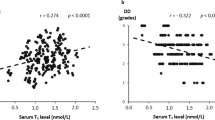
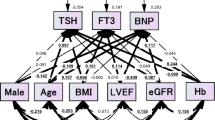
Abstract
The cardiorenal syndrome represents a final common pathway for renal and congestive heart failure and heralds a poor prognosis. Factors that link the failing heart and the failing kidneys—the so-called cardiorenal connectors—are, therefore, of clinical and therapeutic interest. Alterations in the levels and function of thyroid hormones that fit the spectrum of nonthyroidal illnesses could be considered to be cardiorenal connectors as both renal failure and heart failure progress with the development of nonthyroidal illness. In addition, circumstantial evidence suggests that nonthyroidal illness can induce deterioration in the function of the heart and the kidneys via multiple pathways. As a consequence, these reciprocal associations could result in a vicious cycle of deterioration that likely contributes to increased mortality. In this Review, we describe the evidence for a pathophysiological role of nonthyroidal illness in the cardiorenal syndrome. We also discuss the available data from studies that have investigated the efficacy of thyroid hormone replacement therapy in patients with renal failure and the rationale for interventional trials to examine the effects of normalization of the thyroid hormone profile in patients with renal failure and congestive heart failure.
Key Points
-
Cardiac disease and renal disease are frequently accompanied by nonthyroidal illness (that is, alterations in thyroid hormones in the absence of hypothalamic–pituitary–thyroidal disease)
-
General factors (such as inflammation and nutritional deficiencies) and pathophysiological factors that are specific to cardiac and renal disease contribute to the development of nonthyroidal illness
-
The presence of nonthyroidal illness is associated with an increased risk of cardiovascular death in patients with cardiac and/or renal disease
-
Nonthyroidal illness can result in deterioration of cardiovascular and renal function via several pathways
-
The genesis of nonthyroidal illness in cardiac and renal failure as well as its deleterious effects on both organ systems suggest that nonthyroidal illness acts as a cardiorenal connector in the cardiorenal syndrome
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ronco, C. Cardio-renal syndromes: from foggy bottoms to sunny hills. Heart Fail. Rev. 16, 509–517 (2011).
Smith, G. L. et al. Renal impairment and outcomes in heart failure: systematic review and meta-analysis. J. Am. Coll. Cardiol. 47, 1987–1996 (2006).
Bongartz, L. G. et al. The severe cardiorenal syndrome: 'Guyton revisited'. Eur. Heart J. 26, 11–17 (2005).
Hamilton, M. A. Prevalence and clinical implications of abnormal thyroid hormone metabolism in advanced heart failure. Ann. Thorac. Surg. 56, S48–S52 (1993).
Zoccali, C. et al. Low triiodothyronine: a new facet of inflammation in end-stage renal disease. J. Am. Soc. Nephrol. 16, 2789–2795 (2005).
Spratt, D. I. et al. Physiological effects of nonthyroidal illness syndrome in patients after cardiac surgery. Am. J. Physiol. Endocrinol. Metab. 293, E310–E315 (2007).
Iervasi, G. et al. Association between increased mortality and mild thyroid dysfunction in cardiac patients. Arch. Intern. Med. 167, 1526–1532 (2007).
Gerdes, A. M. & Iervasi, G. Thyroid replacement therapy and heart failure. Circulation 122, 385–393 (2010).
Zoccali, C. et al. Low triiodothyronine and survival in end-stage renal disease. Kidney Int. 70, 523–528 (2006).
Carrero, J. J. et al. Clinical and biochemical implications of low thyroid hormone levels (total and free forms) in euthyroid patients with chronic kidney disease. J. Intern. Med. 262, 690–701 (2007).
Meuwese, C. L. et al. Baseline levels and trimestral variation of triiodothyronine and thyroxine and their association with mortality in maintenance hemodialysis patients. Clin. J. Am. Soc. Nephrol. 7, 131–138 (2012).
Warner, M. H. & Beckett, G. J. Mechanisms behind the non-thyroidal illness syndrome: an update. J. Endocrinol. 205, 1–13 (2010).
Peeters, R. P. et al. Reduced activation and increased inactivation of thyroid hormone in tissues of critically ill patients. J. Clin. Endocrinol. Metab. 88, 3202–3211 (2003).
Docter, R., Krenning, E. P., de Jong, M. & Hennemann, G. The sick euthyroid syndrome: changes in thyroid hormone serum parameters and hormone metabolism. Clin. Endocrinol. (Oxf.) 39, 499–518 (1993).
Boelen, A., Kwakkel, J. & Fliers, E. Beyond low plasma T3: local thyroid hormone metabolism during inflammation and infection. Endocr. Rev. 32, 670–693 (2011).
Feingold, K. et al. Altered expression of nuclear hormone receptors and coactivators in mouse heart during the acute-phase response. Am. J. Physiol. Endocrinol. Metab. 286, E201–E207 (2004).
Feingold, K. R. et al. LPS decreases fatty acid oxidation and nuclear hormone receptors in the kidney. J. Lipid Res. 49, 2179–2187 (2008).
Squire, C. R. Methods for the investigation of thyroid function. Methods Mol. Biol. 324, 91–108 (2006).
Song, S. H. et al. The prevalence of low triiodothyronine according to the stage of chronic kidney disease in subjects with a normal thyroid-stimulating hormone. Nephrol. Dial. Transplant. 24, 1534–1538 (2009).
Kaptein, E. M. Thyroid hormone metabolism and thyroid diseases in chronic renal failure. Endocr. Rev. 17, 45–63 (1996).
Kaptein, E. M. et al. The thyroid in end-stage renal disease. Medicine (Baltimore) 67, 187–197 (1988).
Yonemura, K. et al. Low free thyroxine concentrations and deficient nocturnal surge of thyroid-stimulating hormone in haemodialysed patients compared with undialysed patients. Nephrol. Dial. Transplant. 15, 668–672 (2000).
Pagliacci, M. C. et al. Thyroid function tests in patients undergoing maintenance dialysis: characterization of the 'low-T4 syndrome' in subjects on regular hemodialysis and continuous ambulatory peritoneal dialysis. Nephron 46, 225–230 (1987).
Lo, J. C., Chertow, G. M., Go, A. S. & Hsu, C. Y. Increased prevalence of subclinical and clinical hypothyroidism in persons with chronic kidney disease. Kidney Int. 67, 1047–1052 (2005).
Ramirez, G., O'Neill, W. Jr, Jubiz, W. & Bloomer, H. A. Thyroid dysfunction in uremia: evidence for thyroid and hypophyseal abnormalities. Ann. Intern. Med. 84, 672–676 (1976).
Chen, W. L., Huang, W. S., Lin, Y. F. & Shieh, S. D. Changes in thyroid hormone metabolism in exertional heat stroke with or without acute renal failure. J. Clin. Endocrinol. Metab. 81, 625–629 (1996).
Iglesias, P. et al. Thyroid function tests in acute kidney injury. J. Nephrol. 26, 164–172 (2013).
Witzke, O. et al. Differential T4 degradation pathways in young patients with preterminal and terminal renal failure. Horm. Metab. Res. 39, 355–358 (2007).
Meuwese, C. L., Stenvinkel, P., Dekker, F. W. & Carrero, J. J. Monitoring of inflammation in patients on dialysis: forewarned is forearmed. Nat. Rev. Nephrol. 7, 166–176 (2011).
Abo-Zenah, H. A., Shoeb, S. A., Sabry, A. A. & Ismail, H. A. Relating circulating thyroid hormone concentrations to serum interleukins-6 and 10 in association with non-thyroidal illnesses including chronic renal insufficiency. BMC Endocr. Disord. 8, 1 (2008).
Zoccali, C. et al. Low triiodothyronine and cardiomyopathy in patients with end-stage renal disease. J. Hypertens. 24, 2039–2046 (2006).
Hermus, R. M. et al. Continuous infusion of interleukin-1β induces a nonthyroidal illness syndrome in the rat. Endocrinology 131, 2139–2146 (1992).
Carrero, J. J. et al. Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J. Ren. Nutr. 23, 77–90 (2013).
Meuwese, C. L., Carrero, J. J. & Stenvinkel, P. Recent insights in inflammation-associated wasting in patients with chronic kidney disease. Contrib. Nephrol. 171, 120–126 (2011).
Tomoda, F. et al. Effects of erythropoietin treatment on thyroid dysfunction in hemodialysis patients with renal anemia. Nephron 66, 307–311 (1994).
Sato, K. et al. Reversible primary hypothyroidism and elevated serum iodine level in patients with renal dysfunction. Acta Endocrinol. (Copenh.) 126, 253–259 (1992).
Wiederkehr, M. R., Kalogiros, J. & Krapf, R. Correction of metabolic acidosis improves thyroid and growth hormone axes in haemodialysis patients. Nephrol. Dial. Transplant. 19, 1190–1197 (2004).
Wilber, J. F. & Utiger, R. D. The effect of glucocorticoids on thyrotropin secretion. J. Clin. Invest. 48, 2096–2103 (1969).
Wiersinga, W. M. & Touber, J. L. The influence of β-adrenoceptor blocking agents on plasma thyroxine and triiodothyronine. J. Clin. Endocrinol. Metab. 45, 293–298 (1977).
Singh, N., Weisler, S. L. & Hershman, J. M. The acute effect of calcium carbonate on the intestinal absorption of levothyroxine. Thyroid 11, 967–971 (2001).
Napolitano, G. et al. Thyroid function and plasma selenium in chronic uremic patients on hemodialysis treatment. Biol. Trace Elem. Res. 55, 221–230 (1996).
Carrero, J. J. et al. Prevalence and clinical implications of testosterone deficiency in men with end-stage renal disease. Nephrol. Dial. Transplant. 26, 184–190 (2011).
Miyashita, K. et al. Regulation of rat liver type 1 iodothyronine deiodinase mRNA levels by testosterone. Mol. Cell Endocrinol. 115, 161–167 (1995).
Iervasi, G. et al. Low-T3 syndrome: a strong prognostic predictor of death in patients with heart disease. Circulation 107, 708–713 (2003).
Psirropoulos, D. et al. Heart failure accompanied by sick euthyroid syndrome and exercise training. Curr. Opin. Cardiol. 17, 266–270 (2002).
Opasich, C. et al. Sick euthyroid syndrome in patients with moderate-to-severe chronic heart failure. Eur. Heart J. 17, 1860–1866 (1996).
Ascheim, D. D. & Hryniewicz, K. Thyroid hormone metabolism in patients with congestive heart failure: the low triiodothyronine state. Thyroid 12, 511–515 (2002).
Hamilton, M. A., Stevenson, L. W., Luu, M. & Walden, J. A. Altered thyroid hormone metabolism in advanced heart failure. J. Am. Coll. Cardiol. 16, 91–95 (1990).
Pingitore, A. et al. Early activation of an altered thyroid hormone profile in asymptomatic or mildly symptomatic idiopathic left ventricular dysfunction. J. Card. Fail. 12, 520–526 (2006).
Pantos, C. et al. Thyroid hormone is a critical determinant of myocardial performance in patients with heart failure: potential therapeutic implications. Eur. J. Endocrinol. 157, 515–520 (2007).
Shanoudy, H. et al. Early manifestations of “sick euthyroid” syndrome in patients with compensated chronic heart failure. J. Card. Fail. 7, 146–152 (2001).
Iltumur, K. et al. Clinical investigation: thyroid function test abnormalities in cardiac arrest associated with acute coronary syndrome. Crit. Care 9, R416–R424 (2005).
Karga, H. et al. The role of cytokines and cortisol in the non-thyroidal illness syndrome following acute myocardial infarction. Eur. J. Endocrinol. 142, 236–242 (2000).
Wortsman, J., Premachandra, B. N., Chopra, I. J. & Murphy, J. E. Hypothyroxinemia in cardiac arrest. Arch. Intern. Med. 147, 245–248 (1987).
Friberg, L., Werner, S., Eggertsen, G. & Ahnve, S. Rapid down-regulation of thyroid hormones in acute myocardial infarction: is it cardioprotective in patients with angina? Arch. Intern. Med. 162, 1388–1394 (2002).
Ross, R. Atherosclerosis--an inflammatory disease. N. Engl. J. Med. 340, 115–126 (1999).
Torre-Amione, G. et al. Tumor necrosis factor-α and tumor necrosis factor receptors in the failing human heart. Circulation 93, 704–711 (1996).
Mancini, D. M. et al. Contribution of skeletal muscle atrophy to exercise intolerance and altered muscle metabolism in heart failure. Circulation 85, 1364–1373 (1992).
Adams, K. F. Jr et al. Prospective assessment of the occurrence of anemia in patients with heart failure: results from the Study of Anemia in a Heart Failure Population (STAMINA-HFP) Registry. Am. Heart J. 157, 926–932 (2009).
Eskes, S. A. & Wiersinga, W. M. Amiodarone and thyroid. Best Pract. Res. Clin. Endocrinol. Metab. 23, 735–751 (2009).
Stockigt, J. R. et al. Interaction of furosemide with serum thyroxine-binding sites: in vivo and in vitro studies and comparison with other inhibitors. J. Clin. Endocrinol. Metab. 60, 1025–1031 (1985).
Simonides, W. S. et al. Hypoxia-inducible factor induces local thyroid hormone inactivation during hypoxic-ischemic disease in rats. J. Clin. Invest. 118, 975–983 (2008).
Wassen, F. W. et al. Induction of thyroid hormone-degrading deiodinase in cardiac hypertrophy and failure. Endocrinology 143, 2812–2815 (2002).
Boelen, A. et al. Impaired bacterial clearance in type 3 deiodinase-deficient mice infected with Streptococcus pneumoniae. Endocrinology 150, 1984–1990 (2009).
Ueta, C. B. et al. Absence of myocardial thyroid hormone inactivating deiodinase results in restrictive cardiomyopathy in mice. Mol. Endocrinol. 26, 809–818 (2012).
Nomura, S. et al. Reduced peripheral conversion of thyroxine to triiodothyronine in patients with hepatic cirrhosis. J. Clin. Invest. 56, 643–652 (1975).
de Jager, D. J. et al. Cardiovascular and noncardiovascular mortality among patients starting dialysis. JAMA 302, 1782–1789 (2009).
Katzeff, H. L., Powell, S. R. & Ojamaa, K. Alterations in cardiac contractility and gene expression during low-T3 syndrome: prevention with T3 . Am. J. Physiol. 273, E951–E956 (1997).
Sabatino, L. et al. A study of iodothyronine 5′-monodeiodinase activities in normal and pathological tissues in man and their comparison with activities in rat tissues. Life Sci. 68, 191–202 (2000).
Bengel, F. M. et al. Effect of thyroid hormones on cardiac function, geometry, and oxidative metabolism assessed noninvasively by positron emission tomography and magnetic resonance imaging. J. Clin. Endocrinol. Metab. 85, 1822–1827 (2000).
Amidi, M. et al. Effect of the thyroid state on myocardial contractility and ventricular ejection rate in man. Circulation 38, 229–239 (1968).
Klein, I. & Ojamaa, K. Thyroid hormone and the cardiovascular system. N. Engl. J. Med. 344, 501–509 (2001).
Danzi, S. & Klein, I. Thyroid hormone-regulated cardiac gene expression and cardiovascular disease. Thyroid 12, 467–472 (2002).
Danzi, S., Ojamaa, K. & Klein, I. Triiodothyronine-mediated myosin heavy chain gene transcription in the heart. Am. J. Physiol. Heart Circ. Physiol. 284, H2255–H2262 (2003).
Bauab, R. C. et al. Low triiodothyronine (T3) or reverse triiodothyronine (rT3) syndrome modifies gene expression in rats with congestive heart failure. Endocr. Res. 31, 397–405 (2005).
Moalic, J. M. et al. β1 adrenergic receptor and Gα s mRNAs in rat heart as a function of mechanical load and thyroxine intoxication. Cardiovasc. Res. 27, 231–237 (1993).
Murray, J. F. & Kelly, J. J. Jr. The relation of thyroidal homone level to epinephrine response: a diagnostic test for hyperthyroidism. Ann. Intern. Med. 51, 309–321 (1959).
Ojamaa, K., Klein, I., Sabet, A. & Steinberg, S. F. Changes in adenylyl cyclase isoforms as a mechanism for thyroid hormone modulation of cardiac β-adrenergic receptor responsiveness. Metabolism 49, 275–279 (2000).
Jaroszynski, A. J. et al. Low-T3 syndrome and signal-averaged ECG in haemodialysed patients. Physiol. Res. 54, 521–526 (2005).
Fredlund, B. O. & Olsson, S. B. Long QT interval and ventricular tachycardia of “torsade de pointe” type in hypothyroidism. Acta Med. Scand. 213, 231–235 (1983).
Shojaie, M. & Eshraghian, A. Primary hypothyroidism presenting with torsades de pointes type tachycardia: a case report. Cases J. 1, 298 (2008).
Komar, N. N. & Gabrielsen, T. O. Arterial calcification in adult cretins. Am. J. Roentgenol. Radium. Ther. Nucl. Med. 101, 202–203 (1967).
Meuwese, C. L. et al. Nonthyroidal illness: a risk factor for coronary calcification and arterial stiffness in patients undergoing peritoneal dialysis. J. Intern. Med. http://dx.doi.org/10.1111/joim.12107.
Tatar, E. et al. Associations of triiodothyronine levels with carotid atherosclerosis and arterial stiffness in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 6, 2240–2246 (2011).
Tatar, E. et al. The association between thyroid hormones and arterial stiffness in peritoneal dialysis patients. Int. Urol. Nephrol. 44, 601–606 (2011).
Yilmaz, M. I. et al. Low triiodothyronine alters flow-mediated vasodilatation in advanced nondiabetic kidney disease. Am. J. Nephrol. 33, 25–32 (2010).
Malyszko, J., Malyszko, J. S., Pawlak, K. & Mysliwiec, M. Thyroid function, endothelium, and inflammation in hemodialyzed patients: possible relations? J. Ren. Nutr. 17, 30–37 (2007).
Ojamaa, K., Klemperer, J. D. & Klein, I. Acute effects of thyroid hormone on vascular smooth muscle. Thyroid 6, 505–512 (1996).
Mizuno, I. et al. Upregulation of the klotho gene expression by thyroid hormone and during adipose differentiation in 3T3-L1 adipocytes. Life Sci. 68, 2917–2923 (2001).
Sato, Y. et al. Thyroid hormone targets matrix Gla protein gene associated with vascular smooth muscle calcification. Circ. Res. 97, 550–557 (2005).
Bommer, C. et al. D-thyroxine reduces lipoprotein(a) serum concentration in dialysis patients. J. Am. Soc. Nephrol. 9, 90–96 (1998).
Passino, C. et al. Prognostic value of combined measurement of brain natriuretic peptide and triiodothyronine in heart failure. J. Card. Fail. 15, 35–40 (2009).
Montenegro, J. et al. Changes in renal function in primary hypothyroidism. Am. J. Kidney Dis. 27, 195–198 (1996).
Kreisman, S. H. & Hennessey, J. V. Consistent reversible elevations of serum creatinine levels in severe hypothyroidism. Arch. Intern. Med. 159, 79–82 (1999).
Shin, D. H. et al. Preservation of renal function by thyroid hormone replacement therapy in chronic kidney disease patients with subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 97, 2732–2740 (2012).
Shin, D. H. et al. Thyroid hormone replacement therapy attenuates the decline of renal function in chronic kidney disease patients with subclinical hypothyroidism. Thyroid 23, 654–661 (2013).
Kimmel, M., Braun, N. & Alscher, M. D. Influence of thyroid function on different kidney function tests. Kidney Blood Press. Res. 35, 9–17 (2012).
Villabona, C. et al. Blood volumes and renal function in overt and subclinical primary hypothyroidism. Am. J. Med. Sci. 318, 277–280 (1999).
Hlad, C. J. Jr & Bricker, N. S. Renal function and I131 clearance in hyperthyroidism and myxedema. J. Clin. Endocrinol. Metab. 14, 1539–1550 (1954).
Karanikas, G. et al. Isotopic renal function studies in severe hypothyroidism and after thyroid hormone replacement therapy. Am. J. Nephrol. 24, 41–45 (2004).
Gillum, D. M., Falk, S. A., Hammond, W. S. & Conger, J. D. Glomerular dynamics in the hypothyroid rat and the role of the renin-angiotensin system. Am. J. Physiol. 253, F170–F179 (1987).
Allon, M., Harrow, A., Pasque, C. B. & Rodriguez, M. Renal sodium and water handling in hypothyroid patients: the role of renal insufficiency. J. Am. Soc. Nephrol. 1, 205–210 (1990).
Vargas, F. et al. Vascular and renal function in experimental thyroid disorders. Eur. J. Endocrinol. 154, 197–212 (2006).
Moreno, J. M. et al. Role of endothelium-derived relaxing factors in the renal response to vasoactive agents in hypothyroid rats. Am. J. Physiol. Endocrinol. Metab. 285, E182–E188 (2003).
Miell, J. P. et al. Effects of hypothyroidism and hyperthyroidism on insulin-like growth factors (IGFs) and growth hormone and IGF-binding proteins. J. Clin. Endocrinol. Metab. 76, 950–955 (1993).
Davis, R. G., Madsen, K. M., Fregly, M. J. & Tisher, C. C. Kidney structure in hypothyroidism. Am. J. Pathol. 113, 41–49 (1983).
Salomon, M. I. et al. Renal lesions in hypothyroidism: a study based on kidney biopsies. Metabolism 16, 846–852 (1967).
Mariani, L. H. & Berns, J. S. The renal manifestations of thyroid disease. J. Am. Soc. Nephrol. 23, 22–26 (2012).
McDonough, A. A. et al. Thyroid hormone coordinately regulates Na+-K+-ATPase α and β-subunit mRNA levels in kidney. Am. J. Physiol. 254, C323–C329 (1988).
Cadnapaphornchai, M. A. et al. Urinary concentrating defect in hypothyroid rats: role of sodium, potassium, 2-chloride co-transporter, and aquaporins. J. Am. Soc. Nephrol. 14, 566–574 (2003).
Chou, K. M. et al. Correlation of clinical changes with regard to thyroxine replacement therapy in hypothyroid patients—focusing on the change of renal function. Kidney Blood Press. Res. 34, 365–372 (2011).
Reinhardt, W. et al. Triiodothyronine (T3) reflects renal graft function after renal transplantation. Clin. Endocrinol. (Oxf.) 46, 563–569 (1997).
Junik, R. et al. Function, structure, and volume of thyroid gland following allogenic kidney transplantation. Transplant. Proc. 35, 2224–2226 (2003).
Thomas, M. C., Mathew, T. H. & Russ, G. R. Changes in thyroxine requirements in patients with hypothyroidism undergoing renal transplantation. Am. J. Kidney Dis. 39, 354–357 (2002).
Rosolowska-Huszcz, D., Kozlowska, L. & Rydzewski, A. Influence of low protein diet on nonthyroidal illness syndrome in chronic renal failure. Endocrine 27, 283–288 (2005).
Utas, C. et al. Improvement of thyroid hormone profile and thyrotrophin (TSH) surge alterations in hemodialysis patients on erythropoietin treatment. Clin. Nephrol. 55, 471–476 (2001).
Disthabanchong, S. & Treeruttanawanich, A. Oral sodium bicarbonate improves thyroid function in predialysis chronic kidney disease. Am. J. Nephrol. 32, 549–556 (2010).
Sutter, P. M. et al. Beneficial effect of thyroxin in the treatment of ischemic acute renal failure. Pediatr. Nephrol. 2, 1–7 (1988).
Siegel, N. J. et al. Beneficial effect of thyroxin on recovery from toxic acute renal failure. Kidney Int. 25, 906–911 (1984).
Michael, U. F., Logan, J. L. & Meeks, L. A. The beneficial effects of thyroxine on nephrotoxic acute renal failure in the rat. J. Am. Soc. Nephrol. 1, 1236–1240 (1991).
Acker, C. G. et al. A trial of thyroxine in acute renal failure. Kidney Int. 57, 293–298 (2000).
De Groot, L. J. Non-thyroidal illness syndrome is a manifestation of hypothalamic-pituitary dysfunction, and in view of current evidence, should be treated with appropriate replacement therapies. Crit. Care Clin. 22, 57–86 (2006).
Harris, A. R., Fang, S. L., Vagenakis, A. G. & Braverman, L. E. Effect of starvation, nutriment replacement, and hypothyroidism on in vitro hepatic T4 to T3 conversion in the rat. Metabolism 27, 1680–1690 (1978).
Escobar-Morreale, H. F., del Rey, F. E., Obregon, M. J. & de Escobar, G. M. Only the combined treatment with thyroxine and triiodothyronine ensures euthyroidism in all tissues of the thyroidectomized rat. Endocrinology 137, 2490–2502 (1996).
Straub, E. Effects of L-thyroxine in acute renal failure. Res. Exp. Med. (Berl.) 168, 81–87 (1976).
Adamovich, K., Baranyai, Z., Guignard, J. P. & Sulyok, E. Effect of thyroxine administration on renal functions in newborn infants with perinatal asphyxia. Acta Paediatr. Hung. 32, 219–233 (1992).
Lim, V. S., Flanigan, M. J., Zavala, D. C. & Freeman, R. M. Protective adaptation of low serum triiodothyronine in patients with chronic renal failure. Kidney Int. 28, 541–549 (1985).
Lim, V. S., Tsalikian, E. & Flanigan, M. J. Augmentation of protein degradation by L-triiodothyronine in uremia. Metabolism 38, 1210–1215 (1989).
Carter, J. N., Eastman, C. J., Corcoran, J. M. & Lazarus, L. Effects of triiodothyronine administration in patients with chronic renal failure. Aust. N. Z. J. Med. 7, 612–616 (1977).
Acker, C. G. et al. Thyroid hormone in the treatment of post-transplant acute tubular necrosis (ATN). Am. J. Transplant. 2, 57–61 (2002).
Celikyurt, U. et al. Effect of cardiac resynchronization therapy on thyroid function. Clin. Cardiol. 34, 703–705 (2011).
D'Aloia, A. et al. Effect of short-term infusive dobutamine therapy on thyroid hormone profile and hemodynamic parameters in patients with acute worsening heart failure and low-triiodothyronine syndrome. J. Investig. Med. 60, 907–910 (2012).
Hamilton, M. A. et al. Safety and hemodynamic effects of intravenous triiodothyronine in advanced congestive heart failure. Am. J. Cardiol. 81, 443–447 (1998).
Pingitore, A. et al. Acute effects of triiodothyronine (T3) replacement therapy in patients with chronic heart failure and low-T3 syndrome: a randomized, placebo-controlled study. J. Clin. Endocrinol. Metab. 93, 1351–1358 (2008).
Iervasi, G. et al. in Proceedings of the 2nd International Congress on Heart Disease—New Trends in Research, Diagnosis and Treatment (ed. Kimchi, A.) 549–553 (Medimond Medical Publications, 2001).
Moruzzi, P. et al. Usefulness of L-thyroxine to improve cardiac and exercise performance in idiopathic dilated cardiomyopathy. Am. J. Cardiol. 73, 374–378 (1994).
Moruzzi, P., Doria, E. & Agostoni, P. G. Medium-term effectiveness of L-thyroxine treatment in idiopathic dilated cardiomyopathy. Am. J. Med. 101, 461–467 (1996).
Malik, F. S. et al. Intravenous thyroid hormone supplementation in heart failure with cardiogenic shock. J. Card. Fail. 5, 31–37 (1999).
Goldman, S. et al. DITPA (3, 5-diiodothyropropionic acid), a thyroid hormone analog to treat heart failure: phase II trial veterans affairs cooperative study. Circulation 119, 3093–3100 (2009).
Morkin, E. et al. Pilot studies on the use of 3, 5-diiodothyropropionic acid, a thyroid hormone analog, in the treatment of congestive heart failure. Cardiology 97, 218–225 (2002).
Talukder, M. A. et al. Detrimental effects of thyroid hormone analog DITPA in the mouse heart: increased mortality with in vivo acute myocardial ischemia-reperfusion. Am. J. Physiol. Heart Circ. Physiol. 300, H702–H711 (2011).
The coronary drug project research group. The coronary drug project. Findings leading to further modifications of its protocol with respect to dextrothyroxine. JAMA 220, 996–1008 (1972).
[No authors listed] Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. The Expert Panel. Arch. Intern. Med. 148, 36–69 (1988).
Kaptein, E. M., Sanchez, A., Beale, E. & Chan, L. S. Clinical review: thyroid hormone therapy for postoperative nonthyroidal illnesses: a systematic review and synthesis. J. Clin. Endocrinol. Metab. 95, 4526–4534 (2010).
Biondi, B. et al. Endothelial-mediated coronary flow reserve in patients with mild thyroid hormone deficiency. Eur. J. Endocrinol. 161, 323–329 (2009).
Sawin, C. T. et al. Low serum thyrotropin concentrations as a risk factor for atrial fibrillation in older persons. N. Engl. J. Med. 331, 1249–1252 (1994).
Rosner, M. H. & Bolton, W. K. The mortality risk associated with higher hemoglobin: is the therapy to blame? Kidney Int. 74, 695–697 (2008).
Baxter, J. D. & Webb, P. Thyroid hormone mimetics: potential applications in atherosclerosis, obesity and type 2 diabetes. Nat. Rev. Drug Discov. 8, 308–320 (2009).
Van den Berghe, G. et al. Reactivation of pituitary hormone release and metabolic improvement by infusion of growth hormone-releasing peptide and thyrotropin-releasing hormone in patients with protracted critical illness. J. Clin. Endocrinol. Metab. 84, 1311–1323 (1999).
Chopra, I. J., Solomon, D. H., Hepner, G. W. & Morgenstein, A. A. Misleadingly low free thyroxine index and usefulness of reverse triiodothyronine measurement in nonthyroidal illnesses. Ann. Intern. Med. 90, 905–912 (1979).
Surks, M. I. et al. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA 291, 228–238 (2004).
Monzani, F. et al. Effect of levothyroxine replacement on lipid profile and intima-media thickness in subclinical hypothyroidism: a double-blind, placebo-controlled study. J. Clin. Endocrinol. Metab. 89, 2099–2106 (2004).
Acknowledgements
The authors wish to express their gratitude to J. W. M. Plevier (information specialist at The Walaeus Library, Leiden University Medical Center, Netherlands) for her assistance in the literature search. The authors' work and some of the discussed studies are partially supported by the Westman's foundation, by the Center for Gender Medicine at Karolinska Institutet and by the Swedish Medical Research Council (J. J. Carrero).
Author information
Authors and Affiliations
Contributions
C. L. Meuwese and J. J. Carrero researched the data for the article and wrote the manuscript. All authors made a substantial contribution to discussions of the content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Search criteria for Tables 1 and 2 (DOC 32 kb)
Rights and permissions
About this article
Cite this article
Meuwese, C., Dekkers, O., Stenvinkel, P. et al. Nonthyroidal illness and the cardiorenal syndrome. Nat Rev Nephrol 9, 599–609 (2013). https://doi.org/10.1038/nrneph.2013.170
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2013.170
This article is cited by
-
The combination of nonthyroidal illness syndrome and renal dysfunction further increases mortality risk in patients with acute myocardial infarction: a prospective cohort study
BMC Cardiovascular Disorders (2019)
-
Thyroid dysfunction and kidney disease: An update
Reviews in Endocrine and Metabolic Disorders (2017)