Key Points
-
The voltage-gated sodium channel NaV1.7 is expressed in most nociceptive dorsal root ganglion (DRG) neurons and is present peripherally within free nerve endings in the epidermis and centrally within superficial lamina of the dorsal horn in the spinal cord. NaV1.7 is also expressed in sympathetic neurons and olfactory sensory neurons.
-
NaV1.7 produces a rapidly activating and inactivating but slowly repriming current that is sensitive to nanomolar concentrations of tetrodotoxin, and a robust ramp current in response to small, slow depolarizations, suggesting that NaV1.7 acts as a threshold channel.
-
The ability of NaV1.7 to boost subthreshold stimuli, together with its presence at nerve endings in the epidermis and in the dorsal horn, increases the probability of neurons reaching their threshold for firing action potentials, and may facilitate neurotransmitter release onto second-order neurons.
-
Genetic and functional studies have linked gain-of-function mutations of SCN9A with pain disorders such as inherited erythromelalgia and paroxysmal pain disorder, whereas loss-of-function mutations are linked to complete insensitivity to pain that may be accompanied by anosmia. Other gain-of-function variants in NaV1.7 are risk factors for painful small-fibre neuropathy.
-
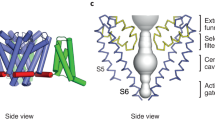
Atomic structural modelling of NaV1.7 based on the crystal structure of other ion channels has identified the activation gate of the NaV1.7, and together with thermodynamic analysis of 'seed' mutations that increase drug responsiveness, may allow the identification of channel variants that enhance responsiveness to pharmacotherapies.
-
The central role of NaV1.7 in pain signalling and the lack of cognitive, motor and cardiac deficits in patients with NaV1.7-linked complete insensitivity to pain has made this channel a major focus for the development of small-molecule blockers and other biological approaches for the treatment of pain.
Abstract
The voltage-gated sodium channel NaV1.7 is preferentially expressed in peripheral somatic and visceral sensory neurons, olfactory sensory neurons and sympathetic ganglion neurons. NaV1.7 accumulates at nerve fibre endings and amplifies small subthreshold depolarizations, poising it to act as a threshold channel that regulates excitability. Genetic and functional studies have added to the evidence that NaV1.7 is a major contributor to pain signalling in humans, and homology modelling based on crystal structures of ion channels suggests an atomic-level structural basis for the altered gating of mutant NaV1.7 that causes pain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Catterall, W. A., Goldin, A. L. & Waxman, S. G. International Union of Pharmacology. XLVII. Nomenclature and structure–function relationships of voltage-gated sodium channels. Pharmacol. Rev. 57, 397–409 (2005). A general review on the sodium channel subfamily of voltage-gated ion channels.
Catterall, W. A. From ionic currents to molecular mechanisms: the structure and function of voltage-gated sodium channels. Neuron 26, 13–25 (2000).
Payandeh, J., Scheuer, T., Zheng, N. & Catterall, W. A. The crystal structure of a voltage-gated sodium channel. Nature 475, 353–358 (2011). The first description of a high-resolution crystal structure of a homotetrameric bacterial voltage-gated sodium channel.
Catterall, W. A. Signaling complexes of voltage-gated sodium and calcium channels. Neurosci. Lett. 486, 107–116 (2010).
Dib-Hajj, S. D. & Waxman, S. G. Isoform-specific and pan-channel partners regulate trafficking and plasma membrane stability; and alter sodium channel gating properties. Neurosci. Lett. 486, 84–91 (2010).
Leterrier, C., Brachet, A., Fache, M. P. & Dargent, B. Voltage-gated sodium channel organization in neurons: protein interactions and trafficking pathways. Neurosci. Lett. 486, 92–100 (2010).
Patino, G. A. & Isom, L. L. Electrophysiology and beyond: multiple roles of Na+ channel β subunits in development and disease. Neurosci. Lett. 486, 53–59 (2010).
Cummins, T. R. et al. NaV1.3 sodium channels: rapid repriming and slow closed-state inactivation display quantitative differences after expression in a mammalian cell line and in spinal sensory neurons. J. Neurosci. 21, 5952–5961 (2001). This study documents the effect of cell background on the biophysical properties of voltage-gated sodium channels and highlights the need to study these channels in their native cell types.
Choi, J. S., Dib-Hajj, S. D. & Waxman, S. Differential slow inactivation and use-dependent inhibition of NaV1.8 channels contribute to distinct firing properties in IB4+ and IB4− DRG neurons. J. Neurophysiol. 97, 1258–1265 (2007).
Rush, A. M. et al. A single sodium channel mutation produces hyper- or hypoexcitability in different types of neurons. Proc. Natl Acad. Sci. USA 103, 8245–8250 (2006). This study demonstrates that the distinct cellular responses of DRG neurons and sympathetic ganglion neurons to expression of mutant Na V 1.7 channel depends on the presence or absence of another sodium channel, Na V 1.8.
Choi, J. S. et al. Alternative splicing may contribute to time-dependent manifestation of inherited erythromelalgia. Brain 133, 1823–1835 (2010).
Dib-Hajj, S. D. et al. Transfection of rat or mouse neurons by biolistics or electroporation. Nature Protoc. 4, 1118–1126 (2009).
Dib-Hajj, S. D., Cummins, T. R., Black, J. A. & Waxman, S. G. Sodium channels in normal and pathological pain. Annu. Rev. Neurosci. 33, 325–347 (2010).
Ahn, H. S. et al. NaV1.7 is the predominant sodium channel in rodent olfactory sensory neurons. Mol. Pain 7, 32 (2011).
Weiss, J. et al. Loss-of-function mutations in sodium channel NaV1.7 cause anosmia. Nature 472, 186–190 (2011).
Muroi, Y. et al. Selective silencing of NaV1.7 decreases excitability and conduction in vagal sensory neurons. J. Physiol. 589, 5663–5676 (2011).
Smith, E. S. et al. The molecular basis of acid insensitivity in the African naked mole-rat. Science 334, 1557–1560 (2011).
Toledo-Aral, J. J. et al. Identification of PN1, a predominant voltage-dependent sodium channel expressed principally in peripheral neurons. Proc. Natl Acad. Sci. USA 94, 1527–1532 (1997). The first study to report the major cellular distribution of Na V 1.7.
Sage, D. et al. NaV1.7 and NaV1.3 are the only tetrodotoxin-sensitive sodium channels expressed by the adult guinea pig enteric nervous system. J. Comp. Neurol. 504, 363–378 (2007).
Kwong, K. et al. Voltage-gated sodium channels in nociceptive versus non-nociceptive nodose vagal sensory neurons innervating guinea pig lungs. J. Physiol. 586, 1321–1336 (2008).
Holm, A. N. et al. Sodium current in human jejunal circular smooth muscle cells. Gastroenterology 122, 178–187 (2002).
Jo, T. et al. Voltage-gated sodium channel expressed in cultured human smooth muscle cells: involvement of SCN9A. FEBS Lett. 567, 339–343 (2004).
Saleh, S., Yeung, S. Y., Prestwich, S., Pucovsky, V. & Greenwood, I. A. Electrophysiological and molecular identification of voltage-gated sodium channels in murine vascular myocytes. J. Physiol. 568, 155–169 (2005).
Djouhri, L. et al. Sensory and electrophysiological properties of guinea-pig sensory neurones expressing NaV1.7 (PN1) Na+ channel α-subunit protein. J. Physiol. 546, 565–576 (2003). This study demonstrates the presence of Na V 1.7 in functionally identified nociceptors.
Felts, P. A., Yokoyama, S., Dib-Hajj, S., Black, J. A. & Waxman, S. G. Sodium channel α-subunit mRNAs I, II, III, NaG, Na6 and HNE (PN1) — different expression patterns in developing rat nervous system. Mol. Brain Res. 45, 71–82 (1997).
Diss, J. K. et al. A potential novel marker for human prostate cancer: voltage-gated sodium channel expression in vivo. Prostate Cancer Prostatic Dis. 8, 266–273 (2005).
Fraser, S. P. et al. Voltage-gated sodium channel expression and potentiation of human breast cancer metastasis. Clin. Cancer Res. 11, 5381–5389 (2005).
Hoffman, J. F., Dodson, A., Wickrema, A. & Dib-Hajj, S. D. Tetrodotoxin-sensitive Na+ channels and muscarinic and purinergic receptors identified in human erythroid progenitor cells and red blood cell ghosts. Proc. Natl Acad. Sci. USA 101, 12370–12374 (2004).
Kis-Toth, K. et al. Voltage-gated sodium channel NaV1.7 maintains the membrane potential and regulates the activation and chemokine-induced migration of a monocyte-derived dendritic cell subset. J. Immunol. 187, 1273–1280 (2011).
Djouhri, L. et al. The TTX-resistant sodium channel NaV1.8 (SNS/PN3): expression and correlation with membrane properties in rat nociceptive primary afferent neurons. J. Physiol. 550, 739–752 (2003).
Persson, A. K., Gasser, A., Black, J. A. & Waxman, S. G. NaV1.7 accumulates and co-localizes with phosphorylated ERK1/2 within transected axons in early experimental neuromas. Exp. Neurol. 230, 273–279 (2011).
Black, J. A., Frezel, N., Dib-Hajj, S. D. & Waxman, S. G. Expression of NaV1.7 in DRG neurons extends from peripheral terminals in the skin to central preterminal branches and terminals in the dorsal horn. Mol. Pain 8, 82 (2012).
Cummins, T. R., Howe, J. R. & Waxman, S. G. Slow closed-state inactivation: a novel mechanism underlying ramp currents in cells expressing the hNE/PN1 sodium channel. J. Neurosci. 18, 9607–9619 (1998). This study shows that Na V 1.7 can produce a robust ramp current, suggesting that Na V 1.7 can amplify subthreshold depolarizations and act as a threshold channel.
Klugbauer, N., Lacinova, L., Flockerzi, V. & Hofmann, F. Structure and functional expression of a new member of the tetrodotoxin-sensitive voltage-activated sodium channel family from human neuroendocrine cells. EMBO J. 14, 1084–1090 (1995). The first report of the isolation and characterization of Na V 1.7 as a TTX-sensitive sodium channel.
Herzog, R. I., Cummins, T. R., Ghassemi, F., Dib-Hajj, S. D. & Waxman, S. G. Distinct repriming and closed-state inactivation kinetics of NaV1.6 and NaV1.7 sodium channels in mouse spinal sensory neurons. J. Physiol. 551, 741–750 (2003).
Rush, A. M., Cummins, T. R. & Waxman, S. G. Multiple sodium channels and their roles in electrogenesis within dorsal root ganglion neurons. J. Physiol. 579, 1–14 (2007).
Dib-Hajj, S. D., Cummins, T. R., Black, J. A. & Waxman, S. G. From genes to pain: NaV1.7 and human pain disorders. Trends Neurosci. 30, 555–563 (2007).
Jarecki, B. W., Piekarz, A. D., Jackson, J. O., 2nd & Cummins, T. R. Human voltage-gated sodium channel mutations that cause inherited neuronal and muscle channelopathies increase resurgent sodium currents. J. Clin. Invest. 120, 369–378 (2010).
Faber, C. G. et al. Gain of function NaV1.7 mutations in idiopathic small fiber neuropathy. Ann. Neurol. 71, 26–39 (2012). This study was the first to show that patients with idiopathic SFN can harbour Na V 1.7 variants; it also shows that these variants cause hyperexcitability of DRG neurons.
Raman, I. M. & Bean, B. P. Resurgent sodium current and action potential formation in dissociated cerebellar Purkinje neurons. J. Neurosci. 17, 4517–4526 (1997). This study documents a state of open channel block, which permits the passing of a current upon hyperpolarization of the cell membrane to negative potentials immediately following a strong depolarizing pulse that fully activates and inactivates the channel.
Raman, I. M., Sprunger, L. K., Meisler, M. H. & Bean, B. P. Altered subthreshold sodium currents and disrupted firing patterns in Purkinje neurons of Scn8a mutant mice. Neuron 19, 881–891 (1997).
Cummins, T. R., Dib-Hajj, S. D., Herzog, R. I. & Waxman, S. G. NaV1.6 channels generate resurgent sodium currents in spinal sensory neurons. FEBS Lett. 579, 2166–2170 (2005).
Nassar, M. A. et al. Nociceptor-specific gene deletion reveals a major role for NaV1.7 (PN1) in acute and inflammatory pain. Proc. Natl Acad. Sci. (USA) 101, 12706–12711 (2004). The first report showing that knockout of Na V 1.7 in DRG neurons impairs acute and inflammatory pain.
Goldberg, Y. et al. Loss-of-function mutations in the NaV1.7 gene underlie congenital indifference to pain in multiple human populations. Clin. Genet. 71, 311–319 (2007).
Nilsen, K. B. et al. Two novel SCN9A mutations causing insensitivity to pain. Pain 143, 155–158 (2009).
Undem, B. J. & Carr, M. J. Targeting primary afferent nerves for novel antitussive therapy. Chest 137, 177–184 (2010).
Schild, J. H. & Kunze, D. L. Experimental and modeling study of Na+ current heterogeneity in rat nodose neurons and its impact on neuronal discharge. J. Neurophysiol. 78, 3198–3209 (1997).
Farrag, K. J., Costa, S. K. & Docherty, R. J. Differential sensitivity to tetrodotoxin and lack of effect of prostaglandin E2 on the pharmacology and physiology of propagated action potentials. Br. J. Pharmacol. 135, 1449–1456 (2002).
Park, T. J. et al. Selective inflammatory pain insensitivity in the African naked mole-rat (Heterocephalus glaber). PLoS Biol. 6, e13 (2008).
Singh, N. A. et al. A role of SCN9A in human epilepsies, as a cause of febrile seizures and as a potential modifier of Dravet syndrome. PLoS Genet. 5, e1000649 (2009).
Drenth, J. P. & Waxman, S. G. Mutations in sodium-channel gene SCN9A cause a spectrum of human genetic pain disorders. J. Clin. Invest. 117, 3603–3609 (2007).
Devor, M. Sodium channels and mechanisms of neuropathic pain. J. Pain 7, S3–S12 (2006).
Lyu, Y. S., Park, S. K., Chung, K. & Chung, J. M. Low dose of tetrodotoxin reduces neuropathic pain behaviors in an animal model. Brain Res. 871, 98–103 (2000).
Black, J. A., Nikolajsen, L., Kroner, K., Jensen, T. S. & Waxman, S. G. Multiple sodium channel isoforms and mitogen-activated protein kinases are present in painful human neuromas. Ann. Neurol. 64, 644–653 (2008). This study demonstrates the presence of sodium channels Na V 1.3, Na V 1.7 and Na V 1.8, and activated MAPK1, MAPK3 and MAPK12 within blind axon terminals of painful human neuromas.
Stamboulian, S. et al. ERK1/2 mitogen-activated protein kinase phosphorylates sodium channel NaV1.7 and alters its gating properties. J. Neurosci. 30, 1637–1647 (2010).
Black, J. A., Liu, S., Tanaka, M., Cummins, T. R. & Waxman, S. G. Changes in the expression of tetrodotoxin-sensitive sodium channels within dorsal root ganglia neurons in inflammatory pain. Pain 108, 237–247 (2004).
Gould, H. J. et al. Ibuprofen blocks changes in NaV1.7 and 1.8 sodium channels associated with complete freund's adjuvant-induced inflammation in rat. J. Pain 5, 270–280 (2004).
Chattopadhyay, M., Mata, M. & Fink, D. J. Continuous δ-opioid receptor activation reduces neuronal voltage-gated sodium channel (NaV1.7) levels through activation of protein kinase C in painful diabetic neuropathy. J. Neurosci. 28, 6652–6658 (2008).
Chattopadhyay, M., Mata, M. & Fink, D. J. Vector-mediated release of GABA attenuates pain-related behaviors and reduces NaV1.7 in DRG neurons. Eur. J. Pain 15, 913–920 (2011).
Yeomans, D. C. et al. Decrease in inflammatory hyperalgesia by Herpes vector-mediated knockdown of NaV1.7 sodium channels in primary afferents. Hum. Gene Ther. 16, 271–277 (2005).
Chattopadhyay, M., Zhou, Z., Hao, S., Mata, M. & Fink, D. J. Reduction of voltage gated sodium channel protein in DRG by vector mediated miRNA reduces pain in rats with painful diabetic neuropathy. Mol. Pain 8, 17 (2012).
Minett, M. S. et al. Distinct NaV1.7-dependent pain sensations require different sets of sensory and sympathetic neurons. Nature Commun. 3, 791 (2012). This study suggests that knockout of Na V 1.7 in neurons from DRG and sympathetic ganglia is needed to attenuate neuropathic pain.
Shields, S. D. et al. Sodium channel NaV1.7 is essential for lowering heat pain threshold after burn injury. J. Neurosci. 32, 10819–10832 (2012).
Yang, Y. et al. Mutations in SCN9A, encoding a sodium channel α subunit, in patients with primary erythermalgia. J. Med. Genet. 41, 171–174 (2004). This report identifies gain-of-function mutations in SCN9A in patients with IEM.
Fertleman, C. R. et al. SCN9A mutations in paroxysmal extreme pain disorder: allelic variants underlie distinct channel defects and phenotypes. Neuron 52, 767–774 (2006). This study identifies and characterizes gain-of-function mutations in SCN9A in patients with PEPD.
Cox, J. J. et al. An. SCN9A channelopathy causes congenital inability to experience pain. Nature 444, 894–898 (2006). This study identifies and characterizes loss-of-function mutations in SCN9A that underlie CIP.
Sheets, P. L., Jackson Ii, J. O., Waxman, S. G., Dib-Hajj, S. & Cummins, T. R. A. NaV1.7 channel mutation associated with hereditary erythromelalgia contributes to neuronal hyperexcitability and displays reduced lidocaine sensitivity. J. Physiol. 581, 1019–1031 (2007).
Cheng, X. et al. Deletion mutation of sodium channel NaV1.7 in inherited erythromelalgia: enhanced slow inactivation modulates dorsal root ganglion neuron hyperexcitability. Brain 134, 1972–1986 (2011).
Fertleman, C. R. et al. Paroxysmal extreme pain disorder (previously familial rectal pain syndrome). Neurology 69, 586–595 (2007).
Choi, J. S. et al. Paroxysmal extreme pain disorder: a molecular lesion of peripheral neurons. Nature Rev. Neurol. 7, 51–55 (2011).
Estacion, M. et al. NaV1.7 gain-of-function mutations as a continuum: A1632E displays physiological changes associated with erythromelalgia and paroxysmal extreme pain disorder mutations and produces symptoms of both disorders. J. Neurosci. 28, 11079–11088 (2008).
Dib-Hajj, S. D. et al. Paroxysmal extreme pain disorder M1627K mutation in human NaV1.7 renders DRG neurons hyperexcitable. Mol. Pain 4, 37 (2008).
Cheng, X. et al. Mutations at opposite ends of the DIII/S4–S5 linker of sodium channel NaV1.7 produce distinct pain disorders. Mol. Pain 6, 24 (2010).
Vasylyev, D. V. & Waxman, S. G. Membrane properties and electrogenesis in the distal axons of small dorsal root ganglion neurons in vitro. J. Neurophysiol. 108, 729–740 (2012).
Han, C. et al. Early- and late-onset inherited erythromelalgia: genotype–phenotype correlation. Brain 132, 1711–1722 (2009).
Harty, T. P. et al. NaV1.7 mutant A863P in erythromelalgia: effects of altered activation and steady-state inactivation on excitability of nociceptive dorsal root ganglion neurons. J. Neurosci. 26, 12566–12575 (2006).
Ahmad, S. et al. A stop codon mutation in SCN9A causes lack of pain sensation. Hum. Mol. Genet. 16, 2114–2121 (2007).
Kurban, M., Wajid, M., Shimomura, Y. & Christiano, A. M. A nonsense mutation in the SCN9A gene in congenital insensitivity to pain. Dermatology 221, 179–183 (2010).
Staud, R. et al. Two novel mutations of SCN9A (NaV1.7) are associated with partial congenital insensitivity to pain. Eur. J. Pain 15, 223–230 (2011).
Reich, D. E. & Lander, E. S. On the allelic spectrum of human disease. Trends Genet. 17, 502–510 (2001).
Estacion, M. et al. A sodium channel gene SCN9A polymorphism that increases nociceptor excitability. Ann. Neurol. 66, 862–866 (2009). This report identifies and characterizes a common variant of SCN9A that is associated with pain.
Reimann, F. et al. Pain perception is altered by a nucleotide polymorphism in SCN9A. Proc. Natl Acad. Sci. USA 107, 5148–5153 (2010).
Estacion, M. et al. Intra- and interfamily phenotypic diversity in pain syndromes associated with a gain-of-function variant of NaV1.7. Mol. Pain 7, 92 (2011).
Choi, J. S. & Waxman, S. G. Physiological interactions between NaV1.7 and NaV1.8 sodium channels: a computer simulation study. J. Neurophysiol. 106, 3173–3184 (2011).
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Jiang, Y. et al. The open pore conformation of potassium channels. Nature 417, 523–526 (2002).
Long, S. B., Campbell, E. B. & Mackinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309, 897–903 (2005).
Ren, D. et al. A prokaryotic voltage-gated sodium channel. Science 294, 2372–2375 (2001).
Charalambous, K. & Wallace, B. A. NaChBac: the long lost sodium channel ancestor. Biochemistry 50, 6742–6752 (2011).
Lampert, A., Dib-Hajj, S. D., Tyrrell, L. & Waxman, S. G. Size matters: erythromelalgia mutation S241T in NaV1.7 alters channel gating. J. Biol. Chem. 281, 36029–36035 (2006).
Tsujino, A. et al. Myasthenic syndrome caused by mutation of the SCN4A sodium channel. Proc. Natl Acad. Sci. USA 100, 7377–7382 (2003).
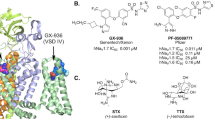
Lampert, A. et al. A pore-blocking hydrophobic motif at the cytoplasmic aperture of the closed-state NaV1.7 channel is disrupted by the erythromelalgia-associated F1449V mutation. J. Biol. Chem. 283, 24118–24127 (2008). An atomic structural modelling of Na V 1.7 based on the potassium channel KcsA crystal structure identifies a putative activation gate.
Burley, S. K. & Petsko, G. A. Aromatic–aromatic interaction: a mechanism of protein structure stabilization. Science 229, 23–28 (1985).
Dib-Hajj, S. D. et al. Gain-of-function mutation in NaV1.7 in familial erythromelalgia induces bursting of sensory neurons. Brain 128, 1847–1854 (2005). The first demonstration that a gain-of-function familial mutation in SCN9A renders DRG neurons hyperexcitable, thus providing the pathophysiological basis for pain in these patients.
Rojas, A., Wu, J., Wang, R. & Jiang, C. Gating of the ATP-sensitive K+ channel by a pore-lining phenylalanine residue. Biochim. Biophys. Acta 1768, 39–51 (2007).
Han, C. et al. Functional profiles of SCN9A variants in dorsal root ganglion neurons and superior cervical ganglion neurons correlate with autonomic symptoms in small fibre neuropathy. Brain 135, 2613–2628 (2012).
Akopian, A. N., Sivilotti, L. & Wood, J. N. A tetrodotoxin-resistant voltage-gated sodium channel expressed by sensory neurons. Nature 379, 257–262 (1996).
Akopian, A. N. et al. The tetrodotoxin-resistant sodium channel SNS has a specialized function in pain pathways. Nature Neurosci. 2, 541–548 (1999). Together with reference 97, these studies were the first to identify and characterize Na V 1.8 from DRG neurons and demonstrates a role for this channel in pain.
Sangameswaran, L. et al. Structure and function of a novel voltage-gated, tetrodoxtoxin-resistant sodium channel specific to sensory neurons. J. Biol. Chem. 271, 5953–5956 (1996).
England, S. & de Groot, M. J. Subtype-selective targeting of voltage-gated sodium channels. Br. J. Pharmacol. 158, 1413–1425 (2009).
Choi, J. S. et al. Mexiletine-responsive erythromelalgia due to a new NaV1.7 mutation showing use-dependent current fall-off. Exp. Neurol. 216, 383–389 (2009).
Fischer, T. Z. et al. A novel NaV1.7 mutation producing carbamazepine-responsive erythromelalgia. Ann. Neurol. 65, 733–741 (2009). This study identifies the SCN9A mutation V400M in patients who responded to treatment with carbamazepine, and demonstrates that this mutation increases responsiveness to carbamazepine without altering the affinity of the channel to the drug.
Yang, Y. et al. Structural modelling and mutant cycle analysis predict pharmacoresponsiveness of a NaV1.7 mutant channel. Nature Commun. 3, 1186 (2012). Using V400M as a 'seed' SCN9A mutation, this atomic structural modelling and thermodynamic coupling analysis predicts and then confirms that a second SCN9A mutation, S241T, is responsive to carbamazepine.
Williams, B. S. et al. Characterization of a new class of potent inhibitors of the voltage-gated sodium channel NaV1.7. Biochemistry 46, 14693–14703 (2007).
London, C. et al. Imidazopyridines: a novel class of hNaV1.7 channel blockers. Bioorg. Med. Chem. Lett. 18, 1696–1701 (2008).
Bregman, H. et al. Identification of a potent, state-dependent inhibitor of NaV1.7 with oral efficacy in the formalin model of persistent pain. J. Med. Chem. 54, 4427–4445 (2011).
Chowdhury, S. et al. Discovery of XEN907, a spirooxindole blocker of NaV1.7 for the treatment of pain. Bioorg. Med. Chem. Lett. 21, 3676–3681 (2011).
Chapman, M. L. et al. Characterization of a novel subtype-selective inhibitor of human NaV1.7 voltage-dependent sodium channels (PT 418). IASP 14th World Congress on Pain [online], (2012).
Rice, A. S. & Hill, R. G. New treatments for neuropathic pain. Annu. Rev. Med. 57, 535–551 (2006).
Dworkin, R. H. et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 132, 237–251 (2007).
Sindrup, S. H. & Jensen, T. S. Are sodium channel blockers useless in peripheral neuropathic pain? Pain 128, 6–7 (2007).
Gerner, P. & Strichartz, G. R. Sensory and motor complications of local anesthetics. Muscle Nerve 37, 421–425 (2008).
Beyreuther, B., Callizot, N. & Stohr, T. Antinociceptive efficacy of lacosamide in a rat model for painful diabetic neuropathy. Eur. J. Pharmacol. 539, 64–70 (2006).
Beyreuther, B. K. et al. Antinociceptive efficacy of lacosamide in rat models for tumor- and chemotherapy-induced cancer pain. Eur. J. Pharmacol. 565, 98–104 (2007).
Hao, J. X., Stohr, T., Selve, N., Wiesenfeld-Hallin, Z. & Xu, X. J. Lacosamide, a new anti-epileptic, alleviates neuropathic pain-like behaviors in rat models of spinal cord or trigeminal nerve injury. Eur. J. Pharmacol. 553, 135–140 (2006).
Stohr, T. et al. Lacosamide, a novel anti-convulsant drug, shows efficacy with a wide safety margin in rodent models for epilepsy. Epilepsy Res. 74, 147–154 (2007).
Doty, P., Rudd, G. D., Stoehr, T. & Thomas, D. Lacosamide. Neurotherapeutics 4, 145–148 (2007).
Rauck, R. L., Shaibani, A., Biton, V., Simpson, J. & Koch, B. Lacosamide in painful diabetic peripheral neuropathy: a phase 2 double-blind placebo-controlled study. Clin. J. Pain 23, 150–158 (2007).
Sheets, P. L., Heers, C., Stoehr, T. & Cummins, T. R. Differential block of sensory neuronal voltage-gated sodium channels by lacosamide [(2R)-2-(acetylamino)-N-benzyl-3-methoxypropanamide], lidocaine, and carbamazepine. J. Pharmacol. Exp. Ther. 326, 89–99 (2008).
Xu, G. Y. & Zhao, Z. Q. Change in excitability and phenotype of substance P and its receptor in cat Aβ sensory neurons following peripheral inflammation. Brain Res. 923, 112–119 (2001).
Dworkin, R. H. et al. Recommendations for the pharmacological management of neuropathic pain: an overview and literature update. Mayo Clin. Proc. 85, S3–S14 (2010).
Wilson, M. J. et al. μ-Conotoxins that differentially block sodium channels NaV1.1 through 1.8 identify those responsible for action potentials in sciatic nerve. Proc. Natl Acad. Sci. USA 108, 10302–10307 (2011).
Lewis, R. J., Dutertre, S., Vetter, I. & Christie, M. J. Conus venom peptide pharmacology. Pharmacol. Rev. 64, 259–298 (2012).
Dib-Hajj, S. D. et al. Voltage-gated sodium channels in pain states: role in pathophysiology and targets for treatment. Brain Res. Rev. 60, 65–83 (2009).
Middleton, R. E. et al. Two tarantula peptides inhibit activation of multiple sodium channels. Biochemistry 41, 14734–14747 (2002).
Smith, J. J., Cummins, T. R., Alphy, S. & Blumenthal, K. M. Molecular interactions of the gating modifier toxin ProTx-II with NaV 1.5: implied existence of a novel toxin binding site coupled to activation. J. Biol. Chem. 282, 12687–12697 (2007).
Peng, K., Shu, Q., Liu, Z. & Liang, S. Function and solution structure of huwentoxin-IV, a potent neuronal tetrodotoxin (TTX)-sensitive sodium channel antagonist from Chinese bird spider Selenocosmia huwena. J. Biol. Chem. 277, 47564–47571 (2002).
Xiao, Y. et al. Tarantula huwentoxin-IV inhibits neuronal sodium channels by binding to receptor site 4 and trapping the domain II voltage sensor in the closed configuration. J. Biol. Chem. 283, 27300–27313 (2008).
McArthur, J. R. et al. Interactions of key charged residues contributing to selective block of neuronal sodium channels by μ-conotoxin KIIIA. Mol. Pharmacol. 80, 573–584 (2011).
Clark, R. J., Akcan, M., Kaas, Q., Daly, N. L. & Craik, D. J. Cyclization of conotoxins to improve their biopharmaceutical properties. Toxicon 59, 446–455 (2012).
Fink, D. J. et al. Gene therapy for pain: results of a phase I clinical trial. Ann. Neurol. 70, 207–212 (2012).
Samad, O. A. et al. Virus-mediated shRNA knockdown of NaV1.3 in rat dorsal root ganglion attenuates nerve injury-induced neuropathic pain. Mol. Ther. 21 Aug 2012 (doi:10.1038/mt.2012.169).
Firestein, S. How the olfactory system makes sense of scents. Nature 413, 211–218 (2001).
Kaupp, U. B. Olfactory signalling in vertebrates and insects: differences and commonalities. Nature Rev. Neurosci. 11, 188–200 (2010).
Rajendra, S., Lynch, J. W. & Barry, P. H. An analysis of Na+ currents in rat olfactory receptor neurons. Pflugers Arch. 420, 342–346 (1992).
Cummins, T. R., Dib-Hajj, S. D. & Waxman, S. G. Electrophysiological properties of mutant NaV1.7 sodium channels in a painful inherited neuropathy. J. Neurosci. 24, 8232–8236 (2004). The first demonstration that mutations in SCN9A from patients with IEM manifest gain-of-function attributes.
Blair, N. T. & Bean, B. P. Roles of tetrodotoxin (TTX)-sensitive Na+ current, TTX-resistant Na+ current, and Ca2+ current in the action potentials of nociceptive sensory neurons. J. Neurosci. 22, 10277–10290 (2002).
Cummins, T. R. & Waxman, S. G. Downregulation of tetrodotoxin-resistant sodium currents and upregulation of a rapidly repriming tetrodotoxin-sensitive sodium current in small spinal sensory neurons after nerve injury. J. Neurosci. 17, 3503–3514 (1997).
Elliott, A. A. & Elliott, J. R. Characterization of TTX-sensitive and TTX-resistant sodium currents in small cells from adult rat dorsal root ganglia. J. Physiol. 463, 39–56 (1993).
Cheng, X., Dib-Hajj, S. D., Tyrrell, L. & Waxman, S. G. Mutation I136V alters electrophysiological properties of the NaV1.7 channel in a family with onset of erythromelalgia in the second decade. Mol. Pain 4, 1 (2008).
Han, C. et al. NaV1.7-related small fiber neuropathy: impaired slow-inactivation and DRG neuron hyperexcitability. Neurology 78, 1635–1643 (2012).
Acknowledgements
The authors thank the members of their group for valuable discussions. Work in the authors' laboratory is supported in part by grants from the Rehabilitation Research and Development Service and Medical Research Service, US Department of Veterans Affairs, and from the Erythromelalgia Association. The Center for Neuroscience and Regeneration Research is a collaboration between the Paralyzed Veterans of America and Yale University, Connecticut, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Pfizer has provided a research grant, administered through Yale University, Connecticut, USA, in support of research funding for S.G.W. and S.D.H.
Related links
Related links
DATABASES
Online Mendelian Inheritance in Man (OMIM)
FURTHER INFORMATION
Center for Neuroscience and Regeneration Research, Yale School of Medicine
Glossary
- Neuropathic pain
-
Pain resulting from lesions or diseases of the somatosensory system.
- Nociceptors
-
Pain-sensing or damage-sensing neurons.
- Repriming
-
Refolding of a channel after opening and inactivating to restore a closed, but available channel. The channel is refractory to additional stimulations during repriming.
- Ramp current
-
Inward current due to transient channel activation in response to the small, slow depolarization of cell membranes.
- Neuroma
-
A collection of demyelinated and dysmyelinated axon sprouts and connective tissue that result from abortive regeneration of transected axons.
- Inherited sodium channelopathies
-
Pathologies linked to mutations or functional variants in sodium channels that can be transmitted to progeny.
- Fast inactivation
-
Inactivation (within milliseconds) of sodium channels occurs by blocking the cytoplasmic vestibule of the channel by a tetrapeptide (inactivation gate) within the linker joining domains III and IV, and terminating the inflow of sodium ions.
- Haploinsufficiency
-
When one functional copy of a gene is not enough to prevent a deficit.
- Atomic structural modelling
-
Construction of a model of a folded protein based on the atom coordinates of a related member of the family whose high-resolution crystal structure is determined and additional constraints derived from studies of distant members of the superfamily.
Rights and permissions
About this article
Cite this article
Dib-Hajj, S., Yang, Y., Black, J. et al. The NaV1.7 sodium channel: from molecule to man. Nat Rev Neurosci 14, 49–62 (2013). https://doi.org/10.1038/nrn3404
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn3404
This article is cited by
-
Neural Mechanisms Underlying the Coughing Reflex
Neuroscience Bulletin (2023)
-
Peripheral temperature dysregulation associated with functionally altered NaV1.8 channels
Pflügers Archiv - European Journal of Physiology (2023)
-
TRP channels: a journey towards a molecular understanding of pain
Nature Reviews Neuroscience (2022)
-
Ion channel long non-coding RNAs in neuropathic pain
Pflügers Archiv - European Journal of Physiology (2022)
-
Inhibition of Nav1.7 channel by a novel blocker QLS-81 for alleviation of neuropathic pain
Acta Pharmacologica Sinica (2021)