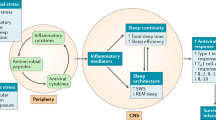
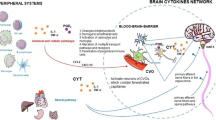
Key Points
-
Serotonin promotes wakefulness but is necessary for sleep.
-
Cytokines, such as interleukin 1 (IL-1) and tumor necrosis factor, are involved in regulating physiological non-rapid eye movement (NREM) sleep.
-
IL-1 contributes to the regulation of NREM sleep in part through interactions with serotonin.
-
Interactions between IL-1 and serotonin are amplified during infection, and sleep is altered.
-
During infection, NREM sleep is fragmented and REM sleep is suppressed.
-
Changes induced in sleep architecture by infectious agents support the generation of fever, and fever imparts survival value.
Abstract
Good sleep is necessary for physical and mental health. For example, sleep loss impairs immune function, and sleep is altered during infection. Immune signalling molecules are present in the healthy brain, where they interact with neurochemical systems to contribute to the regulation of normal sleep. Animal studies have shown that interactions between immune signalling molecules (such as the cytokine interleukin 1) and brain neurochemical systems (such as the serotonin system) are amplified during infection, indicating that these interactions might underlie the changes in sleep that occur during infection. Why should the immune system cause us to sleep differently when we are sick? We propose that the alterations in sleep architecture during infection are exquisitely tailored to support the generation of fever, which in turn imparts survival value.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tononi, G. & Cirelli, C. Sleep and synaptic homeostasis: a hypothesis. Brain Res. Bull. 62, 143–150 (2003).
Krueger, J. M. et al. Sleep as a fundamental property of neuronal assemblies. Nature Rev. Neurosci. 9, 910–919 (2008).
Dantzer, R., O'Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nature Rev. Neurosci. 9, 46–56 (2008).
Centers for Disease Control and Prevention. Percentage of adults who reported an average of ≤6 hours of sleep per 24-hour period, by sex and age group—United States 1985 and 2004. MMWR Morb. Mortal. Wkly Rep. 54, 933 (2005).
Spiegel, K., Sheridan, J. F. & Van Cauter, E. Effect of sleep deprivation on response to immunization. JAMA 288, 1471–1472 (2002).
Lange, T., Perras, B., Fehm, H. L. & Born, J. Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom. Med. 65, 831–835 (2003).
Hasler, G. et al. The association between short sleep duration and obesity in young adults: a 13-year prospective study. Sleep 27, 661–666 (2004).
Taheri, S., Lin, L., Austin, D., Young, T. & Mignot, E. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 1, e62 (2004).
Spiegel, K., Tasali, E., Penev, P. & Van Cauter, E. Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann. Intern. Med. 141, 846–850 (2004).
Gottlieb, D. J. et al. Association of sleep time with diabetes mellitus and impaired glucose tolerance. Arch. Intern. Med. 165, 863–867 (2005).
Ayas, N. T. et al. A prospective study of sleep duration and coronary heart disease in women. Arch. Intern. Med. 163, 205–209 (2003).
Dale, R. C. et al. Encephalitis lethargica syndrome: 20 new cases and evidence of basal ganglia autoimmunity. Brain 127, 21–33 (2004).
Norman, S. E., Chediak, A. D., Kiel, M. & Cohn, M. A. Sleep disturbances in HIV infected homosexual men. AIDS 4, 775–781 (1990).
Buguet, A. et al. The duality of sleeping sickness: focusing on sleep. Sleep Med. Rev. 5, 139–153 (2001).
Lundkvist, G. B., Kristensson, K. & Bentivoglio, M. Why trypanosomes cause sleeping sickness. Physiology (Bethesda) 19, 198–206 (2004).
Drake, C. L. et al. Effects of an experimentally induced rhinovirus cold on sleep, performance, and daytime alertness. Physiol. Behav. 71, 75–81 (2000).
Bettis, R. et al. Impact of influenza treatment with oseltamivir on health, sleep and daily activities of otherwise healthy adults and adolescents. Clin. Drug Investig. 26, 329–340 (2006).
Opp, M. R. Cytokines and sleep. Sleep Med. Rev. 9, 355–364 (2005).
Opp, M., Born, J. & Irwin, M. in Psychoneuroimmunology (ed. Ader, R.) 579–618 (Burlington, Massachusetts, Elsevier Academic Press, 2007).
Krueger, J. M., Obal, F. J., Fang, J., Kubota, T. & Taishi, P. The role of cytokines in physiological sleep regulation. Ann. NY Acad. Sci. 933, 211–221 (2001).
Eriksson, C., Nobel, S., Winblad, B. & Schultzberg, M. Expression of interleukin 1α and β, and interleukin 1 receptor antagonist mRNA in the rat central nervous system after peripheral administration of lipopolysaccharides. Cytokine 12, 423–431 (2000).
Allan, S. M. & Rothwell, N. J. Cytokines and acute neurodegeneration. Nature Rev. Neurosci. 2, 734–744 (2001).
Garden, G. A. & Moller, T. Microglia biology in health and disease. J. Neuroimmune Pharmacol. 1, 127–137 (2006).
Breder, C. D., Dinarello, C. A. & Saper, C. B. Interleukin-1 immunoreactive innervation of the human hypothalamus. Science 240, 321–324 (1988). The first demonstration that neurons contain IL-1.
Marz, P. et al. Sympathetic neurons can produce and respond to interleukin 6. Proc. Natl Acad. Sci. USA 95, 3251–3256 (1998).
Ignatowski, T. A. et al. Neuronal-associated tumor necrosis factor (TNFα): its role in noradrenergic functioning and modification of its expression following antidepressant drug administration. J. Neuroimmunol. 79, 84–90 (1997).
Breder, C. D., Tsujimoto, M., Terano, Y., Scott, D. W. & Saper, C. B. Distribution and characterization of tumor necrosis factor-alpha-like immunoreactivity in the murine central nervous system. J. Comp. Neurol. 337, 543–567 (1993).
Bette, M., Kaut, O., Schafer, M. K. & Weihe, E. Constitutive expression of p55TNFR mRNA and mitogen-specific up-regulation of TNFα and p75TNFR mRNA in mouse brain. J. Comp. Neurol. 465, 417–430 (2003).
Ban, E. M. Interleukin-1 receptors in the brain: characterization by quantitative in situ autoradiography. Immunomethods 5, 31–40 (1994).
Olivadoti, M. D. & Opp, M. R. Effects of i.c.v. administration of interleukin-1 on sleep and body temperature of interleukin-6-deficient mice. Neuroscience 153, 338–348 (2008).
Opp, M. R., Obál, F. & Krueger, J. M. Interleukin-1 alters rat sleep: temporal and dose-related effects. Am. J. Physiol. 260, R52–R58 (1991).
Lancel, M., Mathias, S., Faulhaber, J. & Schiffelholz, T. Effect of interleukin-1β on EEG power density during sleep depends on circadian phase. Am. J. Physiol. 270, R830–R837 (1996).
Imeri, L., Bianchi, S. & Opp, M. R. Inhibition of caspase-1 in rat brain reduces spontaneous nonrapid eye movement sleep and nonrapid eye movement sleep enhancement induced by lipopolysaccharide. Am. J. Physiol. 291, R197–R204 (2006). This report that NREM sleep is reduced when cleavage of biologically active IL-1 from its inactive precursor is impeded provides additional evidence of a role for IL-1 in regulating physiological NREM sleep and in the alterations in NREM sleep that follow host defence activation.
Takahashi, S. & Krueger, J. M. Inhibition of tumor necrosis factor prevents warming-induced sleep responses in rabbits. Am. J. Physiol. 272, R1325–R1329 (1997).
Baracchi, F. & Opp, M. R. Sleep-wake behavior and responses to sleep deprivation of mice lacking both interleukin-1β receptor 1 and tumor necrosis factor-α receptor 1. Brain Behav. Immun. 22, 982–993 (2008).
Opp, M. R. & Krueger, J. M. Anti-interleukin-1β reduces sleep and sleep rebound after sleep deprivation in rats. Am. J. Physiol. 266, R688–R695 (1994).
Opp, M. R. & Krueger, J. M. Interleukin 1-receptor antagonist blocks interleukin 1-induced sleep and fever. Am. J. Physiol. 260, R453–R457 (1991).
Cearley, C., Churchill, L. & Krueger, J. M. Time of day differences in IL1β and TNFα mRNA levels in specific regions of the rat brain. Neurosci. Lett. 352, 61–63 (2003).
Moldofsky, H., Lue, F. A., Eisen, J., Keystone, E. & Gorczynski, R. M. The relationship of interleukin-1 and immune functions to sleep in humans. Psychosom. Med. 48, 309–318 (1986).
Lue, F. A. et al. Sleep and cerebrospinal fluid interleukin-1-like activity in the cat. Int. J. Neurosci. 42, 179–183 (1988).
Opp, M. R. & Toth, L. A. Neural-immune interactions in the regulation of sleep. Front. Biosci. 8, d768–d779 (2003).
Turrin, N. P. et al. Pro-inflammatory and anti-inflammatory cytokine mRNA induction in the periphery and brain following intraperitoneal administration of bacterial lipopolysaccharide. Brain Res. Bull. 54, 443–453 (2001).
Zhang, Y., Gaekwad, J., Wolfert, M. A. & Boons, G. J. Modulation of innate immune responses with synthetic lipid A derivatives. J. Am. Chem. Soc. 129, 5200–5216 (2007).
Datta, S. C. & Opp, M. R. Lipopolysaccharide-induced increases in cytokines in discrete mouse brain regions are detectable using Luminex xMAP technology. J. Neurosci. Methods 175, 119–124 (2008).
Krueger, J. M. & Majde, J. A. Microbial products and cytokines in sleep and fever regulation. Crit. Rev. Immunol. 14, 355–379 (1994).
Imeri, L., Opp, M. R. & Krueger, J. M. An IL-1 receptor and an IL-1 receptor antagonist attenuate muramyl dipeptide- and IL-1-induced sleep and fever. Am. J. Physiol. 265, R907–R913 (1993).
Dunn, A. J. Effects of cytokines and infections on brain neurochemistry. Clin. Neurosci. Res. 6, 52–68 (2006).
Brodie, B. B., Pletscher, A. & Shore, P. A. Evidence that serotonin has a role in brain function. Science 122, 968 (1955).
Jacobs, B. L. & Azmitia, E. C. Structure and function of the brain serotonin system. Physiol. rev. 72, 165–229 (1992).
Ursin, R. Serotonin and sleep. Sleep Med. Rev. 6, 55–69 (2002).
Anisman, H., Merali, Z. & Hayley, S. Neurotransmitter, peptide and cytokine processes in relation to depressive disorder: comorbidity between depression and neurodegenerative disorders. Prog. Neurobiol. 85, 1–74 (2008).
Jouvet, M. Sleep and serotonin: an unfinished story. Neuropsychopharmacology 21, 24s–27s (1999). A historical review of 40 years of investigation into 5-HT's role in regulating arousal state by the investigator who is best known for his contributions to this topic. Data are interpreted within the framework of the hypothesis that 5-HT promotes wakefulness but stimulates the synthesis or release of unknown sleep factors that induce subsequent sleep.
Cespuglio, R., Gomez, M. E., Walker, E. & Jouvet, M. Effets du refroidissement et de la stimulation des noyaux du systeme du raphe sur les etats de vigilance chez le chat. Electroenceph. Clin. Neurophysiol. 47, 289–308 (1979).
Cespuglio, R., Faradji, H., Gomez, M. E. & Jouvet, M. Single unit recordings in the nuclei raphe dorsalis and magnus during sleep-waking cycle of semi-chronic prepared cats. Neurosci. Lett. 24, 133–138 (1981).
Lydic, R., McCarley, R. W. & Hobson, J. A. Serotonin neurons and sleep. I. Long term recordings of dorsal raphe discharge frequency and PGO waves. Arch. Ital. Biol. 125, 317–343 (1987).
McGinty, D. J. & Harper, R. M. Dorsal raphe neurons: depression of firing during sleep in cats. Brain Res. 101, 569–575 (1976). This was the first study to demonstrate state-dependent activity of serotonergic neurons.
Trulson, M. E. & Jacobs, B. L. Raphe unit activity in freely moving cats: correlation with level of behavioral arousal. Brain Res. 163, 135–150 (1979).
Cespuglio, R. et al. Voltammetric detection of the release of 5-hydroxyindole compounds throughout the sleep-waking cycle of the rat. Exp. Brain Res. 80, 121–128 (1990).
Gemma, C., Imeri, L., De Simoni, M. G. & Mancia, M. Interleukin-1 induces changes in sleep, brain temperature, and serotonergic metabolism. Am. J. Physiol. Regul. Integr. Comp. Physiol. 272, R601–R606 (1997).
Portas, C. M. & McCarley, R. W. Behavioral state-related changes of extracellular serotonin concentration in the dorsal raphe nucleus: a microdialysis study in the freely moving cat. Brain Res. 648, 306–312 (1994).
Portas, C. M. et al. On-line detection of extracellular levels of serotonin in dorsal raphe nucleus and frontal cortex over the sleep/wake cycle in the freely moving rat. Neuroscience 83, 807–814 (1998).
Dugovic, C. Functional activity of 5-HT2 receptors in the modulation of the sleep/wakefulness states. J. Sleep Res. 1, 163–168 (1992).
Saper, C. B., Chou, T. C. & Scammell, T. E. The sleep switch: hypothalamic control of sleep and wakefulness. Trends Neurosci. 24, 726–731 (2001).
Jones, B. E. From waking to sleeping: neuronal and chemical substrates. Trends Pharmacol. Sci. 26, 578–586 (2005).
Imeri, L., Mancia, M., Bianchi, M. & Opp, M. R. 5-hydroxytryptophan, but not L-tryptophan, alters sleep and brain temperature in rats. Neuroscience 95, 445–452 (2000). The first of several papers by these authors to demonstrate that activation of the 5-HT system has a biphasic effect on arousal state, with initial wakefulness followed by subsequent NREM sleep.
Imeri, L., Bianchi, S. & Opp, M. R. Antagonism of corticotropin-releasing hormone alters serotonergic-induced changes in brain temperature, but not sleep, of rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R1116–R1123 (2005).
Morrow, J. D., Vikraman, S., Imeri, L. & Opp, M. R. Effects of serotonergic activation by 5-hydroxytryptophan on sleep and body temperature of C57BL/56J and interleukin-6-deficient mice are dose and time related. Sleep 31, 21–33 (2008).
McCarley, R. W. Neurobiology of REM and NREM sleep. Sleep Med. 8, 302–330 (2007).
Lu, J., Sherman, D., Devor, M. & Saper, C. B. A putative flip-flop switch for control of REM sleep. Nature 441, 589–594 (2006).
Adrien, J., Alexandre, C., Boutrel, B. & Popa, D. Contribution of the “knock-out” technology to understanding the role of serotonin in sleep regulations. Arch. Ital. Biol. 142, 369–377 (2004).
Hagan, J. J. et al. Characterization of SB-269970-A, a selective 5-HT7 receptor antagonist. Br. J. Pharmacol. 130, 539–548 (2000).
Hedlund, P. B., Huitron-Resendiz, S., Henriksen, S. J. & Sutcliffe, J. G. 5-HT7 receptor inhibition and inactivation induce antidepressantlike behavior and sleep pattern. Biol. Psychiatry 58, 831–837 (2005).
Obal, F. Jr & Krueger, J. M. Biochemical regulation of non-rapid-eye-movement sleep. Front. Biosci. 8, d520–d550 (2003).
Manfridi, A. et al. Interleukin-1β enhances non-rapid eye movement sleep when microinjected into the dorsal raphe nucleus and inhibits serotonergic neurons in vitro. Eur. J. Neurosci. 18, 1041–1049 (2003).
Brambilla, D., Franciosi, S., Opp, M. R. & Imeri, L. Interleukin-1 inhibits firing of serotonergic neurons in the dorsal raphe nucleus and enhances GABAergic inhibitory post-synaptic potentials. Eur. J. Neurosci. 26, 1862–1869 (2007).
Serantes, R. et al. Interleukin-1β enhances GABAA receptor cell-surface expression by a phosphatidylinositol 3-kinase/Akt pathway: relevance to sepsis-associated encephalopathy. J. Biol. Chem. 281, 14632–14643 (2006).
Miller, L. G., Galpern, W. R., Dunlap, K., Dinarello, C. A. & Turner, T. J. Interleukin-1 augments gamma-aminobutyric acidA receptor function in brain. Mol. Pharmacol. 39, 105–108 (1991).
Tabarean, I. V., Korn, H. & Bartfai, T. Interleukin-1β induces hyperpolarization and modulates synaptic inhibition in preoptic and anterior hypothalamic neurons. Neuroscience 141, 1685–1695 (2006).
Feleder, C., Arias, P., Refojo, D., Nacht, S. & Moguilevsky, J. Interleukin-1 inhibits NMDA-stimulated GnRH secretion: associated effects on the release of hypothalamic inhibitory amino acid neurotransmitters. Neuroimmunomodulation 7, 46–50 (2000).
Zeise, M. L., Madamba, S. & Siggins, G. R. Interleukin-1β increases synaptic inhibition in rat hippocampal pyramidal neurons in vitro. Regul. Pept. 39, 1–7 (1992).
Luk, W. P. et al. Adenosine: a mediator of interleukin-1β-induced hippocampal synaptic inhibition. J. Neurosci. 19, 4238–4244 (1999).
Alam, M. N. et al. Interleukin-1β modulates state-dependent discharge activity of preoptic area and basal forebrain neurons: role in sleep regulation. Eur. J. Neurosci. 20, 207–216 (2004). These authors demonstrated for the first time that IL-1 applied directly to the hypothalamus inhibits wake-active neurons. The paper also reported increases in discharge rates in a subset of sleep-active neurons.
Baker, F. C. et al. Interleukin 1β enhances non-rapid eye movement sleep and increases c-Fos protein expression in the median preoptic nucleus of the hypothalamus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 288, R998–R1005 (2005).
Shintani, F. et al. Interleukin-1β augments release of norepinephrine, dopamine, and serotonin in the rat anterior hypothalamus. J. Neurosci. 13, 3574–3581 (1993).
Denoyer, M., Sallanon, M., Kitahama, K., Aubert, C. & Jouvet, M. Reversibility of para-chlorophenylalanine-induced insomnia by intrahypothalamic microinjection of L-5-hydroxytryptophan. Neuroscience 28, 83–94 (1989). These authors demonstrated that the anterior hypothalamus is the only brain region where 5-HTP rescues sleep in cats made insomniac by depletion of brain 5-HT.
Khateb, A., Fort, P., Alonso, A., Jones, B. E. & Muhlethaler, M. Pharmacological and immunohistochemical evidence for serotonergic modulation of cholinergic nucleus basalis neurons. Eur. J. Neurosci. 5, 541–547 (1993).
Imeri, L., Bianchi, S. & Mancia, M. Muramyl dipeptide and IL-1 effects on sleep and brain temperature after inhibition of serotonin synthesis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 273, R1663–R1668 (1997).
Imeri, L., Mancia, M. & Opp, M. R. Blockade of 5-HT2 receptors alters interleukin-1-induced changes in rat sleep. Neuroscience 92, 745–749 (1999).
Gemma, C., Imeri, L. & Opp, M. R. Serotonergic activation stimulates the pituitary-adrenal axis and alters interleukin-1 mRNA expression in rat brain. Psychoneuroendocrinology 28, 875–884 (2003). This was the first paper to report that activation of the serotonergic system alters IL-1 mRNA expression in the brain.
Dinarello, C. A. et al. Interleukin 1 induces interleukin 1. I. Induction of circulating interleukin 1 in rabbits in vivo and in human mononuclear cells in vitro. J. Immunol. 139, 1902–1910 (1987).
Taishi, P., Churchill, L., De, A., Obal, F. Jr & Krueger, J. M. Cytokine mRNA induction by interleukin-1β or tumor necrosis factor α in vitro and in vivo. Brain Res. 1226, 89–98 (2008).
Turnbull, A. V. & Rivier, C. Regulation of the hypothalamic-pituitary-adrenal axis by cytokines: actions and mechanisms of action. Physiol. rev. 79, 1–71 (1999).
Mignot, E. Why we sleep: the temporal organization of recovery. PLoS Biol. 6, e106 (2008).
Siegel, J. M. Clues to the functions of mammalian sleep. Nature 437, 1264–1271 (2005).
Toth, L. A., Tolley, E. A. & Krueger, J. M. Sleep as a prognostic indicator during infectious disease in rabbits. Proc. Soc. Exp. Biol. Med. 203, 179–192 (1993). This retrospective analysis of data derived from almost 100 rabbits revealed associations between the quality of sleep and clinical symptoms, morbidity and mortality. Survival from infectious pathogens is associated with better quality sleep.
Parmeggiani, P. L. Thermoregulation and sleep. Front. Biosci. 8, s557–s567 (2003).
Kluger, M. J., Kozak, W., Conn, C. A., Leon, L. R. & Soszynski, D. The adaptive value of fever. Infect. Dis. Clin. North Am. 10, 1–21 (1996).
Obál, F. Jr, Rubicsek, G., Sary, G. & Obál, F. Changes in the brain and core temperatures in relation to the various arousal states in rats in the light and dark periods of the day. Pflügers Arch. 404, 73–79 (1985).
Glotzbach, S. F. & Heller, H. C. Central nervous regulation of body temperature during sleep. Science 194, 537–538 (1976).
Kluger, M. J. Fever. It's Biology, Evolution and Function (Princeton Univ. Press, Princeton, 1979).
Pollmacher, T. et al. Experimental immunomodulation, sleep, and sleepiness in humans. Ann. NY Acad. Sci. 917, 488–499 (2000).
Mullington, J. et al. Dose-dependent effects of endotoxin on human sleep. Am. J. Physiol. 278, R947–R955 (2000).
Haack, M., Schuld, A., Kraus, T. & Pollmacher, T. Effects of sleep on endotoxin-induced host responses in healthy men. Psychosom. Med. 63, 568–578 (2001). Along with references 101 and 102, this paper demonstrated the effects on the sleep of healthy human volunteers of host defence activation by injection of endotoxin.
Friess, E., Wiedemann, K., Steiger, A. & Holsboer, F. The hypothalamic-pituitary-adrenocortical system and sleep in man. Adv. Neuroimmunol. 5, 111–125 (1995).
Steiger, A. Sleep and the hypothalamo-pituitary-adrenocortical system. Sleep Med. Rev. 6, 125–138 (2002).
Palmblad, J., Pertrini, B., Wasserman, J. & Kerstedt, T. A. Lymphocyte and granulocyte reactions during sleep deprivation. Psychosom. Med. 41, 273–278 (1979). The first study of human subjects to specifically focus on the impact of sleep loss on immunity.
Dinges, D. F. et al. Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. J. Clin. Invest. 93, 1930–1939 (1994).
Irwin, M. R., Wang, M., Campomayor, C. O., Collado-Hidalgo, A. & Cole, S. Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Arch. Intern. Med. 166, 1756–1762 (2006).
Marshall, L. & Born, J. Brain-immune interactions in sleep. Int. Rev. Neurobiol. 52, 93–131 (2002).
Rada, P. et al. Interleukin-1β decreases acetylcholine measured by microdialysis in the hippocampus of freely moving rats. Brain Res. 550, 287–290 (1991).
Carmeliet, P., Van Damme, J. & Denef, C. Interleukin-1 beta inhibits acetylcholine synthesis in the pituitary corticotropic cell line AtT20. Brain Res. 491, 199–203 (1989).
Li, Y. et al. Neuronal-glial interactions mediated by interleukin-1 enhance neuronal acetylcholinesterase activity and mRNA expression. J. Neurosci. 20, 1–149 (2000).
Fogal, B. & Hewett, S. J. Interleukin-1β: a bridge between inflammation and excitotoxicity? J. Neurochem. 106, 1–23 (2008).
Basheer, R., Strecker, R. E., Thakkar, M. M. & McCarley, R. W. Adenosine and sleep-wake regulation. Prog. Neurobiol. 73, 379–396 (2004).
Sperlagh, B., Baranyi, M., Hasko, G. & Vizi, E. S. Potent effect of interleukin-1β to evoke ATP and adenosine release from rat hippocampal slices. J. Neuroimmunol. 151, 33–39 (2004).
Zhu, G. et al. Involvement of Ca2+-induced Ca2+ releasing system in interleukin-1β-associated adenosine release. Eur. J. Pharmacol. 532, 246–252 (2006).
Rosenbaum, E. Warum müssen wir schlafen? Eine neue Theorie des Schlafes (August Hirschwald, Berlin, 1892).
Tigerstedt, R. & Bergman, P. Niere und kreislauf. Arch. Physiol. 8, 223–271 (1898).
Baylis, W. & Starling, E. The mechanism of pancreatic secretion. J. Physiol. 28, 325–353 (1902).
Ishimori, K. True cause of sleep: a hypnogenic substance as evidenced in the brain of sleep-deprived animals. Tokyo Igakkai Zasshi 23, 429–459 (1909).
Legendre, R. & Piéron, H. Recherches sur le besoin de sommeil consecutif a une vielle prolongee. Z. Allg. Physiol. 14, 235–262 (1913).
Kornmüller, A., Lux, H., Winkel, K. & Klee, M. Neurohumoral ausgelöste schlafzustände an tieren mit gekreuztem kreislfau unter kontrolle von EEG-ableitungen. Naturwissenschaften 14, 503–505 (1961).
Schoenenberger, G. A., Maier, P. F., Tobler, H. J. & Monnier, M. A naturally occuring delta-EEG enhancing nonapeptide in rabbits. Pflügers Arch. 369, 99–109 (1977).
Nagasaki, H., Iriki, M., Inoue, S. & Uchizono, K. Proceedings: Sleep promoting substances in the brain stem of rats. Nippon Seirigaku Zasshi 36, 293 (1974).
Pappenheimer, J. R., Miller, T. B. & Goodrich, C. A. Sleep-promoting effects of cerebrospinal fluid from sleep-deprived goats. Proc. Natl Acad. Sci. USA 58, 513–517 (1967).
Fencl, V., Koski, G. & Pappenheimer, J. R. Factors in cerebrospinal fluid from goats that affect sleep and activity in rats. J. Physiol. 216, 565–589 (1971).
Pappenheimer, J. R., Koski, G., Fencl, V., Karnovsky, M. L. & Krueger, J. Extraction of sleep-promoting factor S from cerebrospinal fluid and from brains of sleep-deprived animals. J. Neurophysiol. 38, 1299–1311 (1975).
Krueger, J. M., Pappenheimer, J. R. & Karnovsky, M. L. Sleep-promoting factor S: purification and properties. Proc. Natl Acad. Sci. USA 75, 5235–5238 (1978).
Krueger, J. M., Pappenheimer, J. R. & Karnovsky, M. L. The composition of sleep-promoting factor isolated from human urine. J. Biol. Chem. 257, 1664–1669 (1982).
Krueger, J. M., Pappenheimer, J. R. & Karnovsky, M. L. Sleep-promoting effects of muramyl peptides. Proc. Natl Acad. Sci. USA 79, 6102–6106 (1982).
Fontana, A., Kristensen, F., Dubs, R., Gemsa, D. & Weber, E. Production of prostaglandin E and an interleukin-1 like factor by cultured astrocytes and C6 glioma cells. J. Immunol. 129, 2413–2419 (1982).
Krueger, J. M., Dinarello, C. A. & Chedid, L. Promotion of slow-wave sleep (SWS) by a purified interleukin-1 (IL-1) preparation. Fed. Proc. 42, 356 (1983).
Krueger, J. M., Walter, J., Dinarello, C. A., Wolff, S. M. & Chedid, L. Sleep-promoting effects of endogenous pyrogen (interleukin-1). Am. J. Physiol. 246, R994–R999 (1984). This and reference 134 were the first research articles to demonstrate the effects of IL-1 on sleep and the electroencephalogram.
Tobler, I., Borbély, A. A., Schwyzer, M. & Fontana, A. Interleukin-1 derived from astrocytes enhances slow wave activity in sleep EEG of the rat. Eur. J. Pharmacol. 104, 191–192 (1984).
Shoham, S., Davenne, D., Cady, A. B., Dinarello, C. A. & Krueger, J. M. Recombinant tumor necrosis factor and interleukin 1 enhance slow-wave sleep. Am. J. Physiol. 253, R142–R149 (1987).
Acknowledgements
The authors were supported by US National Institutes of Health Grants MH64843 and HL080972, the Department of Anesthesiology of the University of Michigan Medical School, and the Ministero dell'Istruzione, dell'Universita' e della Ricerca, Italy.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Encephalitis lethargica
-
An infectious process and an associated inflammatory response involving the brain and characterized by pronounced somnolence. The first cases were described in 1916 by the Viennese neurologist von Economo. The pathology is typical of viral infections and is localized principally to the midbrain, the subthalamus and the hypothalamus. The causative agent has not been definitively determined.
- Trypanosomiasis
-
A disease caused by the protozoan Trypanosoma brucei and transmitted by several species of the tsetse fly. When the brain and meninges become involved, usually in the second year of infection, a chronic progressive neurologic syndrome results. Complete loss of the timing of sleep, alterations in sleep architecture, and later apathy, stupor and coma characterize the syndrome.
- Sleep fragmentation
-
Interruption of sleep bouts by brief arousals such that the duration of the bout is reduced and transitions from one behavioural state to another occur more frequently.
- Lipid A
-
The innermost, hydrophobic, lipid component of lipopolysaccharide. Lipid A anchors the lipopolysaccharide to the outer membrane of the Gram-negative bacterial cell wall.
- Lipopolysaccharide
-
A component (also known as endotoxin) of the outer wall of Gram-negative bacterial cell walls. It is composed of a lipid and polysaccharides joined by covalent bonds. It elicits strong immune responses through signalling pathways coupled to Toll-like receptor 4.
- Muramyl dipeptide
-
The synthetic analogue of muramyl peptides, the monomeric building blocks of bacterial cell wall peptidoglycan. Muramyl peptides are released by mammalian macrophages during the digestion of bacterial cell walls.
- Inhibitory postsynaptic potential
-
(IPSP). Hyperpolarization of the membrane potential of a postsynaptic neuron. IPSPs are induced by a neurotransmitter released by a presynaptic neuron. Hyperpolarization reduces neuronal excitability because it is more difficult to trigger an action potential in a hyperpolarized neuron.
- Acute-phase response
-
The reaction that develops in response to an injury or infection. It is mediated by pro-inflammatory cytokines (such as IL-1) and is characterized by a local response (inflammation) and a systemic component, which includes the production of acute-phase proteins by hepatocytes, leukocytosis, fever and profound changes in lipid, protein and carbohydrate metabolism.
- Sickness behaviour
-
The constellation of symptoms (decreased food intake, depressed activity, loss of interest in usual activities, disappearance of self-maintenance behaviours and altered sleep) that accompany responses to infection.
Rights and permissions
About this article
Cite this article
Imeri, L., Opp, M. How (and why) the immune system makes us sleep. Nat Rev Neurosci 10, 199–210 (2009). https://doi.org/10.1038/nrn2576
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn2576
This article is cited by
-
Sleep characteristics and inflammatory markers in women with post-traumatic stress disorder
BMC Psychiatry (2023)
-
The role of insufficient sleep and circadian misalignment in obesity
Nature Reviews Endocrinology (2023)
-
Heat of the night: sleep disturbance activates inflammatory mechanisms and induces pain in rheumatoid arthritis
Nature Reviews Rheumatology (2023)
-
Association Between Poor Sleep and Myocardial Infarction in Patients with Psoriasis: Findings from a Cross-Sectional Study with the National Psoriasis Foundation
Dermatology and Therapy (2023)
-
Transcutaneous Electrical Acupoint Stimulation Improves Postoperative Sleep Quality in Patients Undergoing Laparoscopic Gastrointestinal Tumor Surgery: A Prospective, Randomized Controlled Trial
Pain and Therapy (2023)