Key Points
-
Probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” and have been studied by the scientific community for more than a century. The vast majority of marketed probiotics belong to the genera Lactobacillus and Bifidobacterium. Although the scientific evidence for the health-promoting effects of probiotics in specific challenged populations is solid (for example, the prevention of antibiotic-associated and acute infectious diarrhoea), the effectiveness of these organisms in other areas, such as the treatment of inflammatory bowel disease, is less established.
-
Several modes of action have been proposed for probiotics, including the strengthening of intestinal epithelial barrier function by stimulation of mucin secretion or enhancement of tight junction function, the clearance of pathogens by competitive binding to receptors presented by epithelial cells, and the synthesis of antimicrobial substances such as bacteriocins. Another key mode of action by which probiotics are proposed to exert their beneficial effects is through modulation of the host immune system in the intestinal mucosa.
-
The intestinal mucosa contains several specialized cell types involved in immunomodulation, and these cells express a range of pattern recognition receptors (PRRs), including Toll-like receptors (TLRs), NOD-like receptors (NLRs) and C-type lectin receptors (CLRs). Upon exposure to microorganism-associated molecular patterns (MAMPs), PRRs respond by activating associated adaptor proteins that are linked to nuclear factor-κB and mitogen-activated protein kinase signalling cascades, resulting in modulated expression of response genes, including genes encoding cytokines, chemokines and antimicrobial peptides.
-
Multiple conserved, polymeric MAMPs are located in the bacterial cell envelope, including peptidoglycan, capsular polysaccharide, wall teichoic acids and lipoteichoic acids (LTAs). Several MAMP–PRR interactions are well documented. However, the strain- and species-specific probiotic effects that are observed hinder the investigation into general probiotic MAMPs and the effects that they induce. Obtaining insight into strain specificity will require comparative structural analyses of the complex MAMP backbones and their modifications.
-
Strain-specific proteinaceous MAMPs have also been identified, further adding to the complexity of intestinal host–microorganism interactions. Overall, the molecular analysis of MAMPs has led to the perception that their functioning is influenced by the cellular context in which they are expressed, and the overall MAMP profile will lead to an integrated response by the host to maintain or regain intestinal homeostasis.
-
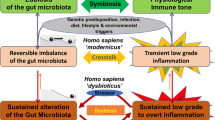
Determination of the changes in the duodenal transcriptomes of healthy volunteers after the consumption of four different lactobacilli revealed species-specific host immunomodulation via distinct signalling pathways, pinpointing the importance of strain selection for specific probiotic applications. Moreover, a remarkably large intrapersonal distance between the transcriptome profiles was observed in comparison to the interpersonal transcriptional changes induced by consumption of different probiotic strains. These observations suggest that, within the concept of the 'bandwidth of human health' that we outline here, different molecular solutions may underlie intestinal homeostasis in healthy individuals, a fact that is likely to affect the physiological consequences of a healthy person's responses to functional foods such as probiotics.
Abstract
Probiotic bacteria can modulate immune responses in the host gastrointestinal tract to promote health. The genomics era has provided novel opportunities for the discovery and characterization of bacterial probiotic effector molecules that elicit specific responses in the intestinal system. Furthermore, nutrigenomic analyses of the response to probiotics have unravelled the signalling and immune response pathways which are modulated by probiotic bacteria. Together, these genomic approaches and nutrigenomic analyses have identified several bacterial factors that are involved in modulation of the immune system and the mucosal barrier, and have revealed that a molecular 'bandwidth of human health' could represent a key determinant in an individual's physiological responsiveness to probiotics. These approaches may lead to improved stratification of consumers and to subpopulation-level probiotic supplementation to maintain or improve health, or to reduce the risk of disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Food and Agriculture Organization–WHO. Guidelines for the evaluation of probiotics in food. (WHO, London, Ontario, 2002).
Kleerebezem, M. et al. The extracellular biology of the lactobacilli. FEMS Microbiol. Rev. 34, 199–230 (2010).
Gordon, S. Elie Metchnikoff: father of natural immunity. Eur. J. Immunol. 38, 3257–3264 (2008).
Metchnikoff, É. The Prolongation of Life: Optimistic Studies (ed. Mitchell, P. C.) (Heinemann, London, 1907).
Tissier, H. Des infections intestinales par la méthode de la flore bactérienne de l'intestin. Crit. Rev. Soc. Biol. 60, 359–361 (1906) (in French).
Lebeer, S., Vanderleyden, J. & De Keersmaecker, S. C. Host interactions of probiotic bacterial surface molecules: comparison with commensals and pathogens. Nature Rev. Microbiol. 8, 171–184 (2010).
Marco, M. L., Pavan, S. & Kleerebezem, M. Towards understanding molecular modes of probiotic action. Curr. Opin. Biotechnol. 17, 204–210 (2006).
Do, V. T., Baird, B. G. & Kockler, D. R. Probiotics for maintaining remission of ulcerative colitis in adults. Ann. Pharmacother. 44, 565–571 (2010).
Trebichavsky, I., Splichal, I., Rada, V. & Splichalova, A. Modulation of natural immunity in the gut by Escherichia coli strain Nissle 1917. Nutr. Rev. 68, 459–464 (2010).
Alfaleh, K., Anabrees, J. & Bassler, D. Probiotics reduce the risk of necrotizing enterocolitis in preterm infants: a meta-analysis. Neonatology 97, 93–99 (2010).
Deshpande, G., Rao, S., Patole, S. & Bulsara, M. Updated meta-analysis of probiotics for preventing necrotizing enterocolitis in preterm neonates. Pediatrics 125, 921–930 (2010).
Kale-Pradhan, P. B., Jassal, H. K. & Wilhelm, S. M. Role of Lactobacillus in the prevention of antibiotic-associated diarrhea: a meta-analysis. Pharmacotherapy 30, 119–126 (2010).
Holubar, S. D., Cima, R. R., Sandborn, W. J. & Pardi, D. S. Treatment and prevention of pouchitis after ileal pouch-anal anastomosis for chronic ulcerative colitis. Cochrane Database Syst. Rev. 2010, CD001176 (2010).
Boyle, R. J., Bath-Hextall, F. J., Leonardi-Bee, J., Murrell, D. F. & Tang, M. L. Probiotics for the treatment of eczema: a systematic review. Clin. Exp. Allergy 39, 1117–1127 (2009).
Rioux, K. P. & Fedorak, R. N. Probiotics in the treatment of inflammatory bowel disease. J. Clin. Gastroenterol. 40, 260–263 (2006).
Moayyedi, P. et al. The efficacy of probiotics in the treatment of irritable bowel syndrome: a systematic review. Gut 59, 325–332 (2010).
Borchers, A. T., Selmi, C., Meyers, F. J., Keen, C. L. & Gershwin, M. E. Probiotics and immunity. J. Gastroenterol. 44, 26–46 (2009).
Pelto, L., Isolauri, E., Lilius, E. M., Nuutila, J. & Salminen, S. Probiotic bacteria downregulate the milk-induced inflammatory response in milk-hypersensitive subjects but have an immunostimulatory effect in healthy subjects. Clin. Exp. Allergy 28, 1474–1479 (1998).
Roessler, A. et al. The immune system in healthy adults and patients with atopic dermatitis seems to be affected differently by a probiotic intervention. Clin. Exp. Allergy 38, 93–102 (2008).
Lebeer, S., Vanderleyden, J. & De Keersmaecker, S. C. Genes and molecules of lactobacilli supporting probiotic action. Microbiol. Mol. Biol. Rev. 72, 728–764 (2008).
Corr, S. C., Hill, C. & Gahan, C. G. Understanding the mechanisms by which probiotics inhibit gastrointestinal pathogens. Adv. Food Nutr. Res. 56, 1–15 (2009).
Mack, D. R., Ahrne, S., Hyde, L., Wei, S. & Hollingsworth, M. A. Extracellular MUC3 mucin secretion follows adherence of Lactobacillus strains to intestinal epithelial cells in vitro. Gut 52, 827–833 (2003).
Schlee, M. et al. Probiotic lactobacilli and VSL#3 induce enterocyte β-defensin 2. Clin. Exp. Immunol. 151, 528–535 (2008).
Schlee, M. et al. Induction of human β-defensin 2 by the probiotic Escherichia coli Nissle 1917 is mediated through flagellin. Infect. Immun. 75, 2399–2407 (2007).
Yan, F. et al. Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 132, 562–575 (2007). The first study to pinpoint the role in probiotic activity for specific proteinaceous components of a well-characterized probiotic strain.
Yan, F. & Polk, D. B. Probiotic bacterium prevents cytokine-induced apoptosis in intestinal epithelial cells. J. Biol. Chem. 277, 50959–50965 (2002).
Anderson, R. C. et al. Lactobacillus plantarum MB452 enhances the function of the intestinal barrier by increasing the expression levels of genes involved in tight junction formation. BMC Microbiol. 10, 316 (2010).
Karczewski, J. et al. Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G851–G859 (2010).
Seth, A., Yan, F., Polk, D. B. & Rao, R. K. Probiotics ameliorate the hydrogen peroxide-induced epithelial barrier disruption by a PKC- and MAP kinase-dependent mechanism. Am. J. Physiol. Gastrointest. Liver Physiol. 294, G1060–G1069 (2008).
O'Flaherty, S., Saulnier, D. M., Pot, B. & Versalovic, J. How can probiotics and prebiotics impact mucosal immunity? Gut Microbes 1, 293–300 (2010).
Booijink, C. C. et al. High temporal and inter-individual variation detected in the human ileal microbiota. Environ. Microbiol. 12, 3213–3227 (2010).
Booijink, C. C., Zoetendal, E. G., Kleerebezem, M. & de Vos, W. M. Microbial communities in the human small intestine: coupling diversity to metagenomics. Future Microbiol. 2, 285–295 (2007).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011).
Maslowski, K. M. et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 461, 1282–1286 (2009).
Veiga, P. et al. Bifidobacterium animalis subsp. lactis fermented milk product reduces inflammation by altering a niche for colitogenic microbes. Proc. Natl Acad. Sci. USA 107, 18132–18137 (2010).
O'Hara, A. M. et al. Functional modulation of human intestinal epithelial cell responses by Bifidobacterium infantis and Lactobacillus salivarius. Immunology 118, 202–215 (2006).
Balda, M. S. & Matter, K. Tight junctions at a glance. J. Cell Sci. 121, 3677–3682 (2008).
McCracken, V. J. & Lorenz, R. G. The gastrointestinal ecosystem: a precarious alliance among epithelium, immunity and microbiota. Cell. Microbiol. 3, 1–11 (2001).
Bevins, C. L. & Salzman, N. H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nature Rev. Microbiol. 9, 356–368 (2011).
Salzman, N. H. Microbiota-immune system interaction: an uneasy alliance. Curr. Opin. Microbiol. 14, 99–105 (2011).
Wells, J. M., Rossi, O., Meijerink, M. & van Baarlen, P. Epithelial crosstalk at the microbiota-mucosal interface. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4607–4614 (2011).
Liew, F. Y. TH1 and TH2 cells: a historical perspective. Nature Rev. Immunol. 2, 55–60 (2002).
Barnes, M. J. & Powrie, F. Regulatory T cells reinforce intestinal homeostasis. Immunity 31, 401–411 (2009).
Kelsall, B. Recent progress in understanding the phenotype and function of intestinal dendritic cells and macrophages. Mucosal Immunol. 1, 460–469 (2008).
Rescigno, M. Intestinal dendritic cells. Adv. Immunol. 107, 109–138 (2010).
Coombes, J. L. & Powrie, F. Dendritic cells in intestinal immune regulation. Nature Rev. Immunol. 8, 435–446 (2008).
Kapsenberg, M. L. Dendritic-cell control of pathogen-driven T-cell polarization. Nature Rev. Immunol. 3, 984–993 (2003).
Sansonetti, P. J. & Medzhitov, R. Learning tolerance while fighting ignorance. Cell 138, 416–420 (2009).
Chung, H. & Kasper, D. L. Microbiota-stimulated immune mechanisms to maintain gut homeostasis. Curr. Opin. Immunol. 22, 455–460 (2010).
Lee, Y. K. & Mazmanian, S. K. Has the microbiota played a critical role in the evolution of the adaptive immune system? Science 330, 1768–1773 (2010).
Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nature Rev. Immunol. 8, 411–420 (2008).
Gaboriau-Routhiau, V. et al. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity 31, 677–689 (2009). A comprehensive study which shows that a non-culturable Clostridium -related species of segmented filamentous bacteria is able to promote differentiation and maturation of mouse T H 1, T H 17 and T Reg cells to similar extents as a complete mouse microbiota.
Sartor, R. B. Therapeutic correction of bacterial dysbiosis discovered by molecular techniques. Proc. Natl Acad. Sci. USA 105, 16413–16414 (2008).
Reid, G. et al. Microbiota restoration: natural and supplemented recovery of human microbial communities. Nature Rev. Microbiol. 9, 27–38 (2011).
Asong, J., Wolfert, M. A., Maiti, K. K., Miller, D. & Boons, G. J. Binding and cellular activation studies reveal that Toll-like receptor 2 can differentially recognize peptidoglycan from Gram-positive and Gram-negative bacteria. J. Biol. Chem. 284, 8643–8653 (2009).
Takeda, K., Kaisho, T. & Akira, S. Toll-like receptors. Annu. Rev. Immunol. 21, 335–376 (2003).
Dziarski, R. & Gupta, D. Staphylococcus aureus peptidoglycan is a Toll-like receptor 2 activator: a reevaluation. Infect. Immun. 73, 5212–5216 (2005).
Travassos, L. H. et al. Toll-like receptor 2-dependent bacterial sensing does not occur via peptidoglycan recognition. EMBO Rep. 5, 1000–1006 (2004).
Volz, T. et al. Natural Staphylococcus aureus-derived peptidoglycan fragments activate NOD2 and act as potent co-stimulators of the innate immune system exclusively in the presence of TLR signals. FASEB J. 24, 4089–4102 (2010).
Macho Fernandez, E. et al. Anti-inflammatory capacity of selected lactobacilli in experimental colitis is driven by NOD2-mediated recognition of a specific peptidoglycan-derived muropeptide. Gut 60, 1050–1059 (2011). An elegant invesitgation that provides insight at the molecular level into why peptidoglycan derived from two Lactobacillus spp. is differentially recognized by NLRP2 and, consequently, leads to different efficacies of these species in the treatment of collitis in a mouse model.
Delcour, J., Ferain, T., Deghorain, M., Palumbo, E. & Hols, P. The biosynthesis and functionality of the cell-wall of lactic acid bacteria. Antonie Van Leeuwenhoek 76, 159–184 (1999).
Foligne, B. et al. A key role of dendritic cells in probiotic functionality. PLoS ONE 2, e313 (2007).
Zeuthen, L. H., Fink, L. N. & Frokiaer, H. Toll-like receptor 2 and nucleotide-binding oligomerization domain-2 play divergent roles in the recognition of gut-derived lactobacilli and bifidobacteria in dendritic cells. Immunology 124, 489–502 (2008).
Xia, G., Kohler, T. & Peschel, A. The wall teichoic acid and lipoteichoic acid polymers of Staphylococcus aureus. Int. J. Med. Microbiol. 300, 148–154 (2010).
Grangette, C. et al. Enhanced antiinflammatory capacity of a Lactobacillus plantarum mutant synthesizing modified teichoic acids. Proc. Natl Acad. Sci. USA 102, 10321–10326 (2005). This work provides the first direct evidence, using both in vitro and in vivo models, that a bacterial cell wall compound (LTA) can modulate the host immune response.
Kim, H. G. et al. Inhibitory effects of Lactobacillus plantarum lipoteichoic acid (LTA) on Staphylococcus aureus LTA-induced tumour necrosis factor-alpha production. J. Microbiol. Biotechnol. 18, 1191–1196 (2008).
Matsuguchi, T. et al. Lipoteichoic acids from Lactobacillus strains elicit strong tumour necrosis factor alpha-inducing activities in macrophages through Toll-like receptor 2. Clin. Diagn. Lab. Immunol. 10, 259–266 (2003).
Mohamadzadeh, M. et al. Regulation of induced colonic inflammation by Lactobacillus acidophilus deficient in lipoteichoic acid. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4623–4630 (2011).
Saber, R. et al. Lipoteichoic acid-deficient Lactobacillus acidophilus regulates downstream signals. Immunotherapy 3, 337–347 (2011).
Hirose, Y. et al. Lipoteichoic acids on Lactobacillus plantarum cell surfaces correlate with induction of interleukin-12p40 production. Microbiol. Immunol. 54, 143–151 (2010).
Perea Velez, M. et al. Functional analysis of D-alanylation of lipoteichoic acid in the probiotic strain Lactobacillus rhamnosus GG. Appl. Environ. Microbiol. 73, 3595–3604 (2007).
Kaji, R., Kiyoshima-Shibata, J., Nagaoka, M., Nanno, M. & Shida, K. Bacterial teichoic acids reverse predominant IL-12 production induced by certain Lactobacillus strains into predominant IL-10 production via TLR2-dependent ERK activation in macrophages. J. Immunol. 184, 3505–3513 (2010).
Ryu, Y. H. et al. Differential immunostimulatory effects of Gram-positive bacteria due to their lipoteichoic acids. Int. Immunopharmacol. 9, 127–133 (2009).
Jang, K. S., Baik, J. E., Han, S. H., Chung, D. K. & Kim, B. G. Multi-spectrometric analyses of lipoteichoic acids isolated from Lactobacillus plantarum. Biochem. Biophys. Res. Commun. 407, 823–830 (2011).
Claes, I. J. et al. Impact of lipoteichoic acid modification on the performance of the probiotic Lactobacillus rhamnosus GG in experimental colitis. Clin. Exp. Immunol. 162, 306–314 (2010).
Wang, Q. et al. A bacterial carbohydrate links innate and adaptive responses through Toll-like receptor 2. J. Exp. Med. 203, 2853–2863 (2006).
Rockel, C., Hartung, T. & Hermann, C. Different Staphylococcus aureus whole bacteria mutated in putative pro-inflammatory membrane components have similar cytokine inducing activity. Immunobiology 216, 316–321 (2011).
Yasuda, E., Serata, M. & Sako, T. Suppressive effect on activation of macrophages by Lactobacillus casei strain Shirota genes determining the synthesis of cell wall-associated polysaccharides. Appl. Environ. Microbiol. 74, 4746–4755 (2008).
Hafez, M. et al. The K5 capsule of Escherichia coli strain Nissle 1917 is important in mediating interactions with intestinal epithelial cells and chemokine induction. Infect. Immun. 77, 2995–3003 (2009).
Lebeer, S. et al. Identification of a gene cluster for the biosynthesis of a long, galactose-rich exopolysaccharide in Lactobacillus rhamnosus GG and functional analysis of the priming glycosyltransferase. Appl. Environ. Microbiol. 75, 3554–3563 (2009).
Lebeer, S., Claes, I. J., Verhoeven, T. L., Vanderleyden, J. & De Keersmaecker, S. C. Exopolysaccharides of Lactobacillus rhamnosus GG form a protective shield against innate immune factors in the intestine. Microb. Biotechnol. 4, 368–374 (2011).
Pilobello, K. T. & Mahal, L. K. Deciphering the glycocode: the complexity and analytical challenge of glycomics. Curr. Opin. Chem. Biol. 11, 300–305 (2007).
Xia, G. et al. Glycosylation of wall teichoic acid in Staphylococcus aureus by TarM. J. Biol. Chem. 285, 13405–13415 (2010).
Vollmer, W. Structural variation in the glycan strands of bacterial peptidoglycan. FEMS Microbiol. Rev. 32, 287–306 (2008).
Tomita, S. et al. Comparison of components and synthesis genes of cell wall teichoic acid among Lactobacillus plantarum strains. Biosci. Biotechnol. Biochem. 74, 928–933 (2010).
Francius, G. et al. Detection, localization, and conformational analysis of single polysaccharide molecules on live bacteria. ACS Nano 2, 1921–1929 (2008).
Marco, M. L. et al. Convergence in probiotic Lactobacillus gut-adaptive responses in humans and mice. ISME J. 4, 1481–1484 (2010). The first report on the in situ transcriptome of a Lactobacillus sp. in the human intestinal tract.
Andre, G. et al. Fluorescence and atomic force microscopy imaging of wall teichoic acids in Lactobacillu s plantarum. ACS Chem. Biol. 6, 366–376 (2011).
Atilano, M. L. et al. Teichoic acids are temporal and spatial regulators of peptidoglycan crosslinking in Staphylococcus aureus. Proc. Natl Acad. Sci. USA 107, 18991–18996 (2010).
Ventura, M. et al. Genome-scale analyses of health-promoting bacteria: probiogenomics. Nature Rev. Microbiol. 7, 61–71 (2009).
Smits, H. H. et al. Selective probiotic bacteria induce IL-10-producing regulatory T cells in vitro by modulating dendritic cell function through dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin. J. Allergy Clin. Immunol. 115, 1260–1267 (2005).
Konstantinov, S. R. et al. S layer protein A of Lactobacillus acidophilus NCFM regulates immature dendritic cell and T cell functions. Proc. Natl Acad. Sci. USA 105, 19474–19479 (2008). The first study to identify the ligand of a pivotal immature-DC receptor that is involved in immunomodulation by a probiotic Lactobacillus strain.
Johnson-Henry, K. C., Hagen, K. E., Gordonpour, M., Tompkins, T. A. & Sherman, P. M. Surface-layer protein extracts from Lactobacillus helveticus inhibit enterohaemorrhagic Escherichia coli O157:H7 adhesion to epithelial cells. Cell. Microbiol. 9, 356–367 (2007).
Corr, S. C. et al. Bacteriocin production as a mechanism for the antiinfective activity of Lactobacillus salivarius UCC118. Proc. Natl Acad. Sci. USA 104, 7617–7621 (2007). An elegantly designed in vivo investigation that robustly proves the probiotic role of Lactobacillus spp. bacteriocins in the prevention or attenuation of intestinal pathogen infection ( Listeria monocytogenes ) in mice.
Molenaar, D. et al. Exploring Lactobacillus plantarum genome diversity by using microarrays. J. Bacteriol. 187, 6119–6127 (2005).
Siezen, R. J. et al. Phenotypic and genomic diversity of Lactobacillus plantarum strains isolated from various environmental niches. Environ. Microbiol. 12, 758–773 (2010).
van Haemert, S. et al. Identification of Lactobacillus plantarum genes modulating the cytokine response of human peripheral blood mononuclear cells. BMC Microbiol. 10, 293 (2010).
Meijerink, M. et al. Identification of genetic loci in Lactobacillus plantarum that modulate the immune response of dendritic cells using comparative genome hybridization. PLoS ONE 5, e10632 (2010).
Bron, P. A., Grangette, C., Mercenier, A., de Vos, W. M. & Kleerebezem, M. Identification of Lactobacillus plantarum genes that are induced in the gastrointestinal tract of mice. J. Bacteriol. 186, 5721–5729 (2004).
Marco, M. L., Bongers, R. S., de Vos, W. M. & Kleerebezem, M. Spatial and temporal expression of Lactobacillus plantarum genes in the gastrointestinal tracts of mice. Appl. Environ. Microbiol. 73, 124–132 (2007).
Marco, M. L. et al. Lifestyle of Lactobacillus plantarum in the mouse caecum. Environ. Microbiol. 11, 2747–2757 (2009).
Pretzer, G. et al. Biodiversity-based identification and functional characterization of the mannose-specific adhesin of Lactobacillus plantarum. J. Bacteriol. 187, 6128–6136 (2005).
Gross, G. et al. Mannose-specific interaction of Lactobacillus plantarum with porcine jejunal epithelium. FEMS Immunol. Med. Microbiol. 54, 215–223 (2008).
Bauerl, C., Perez-Martinez, G., Yan, F., Polk, D. B. & Monedero, V. Functional analysis of the p40 and p75 proteins from Lactobacillus casei BL23. J. Mol. Microbiol. Biotechnol. 19, 231–241 (2010).
Yan, F. et al. Colon-specific delivery of a probiotic-derived soluble protein ameliorates intestinal inflammation in mice through an EGFR-dependent mechanism. J. Clin. Invest. 121, 2242–2253 (2011).
Abu-Qarn, M., Eichler, J. & Sharon, N. Not just for Eukarya anymore: protein glycosylation in Bacteria and Archaea. Curr. Opin. Struct. Biol. 18, 544–550 (2008).
Cario, E. Bacterial interactions with cells of the intestinal mucosa: Toll-like receptors and NOD2. Gut 54, 1182–1193 (2005).
Cario, E., Gerken, G. & Podolsky, D. K. Toll-like receptor 2 enhances ZO-1-associated intestinal epithelial barrier integrity via protein kinase C. Gastroenterology 127, 224–238 (2004).
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004). This article shows that gut commensal bacteria are recognized by TLRs expressed in the intestinal mucosa, and that this recognition plays a crucial part in the maintenance of intestinal epithelial homeostasis during host–microbiota interactions.
Resta-Lenert, S. & Barrett, K. E. Probiotics and commensals reverse TNF-α- and IFN-γ-induced dysfunction in human intestinal epithelial cells. Gastroenterology 130, 731–746 (2006).
Ulluwishewa, D. et al. Regulation of tight junction permeability by intestinal bacteria and dietary components. J. Nutr. 141, 769–776 (2011).
Mangell, P. et al. Lactobacillus plantarum 299v inhibits Escherichia coli-induced intestinal permeability. Dig. Dis. Sci. 47, 511–516 (2002).
Qin, H., Zhang, Z., Hang, X. & Jiang, Y. L. plantarum prevents Enteroinvasive Escherichia coli-induced tight junction proteins changes in intestinal epithelial cells. BMC Microbiol. 9, 63 (2009).
Troost, F. J. et al. Identification of the transcriptional response of human intestinal mucosa to Lactobacillus plantarum WCFS1 in vivo. BMC Genomics 9, 374 (2008).
Mao, Y. et al. The effects of Lactobacillus strains and oat fiber on methotrexate-induced enterocolitis in rats. Gastroenterology 111, 334–344 (1996).
White, J. S. et al. The probiotic bacterium Lactobacillus plantarum species 299 reduces intestinal permeability in experimental biliary obstruction. Lett. Appl. Microbiol. 42, 19–23 (2006).
Christensen, H. R., Frokiaer, H. & Pestka, J. J. Lactobacilli differentially modulate expression of cytokines and maturation surface markers in murine dendritic cells. J. Immunol. 168, 171–178 (2002).
Drakes, M., Blanchard, T. & Czinn, S. Bacterial probiotic modulation of dendritic cells. Infect. Immun. 72, 3299–3309 (2004).
Gately, M. K. et al. The interleukin-12/interleukin-12-receptor system: role in normal and pathologic immune responses. Annu. Rev. Immunol. 16, 495–521 (1998).
Mohamadzadeh, M. et al. Lactobacilli activate human dendritic cells that skew T cells toward T helper 1 polarization. Proc. Natl Acad. Sci. USA 102, 2880–2885 (2005).
Foligne, B. et al. Correlation between in vitro and in vivo immunomodulatory properties of lactic acid bacteria. World J. Gastroenterol. 13, 236–243 (2007).
Latvala, S. et al. Potentially probiotic bacteria induce efficient maturation but differential cytokine production in human monocyte-derived dendritic cells. World J. Gastroenterol. 14, 5570–5583 (2008).
Niers, L. E. et al. Selection of probiotic bacteria for prevention of allergic diseases: immunomodulation of neonatal dendritic cells. Clin. Exp. Immunol. 149, 344–352 (2007).
Vissers, Y. M. et al. Lactobacillus strains differentially modulate cytokine production by hPBMC from pollen-allergic patients. FEMS Immunol. Med. Microbiol. 61, 28–40 (2011).
Peran, L. et al. Preventative effects of a probiotic, Lactobacillus salivarius ssp. salivarius, in the TNBS model of rat colitis. World J. Gastroenterol. 11, 5185–5192 (2005).
Zoumpopoulou, G. et al. Lactobacillus fermentum ACA-DC 179 displays probiotic potential in vitro and protects against trinitrobenzene sulphonic acid (TNBS)-induced colitis and Salmonella infection in murine models. Int. J. Food Microbiol. 121, 18–26 (2008).
Roselli, M. et al. Prevention of TNBS-induced colitis by different Lactobacillus and Bifidobacterium strains is associated with an expansion of γδT and regulatory T cells of intestinal intraepithelial lymphocytes. Inflamm. Bowel Dis. 15, 1526–1536 (2009).
Kwon, H. K. et al. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc. Natl Acad. Sci. USA 107, 2159–2164 (2010).
Mileti, E., Matteoli, G., Iliev, I. D. & Rescigno, M. Comparison of the immunomodulatory properties of three probiotic strains of Lactobacilli using complex culture systems: prediction for in vivo efficacy. PLoS ONE 4, e7056 (2009).
Di Caro, S. et al. The European experience with double-balloon enteroscopy: indications, methodology, safety, and clinical impact. Gastrointest. Endosc. 62, 545–550 (2005).
van Baarlen, P. et al. Differential NF-κB pathways induction by Lactobacillus plantarum in the duodenum of healthy humans correlating with immune tolerance. Proc. Natl Acad. Sci. USA 106, 2371–2376 (2009). Pioneering work that addresses the transcriptional response of the healthy human duodenal mucosa to the oral intake of probiotic-containing products, highlighting the differential responses to different preparations of the same probiotic strain.
van Baarlen, P. et al. Human mucosal in vivo transcriptome responses to three lactobacilli indicate how probiotics may modulate human cellular pathways. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4562–4569 (2011). An in vivo study showing that, in healthy humans, there are differential duodenal transcriptome responses to three commercial probiotic strains; this study also highlights human individuality in baseline transcriptome patterns.
Kalliomaki, M. et al. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet 357, 1076–1079 (2001). This article convincingly shows that consumption of L. rhamnosus str. GG is effective in the prevention of early atopic disease (eczema, rhinitis or asthma) in children at high risk.
Kalliomaki, M., Salminen, S., Poussa, T., Arvilommi, H. & Isolauri, E. Probiotics and prevention of atopic disease: 4-year follow-up of a randomised placebo-controlled trial. Lancet 361, 1869–1871 (2003).
Kalliomaki, M. & Isolauri, E. Role of intestinal flora in the development of allergy. Curr. Opin. Allergy Clin. Immunol. 3, 15–20 (2003).
Schultz, M. et al. Immunomodulatory consequences of oral administration of Lactobacillus rhamnosus strain GG in healthy volunteers. J. Dairy Res. 70, 165–173 (2003).
Gilliland, S. E. Acidophilus milk products: a review of potential benefits to consumers. J. Dairy Sci. 72, 2483–2494 (1989).
Gill, H. S., Rutherfurd, K. J., Prasad, J. & Gopal, P. K. Enhancement of natural and acquired immunity by Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019). Br. J. Nutr. 83, 167–176 (2000).
Paturi, G., Phillips, M. & Kailasapathy, K. Effect of probiotic strains Lactobacillus acidophilus LAFTI L10 and Lactobacillus paracasei LAFTI L26 on systemic immune functions and bacterial translocation in mice. J. Food Prot. 71, 796–801 (2008).
Weiss, G. et al. Lactobacillus acidophilus induces virus immune defence genes in murine dendritic cells by a Toll-like receptor-2-dependent mechanism. Immunology 131, 268–281 (2010).
Bloksma, N., de Heer, E., van Dijk, H. & Willers, J. M. Adjuvanticity of lactobacilli. I. Differential effects of viable and killed bacteria. Clin. Exp. Immunol. 37, 367–375 (1979).
Bloksma, N., Van Dijk, H., Korst, P. & Willers, J. M. Cellular and humoral adjuvant activity of mistletoe extract. Immunobiology 156, 309–318 (1979).
Heinzerling, L., von Baehr, V., Liebenthal, C., von Baehr, R. & Volk, H. D. Immunologic effector mechanisms of a standardized mistletoe extract on the function of human monocytes and lymphocytes in vitro, ex vivo, and in vivo. J. Clin. Immunol. 26, 347–359 (2006).
Karlsson, H., Hessle, C. & Rudin, A. Innate immune responses of human neonatal cells to bacteria from the normal gastrointestinal flora. Infect. Immun. 70, 6688–6696 (2002).
Karlsson, H., Larsson, P., Wold, A. E. & Rudin, A. Pattern of cytokine responses to gram-positive and gram-negative commensal bacteria is profoundly changed when monocytes differentiate into dendritic cells. Infect. Immun. 72, 2671–2678 (2004).
Mane, J. et al. A mixture of Lactobacillus plantarum CECT 7315 and CECT 7316 enhances systemic immunity in elderly subjects: a dose-response, double-blind, placebo-controlled, randomized pilot trial. Nutr. Hosp. 26, 228–235 (2011).
Bouwman, F. G. et al. 2D-electrophoresis and multiplex immunoassay proteomic analysis of different body fluids and cellular components reveal known and novel markers for extended fasting. BMC Med. Genomics 4, 24 (2011).
Rimbach, G. et al. Dietary isoflavones in the prevention of cardiovascular disease: a molecular perspective. Food Chem. Toxicol. 46, 1308–1319 (2008).
Cheng, F. et al. A critical role for Stat3 signalling in immune tolerance. Immunity 19, 425–436 (2003).
Yang, X. O. et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J. Biol. Chem. 282, 9358–9363 (2007).
Chaudhry, A. et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science 326, 986–991 (2009).
Stritesky, G. L. et al. The transcription factor STAT3 is required for T helper 2 cell development. Immunity 34, 39–49 (2011).
de Roos, N. M. & Katan, M. B. Effects of probiotic bacteria on diarrhea, lipid metabolism, and carcinogenesis: a review of papers published between 1988 and 1998. Am. J. Clin. Nutr. 71, 405–411 (2000).
Szajewska, H., Ruszczynski, M. & Radzikowski, A. Probiotics in the prevention of antibiotic-associated diarrhea in children: a meta-analysis of randomized controlled trials. J. Pediatr. 149, 367–372 (2006).
Muller, M. & Kersten, S. Nutrigenomics: goals and strategies. Nature Rev. Genet. 4, 315–322 (2003).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010). A landmark paper that defines the first catalogue of 3.3 million non-redundant genes of the human large-intestinal microbiome.
Arumugam, M. et al. Enterotypes of the human gut microbiome. Nature 473, 174–180 (2011). This article describes a metagenomics-based assessment of the functional composition of the human intestinal microbiome, revealing three distinct host–microorganism symbiotic states, termed enterotypes.
Canani, R. B. et al. Probiotics for treatment of acute diarrhoea in children: randomised clinical trial of five different preparations. BMJ 335, 340 (2007).
Snel, J. et al. Strain-specific immunomodulatory effects of Lactobacillus plantarum strains on birch-pollen-allergic subjects out of season. Clin. Exp. Allergy 41, 232–242 (2011).
Johansson, M. E. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
Walter, J. et al. Identification of Lactobacillus reuteri genes specifically induced in the mouse gastrointestinal tract. Appl. Environ. Microbiol. 69, 2044–2051 (2003).
Denou, E. et al. Gene expression of commensal Lactobacillus johnsonii strain NCC533 during in vitro growth and in the murine gut. J. Bacteriol. 189, 8109–8119 (2007). The first study to investigate a Lactobacillus sp. in situ transcriptome in the mouse intestine.
Willing, B. P., Antunes, L. C., Keeney, K. M., Ferreira, R. B. & Finlay, B. B. Harvesting the biological potential of the human gut microbiome. Bioessays 33, 414–418 (2011).
Zoetendal, E. G., Rajilic-Stojanovic, M. & de Vos, W. M. High-throughput diversity and functionality analysis of the gastrointestinal tract microbiota. Gut 57, 1605–1615 (2008).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl Acad. Sci. USA 105, 16731–16736 (2008).
Atarashi, K. et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 331, 337–341 (2011).
Kunisawa, J. & Kiyono, H. Peaceful mutualism in the gut: revealing key commensal bacteria for the creation and maintenance of immunological homeostasis. Cell Host Microbe 9, 83–84 (2011).
Nelson, K. E. et al. A catalogue of reference genomes from the human microbiome. Science 328, 994–999 (2010).
Gloux, K. et al. Development of high-throughput phenotyping of metagenomic clones from the human gut microbiome for modulation of eukaryotic cell growth. Appl. Environ. Microbiol. 73, 3734–3737 (2007).
Lakhdari, O. et al. Functional metagenomics: a high throughput screening method to decipher microbiota-driven NF-κB modulation in the human gut. PLoS ONE 5, e13092 (2010).
Corcoran, B. M., Stanton, C., Fitzgerald, G. & Ross, R. P. Life under stress: the probiotic stress response and how it may be manipulated. Curr. Pharm. Des. 14, 1382–1399 (2008).
Bron, P. A. & Kleerebezem, M. Engineering lactic acid bacteria for increased industrial functionality. Bioeng. Bugs 2, 1–8 (2011).
Abraham, C. & Medzhitov, R. Interactions between the host innate immune system and microbes in inflammatory bowel disease. Gastroenterology 140, 1729–1737 (2011).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Necrotizing enterocolitis
-
An acute inflammatory condition that is seen primarily in premature infants and results in parts of the bowel dying off.
- Tight junction
-
An area in which the cell membranes from two adjacent vertebrate cells are closely and tightly associated, forming a barrier that is impermeable to liquids and particulate compounds.
- Follicle centres
-
Small, spherical groups of cells; these spheres contain a cavity or centre, in which immune cells (B cells and T cells) proliferate and differentiate.
- Microfold cells
-
(M cells). A cell type that is found in the follicle-associated epithelium of Peyer's patches. M cells have the ability to take up antigens, including bacteria, directly from the small intestinal lumen and to deliver these antigens to the immune cells that are located directly underneath the M cells.
- Peptidoglycan
-
An essential cell wall polymer composed of alternating residues of β-1-4-linked N-acetylmuramic acid and N-acetylglucosamine crosslinked by pentapeptide bridges containing immunomodulatory fragments such as diaminopimelic acid.
- Lipoteichoic acid
-
A D-alanyl- and glycosyl-substituted polyglycerolphosphate polymer that is anchored in the bacterial cytoplasmic membrane through glycolipids.
- Nutrigenomic approaches
-
The application of high-throughput genomic tools in nutrition research to decipher how nutrients affect the production and action of specific gene products and how these in turn affect the response to nutrients.
- Immunological adjuvant
-
An agent that modifies the effect of another agent (often a drug or vaccine) but has few, if any, direct effects when given by itself.
Rights and permissions
About this article
Cite this article
Bron, P., van Baarlen, P. & Kleerebezem, M. Emerging molecular insights into the interaction between probiotics and the host intestinal mucosa. Nat Rev Microbiol 10, 66–78 (2012). https://doi.org/10.1038/nrmicro2690
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2690
This article is cited by
-
Construction of recombinant Lactococcus expressing thymosin and interferon fusion protein and its application as an immune adjuvant
Microbial Cell Factories (2024)
-
Turnover of strain-level diversity modulates functional traits in the honeybee gut microbiome between nurses and foragers
Genome Biology (2023)
-
Changes in intestinal morphology, number of mucus-producing cells and expression of coronavirus receptors APN, DPP4, ACE2 and TMPRSS2 in pigs with aging
Veterinary Research (2023)
-
Gut microbiota-derived 3-phenylpropionic acid promotes intestinal epithelial barrier function via AhR signaling
Microbiome (2023)
-
Artificial-enzymes-armed Bifidobacterium longum probiotics for alleviating intestinal inflammation and microbiota dysbiosis
Nature Nanotechnology (2023)