Abstract
Introduction
Tract-specific myelopathies with distinctive imaging features are uncommon and typically occur with metabolic or paraneoplastic syndromes. We report a unique case of tract-specific myelopathy with neurosyphilis.
Case Presentation
A 53-year-old male presented with a four-month history of flaccid quadriparesis, sensory loss, and bladder dysfunction. His MRIs revealed striking symmetric T2-weighted hyperintensities in the lateral corticospinal tracts and dorsal columns of the cervical spinal cord that extended rostrally into the pyramidal decussation and medial lemnisci of the medulla oblongata. Nerve conduction and needle electromyography studies excluded axonal or demyelinating lower motor neuron disorders. The patient reported previous untreated primary syphilis and was seropositive on the T.pallidum hemagglutination assay. Penicillin therapy resulted in substantial clinical improvement.
Discussion
Although syphilitic meningomyelitis is well-reported, our patient was unique because of the persistent flaccidity (possibly suggesting prolonged spinal shock) and striking tract-specific MRI patterns. These features are novel in syphilitic myelitis and suggest unknown mechanisms of tract-specific tropism and neuronal injury.
Conclusions
“Tract-specific” complete transverse myelopathy with persistent flaccid weakness and areflexia is a novel presentation of neurosyphilis. Early recognition and crystalline penicillin therapy can alleviate morbidity. Our report describes this patient’s findings and discusses the differential diagnoses of tract-specific myelopathies.
Similar content being viewed by others
Introduction
Syphilis, a sexually transmitted disease due to Treponema pallidum, is endemic in low-income countries and increasingly prevalent in other parts of the world [1]. Classic descriptions of neurosyphilis include aseptic meningitis, fulminant meningovascular syphilis, polycranial neuritis, tabes dorsalis, and general paresis [2]. Syphilitic myelopathy is relatively less common but is well-reported in the literature. The five classical syphilitic myelopathy patterns reported by Adams and Meritt in 1944 include syphilitic meningomyelitis, spinal vascular syphilis, syphilitic spinal pachymeningitis (comprising spinal cord gummata and hypertrophic pachymeningitis), syphilitic poliomyelitis, and extradural compression due to vertebral gummata or aortic aneurysms [3, 4]. However, several atypical variations may occur owing to delayed presentations and partial antibiotic therapy, leading to diagnostic challenges [5]. We report a case of syphilitic myelopathy with unique clinical and radiological features and a favourable response to penicillin therapy.
Case presentation
Mr. S, a 53-year-old man, presented with a progressive four-month history of flaccid quadriparesis, sensory loss, bladder dysfunction, and bulbar weakness. Over the first two weeks, he noticed numbness on the feet that ascended upwards to the chest. In the third week, he noticed weakness in the lower limbs and bladder hesitancy, leading to paraparesis and complete urinary retention. He continued to worsen over the next three months. The examination showed a flaccid tone with severe limb and truncal weakness (Medical Research Council grade 2 motor power in the upper limbs and 0 in the lower limbs). Weakness of the palatal arch muscles and faucial pillars (the tensor veli palatini, levator veli palatini, palatopharyngeus, palatoglossus, and the uvula) was marked, resulting in impaired swallowing. Light touch, pin-prick sensation, proprioception, and vibration sense were severely impaired in the upper limbs and absent in the lower limbs, with an abrupt reduction below the T4 spinous process indicating a sensory level. Voluntary anal contraction and deep anal pressure sensation were present, but deep tendon reflexes, plantar responses, and meningeal signs were absent. However, the optic fundus, pupillary reflexes, and other cranial nerves were normal.
Clinical localization and differential diagnosis
The sensory pattern with quadriparesis and bladder dysfunction suggested incomplete myelopathy with a neurological level above C2 (American Spinal Injury Association (ASIA) impairment scale rating C). However, the involvement of the 9th and 10th cranial nerves (bulbar weakness) suggested an extension to the medulla oblongata. Hence, we performed imaging of the brain and the spinal cord and somatosensory evoked potentials (SSEP) to assess dorsal column function. However, persistent flaccidity and hyporeflexia at four months suggested an additional lower motor neuron involvement, such as the anterior horn cells, radicles, or peripheral nerves. Hence, we also performed nerve conduction studies (NCS) with electromyography to clarify the localization.
Our preliminary etiological considerations included inflammatory (immune-mediated and paraneoplastic), metabolic, toxic, and infiltrative disorders. Disorders with a predilection for the spinal cord and brainstem included sarcoidosis, neuro-Bechet’s disease, multiple sclerosis, and infections such as tuberculosis or syphilis. In addition, we considered carcinomatous or lymphomatous arachnoiditis with skull base infiltration. Tumours of the cervico-medullary junction, though uncommon, were another possibility.
Investigation results
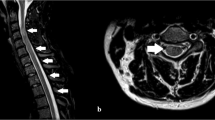
His MRI spine revealed striking symmetric T2-hyperintensities within the lateral corticospinal tracts and posterior columns of the cervical spinal cord. The MRI Brain showed an extension of the hyperintensities into the medulla oblongata, involving the pyramidal decussation and the medial lemnisci, respectively (Fig. 1). There was no enhancement to gadolinium or diffusion restriction. The regions corresponding to grey matter and other white matter pathways were relatively unaffected.
The tibial nerve SSEP showed bilaterally absent cortical responses, confirming the posterior column-medial lemniscus dysfunction. However, the NCS revealed a mild (≤20 percent) reduction in compound muscle action potential amplitudes with normal sensory nerve action potentials. The F-wave latencies were normal, suggesting intact motor root conductions, and needle electromyography showed no spontaneous activity suggestive of axonal denervation. Hence, lower motor neuron aetiologies for weakness were excluded.
Cerebrospinal fluid (CSF) analysis showed two lymphocytes, elevated proteins (62 mg/dl), and normal glucose levels (74 mg/dl). CSF cultures for bacterial, fungal, and tuberculous infections and a multiplex polymerase chain reaction for Herpes Simplex 1 and 2, Varicella-zoster virus, Epstein-Barr Virus, and Cytomegalovirus were negative. He was seronegative for HIV, HTLV, aquaporin-4 (AQP4), myelin oligodendrocyte glycoprotein (MOG), ANA, anti-SSA, anti-SSB, and paraneoplastic antibodies. A fluoro-deoxy-glucose positron emission tomography screening for systemic sarcoidosis, tuberculosis, and malignancies was also negative. Serum rapid plasma reagin (RPR) for syphilis was negative. However, the serum ELISA for Treponema pallidum, and the Treponema pallidum hemagglutination assay (TPHA) returned positive in high titres but was negative in the CSF. On historical review, the patient reported having noticed a painless penile nodule (suggesting a primary syphilitic chancre) after unprotected intercourse with a commercial sex worker over a decade before. His spouse was subsequently tested and found to be TPHA seropositive.
Treatment
In his first month of illness, he had been treated elsewhere for presumed transverse myelitis based on MRI, CSF, and clinical features with 1000 mg of methylprednisolone daily for five days, followed by an oral tapering steroid course, but he had not improved. We initiated empirical treatment for neurosyphilis with high-dose intravenous benzylpenicillin (4 million units every four-hourly for two weeks). Adjunctive steroids (125 mg of methylprednisolone once a week for 12 weeks) were additionally used. He tolerated the treatment without adverse effects and showed subsequent gradual recovery. One year later, he had regained full function of bulbar muscles, upper limbs, trunk, and bladder. He could walk unsupported but had residual lower limb weakness (an improvement from 5 to 2 on the Modified Rankin Scale). The partner was treated as well.
Discussion
Syphilis has earned a moniker as the “great masquerader” owing to its protean clinical manifestations. A resurgence was seen following the HIV epidemic, particularly among high-risk groups [5]. Any part of the neuraxis may be involved at any stage of the infection. Early stages of the disease are often asymptomatic and may occur on unnoticed body parts. This fact and widespread antibiotic use and HIV coinfection can make the diagnosis especially challenging [1].
The diagnosis of neurosyphilis requires a positive CSF serology or serum antibodies accompanied by CSF lymphocytic pleocytosis. However, in our patient, neurosyphilis was probable despite the negative CSF serology and absence of pleocytosis owing to the history of untreated primary syphilis, TPHA seropositivity in the patient and his partner, and the favourable response to penicillin therapy (while not to steroids alone). The absence of lymphocytosis may be attributable to having previously received steroids, which could attenuate the inflammatory response.
While meningovascular forms of neurosyphilis are relatively common, syphilitic myelitis is rare, usually occurring 1 – 30 years from the initial infection. Findings described with syphilitic myelopathy include the “candle-guttering” appearance – an abnormal enhancement to Gadolinium on the superficial spinal cord (like wax dripping down a candle), and the “flip-flop” sign [6, 7]. The latter describes a reversal of signal intensities of the pial surface with respect to the myelon parenchyma between T2-weighted and T1-gadolinium sequences. On T2-weighted sequences, the central part of the involved cord appears hyperintense, while the superficial pia mater appears hypointense. However, Gadolinium injection results in intense pial enhancement. This is believed to result from central spinal cord ischemia together with superficial inflammation causing disruption of the blood–spinal cord barrier [8, 9].
Meningeal inflammation, vasculitis, or compressive pathologies were responsible for the five spinal cord syndromes originally described by Adams and Merrit [4]. However, our patient’s images showed tract-specific tropism, which suggests a novel pathogenetic mechanism. Tract-specific pathology is already described in the context of syphilitic tabes dorsalis, but the symmetric corticospinal tract hyperintensity seen in our patient is novel [10]. A differential diagnosis for tract-specific myelopathy is listed in Table 1.
The persistent flaccid weakness and areflexia at four months is a second unexplained feature. Our patient’s normal motor nerve electromyography and F-wave studies exclude axonal or demyelinating lower motor neuron disorders. Therefore, our patient’s hypotonia must result from abnormal supraspinal mechanisms [11]. This may be analogous to the phenomenon of spinal shock seen after acute myelon injury. Further evidence corroborating these phenomena is necessary.
High-dose crystalline penicillin is the treatment of choice in neurosyphilis, but Doxycycline and Ceftriaxone may be used alternatively. Early treatment is reported to improve outcomes [12]. The adjunctive use of steroids is suggested to enhance recovery and prevent a Jarish-Herxheimer reaction [12].
Conclusions
Neurosyphilis is a great ‘masquerader,’ and atypical presentations are increasingly reported. Longitudinally extensive tract-specific myelopathy with persistent flaccid weakness and areflexia is a novel atypical presentation of neurosyphilis. Syphilitic antibody testing may be considered in endemic regions when unusual myelopathies are encountered since early identification and initiation of therapy with intravenous penicillin can prevent irreversible injury.
References
Hook EW. Syphilis. Lancet. 2017;389:1550–7. https://doi.org/10.1016/S0140-6736(16)32411-4.
Berger JR, Sabet A. Infectious myelopathies. Semin Neurol. 2002;22:133–42. https://doi.org/10.1055/s-2002-36536.
Merritt HH. The Early Clinical and Laboratory Manifestations of Syphilis of the Central Nervous System. N. Engl J Med. 1940;223:446–50.
Merritt HH, Adams RD, Solomon HC: Neurosyphilis. New York, Oxford University Press, 1946, pp. 72–73.
Bhai S, Lyons JL. Neurosyphilis Update: Atypical is the New Typical. Curr Infect Rep. 2015;17:481.
Yuan J-L, Wang W-X, Hu W-L. Clinical features of syphilitic myelitis with longitudinally extensive myelopathy on spinal magnetic resonance imaging. World J Clin Cases. 2019;7:1282–90. https://doi.org/10.12998/wjcc.v7.i11.1282.
Dong H, Liu Z, Duan Y, Li D, Qiu Z, Liu Y. et al. Syphilitic meningomyelitis misdiagnosed as spinal cord tumor: Case and review. J Spinal Cord Med Published Online First. 2019;44:789–93. https://doi.org/10.1080/10790268.2019.1658283.
Kikuchi S, Shinpo K, Niino M, Tashiro K. Subacute syphilitic meningomyelitis with characteristic spinal MRI findings. J Neurol. 2003;250:106–7. https://doi.org/10.1007/s00415-003-0921-7.
Tashiro K, Moriwaka F, Sudo K, Akino M, Abe H. Syphilitic myelitis with its magnetic resonance imaging (MRI) verification and successful treatment. Jpn J Psychiatry Neurol. 1987;41:269–71. https://doi.org/10.1111/j.1440-1819.1987.tb00411.x.
O'keefe E, Schwetye KE, Nazarian J, Perrin R, Schmidt RE, Bucelli R. A 66-Year-Old Woman with a Progressive, Longitudinally Extensive, Tract Specific, Myelopathy. Case Rep. Neurol Med. 2016;2016:e4125294. https://doi.org/10.1155/2016/4125294.
Hiersemenzel L-P, Curt A, Dietz V. From spinal shock to spasticity: Neuronal adaptations to a spinal cord injury. Neurology. 2000;54:1574–82. https://doi.org/10.1212/WNL.54.8.1574.
Wu Y, Wu W. Neurosyphilis presenting with myelitis-case series and literature review. J Infect Chemother. 2020;26:296–9. https://doi.org/10.1016/j.jiac.2019.09.007.
Shah S, Vazquez Do Campo R, Kumar N, McKeon A, Flanagan EP, Klein C. et al. Paraneoplastic Myeloneuropathies. Neurology. 2021;96:e632–9. https://doi.org/10.1212/WNL.0000000000011218.
Flanagan EP, McKeon A, Lennon VA, Kearns J, Weinshenker BG, Krecke KN, et al. Paraneoplastic isolated myelopathy: clinical course and neuroimaging clues. Neurology. 2011;76:2089–95.
Talbott JF, Narvid J, Chazen JL, Chin CT, Shah V. An Imaging-Based Approach to Spinal Cord Infection. Semin Ultrasound CT MRI. 2016;37:411–30. https://doi.org/10.1053/j.sult.2016.05.006.
Weidauer S, Nichtweiss M, Hattingen E. Differential diagnosis of white matter lesions: Nonvascular causes-Part II. Clin Neuroradiol. 2014;24:93–110. https://doi.org/10.1007/s00062-013-0267-1.
West TW, Hess C, Cree BAC. Acute transverse myelitis: demyelinating, inflammatory, and infectious myelopathies. Semin Neurol. 2012;32:97–113. https://doi.org/10.1055/s-0032-1322586.
Funding
This work was funded by internal resources of the Christian Medical College Vellore.
Author information
Authors and Affiliations
Contributions
AMT and RNB prepared the first draft, table, and subsequent revisions, while AMM, AS, and ATP provided clinical consultations and critical reviews. Laboratory and radiological analyses were performed by JAJ and PM, respectively. All authors reviewed and approved the final manuscript and the response to the reviewers.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
The next of kin (the patient’s son) obtained consent due to neurological impairment precluding writing.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miraclin T, A., Mani, A.M., Sivadasan, A. et al. Chronic flaccid quadriparesis from tract specific myelopathy in neurosyphilis. Spinal Cord Ser Cases 9, 4 (2023). https://doi.org/10.1038/s41394-023-00560-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-023-00560-y