).
Volpe, E. et al. A critical function for transforming growth factor-β, interleukin-23 and proinflammatory cytokines in driving and modulating human Th17 responses. Nat. Immunol. 9, 650–957 (2008).
Yang, L. et al. IL-21 and TGF-β are required for differentiation of human TH17 cells. Nature 454, 350–352 (2008).
Cosmi, L. et al. Human interleukin 17-producing cells originate from a CD161+CD4+ T cell precursor. J. Exp. Med. 205, 1903–1916 (2008).
de Beaucoudrey, L. et al. Mutations in STAT3 and IL12RB1 impair the development of human IL-17-producing T cells. J. Exp. Med. 205, 1543–1550 (2008).
Ma, C. S. et al. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J. Exp. Med. 205, 1551–1557 (2008).
Milner, J. D. et al. Impaired Th17 differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 452, 773–776 (2008).
Santarlasci, V. et al. TGF-beta indirectly favors the development of human Th17 cells by inhibiting Th1 cells. Eur. J. Immunol. 39, 207–215 (2009).
Kroenke, M. A., Carlson, T. J., Andjelkovic, A. V. & Segal, B. M. IL-12- and IL-23-modulated T cells induce distinct types of EAE based on histology, CNS chemokine profile, and response to cytokine inhibition. J. Exp. Med. 205, 1535–1541 (2009).
Doodes, P. D. et al. Development of proteoglycan-induced arthritis is independent of IL-17. J. Immunol. 181, 329–337 (2008).
Luger, D. et al. Either a Th17 or a Th1 effector response can drive autoimmunity: conditions of disease induction affect dominant effector category. J. Exp. Med. 205, 799–810 (2008).
Collison, L. W. & Vignali, D. A. Interleukin-35: odd one out or part of the family? Immunol. Rev. 226, 248–262 (2008).
Komiyama, Y. et al. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J. Immunol. 177, 566–573 (2006).
Nakae, S., Nambu, A., Sudo, K. & Iwakura, Y. Suppression of immune induction of collagen-induced arthritis in IL-17-deficient mice. J. Immunol. 171, 6173–6177 (2003).
Hofstetter, H. H. et al. Therapeutic efficacy of IL-17 neutralization in murine experimental autoimmune encephalomyelitis. Cell. Immunol. 237, 123–130 (2005).
Ogawa, A., Andoh, A., Araki, Y., Bamba, T. & Fujiyama, Y. Neutralization of interleukin-17 aggravates dextran sulfate sodium-induced colitis in mice. Clin. Immunol. 110, 55–62 (2004).
Zhang, Z. et al. After interleukin-12p40, are interleukin-23 and interleukin-17 the next therapeutic targets for inflammatory bowel disease? Int. Immunopharmacol. 7, 409–416 (2007).
Duerr, R. H. et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 314, 1461–1463 (2006).
Fuss, I. J. et al. Both IL-12p70 and IL-23 are synthesized during active Crohn's disease and are down-regulated by treatment with anti-IL-12 p40 monoclonal antibody. Inflamm. Bowel Dis. 12, 9–15 (2006).
Annunziato, F. et al. Phenotypic and functional features of human Th17 cells. J. Exp. Med. 204, 1849–1861 (2007).
Lee, Y. K. et al. Late developmental plasticity in the T helper 17 lineage. Immunity 30, 92–107 (2009).
Shi, G. et al. Phenotype switching by inflammation-inducing polarized Th17 cells, but not by Th1 cells. J. Immunol. 181, 7205–7213 (2008).
Bending, D. et al. Highly purified Th17 cells from BDC2.5NOD mice convert into Th1-like cells in NOD/SCID recipient mice. J. Clin. Invest. [doi:10.1172/JCI37865] (2009).
Xu, L., Kitani, A., Fuss, I. & Strober, W. Cutting edge: regulatory T cells induce CD4+CD25−Foxp3− T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-beta. J. Immunol. 178, 6725–6729 (2007).
Koenen, H. J. et al. Human CD25highFoxp3pos regulatory T cells differentiate into IL-17-producing cells. Blood 112, 2340–2352 (2008).
Beriou, G. et al. IL-17 producing human peripheral regulatory T cells retain suppressive function. Blood [doi:10.1182/blood-2008-10-183251] (2009).
Nakae, S. et al. IL-17 production from activated T cells is required for the spontaneous development of destructive arthritis in mice deficient in IL-1 receptor antagonist. Proc. Natl Acad. Sci. USA 100, 5986–5990 (2003).
Hirota, K. et al. T cell self-reactivity forms a cytokine milieu for spontaneous development of IL-17+ Th cells that cause autoimmune arthritis. J. Exp. Med. 204, 41–47 (2007).
Fujimoto, M. et al. Interleukin-6 blockade suppresses autoimmune arthritis in mice by the inhibition of inflammatory Th17 responses. Arthritis Rheum. 58, 3710–3719 (2008).
Young, D. A. et al. Blockade of the interleukin-21/interleukin-21 receptor pathway ameliorates disease in animal models of rheumatoid arthritis. Arthritis Rheum. 56, 1152–1163 (2007).
Mathur, A. N. et al. STAT3 and STAT4 direct development of IL-17-secreting Th cells. J. Immunol. 178, 4901–4907 (2007).
Hildner, K. M. et al. Targeting of the transcription factor STAT4 by antisense phosphorothioate oligonucleotides suppresses collagen-induced arthritis. J. Immunol. 178, 3427–3436 (2007).
Geboes, L. et al. Proinflammatory role of the Th17 cytokine interleukin-22 in collagen-induced arthritis in C57BL/6 mice. Arthritis Rheum. 60, 390–395 (2009).
Zheng, Y. et al. Interleukin-22, a TH17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature 445, 648–651 (2007).
Piskin, G., Sylva-Steenland, R. M., Bos, J. D. & Teunissen, M. B. In vitro and in situ expression of IL-23 by keratinocytes in healthy skin and psoriasis lesions: enhanced expression in psoriatic skin. J. Immunol. 176, 1908–1915 (2006).
Capon, F. et al. Sequence variants in the genes for the interleukin-23 receptor (IL23R) and its ligand (IL12B) confer protection against psoriasis. Hum. Genet. 122, 201–206 (2007).
Nair, R. P. et al. Polymorphisms of the IL12B and IL23R genes are associated with psoriasis. J. Invest. Dermatol. 128, 1653–1661 (2008).
Zaba, L. C. et al. Amelioration of epidermal hyperplasia by TNF inhibition is associated with reduced Th17 responses. J. Exp. Med. 204, 3183–3194 (2007).
Arican, O., Sasmaz, S. & Ciragil, P. Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm. 5, 273–279 (2005).
Leonardi, C. L. et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1). Lancet 371, 1665–1674 (2008).
Papp, K. A. et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet 371, 1675–1684 (2008).
Fossiez, F. et al. T cell interleukin-17 induces stromal cells to produce proinflammatory and hematopoietic cytokines. J. Exp. Med. 183, 2593–2603 (1996).
Sato, K. et al. Regulation of osteoclast differentiation and function by the CaMK-CREB pathway. Nat. Med. 12, 1410–1416 (2006).
Kirkham, B. W. et al. Synovial membrane cytokine expression is predictive of joint damage progression in rheumatoid arthritis: a two-year prospective study (the DAMAGE study cohort). Arthritis Rheum. 54, 1122–1131 (2006).
Bush, K. A., Farmer, K. M., Walker, J. S. & Kirkham, B. W. Reduction of joint inflammation and bone erosion in rat adjuvant arthritis by treatment with interleukin-17 receptor IgG1 Fc fusion protein. Arthritis Rheum. 46, 802–805 (2002).
Lubberts, E. IL-17/Th17 targeting: on the road to prevent chronic destructive arthritis? Cytokine 41, 84–91 (2008).
Aarvak, T., Chabaud, M., Miossec, P. & Natvig, J. B. IL-17 is produced by some proinflammatory Th1/Th0 cells but not by Th2 cells. J. Immunol. 162, 1246–1251 (1999).
Mauritz, N. et al. Treatment with γ-interferon triggers the onset of collagen arthritis in mice. Arthritis Rheum. 31, 1297–1304 (1988).
Cooper, S. M., Sriram, S. & Ranges, G. E. Suppression of murine collagen-induced arthritis with monoclonal anti-Ia antibodies and augmentation with IFN-gamma. J. Immunol. 141, 1958–1962 (1988).
Singh, R., Aggarwal, A. & Misra, R. Th1/Th17 cytokine profiles in patients with reactive arthritis/undifferentiated spondyloarthropathy. J. Rheumatol. 34, 2285–2290 (2007).
Notley, C. A. et al. Blockade of tumor necrosis factor in collagen-induced arthritis reveals a novel immunoregulatory pathway for Th1 and Th17 cells. J. Exp. Med. 205, 2491–2497 (2008).
Martin-Orozco, N., Chung, Y., Chang, S. H., Wang, Y. H. & Dong, C. Th17 cells promote pancreatic inflammation but only induce diabetes efficiently in lymphopenic hosts after conversion into Th1 cells. Eur. J. Immunol. 39, 216–224 (2009).
Acknowledgements
The studies by F. Annunziato, L. Cosmi, F. Liotta, E. Maggi and S. Romagnani discussed in this Review have been supported by funds from the Italian Ministry of Education (40%), the Italian Ministry of Health, the Ente Cassa di Risparmio of Florence, and EU Projects SENS-IT-IV and INNOCHEM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Annunziato, F., Cosmi, L., Liotta, F. et al. Type 17 T helper cells—origins, features and possible roles in rheumatic disease. Nat Rev Rheumatol 5, 325–331 (2009). https://doi.org/10.1038/nrrheum.2009.80
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2009.80
This article is cited by
-
The roles of immune cells in Behçet’s disease
Advances in Rheumatology (2023)
-
Identification and Analysis of DNA Methylation Inflammation-Related Key Genes in Intracerebral Hemorrhage
Biochemical Genetics (2023)
-
CD147-mediated chemotaxis of CD4+CD161+ T cells may contribute to local inflammation in rheumatoid arthritis
Clinical Rheumatology (2018)
-
miR-210 expression in PBMCs from patients with systemic lupus erythematosus and rheumatoid arthritis
Irish Journal of Medical Science (1971 -) (2018)
-
GWAS analysis using interspecific backcross progenies reveals superior blue catfish alleles responsible for strong resistance against enteric septicemia of catfish
Molecular Genetics and Genomics (2018)