Abstract
Amyotrophic lateral sclerosis (ALS) is a relentlessly progressive neurodegenerative disease of motor neurons, resulting in worsening weakness of voluntary muscles until death from respiratory failure occurs after about 3 years. Although great advances have been made in our understanding of the genetic causes of ALS, the contribution of environmental factors has been more difficult to assess. Large-scale studies of the clinical patterns of ALS, individual histories preceding the onset of ALS, and the rates of ALS in different populations and groups have led to improved patient care, but have not yet revealed a replicable, definitive environmental risk factor. In this Review, we outline what is currently known of the environmental and genetic epidemiology of ALS, describe the current state of the art with respect to the different types of ALS, and explore whether ALS should be considered a single disease or a syndrome. We examine the relationship between genetic and environmental risk factors, and propose a disease model in which ALS is considered to be the result of environmental risks and time acting on a pre-existing genetic load, followed by an automatic, self-perpetuating decline to death.
Key Points
-
Amyotrophic lateral sclerosis (ALS) is a syndrome rather than a single disease; understanding which clinical patterns go together is a challenge but will improve our ability to identify the causes
-
The frequency of ALS seems to be constant in European-derived populations worldwide
-
For other populations—where sufficient data are available—the risk appears to be lower than in European-derived populations
-
Environmental risk factors are difficult to study because the search space is infinite
-
No replicable, definitive environmental risk factors for ALS have yet been identified
-
The data are consistent with a model in which a pre-existing genetic load is acted on by time and environmental exposures until a tipping point is reached and neurodegeneration begins
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aran, F. A. Research on an as yet undescribed disease of the muscular system (progressive muscular atrophy). Arch. Gen. Med. 24, 15–35 (1848).
Cruveilhier, J. Sur la paralysie musculaire, progressive, atrophique [French]. Bull. Acad. Med. (Paris) 18, 490–502, 546–583 (1852).
Charcot, J. M. & Joffroy, A. Deux cas d'atrophie musculaire progressive avec lesions de la substance grise et des faisceaux antero-lateraux de la moelle epiniere [French]. Arch. Physiol. Neurol. Pathol. 2, 744 (1869).
Kurland, L. T. & Mulder, D. W. Epidemiologic investigations of amyotrophic lateral sclerosis. 2. Familial aggregations indicative of dominant inheritance. I. Neurology 5, 182–196 (1955).
Kurland, L. T. & Mulder, D. W. Epidemiologic investigations of amyotrophic lateral sclerosis. 2. Familial aggregations indicative of dominant inheritance. II. Neurology 5, 249–268 (1955).
Kew, J. J. et al. The relationship between abnormalities of cognitive function and cerebral activation in amyotrophic lateral sclerosis. A neuropsychological and positron emission tomography study. Brain 116, 1399–1423 (1993).
Goldstein, L. H. & Abrahams, S. Changes in cognition and behaviour in amyotrophic lateral sclerosis: nature of impairment and implications for assessment. Lancet Neurol. 12, 368–380 (2013).
Steele, J. C. & McGeer, P. L. The ALS/PDC syndrome of Guam and the cycad hypothesis. Neurology 70, 1984–1990 (2008).
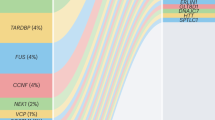
Andersen, P. M. & Al-Chalabi, A. Clinical genetics of amyotrophic lateral sclerosis: what do we really know? Nat. Rev. Neurol. 7, 603–615 (2011).
Abel, O., Powell, J. F., Andersen, P. M. & Al-Chalabi, A. ALSoD: a user-friendly online bioinformatics tool for amyotrophic lateral sclerosis genetics. Hum. Mutat. 33, 1345–1351 (2012).
Brooks, B. R. El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. Subcommittee on Motor Neuron Diseases/Amyotrophic Lateral Sclerosis of the World Federation of Neurology Research Group on Neuromuscular Diseases and the El Escorial “Clinical limits of amyotrophic lateral sclerosis” workshop contributors. J. Neurol. Sci. 124 (Suppl.), 96–107 (1994).
Nodera, H., Izumi, Y. & Kaji, R. New diagnostic criteria of ALS (Awaji criteria) [Japanese]. Brain Nerve 59, 1023–1029 (2007).
Roche, J. C. et al. A proposed staging system for amyotrophic lateral sclerosis. Brain 135, 847–852 (2012).
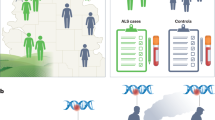
Huisman, M. H. et al. Population based epidemiology of amyotrophic lateral sclerosis using capture-recapture methodology. J. Neurol. Neurosurg. Psychiatry 82, 1165–1170 (2011).
Mortara, P., Chiò, A., Rosso, M. G., Leone, M. & Schiffer, D. Motor neuron disease in the province of Turin, Italy, 1966–1980. Survival analysis in an unselected population. J. Neurol. Sci. 66, 165–173 (1984).
Lee, C. T. et al. Riluzole and prognostic factors in amyotrophic lateral sclerosis long-term and short-term survival: a population-based study of 1149 cases in Taiwan. J. Epidemiol. 23, 35–40 (2013).
Pugliatti, M. et al. Amyotrophic lateral sclerosis in Sardinia, insular Italy, 1995–2009. J. Neurol. 260, 572–579 (2013).
Byrne, S. et al. Cognitive and clinical characteristics of patients with amyotrophic lateral sclerosis carrying a C9orf72 repeat expansion: a population-based cohort study. Lancet Neurol. 11, 232–240 (2012).
Chiò, A. et al. Prognostic factors in ALS: a critical review. Amyotroph. Lateral Scler. 10, 310–323 (2009).
Abhinav, K. et al. Amyotrophic lateral sclerosis in South-East England: a population-based study. The South-East England register for amyotrophic lateral sclerosis (SEALS Registry). Neuroepidemiology 29, 44–48 (2007).
Wijesekera, L. C. et al. Natural history and clinical features of the flail arm and flail leg ALS variants. Neurology 72, 1087–1094 (2009).
Gordon, P. H. et al. The natural history of primary lateral sclerosis. Neurology 66, 647–653 (2006).
Kim, W. K. et al. Study of 962 patients indicates progressive muscular atrophy is a form of ALS. Neurology 73, 1686–1692 (2009).
Van den Berg-Vos, R. M. et al. A long-term prospective study of the natural course of sporadic adult-onset lower motor neuron syndromes. Arch. Neurol. 66, 751–757 (2009).
Ganesalingam, J. et al. Latent cluster analysis of ALS phenotypes identifies prognostically differing groups. PLoS ONE 4, e7107 (2009).
Elamin, M. et al. Cognitive changes predict functional decline in ALS: a population-based longitudinal study. Neurology 80, 1590–1597 (2013).
Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 (2006).
Kwiatkowski, T. J. Jr et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science 323, 1205–1208 (2009).
Vance, C. et al. Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science 323, 1208–1211 (2009).
Al-Sarraj, S. et al. p62 positive, TDP-43 negative, neuronal cytoplasmic and intranuclear inclusions in the cerebellum and hippocampus define the pathology of C9orf72-linked FTLD and MND/ALS. Acta Neuropathol. 122, 691–702 (2011).
Byrne, S. et al. Rate of familial amyotrophic lateral sclerosis: a systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 82, 623–627 (2011).
Byrne, S. et al. Proposed criteria for familial amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 12, 157–159 (2011).
Hanby, M. F. et al. The risk to relatives of patients with sporadic amyotrophic lateral sclerosis. Brain 134, 3454–3457 (2011).
Al-Chalabi, A. & Lewis, C. M. Modelling the effects of penetrance and family size on rates of sporadic and familial disease. Hum. Hered. 71, 281–288 (2011).
Al-Chalabi, A. et al. An estimate of amyotrophic lateral sclerosis heritability using twin data. J. Neurol. Neurosurg. Psychiatry 81, 1324–1326 (2010).
Chiò, A. et al. Epidemiology of ALS in Italy: a 10-year prospective population-based study. Neurology 72, 725–731 (2009).
Donaghy, C. et al. The epidemiology of motor neuron disease in Northern Ireland using capture–recapture methodology. Amyotroph. Lateral Scler. 11, 374–378 (2010).
Wittie, M., Nelson, L. M., Usher, S., Ward, K. & Benatar, M. Utility of capture–recapture methodology to assess completeness of amyotrophic lateral sclerosis case ascertainment. Neuroepidemiology 40, 133–141 (2013).
Donaghy, C. et al. An all-Ireland epidemiological study of MND, 2004–2005. Eur. J. Neurol. 16, 148–153 (2009).
Manjaly, Z. R. et al. The sex ratio in amyotrophic lateral sclerosis: a population based study. Amyotroph. Lateral Scler. 11, 439–442 (2010).
Johnston, C. A. et al. Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London. J. Neurol. 253, 1642–1643 (2006).
Gordon, P. H. et al. Incidence of amyotrophic lateral sclerosis among American Indians and Alaska natives. JAMA Neurol. 70, 476–480 (2013).
Kenna, K. P. et al. Delineating the genetic heterogeneity of ALS using targeted high-throughput sequencing. J. Med. Genet. http://dx.doi.org/10.1136/jmedgenet-2013-101795.
Jang, J. H. et al. Analysis of the C9orf72 hexanucleotide repeat expansion in Korean patients with familial and sporadic amyotrophic lateral sclerosis. Neurobiol. Aging 34, 1311.e7–1311.e9 (2013).
Konno, T. et al. Japanese amyotrophic lateral sclerosis patients with GGGGCC hexanucleotide repeat expansion in C9ORF72. J. Neurol. Neurosurg. Psychiatry 84, 398–401 (2013).
Garcia-Redondo, A. et al. Analysis of the C9orf72 gene in patients with amyotrophic lateral sclerosis in Spain and different populations worldwide. Hum. Mutat. 34, 79–82 (2013).
Smith, B. N. et al. The C9ORF72 expansion mutation is a common cause of ALS+/−FTD in Europe and has a single founder. Eur. J. Hum. Genet. 21, 102–108 (2013).
Tsai, C. P. et al. A hexanucleotide repeat expansion in C9ORF72 causes familial and sporadic ALS in Taiwan. Neurobiol. Aging 33, 2232.e11–2232.e18 (2012).
Gil, J. et al. Prognosis of ALS: comparing data from the Limousin referral centre, France, and a Uruguayan population. Amyotroph. Lateral Scler. 10, 355–360 (2009).
Alonso, V. et al. Increase in motor neuron disease mortality in Spain: temporal and geographical analysis (1990–2005). Amyotroph. Lateral Scler. 12, 192–198 (2011).
Mehal, J. M., Holman, R. C., Schonberger, L. B. & Sejvar, J. J. Amyotrophic lateral sclerosis/motor neuron disease deaths in the United States, 1999–2009. Amyotroph. Lateral Scler. Frontotemporal Degener. 14, 346–352 (2013).
Zaldivar, T. et al. Reduced frequency of ALS in an ethnically mixed population: a population-based mortality study. Neurology 72, 1640–1645 (2009).
Fong, G. C. et al. An epidemiological study of motor neuron disease in Hong Kong. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 6, 164–168 (2005).
Cronin, S., Hardiman, O. & Traynor, B. J. Ethnic variation in the incidence of ALS: a systematic review. Neurology 68, 1002–1007 (2007).
Koerner, D. R. Amyotrophic lateral sclerosis on Guam. Ann. Intern. Med. 37, 1204–1220 (1952).
Arnold, A., Edgren, D. C. & Palladino, V. S. Amyotrophic lateral sclerosis; fifty cases observed on Guam. J. Nerv. Ment. Dis. 117, 135–139 (1953).
Mulder, D. W., Kurland, L. T. & Iriarte, L. L. Neurologic diseases on the island of Guam. U. S. Armed Forces Med. J. 5, 1724–1739 (1954).
Bradley, W. G. & Mash, D. C. Beyond Guam: the cyanobacteria/BMAA hypothesis of the cause of ALS and other neurodegenerative diseases. Amyotroph. Lateral Scler. 10 (Suppl. 2), 7–20 (2009).
Ishiura, H. et al. C9ORF72 repeat expansion in amyotrophic lateral sclerosis in the Kii peninsula of Japan. Arch. Neurol. 69, 1154–1158 (2012).
Logroscino, G. et al. Incidence of amyotrophic lateral sclerosis in Europe. J. Neurol. Neurosurg. Psychiatry 81, 385–390 (2010).
Nalini, A., Thennarasu, K., Gourie-Devi, M., Shenoy, S. & Kulshreshtha, D. Clinical characteristics and survival pattern of 1,153 patients with amyotrophic lateral sclerosis: experience over 30 years from India. J. Neurol. Sci. 272, 60–70 (2008).
Phukan, J. et al. The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J. Neurol. Neurosurg. Psychiatry 83, 102–108 (2012).
Byrne, S. et al. Aggregation of neurologic and neuropsychiatric disease in ALS kindreds: a population based case controlled cohort study of familial and sporadic ALS. Ann. Neurol. http://dx.doi.org/10.1002/ana.23969.
Lewis, M. & Gordon, P. H. Lou Gehrig, Rawhide, and 1938. Neurology 68, 615–618 (2007).
Scarmeas, N., Shih, T., Stern, Y., Ottman, R. & Rowland, L. P. Premorbid weight, body mass, and varsity athletics in ALS. Neurology 59, 773–775 (2002).
Besson, H. et al. Validation of the historical adulthood physical activity questionnaire (HAPAQ) against objective measurements of physical activity. Int. J. Behav. Nutr. Phys. Act. 7, 54 (2010).
Huisman, M. H. et al. Lifetime physical activity and the risk of amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 84, 976–981 (2013).
Eisen, A., Kim, S. & Pant, B. Amyotrophic lateral sclerosis (ALS): a phylogenetic disease of the corticomotoneuron? Muscle Nerve 15, 219–224 (1992).
Turner, M. R. et al. Concordance between site of onset and limb dominance in amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 82, 853–854 (2011).
Vivekananda, U. et al. Low index-to-ring finger length ratio in sporadic ALS supports prenatally defined motor neuronal vulnerability. J. Neurol. Neurosurg. Psychiatry 82, 635–637 (2011).
de Jong, S. W. et al. Smoking, alcohol consumption, and the risk of amyotrophic lateral sclerosis: a population-based study. Am. J. Epidemiol. 176, 233–239 (2012).
Sutedja, N. A. et al. Beneficial vascular risk profile is associated with amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 82, 638–642 (2011).
Gallo, V. et al. Prediagnostic body fat and risk of death from amyotrophic lateral sclerosis: the EPIC cohort. Neurology 80, 829–838 (2013).
O'Reilly, E. J. et al. Premorbid body mass index and risk of amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Frontotemporal Degener. 14, 205–211 (2013).
Beretta, S., Carri, M. T., Beghi, E., Chio, A. & Ferrarese, C. The sinister side of Italian soccer. Lancet Neurol. 2, 656–657 (2003).
Chio, A., Benzi, G., Dossena, M., Mutani, R. & Mora, G. Severely increased risk of amyotrophic lateral sclerosis among Italian professional football players. Brain 128, 472–476 (2005).
Armon, C. Sports and trauma in amyotrophic lateral sclerosis revisited. J. Neurol. Sci. 262, 45–53 (2007).
Wicks, P. et al. Three soccer playing friends with simultaneous amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 8, 177–179 (2007).
Nelson, L. M., McGuire, V., Longstreth, W. T. Jr & Matkin, C. Population-based case–control study of amyotrophic lateral sclerosis in western Washington State. I. Cigarette smoking and alcohol consumption. Am. J. Epidemiol. 151, 156–163 (2000).
Armon, C. Smoking may be considered an established risk factor for sporadic ALS. Neurology 73, 1693–1698 (2009).
Alonso, A., Logroscino, G., Jick, S. S. & Hernán, M. A. Association of smoking with amyotrophic lateral sclerosis risk and survival in men and women: a prospective study. BMC Neurol. 10, 6 (2010).
Weisskopf, M. G. et al. Prospective study of cigarette smoking and amyotrophic lateral sclerosis. Am. J. Epidemiol. 160, 26–33 (2004).
Wang, H. et al. Smoking and risk of amyotrophic lateral sclerosis: a pooled analysis of 5 prospective cohorts. Arch. Neurol. 68, 207–213 (2011).
Sutedja, N. A. et al. Exposure to chemicals and metals and risk of amyotrophic lateral sclerosis: a systematic review. Amyotroph. Lateral Scler. 10, 302–309 (2009).
Sutedja, N. A. et al. What we truly know about occupation as a risk factor for ALS: a critical and systematic review. Amyotroph. Lateral Scler. 10, 295–301 (2009).
Coffman, C. J., Horner, R. D., Grambow, S. C. & Lindquist, J. Estimating the occurrence of amyotrophic lateral sclerosis among Gulf War (1990–1991) veterans using capture–recapture methods. Neuroepidemiology 24, 141–150 (2005).
Horner, R. D. et al. Occurrence of amyotrophic lateral sclerosis among Gulf War veterans. Neurology 61, 742–749 (2003).
Barth, S. K., Kang, H. K., Bullman, T. A. & Wallin, M. T. Neurological mortality among U.S. veterans of the Persian Gulf War: 13-year follow-up. Am. J. Ind. Med. 52, 663–670 (2009).
Horner, R. D., Feussner, J. R. & Kasarskis, E. J. Neurological mortality among Gulf War veterans. Am. J. Ind. Med. 53, 548–549 (2010).
Department of Veterans Affairs. Presumption of service connection for amyotrophic lateral sclerosis. Final rule. Fed. Reg. 74, 57072–57074 (2009).
Board on Population Health and Public Health Practice. Amyotrophic lateral sclerosis in veterans: review of the scientific literature. Institute of Medicine of the National Academies [online], (2006).
Weisskopf, M. G. et al. Prospective study of military service and mortality from ALS. Neurology 64, 32–37 (2005).
Abhinav, K., Al-Chalabi, A., Hortobagyi, T. & Leigh, P. N. Electrical injury and amyotrophic lateral sclerosis: a systematic review of the literature. J. Neurol. Neurosurg. Psychiatry 78, 450–453 (2007).
Snow, J. The cholera near Golden-square, and at Deptford. The John Snow Archive and Research Companion [online], (1854).
Snow, J. On the mode of communication of cholera. The John Snow Archive and Research Companion [online], (1855).
Sabel, C. E. et al. Spatial clustering of amyotrophic lateral sclerosis in Finland at place of birth and place of death. Am. J. Epidemiol. 157, 898–905 (2003).
Veiga-Cabo, J., Almazán-Isla, J., Sendra-Gutiérrez, J. M. & de Pedro-Cuesta, J. Differential features of motor neuron disease mortality in Spain. Int. J. Epidemiol. 26, 1024–1032 (1997).
Scott, K. M. et al. Geographical clustering of amyotrophic lateral sclerosis in South-East England: a population study. Neuroepidemiology 32, 81–88 (2009).
Boumediene, F. et al. Contribution of geolocalisation to neuroepidemiological studies: incidence of ALS and environmental factors in Limousin, France. J. Neurol. Sci. 309, 115–122 (2011).
Turabelidze, G. et al. An epidemiologic investigation of amyotrophic lateral sclerosis in Jefferson County, Missouri, 1998–2002. Neurotoxicology 29, 81–86 (2008).
Uccelli, R. et al. Geographic distribution of amyotrophic lateral sclerosis through motor neuron disease mortality data. Eur. J. Epidemiol. 22, 781–790 (2007).
Migliaretti, G., Berchialla, P., Dalmasso, P., Cavallo, F. & Chio, A. Amyotrophic lateral sclerosis in Piedmont (Italy): a Bayesian spatial analysis of the incident cases. Amyotroph. Lateral Scler. Frontotemporal Degener. 14, 58–65 (2013).
Doi, Y. et al. Temporal trends and geographic clusters of mortality from amyotrophic lateral sclerosis in Japan, 1995–2004. J. Neurol. Sci. 298, 78–84 (2010).
Banack, S. A. & Cox, P. A. Biomagnification of cycad neurotoxins in flying foxes: implications for ALS–PDC in Guam. Neurology 61, 387–389 (2003).
Cox, P. A., Banack, S. A. & Murch, S. J. Biomagnification of cyanobacterial neurotoxins and neurodegenerative disease among the Chamorro people of Guam. Proc. Natl Acad. Sci. USA 100, 13380–13383 (2003).
Bienfang, P. K. et al. Prominent human health impacts from several marine microbes: history, ecology, and public health implications. Int. J. Microbiol. 2011, 152815 (2011).
Chiò, A. et al. Large proportion of amyotrophic lateral sclerosis cases in Sardinia due to a single founder mutation of the TARDBP gene. Arch. Neurol. 68, 594–598 (2011).
van Blitterswijk, M., DeJesus-Hernandez, M. & Rademakers, R. How do C9ORF72 repeat expansions cause amyotrophic lateral sclerosis and frontotemporal dementia: can we learn from other noncoding repeat expansion disorders? Curr. Opin. Neurol. 25, 689–700 (2012).
Byrne, S. et al. Rate of familial amyotrophic lateral sclerosis: a systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 82, 623–627 (2011).
Byrne, S., Jordan, I., Elamin, M. & Hardiman, O. Age at onset of amyotrophic lateral sclerosis is proportional to life expectancy. Amyotroph. Lateral Scler. Frontotemporal Degener. http://dx.doi.org/10.3109/21678421.2013.809122.
Gene report of major ALS genes in ALSoD. ALS Online Genetics Database [online], (2013).
Acknowledgements
We thank the Motor Neuron Disease Association of Great Britain and Northern Ireland, the ALS Association of America, the Irish Health Research Board, Research Motor Neuron, The Irish Motor Neuron Disease Association, The Irish Institute of Clinical Neuroscience, the Angel Fund, and the ALS Therapy Alliance for support. A. Al-Chalabi receives salary support from the National Institute for Health Research (NIHR) Dementia Biomedical Research Unit at South London and Maudsley NHS Foundation Trust and King's College London. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health. The work leading up to this publication was funded by the European Community's Health Seventh Framework Programme (FP7/2007–2013; grant agreement number 259867).
Author information
Authors and Affiliations
Contributions
Both authors researched the data for the article, discussed the content, wrote the article, and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
A. Al-Chalabi has consulted for Cytokinetics and Biogen Idec and receives royalties for the books The Brain (Oneworld Publications) and Genetics of Complex Human Diseases (Cold Spring Harbor Laboratory Press). O. Hardiman has consulted for Cytokinetics, Biogen Idec and Novartis.
Rights and permissions
About this article
Cite this article
Al-Chalabi, A., Hardiman, O. The epidemiology of ALS: a conspiracy of genes, environment and time. Nat Rev Neurol 9, 617–628 (2013). https://doi.org/10.1038/nrneurol.2013.203
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2013.203
This article is cited by
-
Generation of human iPSC-derived phrenic-like motor neurons to model respiratory motor neuron degeneration in ALS
Communications Biology (2024)
-
Desloratadine alleviates ALS-like pathology in hSOD1G93A mice via targeting 5HTR2A on activated spinal astrocytes
Acta Pharmacologica Sinica (2024)
-
Incidence of amyotrophic lateral sclerosis-associated genetic variants: a clinic-based study
Neurological Sciences (2024)
-
Genetics screening in an Italian cohort of patients with Amyotrophic Lateral Sclerosis: the importance of early testing and its implication
Journal of Neurology (2024)
-
Liquid–liquid phase separation in Alzheimer’s disease
Journal of Molecular Medicine (2024)