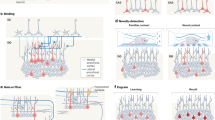
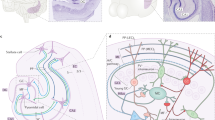
Key Points
-
As a main component of the medial temporal lobe, the hippocampus has been shown to be crucial for the formation of declarative memory in humans. In rodents, the hippocampus has several features that facilitate a multilevel analysis of its function: it is required for spatial learning, which can be directly assessed with tasks such as the Morris water maze task; it demonstrates changes in synaptic efficacy, such long-term potentiation (LTP), after high-frequency input; and individual hippocampal pyramidal cells fire in a place-specific manner as an animal moves through an environment, allowing direct observation of the quality of spatial encoding.
-
The N-methyl-D-aspartate receptor (NMDAR), which is highly expressed in the hippocampus, has been identified as an ideal molecular coincidence detector owing to its voltage-dependent magnesium block, high calcium permeability and slow activation and deactivation kinetics. The demonstration that the induction of hippocampal LTP depends on the activation of NMDARs further strengthened the link between LTP and Hebb's synaptic hypothesis for memory storage, in which modifications of synaptic efficacy by coincident input was the central theme
-
Both pharmacological and genetic approaches have shown that hippocampal NMDARs, particularly in region CA1, are required for the acquisition of spatial memories. Furthermore, deletion of NMDARs specifically in CA1 pyramidal cells leads to a loss of the coordinated activity of CA1 place cells, with overlapping fields and a disruption of the ensemble code for space.
-
NMDAR-mediated plasticity in the recurrent connections in area CA3 of the hippocampus is crucial for the rapid encoding of novel experiences, in a process that might be akin to episodic memory formation in humans. CA3-NR1-knockout mice are deficient in acquiring novel place/reward location information, and CA1 place cells in these mice were significantly impaired when recorded in a novel environment (with enlarged place fields and an augmented integrated firing rate).
-
Memory retrieval is an associative process that might involve recurrent network activation as a means of pattern completion — the recall of an entire memory based on exposure to a partial set of cues. A loss of synaptic plasticity in the recurrent collaterals of area CA3 prevents this form of memory retrieval, as assessed by a spatial learning task in CA3-NR1-knockout mice, and is manifested as uncoordinated, spatially less-tuned CA1 place-cell activity under partial-cue conditions.
-
Hippocampal place cells can be viewed as memory traces at the neuronal ensemble level, showing stable patterns of firing that can develop quickly and can be reactivated independently of behaviour. CA3-NR1-knockout mice have allowed the direct observation of a fourth important property of a putative memory trace — the experience-dependent formation of the spatial specificity of the fields.
-
Progress has been made in understanding the part that NMDARs play in hippocampal memory, both generally, and specifically in terms of where (which subfield/cell type) and when (which phase of memory) the receptors are needed. Furthermore, the development of genetic techniques, including inducible and reversible cell-type-specific gene activation, and the establishment of more refined behavioural and physiological analyses will lead to even deeper comprehension of the relationship between plasticity, memory and space in the hippocampus.
Abstract
N-methyl-D-aspartate receptors (NMDARs) in the rodent hippocampus have been shown to be essential for spatial learning and memory, and for the induction of long-term synaptic plasticity at various hippocampal synapses. In this review, we examine the evidence concerning the role of NMDARs in hippocampal memory processes, with an emphasis on the function of NMDARs in area CA1 of the hippocampus in memory acquisition, and the unique role of NMDARs in area CA3 in the rapid acquisition and associative retrieval of spatial information. Finally, we discuss the data that have emerged from in vivo hippocampal recording studies that indicate that the activity of hippocampal place cells during behaviour is an expression of a memory trace.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Scoville, W. B. & Milner, B. Loss of recent memory after bilateral hippocampal lesions. J. Neurol. Neurosurg. Psychiatry 20, 11–21 (1957).
Schmolck, H., Kensinger, E. A., Corkin, S. & Squire, L. R. Semantic knowledge in patient H. M. and other patients with bilateral medial and lateral temporal lobe lesions. Hippocampus 12, 520–533 (2002).
Rempel-Clower, N. L., Zola, S. M., Squire, L. R. & Amaral, D. G. Three cases of enduring memory impairment after bilateral damage limited to the hippocampal formation. J. Neurosci. 16, 5233–5255 (1996).
Zola, S. M. & Squire, L. R. Relationship between magnitude of damage to the hippocampus and impaired recognition memory in monkeys. Hippocampus 11, 92–98 (2001).
Jarrard, L. E. On the role of the hippocampus in learning and memory in the rat. Behav. Neural Biol. 60, 9–26 (1993).
Morris, R. G. M., Garrud, P., Rawlins, J. N. P. & O'Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 297, 681–683 (1982).
Silva, A. J., Giese, K. P., Fedorov, N. B., Frankland, P. W. & Kogan, J. H. Molecular, cellular, and neuroanatomical substrates of place learning. Neurobiol. Learn. Mem. 70, 44–61 (1998).
O'Keefe, J. & Dostrovsky, J. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res. 34, 171–175 (1971).
Bliss, T. V. P. & Lømo, T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J. Physiol. (Lond.) 232, 331–356 (1973).
Hebb, D. O. The Organization of Behavior: A Neuropsychological Theory (John Wiley, New York, 1949).
Collingridge, G. L., Kehl, S. J. & McLennon, H. Excitatory amino acids in synaptic transmission in the Schaffer collateral-commissural pathway of the rat hippocampus. J. Physiol. (Lond.) 334, 33–46 (1983). The first demonstration that long-term potentiation in the hippocampus is NMDAR-dependent.
Mayer, M. L., Westbrook, G. L. & Guthrie, P. B. Voltage-dependent block by Mg2+ of NMDA responses in spinal cord neurones. Nature 309, 261–263 (1984).
MacDermott, A. B., Mayer, M. L., Westbrook, G. L., Smith, S. J. & Barker, J. L. NMDA-receptor activation increases cytoplasmic calcium concentration in cultured spinal cord neurones. Nature 321, 519–522 (1986).
Nowak, L., Bregestovski, P., Ascher, P., Herbert, A. & Prochiantz, A. Magnesium gates glutamate-activated channels in mouse central neurones. Nature 307, 462–465 (1984). This study utilized the patch-clamp technique to provide the first evidence of a voltage-dependent Mg2+ block of the NMDA Ca2+ channel
Lester, R. A., Clements, J. D., Westbrook, G. L. & Jahr, C. E. Channel kinetics determine the time course of NMDA receptor-mediated synaptic currents. Nature 346, 565–567 (1990).
Morris, R. G. M., Anderson, E., Lynch, G. S. & Baudry, M. Selective impairment of learning and blockade of long-term potentiation by an N-methyl-D-aspartate receptor antagonist, AP5. Nature 319, 774–776 (1986). This paper was the first to show that NMDAR activity is needed for spatial learning and to draw parallels between NMDAR function, hippocampal synaptic plasticity and spatial learning.
Moriyoshi, K. et al. Molecular cloning and characterization of the rat NMDA receptor. Nature 354, 31–37 (1991). The first identification of a gene encoding a subunit of the NMDAR, which paved the way for much of the later genetic and structural work.
Dunah, A. W. et al. Biochemical studies of the structure and function of the N-methyl-D-aspartate subtype of glutamate receptors. Mol. Neurobiol. 19, 151–179 (1999).
Cull-Candy, S., Brickley, S. & Farrant, M. NMDA receptor subunits: diversity, development and disease. Curr. Opin. Neurobiol. 11, 327–335 (2001).
Li, Y., Erzurumulu, R., Chen, C., Jhaveri, S. & Tonegawa, S. Whisker-related neuronal patterns fail to develop in the trigeminal brainstem nuclei of NMDAR1 knockout mice. Cell 76, 427–437 (1994).
Forrest, D. et al. Targeted disruption of NMDA receptor 1 gene abolishes NMDA response and results in neonatal death. Neuron 13, 325–338 (1994).
Amaral, D. G. & Witter, M. P. The three-dimensional organization of the hippocampal formation: a review of anatomical data. Neuroscience 31, 571–591 (1989).
Miles, R. & Wong, R. K. Excitatory synaptic interactions between CA3 neurones in the guinea-pig hippocampus. J. Physiol. (Lond.) 373, 397–418 (1986).
MacVicar, B. A. & Dudek, F. E. Local synaptic circuits in rat hippocampus: interactions between pyramidal cells. Brain Res. 184, 220–223 (1980).
McClelland, J. L. & Goddard, N. H. Considerations arising from a complementary learning systems perspective on hippocampus and neocortex. Hippocampus 6, 654–665 (1996).
McNaughton, B. L. & Morris, R. G. M. Hippocampal synaptic enhancement and information storage within a distributed memory system. Trends Neurosci. 10, 408–415 (1987).
O'Reilly, R. C. & McClelland, J. L. Hippocampal conjunctive encoding, storage, and recall: avoiding a trade-off. Hippocampus 4, 661–682 (1994).
Marr, D. Simple memory: a theory for archicortex. Philos. Trans. R. Soc. London B 262, 23–81 (1971). A seminal work that laid the foundation of much of our current understanding of hippocampal computation. Marr's models include a discussion of partial-cue-based recall and experience-dependent modifications of synaptic strength.
O'Reilly, R. C. & Rudy, J. W. Conjunctive representations in learning and memory: principles of cortical and hippocampal function. Psychol. Rev. 108, 311–345 (2001).
Morris, R. G. M. Spatial localization does not require the presence of local cues. Learn. Motiv. 12, 239–260 (1981).
Davis, S., Butcher, S. P. & Morris, R. G. M. The NMDA receptor antagonist D-2-amino-5-phosphonopentanoate (D-AP5) impairs spatial learning and LTP in vivo at intracerebral concentrations comparable to those that block LTP in vitro. J. Neurosci. 12, 21–34 (1992).
Morris, R. G. M. Synaptic plasticity and learning: selective impairment of learning in rats and blockade of long-term potentiation in vivo by the N-methyl-D-aspartate receptor antagonist AP5. J. Neurosci. 9, 3040–3057 (1989).
Riedel, G. et al. Reversible neural inactivation reveals hippocampal participation in several memory processes. Nature Neurosci. 2, 898–905 (1999).
Abraham, W. C. & Kairiss, E. W. Effects of the NMDA antagonist 2AP5 on complex spike discharge by hippocampal pyramidal cells. Neurosci. Lett. 89, 36–42 (1988).
Errington, M. L., Lynch, M. A. & Bliss, T. V. Long-term potentiation in the dentate gyrus: induction and increased glutamate release are blocked by D(–)aminophosphonovalerate. Neuroscience 20, 279–284 (1987).
Morris, R. G. M., Davis, S. & Butcher, S. P. Hippocampal synaptic plasticity and NMDA receptors: a role in information storage? Phil. Trans. R. Soc. Lond. B 329, 187–204 (1990).
Hargreaves, E. L. & Cain, D. P. Hyperactivity, hyper-reactivity, and sensorimotor deficits induced by low doses of the N-methyl-D-aspartate non-competitive channel blocker MK801. Behav. Brain Res. 47, 23–33 (1992).
Sternberg, N., Hamilton, D. & Hoess, R. Bacteriophage P1 site-specific recombination. II. Recombination between loxP and the bacterial chromosome. J. Mol. Biol. 150, 487–507 (1981).
Hoess, R. H., Ziese, M. & Sternberg, N. P1 site-specific recombination: nucleotide sequence of the recombining sites. Proc. Natl Acad. Sci. USA 79, 3398–3402 (1982).
Tsien, J. Z., Huerta, P. T. & Tonegawa, S. The essential role of hippocampal CA1 NMDA receptor-dependent synaptic plasticity in spatial memory. Cell 87, 1327–1338 (1996). This paper offered the first genetic demonstration of the effect of a brain-subregion-restricted NMDA receptor knockout on spatial memory.
McHugh, T. J., Blum, K. I., Tsien, J. Z., Tonegawa, S. & Wilson, M. A. Impaired hippocampal representation of space in CA1-specific NMDAR1 knockout mice. Cell 87, 1339–1349 (1996). This study was the first to show impaired coherent firing of CA1 place cells using genetically engineered NMDAR-knockout mice, demonstrating the powerful combination of conditional transgenic technology and multi-electrode recording.
Fukaya, M., Kato, A., Lovett, C., Tonegawa, S. & Watanabe, M. Retention of NMDA receptor NR2 subunits in the lumen of endoplasmic reticulum in targeted NR1 knockout mice. Proc. Natl Acad. Sci. USA 100, 4855–4860 (2003).
Huerta, P. T., Sun, L. D., Wilson, M. A. & Tonegawa, S. Formation of temporal memory requires NMDA receptors within CA1 pyramidal neurons. Neuron 25, 473–480 (2000).
Rondi-Reig, L., Libbey, M., Eichenbaum, H. & Tonegawa, S. CA1-specific N-methyl-D-aspartate receptor knockout mice are deficient in solving a nonspatial transverse patterning task. Proc. Natl Acad. Sci. USA 98, 3543–3548 (2001).
Rampon, C. et al. Enrichment induces structural changes and recovery from nonspatial-memory deficits in CA1-NMDAR1-knockout mice. Nature Neurosci. 3, 238–244 (2000).
Bannerman, D. M., Good, M. A., Butcher, S. P., Ramsay, M. & Morris, R. G. Distinct components of spatial learning revealed by prior training and NMDA receptor blockade. Nature 378, 182–186 (1995).
Saucier, D. & Cain, D. P. Spatial learning without NMDA receptor-dependent long-term potentiation. Nature 378, 186–189 (1995). References 46 and 47 provided strong evidence that NMDARs could be made dispensable for learning the water maze task if animals were pretrained on an identical spatial or even similar, non-spatial task. This was the first real evidence that the water maze task might involve several types of learning, some of which require NMDARs in the hippocampus and some of which do not.
Roesler, R. et al. Intrahippocampal infusion of the NMDA receptor antagonist AP5 impairs retention of an inhibitory avoidance task: protection from impairment by pretraining or preexposure to the task apparatus. Neurobiol. Learn. Mem. 69, 87–91 (1998).
Hoh, T., Beiko, J., Boon, F., Weiss, S. & Cain, D. P. Complex behavioral strategy and reversal learning in the watermaze without NMDA receptor-dependent long-term potentiation. J. Neurosci. 19, RC2(1–5) (1999).
Steele, R. J. & Morris, R. G. M. Delay-dependent impairment of a matching-to-place task with chronic and intrahippocampal infusion of the NMDA-antagonist D-AP5. Hippocampus 9, 118–136 (1999). This study showed that a delayed matching-to-place task in the water maze requires hippocampal NMDA receptors in rats, providing new insights into the involvement of the hippocampus in episodic-like memory.
Tulving, E. in Organization of Memory (eds Tulving, E. & Donaldson, W.) 381–403 (Academic, New York, 1972).
Tulving, E. Episodic memory: from mind to brain. Annu. Rev. Psychol. 53, 1–25 (2002).
Tulving, E. & Markowitsch, H. J. Episodic and declarative memory: role of the hippocampus. Hippocampus 8, 198–204 (1998).
Clayton, N. S. & Dickinson, A. Episodic-like memory during cache recovery by scrub jays. Nature 395, 272–274 (1998). Taking advantage of the natural caching behaviour of the scrub jay, these authors provide the strongest evidence for the existence of 'what, where and when (episodic-like) memory' in animals.
Clayton, N. S., Bussey, T. J. & Dickinson, A. Can animals recall the past and plan for the future? Nature Rev. Neurosci. 4, 685–691 (2003).
Morris, R. G. M. Episodic-like memory in animals: psychological criteria, neural mechanisms and the value of episodic-like tasks to investigate animal models of neurodegenerative diseases. Philos. Trans. R. Soc. Lond. B 356, 1453–1465 (2001).
Moser, M. B. & Moser, E. I. Pretraining and the function of hippocampal long-term potentiation. Neuron 26, 559–561 (2000).
Miles, R. & Traub, R. Excitatory synaptic interactions between CA3 neurons in the guinea-pig hippocampus. J. Physiol. (Lond.) 373, 397–418 (1986).
MacVicar, B. & Dudek, F. Local synaptic circuits in rat hippocampus: interaction between pyramidal cells. Brain Res. 184, 220–223 (1980).
Harris, E. W. & Cotman, C. W. Long-term potentiation of guinea pig mossy fiber response is not blocked by N-methyl-D-aspartate antagonist. Neurosci. Lett. 70, 132–137 (1986).
Williams, S. & Johnston, D. Muscarinic depression of long-term potentiation in CA3 hippocampal neurons. Science 242, 84–87 (1988).
Zalutsky, R. A. & Nicoll, R. A. Comparison of two forms of long-term potentiation in single hippocampal neurons. Science 248, 1619–1624 (1990).
Berger, T. W. & Yeckel, M. F. in Long-Term Potentiation: A Debate of Current Issues (eds Baudry, M. & Davis, J. L.) 327–356 (MIT Press, Cambridge, Massachusetts, 1991).
Berzhanskaya, J., Urban, N. N. & Barrionuevo, G. Electrophysiological and pharmacological characterization of the direct perforant path input to hippocampal area CA3. J. Neurophysiol. 79, 2111–2118 (1998).
McClelland, J. L., McNaughton, B. L., O'Reilly, R. C. & Nadel, L. Complementary roles of hippocampus and neocortex in learning and memory. Soc. Neurosci. Abstr. 18, 1216 (1992).
McClelland, J. L., McNaughton, B. L. & O'Reilly, R. C. Why there are complementary learning systems in the hippocampus and neocortex: insights from the success and failures of connectionist models of learning and memory. Psychol. Rev. 102, 419–457 (1995). The authors of references 65 and 66 formulated the theoretical basis of complementary learning systems of hippocampus and neocortex, which allows the brain to rapidly learn new on-line events without disrupting old off-line information, and to integrate these new events properly with previously stored experiences.
Lee, I. & Kesner, R. P. Differential contribution of NMDA receptors in hippocampal subregions to spatial working memory. Nature Neurosci. 5, 162–168 (2002).
Nakazawa, K. et al. Requirement for hippocampal CA3 NMDA receptors in acquisition and recall of associative memory. Science 297, 211–218 (2002). This study combines regionally restricted genetic modification with in vivo physiology and behaviour to provide strong evidence that NMDARs in region CA3 are required for memory recall by pattern completion.
Nakazawa, K. et al. Hippocampal CA3 NMDA receptors are crucial for memory acquisition of one-time experience. Neuron 38, 305–315 (2003).
Vinogradova, O. S. in The Hippocampus (eds Isaacson, R. L. & Pribram, K. H.) 3–69 (Plenum, New York and London, 1975).
McNaughton, B. L., Barnes, C. A., Meltzer, J. & Sutherland, R. J. Hippocampal granule cells are necessary for normal spatial learning but not for spatially-selective pyramidal cell discharge. Exp. Brain Res. 76, 485–496 (1989).
Brun, V. H. et al. Place cells and place recognition maintained by direct entorhinal-hippocampal circuitry. Science 296, 2243–2246 (2002).
Barnes, C. A., McNaughton, B. L., Mizumori, S. J., Leonard, B. W. & Lin, L. -H. Comparison of spatial and temporal characteristics of neuronal activity in sequential stages of hippocampal processing. Prog. Brain Res. 83, 287–300 (1990).
Frank, L. M., Brown, E. N. & Wilson, M. A. Trajectory encoding in the hippocampus and entorhinal cortex. Neuron 27, 169–178 (2000).
Quirk, G. J., Muller, R. U., Kubie, J. L. & Ranck, J. B. Jr. The positional firing properties of medial entorhinal neurons: description and comparison with hippocampal place cells. J. Neurosci. 12, 1945–1963 (1992).
Moser, E. I. & Moser, M. B. One-shot memory in hippocampal CA3 networks. Neuron 38, 147–148 (2003).
Day, M., Langston, R. & Morris, R. G. Glutamate-receptor-mediated encoding and retrieval of paired-associate learning. Nature 424, 205–209 (2003).
Ebbinghaus, H. Memory: A Contribution to Experimental Psychology (Dover, New York, 1885).
Müller, G. E. & Pilzecker, A. Experimentelle Beitrage zur Lehre vom Gedachtnis. Z. Psychol. Erganzungsband 1, 1–300 (1900).
McGaugh, J. L. Time-dependent processes in memory storage. Science 153, 1351–1358 (1966).
McGaugh, J. L. Memory: a century of consolidation. Science 287, 248–251 (2000).
Nader, K. Memory traces unbound. Trends Neurosci. 26, 65–72 (2003).
Squire, L. R. Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans. Psychol. Rev. 99, 195–231 (1992).
Squire, L. R. & Alvarez, P. Retrograde amnesia and memory consolidation: a neurobiological perspective. Curr. Opin. Neurobiol. 5, 169–177 (1995).
Nadel, L. & Moscovitch, M. Memory consolidation, retrograde amnesia and the hippocampal complex. Curr. Opin. Neurobiol. 7, 217–227 (1997).
Bontempi, B., Lauren-Demir, C., Destrade, C. & Jaffard, R. Time-dependent reorganization of brain circuitry underlying long-term memory storage. Nature 400, 671–675 (1999).
Davis, H. P. & Squire, L. R. Protein synthesis and memory: a review. Psych. Bull. 96, 518–559 (1984).
DeZazzo, J. & Tully, T. Dissection of memory formation: from behavioral pharmacology to molecular genetics. Trends Neurosci. 18, 212–218 (1995).
Bailey, C. H., Bartsch, D. & Kandel, E. R. Toward a molecular definition of long-term memory storage. Proc. Natl Acad. Sci. USA 93, 13445–13452 (1996).
Nader, K., Schafem, G. E. & LeDoux, J. E. The labile nature of consolidation theory. Nature Rev. Neurosci. 1, 216–219 (2000).
Abel, T. & Kandel, E. Positive and negative regulatory mechanisms that mediate long-term memory storage. Brain Res. Rev. 26, 360–378 (1998).
Adams, J. P. & Sweatt, J. D. Molecular psychology: roles for the ERK MAP kinase cascade in memory. Annu. Rev. Pharmacol. Toxicol. 42, 135–163 (2002).
Dudai, Y. Molecular bases of long-term memories: a question of persistence. Curr. Opin. Neurobiol. 12, 212–216 (2002).
Bozon, B. et al. MAPK, CREB and zif268 are all required for the consolidation of recognition memory. Philos. Trans. R. Soc. Lond. B 358, 805–814 (2003).
Kang, H. et al. An important role of neural activity-dependent CaMKIV signaling in the consolidation of long-term memory. Cell 106, 771–783 (2001).
Kida, S. et al. CREB required for the stability of new and reactivated fear memories. Nature Neurosci. 5, 348–355 (2002).
Schafe, G. E., Nader, K., Blair, H. T. & LeDoux, J. E. Memory consolidation of Pavlovian fear conditioning: a cellular and molecular perspective. Trends Neurosci. 24, 540–546 (2001).
Taubenfeld, S. M., Milekic, M. H., Monti, B. & Alberini, C. M. The consolidation of new but not reactivated memory requires hippocampal C/EBPb. Nature Neurosci. 4, 813–818 (2001).
Abel, T. & Lattal, K. M. Molecular mechanisms of memory acquisition, consolidation and retrieval. Curr. Opin. Neurobiol. 11, 180–187 (2001).
Izquierdo, I. & Medina, J. H. Memory formation: the sequence of biochemical events in the hippocampus and its connection to activity in other brain structures. Neurobiol. Learn. Mem. 68, 285–316 (1997).
Steward, O. & Schuman, E. M. Protein synthesis at synaptic sites on dendrites. Annu. Rev. Neurosci. 24, 299–325 (2001).
Kelleher, R. J., Govindarajan, A., Jung H. -Y., Kang, H. & Tonegawa, S. Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell 116, 467–479 (2004).
Wu, L. et al. CPEB-mediated cytoplasmic polyadenylation and the regulation of experience-dependent translation of α-CaMKII mRNA at synapses. Neuron 21, 1129–1139 (1998).
Heale, V. & Harley, C. MK-801 and AP5 impair acquisition, but not retention, of the Morris milk maze. Pharmacol. Biochem. Behav. 36, 145–149 (1990).
Norris, C. M. & Foster, T. C. MK-801 improves retention in aged rats: implications for altered neural plasticity in age-related memory deficits. Neurobiol. Learn. Mem. 71, 194–206 (1999).
Packard, M. G. & Teather, L. A. Double dissociation of hippocampal and dorsal-striatal memory systems by posttraining intracerebral injections of 2-amino-5-phosphonopentanoic acid. Behav. Neurosci. 111, 543–551 (1997).
Packard, M. G. & Teather, L. A. Posttraining injections of MK-801 produce a time-dependent impairment of memory in two water maze tasks. Neurobiol. Learn. Mem. 68, 42–50 (1997).
Shimizu, E., Tang, Y. P., Rampon, C. & Tsien, J. Z. NMDA receptor-dependent synaptic reinforcement as a crucial process for memory consolidation. Science 290, 1170–1174 (2001).
Gossen, M. & Bujard, H. Anhydrotetracycline, a novel effector for tetracycline controlled gene expression systems in eukaryotic cells. Nucleic Acids Res. 21, 4411–4412 (1993).
Stäubli, U., Thibault, O., DiLorenzo, M. & Lynch, G. Antagonism of NMDA receptors impairs acquisition but not retetion of olfactory memory. Behav. Neurosci. 103, 54–60 (1989).
Parada-Turska, J. & Turski, W. A. Excitatory amino acid antagonists and memory: effect of drugs acting at N-methyl-D-aspartate receptors in learning and memory tasks. Neuropharmacology 29, 1111–1116 (1990).
Venable, N. & Kelly, P. H. Effects of NMDA receptor antagonists on passive avoidance learning and retrieval in rats and mice. Psychopharmacology (Berl.) 100, 215–221 (1990).
Szapiro, G. et al. Participation of hippocampal metabotropic glutamate receptors, protein kinase A and mitogen-activated protein kinases in memory retrieval. Neuroscience 99, 1–5 (2000).
Liang, K. C., Hon, W. & Davis, M. Pre- and posttraining infustion of N-methyl-D-aspartate receptor antagonists into the amygdala impair memory in an inhibitory avoidance task. Behav. Neurosci. 108, 241–253 (1994).
Martin, S. J., Grimwood, P. D. & Morris, R. G. M. Synaptic plasticity and memory: an evaluation of the hypothesis. Annu. Rev. Neurosci. 23, 649–711 (2000).
Martin, S. J. & Morris, R. G. M. New life in an old idea: the synaptic plasticity and memory hypothesis revisited. Hippocampus 12, 609–636 (2002).
Chomsky, N. Syntactic Structures (Mouton, The Hague, 1957).
Gardner-Medwin, A. R. The recall of events through the learning of associations between their parts. Proc. R. Soc. Lond. B 194, 375–402 (1976).
Hopfield, J. J. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl Acad. Sci. USA 79, 2554–2558 (1982).
Rolls, E. T. in The Computing Neuron (eds Durbin, R., Maill, C. & Mitchison, G.) 125–159 (Addison-Wesley, Wokingham, UK, 1989).
Willshaw, D. J. & Buckingham, J. T. An assessment of Marr's theory of the hippocampus as a temporary memory storage. Phil. Trans. R. Soc. Lond. B 329, 205–215 (1990).
Hasselmo, M. E., Schnell, E. & Barkai, E. Dynamics of learning and recall at excitatory recurrent synapses and cholinergic modulation in rat hippocampal region CA3. J. Neurosci. 15, 5249–5262 (1995).
O'Keefe, J. & Nadel, L. The Hippocampus as a Cognitive Map (Clarendon, Oxford, 1978). This book uses both the animal and the human literature to outline a theory concerning the role of the hippocampus in spatial representation. The ideas outlined in this work have been the basis for countless experiments in the field and continue to generate important discussion.
Wilson, M. A. & McNaughton, B. L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676–679 (1994).
Wilson, M. A. & McNaughton, B. L. Dynamics of the hippocampal ensemble code for space. Science 261, 1055–1058 (1993). Utilizing multiple recording tetrodes, these authors monitored the simultaneous activity of more than one-hundred CA1 neurons as a rat explored the environment. This study was the first to look at hippocampal spatial activity on the level of a network and also the first to demonstrate the establishment of a spatial code in a novel environment.
Eichenbaum, H., Dudchenko, P., Wood, W., Shapiro, M. & Tanila, H. The hippocampus, memory, and place cells: is it spatial memory or a memory space? Neuron 23, 209–226 (1999).
Lee, A. K. & Wilson, M. A. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron 36, 1183–1194 (2002).
Louie, K. & Wilson, M. A. Temporally structured replay of awake hippocampal ensemble activity during rapid eye movement sleep. Neuron 29, 145–156 (2001).
Poucet, B., Save, E. & Lenck-Santini, P. P. Sensory and memory properties of hippocampal place cells. Rev. Neurosci. 11, 95–111 (2000).
Moser, E. I. & Paulsen, O. New excitement in cognitive space: between place cells and spatial memory. Curr. Opin. Neurobiol. 11, 745–751 (2001). The authors reviewed recent evidence connecting hippocampal place cell activity with its mnemonic function.
Skaggs, W. E. & McNaughton, B. L. Replay of neuronal firing sequences in rat hippocampus during sleep following spatial experience. Science 271, 1870–1873 (1996).
Kudrimoti, H., Barnes, C. A. & McNaughton, B. L. Reactivation of hippocampal cell assemblies: effects of behavioral state, experience, and EEG dynamics. J. Neurosci. 19, 4090–4101 (1999).
Nadasdy, Z., Hirase, H., Czurko, A., Csicsvari, J. & Buzsaki, G. Replay and time compression of recurring spike sequences in the hippocampus. J. Neurosci. 19, 9497–9507 (1999).
Muller, R. U. & Kubie, J. L. The effects of changes in the environment on the spatial firing of hippocampal complex-spike cells. J. Neurosci. 7, 1951–1968 (1987).
O'Keefe, J. & Conway, D. H. Hippocampal place units in the freely moving rat: why they fire where they fire. Exp. Brain. Res. 31, 573–590 (1978).
O'Keefe, J. & Speakman, A. Single unit activity in the rat hippocampus during a spatial memory task. Exp. Brain Res. 68, 1–27 (1987).
Pico, R. M., Gerbrandt, L. K., Pondel, M. & Ivy, G. During stepwise cue deletion, rat place behaviors correlate with place unit responses. Brain Res. 330, 369–372 (1985).
Mehta, M. R., Barnes, C. A. & McNaughton, B. L. Experience-dependent, asymmetric expansion of hippocampal place fields. Proc. Natl Acad. Sci. USA 94, 8918–8921 (1997).
Mehta, M. R., Quirk, M. C. & Wilson, M. A. Experience-dependent asymmetric shape of hippocampal receptive fields. Neuron 25, 707–715 (2000).
Kentros, C. et al. Abolition of long-term stability of new hippocampal place cell maps by NMDA receptor blockade. Science 280, 2121–2126 (1998). Combining pharmacology and in vivo recording, the authors show that acute blockade of the NMDAR during the exploration of a novel environment impairs the long-term stability of the newly encoded place fields.
Ekstrom, A. D., Meltzer, J., McNaughton, B. L. & Barnes, C. A. NMDA receptor antagonism blocks experience-dependent expansion of hippcampal 'place fields'. Neuron 31, 631–638 (2001). The authors demonstrated, using the NMDAR antagonist CPP, that experience-dependent asymmetric place field expansion in hippocampal CA1 is NR-dependent, but phase precession on the theta wave is not.
Rotenberg, A., Mayford, M., Hawkins, R. D., Kandel, E. R. & Muller, R. U. Mice expressing activated CaMKII lack low frequency LTP and do not form stable place cells in the CA1 region of the hippocampus. Cell 87, 1351–1361 (1996).
Rotenberg, A., Abel, T., Hawkins, R. D., Kandel, E. R. & Muller, R. U. Parallel instabilities of long-term potentiation, place cells, and learning caused by decreased protein kinase A activity. J. Neurosci. 20, 8096–8102 (2000).
Yan, J. et al. Place-cell impairment in glutamate receptor 2 mutant mice. J. Neurosci. 22, RC204 (2002).
Cho, Y. H., Giese, K. P., Tanila, H., Silva, A. J. & Eichenbaum, H. Abnormal hippocampal spatial representations in aCaMKIIT286A and CREBα/− mice. Science 279, 867–869 (1998).
Muller, R. U., Bostock, E., Taube, J. S. & Kubie, J. L. On the directional firing properties of hippocampal place cells. J. Neurosci. 14, 7235–7251 (1994).
Anderson, M. I. & Jeffery, K. J. Heterogeneous modulation of place cell firing by changes in context. J. Neurosci. 23, 8827–8835 (2003).
Wood, E. R., Dudchenko, P. A., Robitsek, R. J. & Eichenbaum, H. Hippocampal neurons encode information about different types of memory episodes occurring in the same location. Neuron 27, 623–633 (2000).
Young, B. J., Fox, G. D. & Eichenbaum, H. Correlates of hippocampal complex-spike cell activity in rats performing a nonspatial radial maze task. J. Neurosci. 14, 6553–6563 (1994).
Hollup, S. A., Molden, S., Donnett, J. G., Moser, M. B. & Moser, E. I. Accumulation of hippocampal place fields at the goal location in an annular watermaze task. J. Neurosci. 21, 1635–1644 (2001).
Fyhn, M., Molden, S., Hollup, S., Moser, M. -B. & Moser, E. I. Hippocampal neurons responding to first-time dislocation of a target object. Neuron 35, 555–566 (2002).
Rolls, E. T. A theory of hippocampal function in memory. Hippocampus 6, 601–620 (1996).
O'Keefe, J. Hippocampus, theta, and spatial memory. Curr. Opin. Neurobiol. 3, 917–924 (1993).
O'Keefe, J. A review of the hippocampal place cells. Prog. Neurobiol. 13, 419–349 (1979).
Muller, R. U., Stead, M. & Pach, J. The hippocampus as a cognitive graph. J. Gen. Physiol. 107, 663–694 (1996).
Best, P. J., White, A. M. & Minai, A. Spatial processing in the brain: the activity of hippocampal place cells. Annu. Rev. Neurosci. 24, 459–486 (2001).
Thompson, L. T. & Best, P. J. Long-term stability of the place-field activity of single units recorded from the dorsal hippocampus of freely behaving rats. Brain Res. 509, 299–308 (1990).
Muller, R. A quarter of a century of place cells. Neuron 17, 813–822 (1996).
Redish, A. D. et al. Independence of firing correlates of anatomically proximate hippocampal pyramidal cells. J. Neurosci. 21, RC134 (2001).
Branda, C. S. & Dymecki, S. M. Talking about a revolution: The impact of site-specific recombinases on genetic analyses in mice. Dev. Cell 6, 7–28 (2004).
Morozov, A., Kellendonk, C., Simpson, E. & Tronche, F. Using conditional mutagenesis to study the brain. Biol. Psychiatry 54, 1125–1133 (2003).
Grant, S. G. N. et al. Impaired long-term potentiation, spatial learning, and hippocampal development in fyn mutant mice. Science 258, 1903–1909 (1992).
Blendy, J. A., Kaestner, K. H., Schmid, W., Gass, P. & Schutz, G. Targeting of the CREB gene leads to up-regulation of a novel CREB mRNA isoform. EMBO J. 15, 1098–1106 (1996).
Hummler, E. et al. Targeted mutation of the CREB gene: compensation within the CREB/ATF family of transcription factors. Proc. Natl Acad. Sci. USA 91, 5647–5651 (1994).
Mills, A. A. Changing colors in mice: an inducible system that delivers. Genes Dev. 15, 1461–1467 (2001).
Schonig, K. & Bujard, H. Generating conditional mouse mutants via tetracycline-controlled gene expression. Methods Mol. Biol. 209, 69–104 (2003).
Gross, C. et al. Serotonin1A receptor acts during development to establish normal anxiety-like behaviour in the adult. Nature 416, 396–400 (2002).
Chen, J. et al. Transgenic animals with inducible, targeted gene expression in brain. Mol. Pharmacol. 54, 495–503 (1998).
Mayford, M. et al. Control of memory formation through regulated expression of a CaMKII transgene. Science 274, 1678–1683 (1996).
Acknowledgements
This work was supported by an individual NIH grant to S.T. and an NIH Silvio O. Conte Center grant to S.T. and M.A.W., and by grants from the Howard Hughes Medical Institute to S.T. and from the RIKEN-MIT Neuroscience Research Center to S.T. and M.A.W.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- FEAR CONDITIONING
-
A form of Pavlovian (classical) conditioning in which the animal learns that an innocuous stimulus (for example, an auditory tone — the conditioned stimulus or CS), reliably predicts the occurrence of a noxious stimulus (for example, foot shock — the unconditioned stimulus or US) following their repeated paired presentation. As a result of this procedure, presentation of the CS alone elicits conditioned fear responses previously associated with the noxious stimulus only.
- TRANSVERSE PATTERN LEARNING
-
A task in which animals must encode overlapping relationships between cues. A typical stimulus set is A+B−; B+C−; C+A−, where + signifies which cue is rewarded in each configuration.
- STEP-DOWN INHIBITORY AVOIDANCE TASK
-
A form of conditioning in which a rat is placed on a platform and receives a shock when it steps off the platform. Memory for the shock is measured as an increased latency to step off the platform on subsequent trials.
- PAIRED-ASSOCIATE TASK
-
A task that involves the arbitrary association of two stimuli (such as word pairs in humans, or a place and an odour or food item in animals). After exposure to the pair, the subject is presented with one stimulus and tested for recall of the second. It can be used to test declarative memory in humans, or 'episodic-like' memory in animals.
Rights and permissions
About this article
Cite this article
Nakazawa, K., McHugh, T., Wilson, M. et al. NMDA receptors, place cells and hippocampal spatial memory. Nat Rev Neurosci 5, 361–372 (2004). https://doi.org/10.1038/nrn1385
Issue Date:
DOI: https://doi.org/10.1038/nrn1385
This article is cited by
-
A new AAV tool for highly preferentially targeting hippocampal CA2
Molecular Brain (2023)
-
Requirement for hippocampal CA3 NMDA receptors in artificial association of memory events stored in CA3 cell ensembles
Molecular Brain (2023)
-
Blood tissue Plasminogen Activator (tPA) of liver origin contributes to neurovascular coupling involving brain endothelial N-Methyl-D-Aspartate (NMDA) receptors
Fluids and Barriers of the CNS (2023)
-
Loss of Grin2a causes a transient delay in the electrophysiological maturation of hippocampal parvalbumin interneurons
Communications Biology (2023)
-
Platelet factors are induced by longevity factor klotho and enhance cognition in young and aging mice
Nature Aging (2023)