Key Points
-
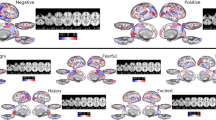
Most humans develop a capacity to recognize the emotional signals of different facial expressions. This capacity is mediated by a brain network that involves emotion-related brain circuits (including the amygdala and the orbitofrontal cortex) and higher-level visual representation areas (the fusiform gyrus and the superior temporal sulcus).
-
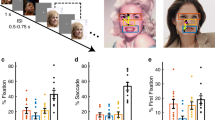
Human infants start to discriminate facial expressions by the second half of their first year. At approximately the same age, they begin to exhibit adult-like attentional bias towards certain salient facial expressions (such as expressions of fear), as well as enhanced vision- and attention-related event-related brain potentials to fearful facial expressions.
-
These findings, together with experimental lesion and anatomical tracing studies in other species, suggest that the key components of the emotion-processing network and their interconnections are established and become functional early in postnatal life.
-
Developmental studies in humans and monkeys have further shown that face-processing mechanisms are initially broadly tuned (activated by a broad range of stimuli, such as human and monkey faces) but narrow and become more specialized for specific types of perceptual discriminations with experience (discrimination of information in human faces).
-
Collectively, these data suggest that the acquisition of representations of facial expressions may reflect the functional emergence of an experience-expectant mechanism by the second half of the first year and rapid experience-driven attunement of this mechanism to species-typical facial expressions.
-
Specifically, components of the emotion-processing network (such as the amygdala) may be to a limited extent prepared for processing and storing information about biologically salient cues, but they require exposure to facial expressions at a specific developmental time point ('expected experience') in order to be refined and develop towards more mature forms.
-
Although the basic organization of the emotion-recognition networks is specified by an experience-expectant neural circuitry that emerges during a sensitive period, representations of facial expressions are likely to be continually fine-tuned by individual-specific experiences (reflecting the experience-dependent development of facial-expression processing).
-
Genetically driven differences in the reactivity of emotion-related brain circuits (for example, polymorphisms that affect serotonin transmission and amygdala reactivity) might, in combination with environmental factors (such as exposure to negative emotions), bias the developmental process towards heightened sensitivity to signals of certain negative emotions.
Abstract
Humans in different cultures develop a similar capacity to recognize the emotional signals of diverse facial expressions. This capacity is mediated by a brain network that involves emotion-related brain circuits and higher-level visual-representation areas. Recent studies suggest that the key components of this network begin to emerge early in life. The studies also suggest that initial biases in emotion-related brain circuits and the early coupling of these circuits and cortical perceptual areas provide a foundation for a rapid acquisition of representations of those facial features that denote specific emotions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ekman, P., Sorenson, E. R. & Friesen, W. V. Pan-cultural elements in facial displays of emotion. Science 164, 86–88 (1969).
Ekman, P. & Friesen, W. V. A new pan-cultural facial expression of emotion. Motiv. Emot. 10, 159–168 (1986).
Levenson, R. W. in Handbook of Affective Sciences (eds Davidson, R. J., Scherer, K. R. & Goldsmith, H. H.) 212–224 (Oxford Univ. Press, New York, 2003).
Vuilleumier, P. How brains beware: neural mechanisms of emotional attention. Trends Cogn. Sci. 9, 585–594 (2005). Presents an overview of studies showing that the respective influences of voluntary attention (task relevance) and affective significance on sensory processing are dissociable and mediated by distinct neural mechanisms.
Pourtois, G., Grandjean, D., Sander, D. & Vuilleumier, P. Electrophysiological correlates of rapid spatial orienting towards fearful faces. Cereb. Cortex 14, 619–633 (2004).
Milders, M., Sahraie, A., Logan, S. & Donnellon, N. Awareness of faces is modulated by their emotional meaning. Emotion 6, 10–17 (2006).
Yang, E., Zald, D. H. & Blake, R. Fearful expressions gain preferential access to awareness during continuous flash suppression. Emotion 7, 882–886 (2007).
Georgiou, G. A. et al. Focusing on fear: attentional disengagement from emotional faces. Vis. Cogn. 12, 145–158 (2005).
Leppänen, J. M., Hietanen, J. K. & Koskinen, K. Differential early ERPs to fearful versus neutral facial expressions: a response to the salience of the eyes? Biol. Psychol. 78, 150–158 (2008).
Batty, M. & Taylor, M. J. Early processing of the six basic facial emotional expressions. Cogn. Brain Res. 17, 613–620 (2003).
Morris, J. S. et al. A neuromodulatory role for the human amygdala in processing emotional facial expressions. Brain 121, 47–57 (1998).
Hadj-Bouziane, F., Bell, A. H., Knusten, T. A., Ungerleider, L. G. & Tootell, R. B. H. Perception of emotional expressions is independent of face selectivity in monkey inferior temporal cortex. Proc. Natl Acad. Sci. USA 105, 5591–5596 (2008).
Hooker, C. I., Germine, L. T., Knight, R. T. & D'Esposito, M. Amygdala response to facial expressions reflects emotional learning. J. Neurosci. 26, 8915–8922 (2006).
Adolphs, R. Neural systems for recognizing emotion. Curr. Opin. Neurobiol. 12, 169–177 (2002).
Luo, Q., Holroyd, T., Jones, M., Hendler, T. & Blair, J. Neural dynamics for facial threat processing as revealed by gamma band synchronization using MEG. Neuroimage 34, 839–847 (2007).
Vuilleumier, P., Armony, J. L., Driver, J. & Dolan, R. J. Distinct spatial frequency sensitivities for processing faces and emotional expressions. Nature Neurosci. 6, 624–631 (2003).
Vuilleumier, P., Richardson, M. P., Armony, J. L., Driver, J. & Dolan, R. J. Distant influences of amygdala lesion on visual cortical activation during emotional face processing. Nature Neurosci. 7, 1271–1278 (2004). Showed that the amygdala is critically involved in enhanced perceptual processing of emotionally expressive faces.
Anderson, A. K. & Phelps, E. A. Lesions of the human amygdala impair enhanced perception of emotionally salient events. Nature 411, 305–309 (2001).
Freese, J. L. & Amaral, D. G. The organization of projections from the amygdala to visual cortical areas TE and V1 in the macaque monkey. J. Comp. Neurol. 486, 295–317 (2005).
Freese, J. L. & Amaral, D. G. Synaptic organization of projections from the amygdala to visual cortical areas TE and V1 in the macaque monkey. J. Comp. Neurol. 496, 655–667 (2006).
Amaral, D. G., Behniea, H. & Kelly, J. L. Topographic organization of projections from the amygdala to the visual cortex in the macaque monkey. Neuroscience 118, 1099–1120 (2003).
Kapp, B. S., Supple, W. F. & Whalen, P. J. Effects of electrical stimulation of the amygdaloid central nucleus on neocortical arousal in the rabbit. Behav. Neurosci. 108, 81–93 (1994).
Bentley, P., Vuilleumier, P., Thiel, C. M., Driver, J. & Dolan, R. J. Cholinergic enhancement modulates neural correlates of selective attention and emotional processing. Neuroimage 20, 58–70 (2003).
Whalen, P. J. Fear, vigilance, and ambiguity: initial neuroimaging studies of the human amygdala. Curr. Dir. Psychol. Sci. 7, 177–188 (1998).
Adolphs, R. Fear, faces, and the human amygdala. Curr. Opin. Neurobiol. 18, 166–172 (2008).
Paton, J. J., Belova, M. A., Morrison, S. E. & Salzman, C. D. The primate amygdala represents the positive and negative value of visual stimuli during learning. Nature 439, 865–870 (2006).
Hornak, J. et al. Changes in emotion after circumscribed surgical lesions of the orbitofrontal and cingulate cortices. Brain 126, 1691–1712 (2003).
O'Doherty, J. et al. Beauty in a smile: the role of medial orbitofrontal cortex in facial attractiveness. Neuropsychologia 41, 147–155 (2003).
Blair, R. J. R., Morris, J. S., Frith, C. D., Perrett, D. I. & Dolan, R. J. Dissociable neural responses to facial expressions of sadness and anger. Brain 122, 883–893 (1999).
Rolls, E. T. The representation of information about faces in the temporal and frontal lobes. Neuropsychologia 45, 124–143 (2007).
Hooker, C. I., Verosky, S. C., Miyakawa, A., Knight, R. T. & D'Esposito, M. The influence of personality on neural mechanisms of observational fear and reward learning. Neuropsychologia 46, 2709–2724 (2008).
Cavada, C., Company, T., Tejedor, J., Cruz-Rizzolo, R. J. & Reinoso-Suarez, F. The anatomical connections of the macaque monkey orbitofrontal cortex: a review. Cereb. Cortex 10, 220–242 (2000).
Bar, M. et al. Top-down facilitation of visual recognition. Proc. Natl Acad. Sci. USA 103, 449–454 (2006).
Kringelbach, M. L. et al. A specific and rapid neural signature for parental instinct. PLoS ONE 3, e1664 (2008).
Canli, T. Toward a neurogenetic theory of neuroticism. Ann. NY Acad. Sci. 1129, 153–174 (2008).
Fox, E., Mathews, A., Calder, A. J. & Yiend, J. Anxiety and sensitivity to gaze direction in emotionally expressive faces. Emotion 7, 478–486 (2007).
Etkin, A. et al. Individual differences in trait anxiety predict the response of the basolateral amygdala to unconsciously processed fearful faces. Neuron 44, 1043–1055 (2004).
Stein, M. B., Simmons, A. N., Feinstein, J. S. & Paulus, M. P. Increased amygdala and insula activation during emotion processing in anxiety-prone subjects. Am. J. Psychiatry 164, 318–327 (2007).
Hare, T. A. et al. Biological substrates of emotional reactivity and regulation in adolescence during an emotional go-nogo task. Biol. Psychiatry 63, 927–934 (2008).
Sorce, F. F., Emde, R. N., Campos, J. J. & Klinnert, M. D. Maternal emotional signaling: its effect on the visual cliff behavior of 1-year-olds. Dev. Psychol. 21, 195–200 (1985).
Johnson, M. H. Subcortical face processing. Nature Rev. Neurosci. 6, 766–774 (2005).
Banks, M. S. & Salapatek, P. in Handbook of Child Psychology (eds Haith, M. M. & Campos, J. J.) 435–571 (Wiley, New York, 1983).
Bushnell, I. W. R. Modification of the externality effect in young infants. J. Exp. Child Psychol. 28, 211–229 (1979).
Maurer, D. & Salapatek, P. Developmental changes in the scanning of faces by young infants. Child Dev. 47, 523–527 (1976).
Field, T. M., Woodson, R. W., Greenberg, R. & Cohen, C. Discrimination and imitation of facial expressions by neonates. Science 218, 179–181 (1982).
Kestenbaum, R. & Nelson, C. A. The recognition and categorization of upright and inverted emotional expressions by 7-month-old infants. Infant Behav. Dev. 13, 497–511 (1990).
Bornstein, M. H. & Arterberry, M. E. Recognition, discrimination and categorization of smiling by 5-month-old infants. Dev. Sci. 6, 585–599 (2003).
Ludemann, P. M. & Nelson, C. A. Categorical representation of facial expressions by 7-month-old infants. Dev. Psychol. 24, 492–501 (1988).
Nelson, C. A., Morse, P. A. & Leavitt, L. A. Recognition of facial expressions by 7-month-old infants. Child Dev. 50, 1239–1242 (1979). One of the first papers to report that infants can perceptually discriminate certain facial expressions from one another (specifically, happy and fearful expressions) and prefer to look longer at fearful faces than happy faces.
Bahrick, L. E., Lickliter, R. & Flom, R. Intersensory redundancy guides the development of selective attention, perception, and cognition. Curr. Dir. Psychol. Sci. 13, 99–102 (2004).
Flom, R. & Bahrick, L. E. The development of infant discrimination of affect in multimodal and unimodal stimulation: the role of intersensory redundancy. Dev. Psychol. 43, 238–252 (2007).
Walker-Andrews, A. S. Infants' perception of expressive behaviors: differentiation of multimodal information. Psychol. Bull. 121, 437–456 (1997).
Kotsoni, E., de Haan, M. & Johnson, M. H. Categorical perception of facial expressions by 7-month-old infants. Perception 30, 1115–1125 (2001).
Nelson, C. A. & Dolgin, K. The generalized discrimination of facial expressions by 7-month-old infants. Child Dev. 56, 58–61 (1985).
Peltola, M. J., Leppänen, J. M., Palokangas, T. & Hietanen, J. K. Fearful faces modulate looking duration and attention disengagement in 7-month-old infants. Dev. Sci. 11, 60–68 (2008). Provided evidence for adult-like emotion–attention interactions in infants and showed that the influence of fearful faces on attention in infants is not simply attributable to the novelty of these faces.
de Haan, M., Johnson, M. H. & Halit, H. Development of face-sensitive event-related potentials during infancy: a review. Int. J. Psychophysiol. 51, 45–58 (2003).
de Haan, M. & Nelson, C. A. Brain activity differentiates face and object processing in 6-month-old infants. Dev. Psychol. 35, 1113–1121 (1999). Provided electrophysiological evidence that the brain of the typically developing 6-month-old infant distinguishes between faces and non-face objects.
de Haan, M., Pascalis, O. & Johnson, M. H. Specialization of neural mechanisms underlying face recognition in human infants. J. Cogn. Neurosci. 14, 199–209 (2002).
Halit, H., Csibra, G., Volein, Á . & Johnson, M. H. Face-sensitive cortical processing in early infancy. J. Child Psychol. Psychiatry 45, 1228–1234 (2004).
Halit, H., de Haan, M. & Johnson, M. H. Cortical specialization for face processing: face-sensitive event-related potential components in 3- and 12-month-old infants. Neuroimage 19, 1180–1193 (2003).
Tzourio-Mazoyer, N. et al. Neural correlates of woman face processing by 2-month-old infants. Neuroimage 15, 454–461 (2002).
Otsuka, Y. et al. Neural activation to upright and inverted faces in infants measured by near infrared spectroscopy. Neuroimage 34, 399–406 (2007).
Amaral, D. G. & Bennett, J. Development of amygdalo-cortical connection in the macaque monkey. Soc. Neurosci. Abstr. 26, 17–26 (2000).
Machado, C. J. & Bachevalier, J. Non-human primate models of childhood psychopathology: the promise and the limitations. J. Child Psychol. Psychiatry 44, 64–87 (2003).
Minagawa-Kawai, Y. et al. Prefrontal activation associated with social attachment: facial emotion recognition in mothers and infants. Cereb. Cortex 30 May 2008 (doi:10.1093/cercor/bhn081). Reported an optical-imaging study which showed that the orbitofrontal cortex is involved in processing happy facial expressions in infants.
Leppänen, J. M., Moulson, M. C., Vogel-Farley, V. K. & Nelson, C. A. An ERP study of emotional face processing in the adult and infant brain. Child Dev. 78, 232–245 (2007). Reported evidence that affective significance modulates the early stages of cortical face processing in adults and 7-month-old infants.
Nelson, C. A. & de Haan, M. Neural correlates of infants' visual responsiveness to facial expressions of emotions. Dev. Psychobiol. 29, 577–595 (1996). Examined the neural correlates of infants' ability to discriminate facial expressions of emotion.
de Haan, M. in Infant EEG and event-related potentials (ed. de Haan, M.) 101–143 (Psychology Press, New York, 2007).
Nelson, C. A. in Human Behavior and the Developing Brain (eds Dawson, G. & Fischer, K.) 269–313 (Guilford Press, New York, 1994).
Reynolds, G. D. & Richards, J. E. Familiarization, attention, and recognition memory in infancy: an event-related-potential and cortical source localization study. Dev. Psychol. 41, 598–615 (2005).
Bush, G., Luu, P. & Posner, M. I. Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn. Sci. 4, 215–222 (2000).
Hoehl, S., Palumbo, L., Heinisch, C. & Striano, T. Infants' attention is biased by emotional expressions and eye gaze direction. Neuroreport 19, 579–582 (2008).
Hoehl, S., Wiese, L. & Striano, T. Young infants' neural processing of objects is affected by eye gaze direction and emotional expression. PLoS ONE 3, e2389 (2008). Provided evidence that infants use information from adults gaze direction and emotional expression to enhance attention towards certain objects.
Bauman, M. D., Lavenex, P., Mason, W. A., Capitanio, J. P. & Amaral, D. G. The development of social behavior following neonatal amygdala lesions in rhesus monkeys. J. Cogn. Neurosci. 16, 1388–1411 (2004).
Moriceau, S. & Sullivan, R. M. Maternal presence serves as a switch between learning fear and attraction in infancy. Nature Neurosci. 9, 1004–1006 (2004).
Thompson, J. V., Sullivan, R. M. & Wilson, D. A. Developmental emergence of fear learning corresponds with changes in amygdala synaptic plasticity. Brain Res. 1200, 58–65 (2008).
Farroni, T., Menon, E., Rigato, S. & Johnson, M. H. The perception of facial expressions in newborns. Eur. J. Dev. Psychol. 4, 2–13 (2007).
Greenough, W. T., Black, J. E. & Wallace, C. S. Experience and brain development. Child Dev. 58, 539–559 (1987). A theoretical review that illustrates the differences between experience-expectant and experience-dependent plasticity and learning.
Pascalis, O., de Haan, M. & Nelson, C. A. Is face processing species-specific during the first year of life? Science 296, 1321–1323 (2002). The first study to show that face-processing mechanisms are initially broadly tuned but narrow with experience.
Pascalis, O. et al. Plasticity of face processing in infancy. Proc. Natl Acad. Sci. USA 102, 5297–5300 (2005). Demonstrated that one can rescue the sensitive period for discriminating monkey faces by providing infants with distributed exposure to monkey faces.
Lewkowicz, D. J. & Ghazanfar, A. A. The decline of cross-species intersensory perception in human infants. Proc. Natl Acad. Sci. USA 103, 6771–6774 (2006).
Weikum, W. M. et al. Visual language discrimination in infancy. Science 316, 1159 (2007).
Scott, L. S., Pascalis, O. & Nelson, C. A. A domain general theory of the development of perceptual discrimination. Curr. Dir. Psychol. Sci. 16, 197–201 (2007).
Sugita, Y. Face perception in monkeys reared with no exposure to faces. Proc. Natl Acad. Sci. USA 105, 394–398 (2008).
Pollak, S. D., Cicchetti, D., Hornung, K. & Reed, A. Recognizing emotion in faces: developmental effects of child abuse and neglect. Dev. Psychol. 36, 679–688 (2000).
Pollak, S. D. & Kistler, D. J. Early experience is associated with the development of categorical representations for facial expression of emotions. Proc. Natl Acad. Sci. USA 88, 7572–7575 (2002). Reported evidence of experience-dependent plasticity in brain networks underlying perception of universal facial expressions.
Pollak, S. D., Klorman, R., Thatcher, J. E. & Cicchetti, D. P3b reflects maltreated children's reactions to facial displays of emotion. Psychophysiology 38, 264–274 (2001).
Pollak, S. D. & Sinha, P. Effects of early experience on children's recognition of facial displays of emotion. Dev. Psychol. 38, 784–791 (2002).
Pollak, S. D. & Tolley-Schell, S. A. Selective attention to facial emotion in physically abused children. J. Abnorm. Psychol. 112, 323–338 (2003).
Salzman, C. D., Paton, J. J., Belova, M. A. & Morrison, S. E. Flexible neural representations of value in the primate brain. Ann. NY Acad. Sci. 1121, 336–354 (2007).
Weinberger, N. M. Specific long-term memory traces in primary auditory cortex. Nature Rev. Neurosci. 5, 279–290 (2004).
Keil, A., Stolarova, M., Moratti, S. & Ray, W. J. Adaptation in human visual cortex as a mechanism for rapid discrimination of aversive stimuli. Neuroimage 36, 472–479 (2007).
Lim, S. L. & Pessoa, L. Affective learning increases sensitivity to graded emotional faces. Emotion 8, 96–103 (2008).
Pizzagalli, D. A., Greischar, L. L. & Davidson, R. J. Spatio-temporal dynamics of brain mechanisms in aversive classical conditioning: high-density event-related potential and brain electrical tomography analyses. Neuropsychologia 41, 184–194 (2003).
Olsson, A. & Phelps, E. A. Social learning of fear. Nature Neurosci. 10, 1095–1102 (2007).
Mathews, A. & MacLeod, C. Cognitive vulnerability to emotional disorders. Annu. Rev. Clin. Psychol. 1, 167–195 (2005).
Hariri, A. R. et al. Serotonin transporter genetic variation and the response of the human amygdala. Science 297, 400–403 (2002).
Caspi, A. et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301, 386–389 (2003).
Hariri, A. R. & Holmes, A. Genetics of emotional regulation: the role of the serotonin transporter in neural function. Trends Cogn. Sci. 10, 182–191 (2006).
Lakatos, K. et al. Association of D4 dopamine receptor gene and serotonin transporter promoter polymorphisms with infants' response to novelty. Mol. Psychiatry 8, 90–97 (2003).
Bishop, S. J. Neurocognitive mechanisms of anxiety: an integrative account. Trends Cogn. Sci. 11, 307–317 (2007).
Moulson, M. C. & Nelson, C. A. Early adverse experiences and the neurobiology of facial emotion processing. Dev. Psychol. (in the press).
Nelson, C. A., Parker, S. W. & Guthrie, D. The discrimination of facial expressions by typically developing infants and toddlers and those experiencing early institutional care. Infant Behav. Dev. 29, 210–219 (2006).
Widen, S. C. & Russell, J. A. A closer look at preschoolers' freely produced labels for facial expressions. Dev. Psychol. 39, 114–128 (2003).
Cunningham, M. G., Bhattacharyya, S. & Benes, F. M. Amygdalo-cortical sprouting continues into early adulthood: implications for the development of normal and abnormal function during adolescence. J. Comp. Neurol. 453, 116–130 (2002).
Hariri, A. R., Bookheimer, S. Y. & Mazziotta, J. C. Modulating emotional responses: effects of a neocortical network on the limbic system. Neuroreport 11, 43–48 (2000).
Kim, H. et al. Contextual modulation of amygdala responsivity to surprised faces. J. Cogn. Neurosci. 16, 1730–1743 (2004).
Cohen Kadosh, K. & Johnson, M. H. Developing a cortex specialized for face perception. Trends Cogn. Sci. 11, 367–369 (2007).
Humphrey, T. The development of the human amygdala during early embryonic life. J. Comp. Neurol. 132, 135–165 (1968).
Kordower, J. H., Piecinski, P. & Rakic, P. Neurogenesis of the amygdaloid nuclear complex in the rhesus monkey. Brain Res. Dev. Brain Res. 68, 9–15 (1992).
Nikolic, I. & Kostovic, I. Development of the lateral amygdaloid nucleus in the human fetus: transient presence of discrete cytoarchitectonic units. Anat. Embryol. (Berl.) 174, 355–360 (1986).
Bachevalier, J., Ungerleider, L. G., O'Neill, J. B. & Friedman, D. P. Regional distribution of [3H] naloxone binding in the brain of a newborn rhesus monkey. Brain Res. 390, 302–308 (1986).
Berger, B., Febvret, A., Greengard, P. & Goldman-Rakic, P. S. DARPP-32, a phosphoprotein enriched in dopaminoceptive neurons bearing dopamine D1 receptors: distribution in the cerebral cortex of the newborn and adult rhesus monkey. J. Comp. Neurol. 299, 327–348 (1990).
Prather, M. D. & Amaral, D. G. The development and distribution of serotonergic fibers in the macaque monkey amygdala. Soc. Neurosci. Abstr. 26, 1727 (2000).
Webster, M. J., Bachevalier, J. & Ungerleider, L. G. Connections of inferior temporal area TEO and TE with parietal and frontal cortex in macaque monkeys. Cereb. Cortex 5, 470–483 (1994).
Webster, M. J., Ungerleider, L. G. & Bachevalier, J. Connections of inferior temporal areas TE and TEO with medial temporal-lobe structures in infant and adult monkeys. J. Neurosci. 11, 1095–1116 (1991).
Rodman, H. R. & Consuelos, M. J. Cortical projections to anterior inferior temporal cortex in infant macaque monkeys. Vis. Neurosci. 11, 119–133 (1994).
Afraz, S.-R., Kiani, R. & Esteky, H. Microstimulation of inferotemporal cortex influences face categorization. Nature 442, 692–695 (2006).
Hasselmo, M. E., Rolls, E. T. & Baylis, G. C. The role of expression and identity in the face-selective responses of neurons in the temporal visual cortex of the monkey. Behav. Brain Res. 32, 203–218 (1989).
Sugase, Y., Yamane, S., Ueno, S. & Kawano, K. Global and fine information coded by single neurons in the temporal visual cortex. Nature 400, 869–873 (1999).
Tsao, D. Y., Freiwald, W. A., Tootell, R. B. H. & Livingstone, M. S. A cortical region consisting entirely of face-selective cells. Science 311, 670–674 (2006).
Bentin, S., Allison, T., Puce, A., Perez, E. & McCarthy, G. Electrophysiological studies of face perception in humans. J. Cogn. Neurosci. 8, 551–565 (1996).
Hoffman, E. A. & Haxby, J. V. Distinct representations of eye gaze and identity in the distributed human neural system for face perception. Nature Neurosci. 3, 80–84 (2000).
Kanwisher, N., McDermott, J. & Chun, M. M. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J. Neurosci. 17, 4302–4311 (1997).
LaBar, K. S., Crupain, M. J., Voyvodic, J. T. & McCarthy, G. Dynamic perception of facial affect and identity in the human brain. Cereb. Cortex 13, 1023–1033 (2003).
Engell, A. D. & Haxby, J. V. Facial expression and gaze-direction in human superior temporal sulcus. Neuropsychologia 45, 3234–3241 (2007).
Haxby, J. V., Hoffman, E. A. & Gobbini, M. I. Human neural systems for face recognition and social communication. Biol. Psychiatry 15, 59–67 (2002).
Calder, A. J. & Young, A. W. Understanding the recognition of facial identity and facial expression. Nature Rev. Neurosci. 6, 641–651 (2005).
Calder, A. J., Young, A. W., Keane, J. & Dean, M. Configural information in facial expression perception. J. Exp. Psychol. Hum. Percept. Perform. 26, 527–551 (2000).
Ambadar, Z., Schooler, J. W. & Cohn, J. F. Deciphering the enigmatic face: the importance of facial dynamics in interpreting subtle facial expressions. Psychol. Sci. 16, 403–410 (2005).
Adams, R. B. J. & Kleck, R. E. Perceived gaze direction and the processing of facial displays of emotion. Psychol. Sci. 14, 644–647 (2003).
Campanella, S. & Belin, P. Integrating face and voice in person perception. Trends Cogn. Sci. 11, 535–543 (2007).
Kreifelts, B., Ethofer, T., Grodd, W., Erb, M. & Wildgruber, D. Audiovisual integration of emotional signals in voice and face: an event-related fMRI study. Neuroimage 37, 1445–1456 (2007).
Puce, A. & Perrett, D. Electrophysiology and brain imaging of biological motion. Philos. Trans. R. Soc. Lond. B Biol. Sci. 358, 435–445 (2003).
Möttönen, R. et al. Perceiving identical sounds as speech or non-speech modulates activity in the left posterior superior temporal sulcus. Neuroimage 30, 563–569 (2006).
Hein, G. & Knight, R. T. Superior temporal sulcus - it's my area: or is it? J. Cogn. Neurosci. 20, 1–12 (2008).
Redcay, E. The superior temporal sulcus performs a common function for social and speech perception: implications for the emergence of autism. Neurosci. Biobehav. Rev. 32, 123–142 (2008).
Phelps, E. A., Ling, S. & Carrasco, M. Emotion facilitates perception and potentiates the perceptual benefits of attention. Psychol. Sci. 17, 292–299 (2006).
Acknowledgements
C.A.N. gratefully acknowledges support from the US National Institutes of Health (MH078829) and the Richard David Scott endowment; J.M.L. acknowledges financial support from the Academy of Finland (grant number 1115536). The authors thank J. Kagan for his comments on an earlier draft of this article.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Universal facial expressions
-
A limited set of emotional facial expressions that seem to be recognized universally (by members of different cultures).
- Magnocellular pathway
-
A system of vision-responsive cells that are characterized by large axons and the rapid transmission of nerve impulses.
- Near-infra-red spectroscopy
-
A technique used for measuring changes in haemoglobin concentrations and oxygenation levels that can be used to study changes in brain activity in localized cortical regions.
- Experience-expectant mechanisms
-
Evolved neural mechanisms and plasticity for processing and storing information that is expected to be common to all members of the species; for example, patterned light, faces, speech and language.
- Experience-dependent mechanisms
-
Neural mechanisms and plasticity for processing information that is unique to the individual; for example, individual differences in processing cognitive, linguistic or social challenges. In the cognitive sphere, for example, learning and memory reflect an experience-dependent process.
- Perceptual narrowing
-
A developmental process in which discrimination of frequently encountered stimulus features is maintained and improved whereas discrimination of less frequently encountered stimulus features is diminished or lost; for example, the gradual loss of the ability to discriminate non-native speech contrasts while retaining the ability to discriminate native speech contrasts.
Rights and permissions
About this article
Cite this article
Leppänen, J., Nelson, C. Tuning the developing brain to social signals of emotions. Nat Rev Neurosci 10, 37–47 (2009). https://doi.org/10.1038/nrn2554
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn2554
This article is cited by
-
Seeing emotions in the eyes: a validated test to study individual differences in the perception of basic emotions
Cognitive Research: Principles and Implications (2023)
-
Effects of COVID-19 pandemic on structural brain development in early adolescence
Scientific Reports (2023)
-
Parent to Offspring Fear Transmission via Modeling in Early Life: A Systematic Review and Meta-Analysis
Clinical Child and Family Psychology Review (2023)
-
The role of maternal trauma and discipline types in emotional processing among Syrian refugee children
European Child & Adolescent Psychiatry (2023)
-
Does the Emotional Modulation of Visual Experience Entail the Cognitive Penetrability of Early Vision?
Review of Philosophy and Psychology (2023)