Key Points
-
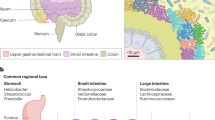
The gut microbiota is spatially stratified along the longitudinal and cross-sectional axes of the gut. Chemical and nutrient gradients, antimicrobial peptides and physical features of the gut contribute to differences in microbial community composition in different locations.
-
The mucosal and lumenal microbiota of the gut represent distinct microbial communities. On a smaller scale, patchiness within these communities suggests that they are highly spatially organized.
-
Diet imparts a large effect on microbial colonization and relative abundance, but some bacteria can thrive independently of dietary changes by living on host-derived nutrients such as mucin glycans. Therefore, the mucus layer can harbour a reservoir of bacteria that is maintained regardless of food intake. The appendix and colonic crypts may also be examples of such microbial reservoirs.
-
Only a subset of gut symbionts are able to access the epithelial surface. Mucus, antimicrobial peptides and adaptive immune activity limit tissue accessibility. Direct interfacing between the host and microbial symbionts may be important for the maintenance of homeostasis.
-
Immunomodulation by certain symbionts allows the host to tolerate intimate relationships with potentially beneficial microorganisms. This may be a way in which commensals distinguish themselves from pathogens and prevent their elimination by the immune system.
-
Although many diseases have been associated with dysbiosis, an understanding of the function of the microbiota in health and disease requires the biogeography of the community to be considered. Recent studies in humans have found differences specific to the mucosal community in cases of inflammatory bowel disease and hepatic encephalopathy.
Abstract
Animals assemble and maintain a diverse but host-specific gut microbial community. In addition to characteristic microbial compositions along the longitudinal axis of the intestines, discrete bacterial communities form in microhabitats, such as the gut lumen, colonic mucus layers and colonic crypts. In this Review, we examine how the spatial distribution of symbiotic bacteria among physical niches in the gut affects the development and maintenance of a resilient microbial ecosystem. We consider novel hypotheses for how nutrient selection, immune activation and other mechanisms control the biogeography of bacteria in the gut, and we discuss the relevance of this spatial heterogeneity to health and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dominguez-Bello, M. G. et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl Acad. Sci. USA 107, 11971–11975 (2010).
Hasegawa, M. et al. Transitions in oral and intestinal microflora composition and innate immune receptor-dependent stimulation during mouse development. Infect. Immun. 78, 639–650 (2010).
La Rosa, P. S. et al. Patterned progression of bacterial populations in the premature infant gut. Proc. Natl Acad. Sci. USA 111, 12522–12527 (2014).
Bevins, C. L. & Salzman, N. H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nat. Rev. Microbiol. 9, 356–368 (2011).
Peterson, D. A., McNulty, N. P., Guruge, J. L. & Gordon, J. I. IgA response to symbiotic bacteria as a mediator of gut homeostasis. Cell Host Microbe 2, 328–339 (2007).
Round, J. L. & Mazmanian, S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 9, 313–323 (2009).
Fernández, L. et al. The human milk microbiota: origin and potential roles in health and disease. Pharmacol. Res. 69, 1–10 (2013).
Rogier, E. W. et al. Secretory antibodies in breast milk promote long-term intestinal homeostasis by regulating the gut microbiota and host gene expression. Proc. Natl Acad. Sci. USA 111, 3074–3079 (2014).
Yu, Z.-T., Chen, C. & Newburg, D. S. Utilization of major fucosylated and sialylated human milk oligosaccharides by isolated human gut microbes. Glycobiology 23, 1281–1292 (2013).
Yu, Z.-T. et al. The principal fucosylated oligosaccharides of human milk exhibit prebiotic properties on cultured infant microbiota. Glycobiology 23, 169–177 (2013).
Marcobal, A. et al. Bacteroides in the infant gut consume milk oligosaccharides via mucus-utilization pathways. Cell Host Microbe 10, 507–514 (2011).
Lewis, Z. T. et al. Maternal fucosyltransferase 2 status affects the gut bifidobacterial communities of breastfed infants. Microbiome 3, 425 (2015).
Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Ley, R. E., Lozupone, C. A., Hamady, M., Knight, R. & Gordon, J. I. Worlds within worlds: evolution of the vertebrate gut microbiota. Nat. Rev. Microbiol. 6, 776–788 (2008).
Seedorf, H. et al. Bacteria from diverse habitats colonize and compete in the mouse gut. Cell 159, 253–266 (2014).
Martínez, I., Muller, C. E. & Walter, J. Long-term temporal analysis of the human fecal microbiota revealed a stable core of dominant bacterial species. PLoS ONE 8, e69621 (2013).
Faith, J. J. et al. The long-term stability of the human gut microbiota. Science 341, 1237439 (2013).
Dethlefsen, L. & Relman, D. A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl Acad. Sci. USA 108, 4554–4561 (2011).
Islam, K. B. M. S. et al. Bile acid is a host factor that regulates the composition of the cecal microbiota in rats. Gastroenterology 141, 1773–1781 (2011).
Zoetendal, E. G. et al. The human small intestinal microbiota is driven by rapid uptake and conversion of simple carbohydrates. ISME J. 6, 1415–1426 (2012).
Gu, S. et al. Bacterial community mapping of the mouse gastrointestinal tract. PLoS ONE 8, e74957 (2013).
Nava, G. M., Friedrichsen, H. J. & Stappenbeck, T. S. Spatial organization of intestinal microbiota in the mouse ascending colon. ISME J. 5, 627–638 (2011). Laser capture microdissection and 16S sequencing are used to profile the microbiome of the inter-fold regions of the proximal colon, revealing a community distinct from that of the central lumen.
Pédron, T. et al. A crypt-specific core microbiota resides in the mouse colon. mBio 3, e00116-12 (2012). The first 16S sequencing study of the colonic crypt microbiome, demonstrating that the crypt community includes many aerobic bacteria and has a distinct profile relative to lumenal bacteria.
Johansson, M. E. V. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
Swidsinski, A., Loening-Baucke, V., Verstraelen, H., Osowska, S. & Doerffel, Y. Biostructure of fecal microbiota in healthy subjects and patients with chronic idiopathic diarrhea. Gastroenterology 135, 568–579 (2008).
Swidsinski, A. et al. Viscosity gradient within the mucus layer determines the mucosal barrier function and the spatial organization of the intestinal microbiota. Inflamm. Bowel Dis. 13, 963–970 (2007).
Smith, H. F. et al. Comparative anatomy and phylogenetic distribution of the mammalian cecal appendix. J. Evol. Biol. 22, 1984–1999 (2009).
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638 (2005).
Yasuda, K. et al. Biogeography of the intestinal mucosal and lumenal microbiome in the rhesus macaque. Cell Host Microbe 17, 385–391 (2015). A detailed investigation of differences in the lumenal and mucosal communities along the gastrointestinal tract of macaques shows that many taxa have preferred spatial habitats.
Wang, Y. et al. Regional mucosa-associated microbiota determine physiological expression of TLR2 and TLR4 in murine colon. PLoS ONE 5, e13607 (2010).
Malmuthuge, N., Griebel, P. J. & Guan, L. L. Taxonomic identification of commensal bacteria associated with the mucosa and digesta throughout the gastrointestinal tracts of preweaned calves. Appl. Environ. Microbiol. 80, 2021–2028 (2014).
Lu, H.-P. et al. Spatial heterogeneity of gut microbiota reveals multiple bacterial communities with distinct characteristics. Sci. Rep. 4, 6185 (2014).
Albenberg, L. et al. Correlation between intraluminal oxygen gradient and radial partitioning of intestinal microbiota in humans and mice. Gastroenterology 147, 1055–1063.e8 (2014). Careful measurements of oxygen content in the gut reveal a steep oxygen gradient in the mucus that is predictive of community membership on the basis of bacterial ability to tolerate oxygen.
Berry, D. et al. Host-compound foraging by intestinal microbiota revealed by single-cell stable isotope probing. Proc. Natl Acad. Sci. USA 110, 4720–4725 (2013). Isotope labelling of mucosal proteins in the gut followed by nanoscale secondary ion mass spectrometry (nanoSIMS) detection in conjunction with FISH identifies mucosal bacteria that consume host-derived proteins. This powerful method provides in situ support for the theory that certain mucin-degrading bacteria largely forage host-derived nutrients.
Png, C. W. et al. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am. J. Gastroenterol. 105, 2420–2428 (2010).
Hong, P.-Y., Croix, J. A., Greenberg, E., Gaskins, H. R. & Mackie, R. I. Pyrosequencing-based analysis of the mucosal microbiota in healthy individuals reveals ubiquitous bacterial groups and micro-heterogeneity. PLoS ONE 6, e25042 (2011).
Zhang, Z. et al. Spatial heterogeneity and co-occurrence patterns of human mucosal-associated intestinal microbiota. ISME J. 8, 881–893 (2013).
Nava, G. M., Carbonero, F., Croix, J. A., Greenberg, E. & Gaskins, H. R. Abundance and diversity of mucosa-associated hydrogenotrophic microbes in the healthy human colon. ISME J. 6, 57–70 (2012).
Tong, M. et al. A modular organization of the human intestinal mucosal microbiota and its association with inflammatory bowel disease. PLoS ONE 8, e80702 (2013).
Davis, C. P., Mulcahy, D., Takeuchi, A. & Savage, D. C. Location and description of spiral-shaped microorganisms in the normal rat cecum. Infect. Immun. 6, 184–192 (1972).
Savage, D. C. & Blumershine, R. V. Surface–surface associations in microbial communities populating epithelial habitats in the murine gastrointestinal ecosystem: scanning electron microscopy. Infect. Immun. 10, 240–250 (1974).
Palestrant, D. et al. Microbial biofilms in the gut: visualization by electron microscopy and by acridine orange staining. Ultrastruct. Pathol. 28, 23–27 (2004).
Swidsinski, A., Loening-Baucke, V., Lochs, H. & Hale, L.-P. Spatial organization of bacterial flora in normal and inflamed intestine: a fluorescence in situ hybridization study in mice. World J. Gastroenterol. 11, 1131–1140 (2005).
Swidsinski, A., Weber, J., Loening-Baucke, V., Hale, L.-P. & Lochs, H. Spatial organization and composition of the mucosal flora in patients with inflammatory bowel disease. J. Clin. Microbiol. 43, 3380–3389 (2005).
Round, J. L. et al. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 332, 974–977 (2011).
Lee, S. M. et al. Bacterial colonization factors control specificity and stability of the gut microbiota. Nature 501, 426–429 (2013). A glycan binding and import system is identified in Bacteroides spp. and found to determine their species-specific niche, localization in colonic crypts and resilience in the face of intestinal perturbations.
Koropatkin, N. M., Cameron, E. A. & Martens, E. C. How glycan metabolism shapes the human gut microbiota. Nat. Rev. Microbiol. 10, 323–335 (2012).
Kaoutari, A. E., Armougom, F., Gordon, J. I., Raoult, D. & Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 11, 497–504 (2013).
Reeves, A. R., Wang, G. R. & Salyers, A. A. Characterization of four outer membrane proteins that play a role in utilization of starch by Bacteroides thetaiotaomicron. J. Bacteriol. 179, 643–649 (1997).
Sonnenburg, J. L. et al. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science 307, 1955–1959 (2005). Transcriptional profiling of B. thetaiotaomicron in the gut of gnotobiotic animals identifies genes involved in the utilization of diet-derived and host-derived nutrients.
Martens, E. C., Koropatkin, N. M., Smith, T. J. & Gordon, J. I. Complex glycan catabolism by the human gut microbiota: the Bacteroidetes Sus-like paradigm. J. Biol. Chem. 284, 24673–24677 (2009).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2013).
Sonnenburg, E. D. et al. Specificity of polysaccharide use in intestinal Bacteroides species determines diet-induced microbiota alterations. Cell 141, 1241–1252 (2010).
Kashyap, P. C. et al. Genetically dictated change in host mucuscarbohydrate landscape exerts a diet-dependent effect on the gut microbiota. Proc. Natl Acad. Sci. USA 110, 17059–17064 (2013).
Cuskin, F. et al. Human gut Bacteroidetes can utilize yeast mannan through a selfish mechanism. Nature 517, 165–169 (2015).
Wadolkowski, E. A., Laux, D. C. & Cohen, P. S. Colonization of the streptomycin-treated mouse large intestine by a human fecal Escherichia coli strain: role of growth in mucus. Infect. Immun. 56, 1030–1035 (1988).
Gries, D. M., Pultz, N. J. & Donskey, C. J. Growth in cecal mucus facilitates colonization of the mouse intestinal tract by methicillin-resistant Staphylococcus aureus. J. Infect. Dis. 192, 1621–1627 (2005).
Larsson, J. M. H., Karlsson, H., Sjövall, H. & Hansson, G. C. A complex, but uniform O-glycosylation of the human MUC2 mucin from colonic biopsies analyzed by nanoLC/MSn. Glycobiology 19, 756–766 (2009).
Thomsson, K. A. et al. Detailed O-glycomics of the Muc2 mucin from colon of wild-type, core 1- and core 3-transferase-deficient mice highlights differences compared with human MUC2. Glycobiology 22, 1128–1139 (2012).
Schluter, J. & Foster, K. R. The evolution of mutualism in gut microbiota via host epithelial selection. PLoS Biol. 10, e1001424 (2012). A mathematical modelling study demonstrates that positive selection through the presentation of nutrients is a more effective way for hosts to control surface bacterial communities than negative selection by antimicrobial compounds.
Sonoyama, K. et al. Response of gut microbiota to fasting and hibernation in Syrian hamsters. Appl. Environ. Microbiol. 75, 6451–6456 (2009).
Carey, H. V., Walters, W. A. & Knight, R. Seasonal restructuring of the ground squirrel gut microbiota over the annual hibernation cycle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 304, R33–R42 (2013).
Schwab, C. et al. Longitudinal study of murine microbiota activity and interactions with the host during acute inflammation and recovery. ISME J. 8, 1101–1114 (2014).
Martens, E. C., Chiang, H. C. & Gordon, J. I. Mucosal glycan foraging enhances fitness and transmission of a saccharolytic human gut bacterial symbiont. Cell Host Microbe 4, 447–457 (2008). Bacteria with mutations in genes involved in mucin O -glycan utilization are shown to be defective in colonization when host animals are fed diets without plant polysaccharides, and also in vertical transmission from mother to pup. This demonstrates the importance of host-derived nutrients in the mucus for stable and long-term colonization.
Sommer, F. et al. Altered mucus glycosylation in core 1 O-glycan-deficient mice affects microbiota composition and intestinal architecture. PLoS ONE 9, e85254 (2014).
Bergström, A. et al. Nature of bacterial colonization influences transcription of mucin genes in mice during the first week of life. BMC Res. Notes 5, 402 (2012).
Needham, B. D. & Trent, M. S. Fortifying the barrier: the impact of lipid A remodelling on bacterial pathogenesis. Nat. Rev. Microbiol. 11, 467–481 (2013).
Cullen, T. W. et al. Antimicrobial peptide resistance mediates resilience of prominent gut commensals during inflammation. Science 347, 170–175 (2015).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011). Bacterial sensing by the epithelium and REGIIIγ secretion by Paneth cells are shown to be necessary for the prevention of microbial overgrowth on the epithelial surface in the small intestine.
Gallo, R. L. & Hooper, L. V. Epithelial antimicrobial defence of the skin and intestine. Nat. Rev. Immunol. 12, 503–516 (2012).
Baughn, A. D. & Malamy, M. H. The strict anaerobe Bacteroides fragilis grows in and benefits from nanomolar concentrations of oxygen. Nature 427, 441–444 (2004).
Miyoshi, A. et al. Oxidative stress in Lactococcus lactis. Genet. Mol. Res. 2, 348–359 (2003).
Johansson, M. E. V., Larsson, J. M. H. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host–microbial interactions. Proc. Natl Acad. Sci. USA 108, S4659–S4665 (2011). A detailed investigation of the protein content of colonic mucus finds distinguishing factors between the outer and inner layers. MUC2 is shown to be required to prevent bacterial overgrowth on the epithelial surface and invasion of tissue.
Pelaseyed, T. et al. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol. Rev. 260, 8–20 (2014).
Cullender, T. C. et al. Innate and adaptive immunity interact to quench microbiome flagellar motility in the gut. Cell Host Microbe 14, 571–581 (2013).
Stecher, B. et al. Flagella and chemotaxis are required for efficient induction of Salmonella enterica serovar Typhimurium colitis in streptomycin-pretreated mice. Infect. Immun. 72, 4138–4150 (2004).
Navarro-Garcia, F. et al. Pic, an autotransporter protein secreted by different pathogens in the Enterobacteriaceae family, is a potent mucus secretagogue. Infect. Immun. 78, 4101–4109 (2010).
Nakjang, S., Ndeh, D. A., Wipat, A., Bolam, D. N. & Hirt, R. P. A novel extracellular metallopeptidase domain shared by animal host-associated mutualistic and pathogenic microbes. PLoS ONE 7, e30287 (2012).
Luo, Q. et al. Enterotoxigenic Escherichia coli secretes a highly conserved mucin-degrading metalloprotease to effectively engage intestinal epithelial cells. Infect. Immun. 82, 509–521 (2014).
Mahdavi, J. et al. Helicobacter pylori SabA adhesin in persistent infection and chronic inflammation. Science 297, 573–578 (2002).
Davis, C. P. & Savage, D. C. Habitat, succession, attachment, and morphology of segmented, filamentous microbes indigenous to the murine gastrointestinal tract. Infect. Immun. 10, 948–956 (1974).
Yin, Y. et al. Comparative analysis of the distribution of segmented filamentous bacteria in humans, mice and chickens. ISME J. 7, 615–621 (2013).
Schnupf, P. et al. Growth and host interaction of mouse segmented filamentous bacteria in vitro. Nature 520, 99–103 (2015).
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
Lee, Y. K., Menezes, J. S., Umesaki, Y. & Mazmanian, S. K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 108, S4615–S4622 (2011).
Wu, H.-J. et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunology 32, 815–827 (2010).
Sansonetti, P. J. War and peace at mucosal surfaces. Nat. Rev. Immunol. 4, 953–964 (2004).
Taylor, R. K., Miller, V. L., Furlong, D. B. & Mekalanos, J. J. Use of phoA gene fusions to identify a pilus colonization factor coordinately regulated with cholera toxin. Proc. Natl Acad. Sci. USA 84, 2833–2837 (1987).
Bhowmick, R. et al. Intestinal adherence of Vibrio cholerae involves a coordinated interaction between colonization factor GbpA and mucin. Infect. Immun. 76, 4968–4977 (2008).
Mouricout, M. Interactions between the enteric pathogen and the host. Adv. Exp. Med. Biol. 412, 109–123 (1997).
Lecuit, M. et al. A transgenic model for listeriosis: role of internalin in crossing the intestinal barrier. Science 292, 1722–1725 (2001).
McCormick, B. A., Colgan, S. P., Delp-Archer, C., Miller, S. I. & Madara, J. L. Salmonella typhimurium attachment to human intestinal epithelial monolayers: transcellular signalling to subepithelial neutrophils. J. Cell Biol. 123, 895–907 (1993).
Winter, S. E. et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 467, 426–429 (2010).
Savage, D. C. Microbial interference between indigenous yeast and lactobacilli in the rodent stomach. J. Bacteriol. 98, 1278–1283 (1969).
Morotomi, M., Watanabe, T., Suegara, N., Kawai, Y. & Mutai, M. Distribution of indigenous bacteria in the digestive tract of conventional and gnotobiotic rats. Infect. Immun. 11, 962–968 (1975).
Sengupta, R. et al. The role of cell surface architecture of lactobacilli in host–microbe interactions in the gastrointestinal tract. Mediators Inflamm. 2013, 237921–237916 (2013).
Mackenzie, D. A. et al. Strain-specific diversity of mucus-binding proteins in the adhesion and aggregation properties of Lactobacillus reuteri. Microbiology 156, 3368–3378 (2010).
Frese, S. A. et al. Molecular characterization of host-specific biofilm formation in a vertebrate gut symbiont. PLoS Genet. 9, e1004057 (2013).
von Ossowski, I. et al. Mucosal adhesion properties of the probiotic Lactobacillus rhamnosus GG SpaCBA and SpaFED pilin subunits. Appl. Environ. Microbiol. 76, 2049–2057 (2010).
Turroni, F. et al. Role of sortase-dependent pili of Bifidobacterium bifidum PRL2010 in modulating bacterium–host interactions. Proc. Natl Acad. Sci. USA 110, 11151–11156 (2013).
Kubinak, J. L. et al. MyD88 signaling in T cells directs IgA-mediated control of the microbiota to promote health. Cell Host Microbe 17, 153–163 (2015).
Palm, N. W. et al. Immunoglobulin A coating identifies colitogenic bacteria in inflammatory bowel disease. Cell 158, 1000–1010 (2014).
Mathias, A. & Corthésy, B. N-glycans on secretory component: mediators of the interaction between secretory IgA and Gram-positive commensals sustaining intestinal homeostasis. Gut Microbes 2, 287–293 (2011).
Peterson, D. A. et al. Characterizing the interactions between a naturally-primed immunoglobulin A and its conserved Bacteroides thetaiotaomicron species-specific epitope in gnotobiotic mice. J. Biol. Chem. 290, 12630–12649 (2015).
Round, J. L. & Mazmanian, S. K. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl Acad. Sci. USA 107, 12204–12209 (2010).
Coyne, M. J., Reinap, B., Lee, M. M. & Comstock, L. E. Human symbionts use a host-like pathway for surface fucosylation. Science 307, 1778–1781 (2005).
Fanning, S. et al. Bifidobacterial surface-exopolysaccharide facilitates commensal-host interaction through immune modulation and pathogen protection. Proc. Natl Acad. Sci. USA 109, 2108–2113 (2012).
Jeon, S. G. et al. Probiotic Bifidobacterium breve induces IL-10-producing Tr1 cells in the colon. PLoS Pathog. 8, e1002714 (2012).
Atarashi, K. et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 331, 337–341 (2011).
Geuking, M. B. et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunology 34, 794–806 (2011).
Arpaia, N. et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504, 451–455 (2013).
Smith, P. M. et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341, 569–573 (2013).
Shan, M. et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science 342, 447–453 (2013).
Monack, D. M., Mueller, A. & Falkow, S. Persistent bacterial infections: the interface of the pathogen and the host immune system. Nat. Rev. Microbiol. 2, 747–765 (2004).
Randal Bollinger, R., Barbas, A. S., Bush, E. L., Lin, S. S. & Parker, W. Biofilms in the large bowel suggest an apparent function of the human vermiform appendix. J. Theor. Biol. 249, 826–831 (2007). This study proposes the hypothesis that the appendix harbours a protected reservoir of bacterial cells that could re-populate the caecum and large intestine.
Hanson, N. B. & Lanning, D. K. Microbial induction of B and T cell areas in rabbit appendix. Dev. Comp. Immunol. 32, 980–991 (2008).
Gophna, U., Sommerfeld, K., Gophna, S., Doolittle, W. F. & Veldhuyzen van Zanten, S. J. O. Differences between tissue-associated intestinal microfloras of patients with Crohn's disease and ulcerative colitis. J. Clin. Microbiol. 44, 4136–4141 (2006).
Manichanh, C. et al. Reduced diversity of faecal microbiota in Crohn's disease revealed by a metagenomic approach. Gut 55, 205–211 (2006).
Walker, A. W. et al. High-throughput clone library analysis of the mucosa-associated microbiota reveals dysbiosis and differences between inflamed and non-inflamed regions of the intestine in inflammatory bowel disease. BMC Microbiol. 11, 7 (2011).
Ott, S. J. et al. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut 53, 685–693 (2004).
Abrahamsson, T. R. et al. Low gut microbiota diversity in early infancy precedes asthma at school age. Clin. Exp. Allergy 44, 842–850 (2014).
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Microbial ecology: human gut microbes associated with obesity. Nature 444, 1022–1023 (2006).
Garcovich, M., Zocco, M. A., Roccarina, D., Ponziani, F. R. & Gasbarrini, A. Prevention and treatment of hepatic encephalopathy: focusing on gut microbiota. World J. Gastroenterol. 18, 6693–6700 (2012).
Henao-Mejia, J. et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482, 179–185 (2012).
Zhu, Q., Gao, R., Wu, W. & Qin, H. The role of gut microbiota in the pathogenesis of colorectal cancer. Tumour Biol. 34, 1285–1300 (2013).
Wu, N. et al. Dysbiosis signature of fecal microbiota in colorectal cancer patients. Microb. Ecol. 66, 462–470 (2013).
Collins, S. M., Surette, M. & Bercik, P. The interplay between the intestinal microbiota and the brain. Nat. Rev. Microbiol. 10, 735–742 (2012).
Gevers, D. et al. The treatment-naive microbiome in new-onset Crohn's disease. Cell Host Microbe 15, 382–392 (2014). A 16S sequencing analysis of faecal, ileal mucosa and rectal mucosa samples from patients with early-stage Crohn disease before treatment shows dysbiosis in the mucosal samples and no difference in the faecal samples. Following treatment, there are unrelated differences in the faecal samples, suggesting that the faecal dysbiosis observed in earlier studies may be a secondary effect.
Petersen, C. & Round, J. L. Defining dysbiosis and its influence on host immunity and disease. Cell. Microbiol. 16, 1024–1033 (2014).
Swidsinski, A. et al. Mucosal flora in inflammatory bowel disease. Gastroenterology 122, 44–54 (2002).
Baumgart, M. et al. Culture independent analysis of ileal mucosa reveals a selective increase in invasive Escherichia coli of novel phylogeny relative to depletion of Clostridiales in Crohn's disease involving the ileum. ISME J. 1, 403–418 (2007).
Rowan, F. et al. Bacterial colonization of colonic crypt mucous gel and disease activity in ulcerative colitis. Ann. Surg. 252, 869–875 (2010).
Bajaj, J. S. et al. Colonic mucosal microbiome differs from stool microbiome in cirrhosis and hepatic encephalopathy and is linked to cognition and inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 303, G675–G685 (2012).
Rosebury, T. Microorganisms Indigenous to Man (McGraw-Hill, 1962).
Freter, R., Brickner, H., Botney, M., Cleven, D. & Aranki, A. Mechanisms that control bacterial populations in continuous-flow culture models of mouse large intestinal flora. Infect. Immun. 39, 676–685 (1983).
Maltby, R., Leatham-Jensen, M. P., Gibson, T., Cohen, P. S. & Conway, T. Nutritional basis for colonization resistance by human commensal Escherichia coli strains HS and Nissle 1917 against E. coli O157:H7 in the mouse intestine. PLoS ONE 8, e53957 (2013).
Wilson, K. H. & Perini, F. Role of competition for nutrients in suppression of Clostridium difficile by the colonic microflora. Infect. Immun. 56, 2610–2614 (1988).
Acknowledgements
The authors thank E. Hsiao, B. Needham and T. Sampson for critical comments on the manuscript. G.P.D. is supported by a US National Science Foundation Graduate Research Fellowship (grant DGE-1144469). Work in the S.K.M. laboratory is supported by funding from the US National Institutes of Health (grants GM099535, DK078938 and MH100556), the Emerald Foundation and the Simons Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Microbiota
-
The collection of microorganisms (including bacteria, viruses, fungi and single-celled eukaryotes) that inhabit a particular habitat, such as an animal.
- Syntrophic interactions
-
Metabolic relationships in which one member provides nutrients to another.
- Secreted immunoglobulin A
-
(sIgA). By far the most abundant isotype of antibody found in the gut.
- Prebiotic
-
A molecule that serves as a nutrient which stimulates the growth of commensal or mutualistic gut bacteria. In many cases, prebiotics can be specific for discrete groups of bacteria on the basis of their metabolic (that is, nutritional) requirements.
- Symbionts
-
In ecology: organisms that participate in a close relationship with other organisms. The term encompasses organisms that participate in different types of relationship, including mutualists, commensals and parasites.
- Commensals
-
In ecology: organisms that participate in a symbiotic relationship in which one party benefits from the other without affecting the other party. Historically, commensals is also used as a term for the resident gut bacteria, although many of these may be mutualists.
- Mutualists
-
In ecology: organisms that participate in a symbiotic relationship in which both parties benefit.
- Gnotobiotic animals
-
Formerly germ-free animals that now carry a defined microbiota. The composition of the microbiota in these animals is usually determined experimentally.
- Digesta
-
The bulk of dietary fibres that is digested as it transits through the gastrointestinal tract.
- Goblet cells
-
Specialized epithelial cells throughout the gastrointestinal tract that secrete gel-forming mucins. Goblet cells can also be present in other mucosal epithelial surfaces throughout the body.
- Biofilm
-
An aggregation of bacteria that are colocalized in a matrix and reside on a surface. Biofilms may include single species of bacteria or polymicrobial communities.
- Indigenous organisms
-
Organisms that are native to a particular habitat (also termed autochthonous), as distinct from organisms that are simply passing through a habitat (allochthonous)
- Mucin 2
-
(MUC2). The most abundant mucin protein in the human gut; the mouse homologue is also the most abundant mouse gut mucin.
- Colonization resistance
-
The prevention of invasion of an exogenous species into a microbial community. In the gut, colonization resistance may be a result of resource competition, spatial exclusion or direct inhibition by commensal microorganisms, or of selection mediated by host factors.
- Paneth cells
-
Specialized epithelial cells that are found at the base of crypts in the small intestine and that secrete antimicrobial peptides.
- Pathobiont
-
A symbiont with the potential to promote pathology under conditions that deviate from homeostasis, such as in immunocompromised or nutrient-deprived individuals.
- Microaerophilic
-
Pertaining to a microorganism: obligately aerobic. These microorganisms thrive only in environments with low oxygen concentrations, such as at the epithelial surface in the gut.
- Dysbiosis
-
A deviation from a normal microbial community, such as an imbalance in the abundance, membership or localization of microorganisms.
Rights and permissions
About this article
Cite this article
Donaldson, G., Lee, S. & Mazmanian, S. Gut biogeography of the bacterial microbiota. Nat Rev Microbiol 14, 20–32 (2016). https://doi.org/10.1038/nrmicro3552
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro3552
This article is cited by
-
Potential roles of the interactions between gut microbiota and metabolites in LPS-induced intrauterine inflammation (IUI) and associated preterm birth (PTB)
Journal of Translational Medicine (2024)
-
Disentangling direct vs indirect effects of microbiome manipulations in a habitat-forming marine holobiont
npj Biofilms and Microbiomes (2024)
-
The gut microbiota and its biogeography
Nature Reviews Microbiology (2024)
-
Microbial butyrate capacity is reduced in inflamed mucosa in patients with ulcerative colitis
Scientific Reports (2024)
-
Pioneering gut health improvements in piglets with phytogenic feed additives
Applied Microbiology and Biotechnology (2024)