Abstract
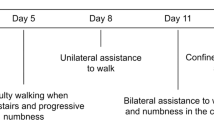
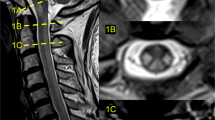
Longitudinal extensive transverse myelitis (LETM) is defined as a spinal cord lesion that extends over three or more vertebrae, as seen on MRI of the spine. The clinical presentation of a patient with LETM is often dramatic and can consist of paraparesis or tetraparesis, sensory disturbances, and gait, bladder, bowel and/or sexual dysfunction. LETM is a characteristic feature of neuromyelitis optica, but such spinal lesions can also occur in various other autoimmune and inflammatory diseases that involve the CNS—such as multiple sclerosis, sarcoidosis or Sjögren syndrome—or in infectious diseases with CNS involvement. Patients with a neoplastic disorder or traumatic spinal cord injury can also present with longitudinal spinal lesions. In this Review, the signs and symptoms that suggest various etiologies and differential diagnoses of LETM are described, and illustrated by educational case studies. The best therapeutic options for patients with each diagnosis are also discussed.
Key Points
-
Longitudinal extensive transverse myelitis (LETM) is a rare but disabling condition, defined as a lesion of the spinal cord that extends over three or more vertebrae on MRI
-
A thorough work-up of patients with suspected LETM is essential to determine the underlying cause of the lesion
-
Evaluation of a patient with LETM should include MRI of the spinal cord and the brain, investigation of cerebrospinal fluid, and detection of cause-specific markers in the blood
-
Therapeutic strategies for patients with LETM will depend on the underlying cause of the lesion, and appropriate treatment should be initiated as early as possible
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Transverse Myelitis Consortium Working Group. Proposed diagnostic criteria and nosology of acute transverse myelitis. Neurology 59, 499–505 (2002).
Jacob, A. & Weinshenker, B. G. An approach to the diagnosis of acute transverse myelitis. Semin. Neurol. 28, 105–120 (2008).
Frohman, E. M. & Wingerchuk, D. M. Clinical practice. Transverse myelitis. N. Engl. J. Med. 363, 564–572 (2010).
Lennon, V. A. et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 364, 2106–2112 (2004).
Wingerchuk, D. M., Lennon, V. A., Pittock, S. J., Lucchinetti, C. F. & Weinshenker, B. G. Revised diagnostic criteria for neuromyelitis optica. Neurology 66, 1485–1489 (2006).
Bou-Haidar, P., Peduto, A. J. & Karunaratne, N. Differential diagnosis of T2 hyperintense spinal cord lesions: part A. J. Med. Imaging Radiat. Oncol. 52, 535–543 (2008).
Bou-Haidar, P., Peduto, A. J. & Karunaratne, N. Differential diagnosis of T2 hyperintense spinal cord lesions: part B. J. Med. Imaging Radiat. Oncol. 53, 152–159 (2009).
Do-Dai, D. D., Brooks, M. K., Goldkamp, A., Erbay, S. & Bhadelia, R. A. Magnetic resonance imaging of intramedullary spinal cord lesions: a pictorial review. Curr. Probl. Diagn. Radiol. 39, 160–185 (2010).
Jarius, S. et al. Cerebrospinal fluid findings in aquaporin-4 antibody positive neuromyelitis optica: results from 211 lumbar punctures. J. Neurol. Sci. 306, 82–90 (2011).
Pittock, S. J. et al. Brain abnormalities in neuromyelitis optica. Arch. Neurol. 63, 390–396 (2006).
Pittock, S. J. et al. Neuromyelitis optica brain lesions localized at sites of high aquaporin 4 expression. Arch. Neurol. 63, 964–968 (2006).
Wingerchuk, D. M. Diagnosis and treatment of neuromyelitis optica. Neurologist 13, 2–11 (2007).
Cassinotto, C. et al. MRI of the spinal cord in neuromyelitis optica and recurrent longitudinal extensive myelitis. J. Neuroradiol. 36, 199–205 (2009).
Weinshenker, B. G. et al. Neuromyelitis optica IgG predicts relapse after longitudinally extensive transverse myelitis. Ann. Neurol. 59, 566–569 (2006).
Matiello, M. et al. NMO-IgG predicts the outcome of recurrent optic neuritis. Neurology 70, 2197–2200 (2008).
McKeon, A. et al. Coexistence of myasthenia gravis and serological markers of neurological autoimmunity in neuromyelitis optica. Muscle Nerve 39, 87–90 (2009).
Wandinger, K. P. et al. Autoantibodies against aquaporin-4 in patients with neuropsychiatric systemic lupus erythematosus and primary Sjögren's syndrome. Arthritis Rheum. 62, 1198–1200 (2010).
Vitali, C. et al. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 61, 554–558 (2002).
Delalande, S. et al. Neurologic manifestations in primary Sjögren syndrome: a study of 82 patients. Medicine (Baltimore) 83, 280–291 (2004).
Bertsias, G. K. et al. EULAR recommendations for the management of systemic lupus erythematosus with neuropsychiatric manifestations: report of a task force of the EULAR standing committee for clinical affairs. Ann. Rheum. Dis. 69, 2074–2082 (2010).
Luyendijk, J. et al. Neuropsychiatric systemic lupus erythematosus: lessons learned from magnetic resonance imaging. Arthritis Rheum. 63, 722–732 (2011).
Schulz, S. W. et al. Initial presentation of acute transverse myelitis in systemic lupus erythematosus: demographics, diagnosis, management and comparison to idiopathic cases. Rheumatol. Int. http://dx.doi.org/10.1007/s00296-011-2053-1.
Heinlein, A. C. & Gertner, E. Marked inflammation in catastrophic longitudinal myelitis associated with systemic lupus erythematosus. Lupus 16, 823–826 (2007).
Mornas, A. R., Thomas, T., Pallot, P. B., Chopin, F. & Raoux, D. Longitudinal myelitis in a patient with systemic lupus erythematosus. Joint Bone Spine 77, 181–183 (2010).
Costabel, U. Sarcoidosis: clinical update. Eur. Respir. J. Suppl. 32, 56s–68s (2001).
Stern, B. J., Krumholz, A., Johns, C., Scott, P. & Nissim, J. Sarcoidosis and its neurological manifestations. Arch. Neurol. 42, 909–917 (1985).
Bolat, S., Berding, G., Dengler, R., Stangel, M. & Trebst, C. Fluorodeoxyglucose positron emission tomography (FDG-PET) is useful in the diagnosis of neurosarcoidosis. J. Neurol. Sci. 287, 257–259 (2009).
Calamia, K. T. & Kaklamanis, P. G. Behçet's disease: recent advances in early diagnosis and effective treatment. Curr. Rheumatol. Rep. 10, 349–355 (2008).
[No authors listed] Criteria for diagnosis of Behçet's disease. International Study Group for Behçet's Disease. Lancet 335, 1078–1080 (1990).
Akman-Demir, G., Serdaroglu, P. & Tasci, B. Clinical patterns of neurological involvement in Behçet's disease: evaluation of 200 patients. The Neuro-Behçet Study Group. Brain 122, 2171–2182 (1999).
Fukae, J. et al. Subacute longitudinal myelitis associated with Behçet's disease. Intern. Med. 49, 343–347 (2010).
Yesilot, N. et al. Clinical characteristics and course of spinal cord involvement in Behçet's disease. Eur. J. Neurol. 14, 729–737 (2007).
Tartaglino, L. M. et al. Multiple sclerosis in the spinal cord: MR appearance and correlation with clinical parameters. Radiology 195, 725–732 (1995).
Qiu, W. et al. Longitudinally extensive myelopathy in Caucasians: a West Australian study of 26 cases from the Perth Demyelinating Diseases Database. J. Neurol. Neurosurg. Psychiatry 81, 209–212 (2010).
Jarius, S. et al. Polyspecific, antiviral immune response distinguishes multiple sclerosis and neuromyelitis optica. J. Neurol. Neurosurg. Psychiatry 79, 1134–1136 (2008).
Polman, C. H. et al. Diagnostic criteria for multiple sclerosis: revisions to the McDonald criteria. Ann. Neurol. 69, 292–302 (2011) (2010).
Banwell, B. et al. Neuromyelitis optica-IgG in childhood inflammatory demyelinating CNS disorders. Neurology 70, 344–352 (2008).
Young, N. P., Weinshenker, B. G. & Lucchinetti, C. F. Acute disseminated encephalomyelitis: current understanding and controversies. Semin. Neurol. 28, 84–94 (2008).
Berger, J. R. & Sabet, A. Infectious myelopathies. Semin. Neurol. 22, 133–142 (2002).
Sellner, J., Hemmer, B. & Muhlau, M. The clinical spectrum and immunobiology of parainfectious neuromyelitis optica (Devic) syndromes. J. Autoimmun. 34, 371–379 (2010).
Umehara, F. et al. Abnormalities of spinal magnetic resonance images implicate clinical variability in human T-cell lymphotropic virus type I-associated myelopathy. J. Neurovirol. 13, 260–267 (2007).
Delgado, S. R., Sheremata, W. A., Brown, A. D. & McCarthy, M. Human T-lymphotropic virus type I or II (HTLV-I/II) associated with recurrent longitudinally extensive transverse myelitis (LETM): two case reports. J. Neurovirol. 16, 249–253 (2010).
Meurs, L., Labeye, D., Declercq, I., Pieret, F. & Gille, M. Acute transverse myelitis as a main manifestation of early stage II neuroborreliosis in two patients. Eur. Neurol. 52, 186–188 (2004).
Chilver-Stainer, L., Fischer, U., Hauf, M., Fux, C. A. & Sturzenegger, M. Syphilitic myelitis: rare, nonspecific, but treatable. Neurology 72, 673–675 (2009).
Kikuchi, S., Shinpo, K., Niino, M. & Tashiro, K. Subacute syphilitic meningomyelitis with characteristic spinal MRI findings. J. Neurol. 250, 106–107 (2003).
Saleem, S., Belal, A. I. & el Ghandour, N. M. Spinal cord schistosomiasis: MR imaging appearance with surgical and pathologic correlation. AJNR Am. J. Neuroradiol. 26, 1646–1654 (2005).
Seo, H. S. et al. Nonenhancing intramedullary astrocytomas and other MR imaging features: a retrospective study and systematic review. AJNR Am. J. Neuroradiol. 31, 498–503 (2010).
Flanagan, E. P. et al. Paraneoplastic isolated myelopathy: clinical course and neuroimaging clues. Neurology 76, 2089–2095 (2011).
Ducray, F. et al. Devic's syndrome-like phenotype associated with thymoma and anti-CV2/CRMP5 antibodies. J. Neurol. Neurosurg. Psychiatry 78, 325–327 (2007).
Graus, F. et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J. Neurol. Neurosurg. Psychiatry 75, 1135–1140 (2004).
Pittock, S. J. & Lucchinetti, C. F. Inflammatory transverse myelitis: evolving concepts. Curr. Opin. Neurol. 19, 362–368 (2006).
Pittock, S. J. et al. Amphiphysin autoimmunity: paraneoplastic accompaniments. Ann. Neurol. 58, 96–107 (2005).
Pittock, S. J. et al. Glutamic acid decarboxylase autoimmunity with brainstem, extrapyramidal, and spinal cord dysfunction. Mayo Clin. Proc. 81, 1207–1214 (2006).
Titulaer, M. J. et al. Screening for tumours in paraneoplastic syndromes: report of an EFNS task force. Eur. J. Neurol. 18, 19-e3 (2011).
Krampla, W. et al. Spinal cord lesions in patients with neuromyelitis optica: a retrospective long-term MRI follow-up study. Eur. Radiol. 19, 2535–2543 (2009).
Mateen, F. J., Monrad, P. A., Hunderfund, A. N., Robertson, C. E. & Sorenson, E. J. Clinically suspected fibrocartilaginous embolism: clinical characteristics, treatments, and outcomes. Eur. J. Neurol. 18, 218–225 (2011).
Krings, T., Mull, M., Gilsbach, J. M. & Thron, A. Spinal vascular malformations. Eur. Radiol. 15, 267–278 (2005).
Mull, M. et al. Value and limitations of contrast-enhanced MR angiography in spinal arteriovenous malformations and dural arteriovenous fistulas. AJNR Am. J. Neuroradiol. 28, 1249–1258 (2007).
Misra, U. K., Kalita, J. & Das, A. Vitamin B12 deficiency neurological syndromes: a clinical, MRI and electrodiagnostic study. Electromyogr. Clin. Neurophysiol. 43, 57–64 (2003).
Goodman, B. P., Chong, B. W., Patel, A. C., Fletcher, G. P. & Smith, B. E. Copper deficiency myeloneuropathy resembling B12 deficiency: partial resolution of MR imaging findings with copper supplementation. AJNR Am. J. Neuroradiol. 27, 2112–2114 (2006).
Jaiser, S. R. & Winston, G. P. Copper deficiency myelopathy. J. Neurol. 257, 869–881 (2010).
McKeon, A. et al. Diagnosis of neuromyelitis spectrum disorders: comparative sensitivities and specificities of immunohistochemical and immunoprecipitation assays. Arch. Neurol. 66, 1134–1138 (2009).
Wang, K. C., Wang, S. J., Lee, C. L., Chen, S. Y. & Tsai, C. P. The rescue effect of plasma exchange for neuromyelitis optica. J. Clin. Neurosci. 18, 43–46 (2011).
Sellner, J. et al. EFNS guidelines on diagnosis and management of neuromyelitis optica. Eur. J. Neurol. 17, 1019–1032 (2010).
Trebst, C. et al. Diagnosis and treatment of neuromyelitis optica: consensus recommendations of the Neuromyelitis Optica Study Group. Nervenarzt 82, 768–777 (2011).
Taylor, J. K. & McMurray, R. V. Medical therapy for systemic lupus erythematosus. J. Miss. State Med. Assoc. 52, 39–43 (2011).
Jounieaux, F. et al. Infliximab treatment for chronic sarcoidosis—a case series. Rev. Mal. Respir. 27, 685–692 (2010).
Vargas, D. L. & Stern, B. J. Neurosarcoidosis: diagnosis and management. Semin. Respir. Crit. Care Med. 31, 419–427 (2010).
Chintamaneni, S., Patel, A. M., Pegram, S. B., Patel, H. & Roppelt, H. Dramatic response to infliximab in refractory neurosarcoidosis. Ann. Indian Acad. Neurol. 13, 207–210 (2010).
Kikuchi, H., Aramaki, K. & Hirohata, S. Effect of infliximab in progressive neuro-Behçet's syndrome. J. Neurol. Sci. 272, 99–105 (2008).
Wiendl, H. et al. Basic and escalating immunomodulatory treatments in multiple sclerosis: current therapeutic recommendations. J. Neurol. 255, 1449–1463 (2008).
Polman, C. H. et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N. Engl. J. Med. 354, 899–910 (2006).
Kappos, L. et al. A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N. Engl. J. Med. 362, 387–401 (2010).
Bernaerts, A. et al. Tuberculosis of the central nervous system: overview of neuroradiological findings. Eur. Radiol. 13, 1876–1890 (2003).
Acknowledgements
We thank V. Westphal for help with figure preparation. Written consent for publication was obtained from the patients described in the case studies or their responsible relatives.
Author information
Authors and Affiliations
Contributions
All authors contributed to researching data for the article and made substantial contributions to discussions of the content. C. Trebst, P. Raab, E. V. Voss and M. Stangel wrote the article, and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Trebst, C., Raab, P., Voss, E. et al. Longitudinal extensive transverse myelitis—it's not all neuromyelitis optica. Nat Rev Neurol 7, 688–698 (2011). https://doi.org/10.1038/nrneurol.2011.176
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2011.176
This article is cited by
-
Coexistence of longitudinally extensive transverse myelitis and diffuse midline glioma in the brainstem in an adolescent boy with acute flaccid paralysis
Child's Nervous System (2024)
-
Longitudinally extensive transverse myelitis with mycobacterium tuberculosis infection
Acta Neurologica Belgica (2023)
-
Update on the diagnosis and treatment of neuromyelits optica spectrum disorders (NMOSD) – revised recommendations of the Neuromyelitis Optica Study Group (NEMOS). Part I: Diagnosis and differential diagnosis
Journal of Neurology (2023)
-
Tuberculous myelitis: a prospective follow-up study
Neurological Sciences (2022)
-
Longitudinal extensive transverse myelitis following ChAdOx1 nCOV-19 vaccine: a case report
BMC Neurology (2021)