Key Points
-
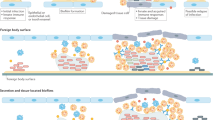
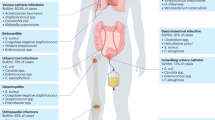
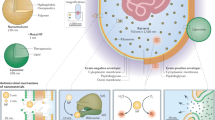
Biofilms harbour complex structural and biological attributes, such as the presence of an extracellular polymeric matrix, physical and chemical heterogeneity and drug tolerance, which provide remarkable therapeutic challenges.
-
Biofilm drug tolerance is a consequence of complex physicochemical and biological properties with multiple microbial genetic and molecular factors, often involving polymicrobial interactions.
-
The challenges to existing antimicrobial or monotherapeutic approaches by the multifactorial nature of biofilm development, combined with drug tolerance, requires robust effective multitargeted or combinatorial therapies.
-
Combinatorial strategies are needed to eliminate existing biofilms by targeting vital structural and functional traits of biofilms, such as the EPS matrix and dormant cells, as well as approaches exploiting host–pathogen interactions.
-
Promising technologies based on 'smart release' or 'on-demand activation' of bioactive agents when triggered by biofilm-derived cues can degrade the matrix and kill resident bacteria, and have the potential to eradicate the pathogenic niche with precision and minimal cytotoxicity to surrounding tissues.
-
Validation of proof-of-concept studies using clinically relevant animal models, as well as clinical trials, are needed for rigorous evaluation.
Abstract
Biofilm formation is a key virulence factor for a wide range of microorganisms that cause chronic infections. The multifactorial nature of biofilm development and drug tolerance imposes great challenges for the use of conventional antimicrobials and indicates the need for multi-targeted or combinatorial therapies. In this Review, we focus on current therapeutic strategies and those under development that target vital structural and functional traits of microbial biofilms and drug tolerance mechanisms, including the extracellular matrix and dormant cells. We emphasize strategies that are supported by in vivo or ex vivo studies, highlight emerging biofilm-targeting technologies and provide a rationale for multi-targeted therapies aimed at disrupting the complex biofilm microenvironment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Flemming, H. C. et al. Biofilms: an emergent form of bacterial life. Nat. Rev. Microbiol. 14, 563–575 (2016).
Stacy, A., McNally, L., Darch, S. E., Brown, S. P. & Whiteley, M. The biogeography of polymicrobial infection. Nat. Rev. Microbiol. 14, 93–105 (2016).
Magin, C. M., Cooper, S. P. & Brennan, A. B. Non-toxic antifouling strategies. Mater. Today 13, 36–44 (2010).
Hobley, L., Harkins, C., MacPhee, C. E. & Stanley-Wall, N. R. Giving structure to the biofilm matrix: an overview of individual strategies and emerging common themes. FEMS Microbiol. Rev. 39, 649–669 (2015).
Peterson, B. W. et al. Viscoelasticity of biofilms and their recalcitrance to mechanical and chemical challenges. FEMS Microbiol. Rev. 39, 234–245 (2015).
Van Acker, H., Van Dijck, P. & Coenye, T. Molecular mechanisms of antimicrobial tolerance and resistance in bacterial and fungal biofilms. Trends Microbiol. 22, 326–333 (2014).
Lebeaux, D., Ghigo, J.-M. & Beloin, C. Biofilm-related infections: bridging the gap between clinical management and fundamental aspects of recalcitrance toward antibiotics. Microbiol. Mol. Biol. Rev. 78, 510–543 (2014).
Koo, H. & Yamada, K. M. Dynamic cell–matrix interactions modulate microbial biofilm and tissue 3D microenvironments. Curr. Opin. Cell Biol. 42, 102–112 (2016).
Hoiby, N. et al. ESCMID guideline for the diagnosis and treatment of biofilm infections 2014. Clin. Microbiol. Infect. 21 (Suppl. 1), S1–25 (2015).
Velkov, T., Roberts, K. D. & Li, J. Rediscovering the octapeptins. Nat. Prod. Rep. 34, 295–309 (2017).
Raad, I. et al. Successful salvage of central venous catheters in patients with catheter-related or central line-associated bloodstream infections by using a catheter lock solution consisting of minocycline, EDTA, and 25% ethanol. Antimicrob. Agents Chemother. 60, 3426–3432 (2016).
Mistry, S. et al. A novel, multi-barrier, drug eluting calcium sulfate/biphasic calcium phosphate biodegradable composite bone cement for treatment of experimental MRSA osteomyelitis in rabbit model. J. Control. Release 239, 169–181 (2016).
Lemire, J. A., Harrison, J. J. & Turner, R. J. Antimicrobial activity of metals: mechanisms, molecular targets and applications. Nat. Rev. Micro 11, 371–384 (2013).
Castaneda, P., McLaren, A., Tavaziva, G. & Overstreet, D. Biofilm antimicrobial susceptibility increases with antimicrobial exposure time. Clin. Orthop. Relat. Res. 474, 1659–1664 (2016). This article illustrates the importance of extended exposure time as well antibiotic concentration required to significantly reduce or eradicate biofilms. Conventional MIC and MBC are based on a 24 h exposure time, but the susceptibility of biofilm bacteria decreased with prolonged exposure.
Howlin, R. P. et al. Antibiotic-loaded synthetic calcium sulfate beads for prevention of bacterial colonization and biofilm formation in periprosthetic infections. Antimicrob. Agents Chemother. 59, 111–120 (2015).
Besinis, A., De Peralta, T., Tredwin, C. J. & Handy, R. D. Review of nanomaterials in dentistry: interactions with the oral microenvironment, clinical applications, hazards, and benefits. ACS Nano 9, 2255–2289 (2015).
Fabbri, S. et al. Streptococcus mutans biofilm transient viscoelastic fluid behaviour during high-velocity microsprays. J. Mech. Behav. Biomed. Mater. 59, 197–206 (2016).
Urish, K. L., DeMuth, P. W., Craft, D. W., Haider, H. & Davis, C. M. Pulse lavage is inadequate at removal of biofilm from the surface of total knee arthroplasty materials. J. Arthroplasty 29, 1128–1132 (2014).
Flemming, H.-C. & Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 8, 623–633 (2010).
Gunn, J. S., Bakaletz, L. O. & Wozniak, D. J. What's on the outside matters: the role of the extracellular polymeric substance of Gram-negative biofilms in evading host immunity and as a target for therapeutic intervention. J. Biol. Chem. 291, 12538–12546 (2016).
Peng, X., Zhang, Y., Bai, G., Zhou, X. & Wu, H. Cyclic di-AMP mediates biofilm formation. Mol. Microbiol. 99, 945–959 (2016).
Mann, E. E. & Wozniak, D. J. Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol. Rev. 36, 893–916 (2012).
Teschler, J. K. et al. Living in the matrix: assembly and control of Vibrio cholerae biofilms. Nat. Rev. Microbiol. 13, 255–268 (2015).
Fernicola, S. et al. In silico discovery and in vitro validation of catechol-containing sulfonohydrazide compounds as potent inhibitors of the diguanylate cyclase PleD. J. Bacteriol. 198, 147–156 (2015).
Sambanthamoorthy, K. et al. Identification of small molecules that antagonize diguanylate cyclase enzymes to inhibit biofilm formation. Antimicrob. Agents Chemother. 56, 5202–5211 (2012).
Falsetta, M. L. et al. Novel antibiofilm chemotherapy targets exopolysaccharide synthesis and stress tolerance in Streptococcus mutans to modulate virulence expression in vivo. Antimicrob. Agents Chemother. 56, 6201–6211 (2012).
Ren, Z. et al. Molecule targeting glucosyltransferase inhibits Streptococcus mutans biofilm formation and virulence. Antimicrob. Agents Chemother. 60, 126–135 (2015).
Totsika, M. et al. A FimH inhibitor prevents acute bladder infection and treats chronic cystitis caused by multidrug-resistant uropathogenic Escherichia coli ST131. J. Infect. Dis. 208, 921–928 (2013).
Nett, J. E., Cabezas-Olcoz, J., Marchillo, K., Mosher, D. F. & Andes, D. R. Targeting fibronectin to disrupt in vivo Candida albicans biofilms. Antimicrob. Agents Chemother. 60, 3152–3155 (2016).
Guiton, P. S. et al. Combinatorial small-molecule therapy prevents uropathogenic Escherichia coli catheter-associated urinary tract infections in mice. Antimicrob. Agents Chemother. 56, 4738–4745 (2012).
Spaulding, C. N. et al. Selective depletion of uropathogenic E. coli from the gut by a FimH antagonist. Nature 546, 528–532 (2017).
Mydock-McGrane, L. et al. Antivirulence C-mannosides as antibiotic-sparing, oral therapeutics for urinary tract infections. J. Med. Chem. 59, 9390–9408 (2016).
Cegelski, L. et al. Small-molecule inhibitors target Escherichia coli amyloid biogenesis and biofilm formation. Nat. Chem. Biol. 5, 913–919 (2009).
Cozens, D. & Read, R. C. Anti-adhesion methods as novel therapeutics for bacterial infections. Expert Rev. Anti Infect. Ther. 10, 1457–1468 (2012).
Kaplan, J. B. Biofilm matrix-degrading enzymes. Methods Mol. Biol. 1147, 203–213 (2014).
Pleszczynska, M., Wiater, A., Janczarek, M. & Szczodrak, J. (1,3)-α-d-Glucan hydrolases in dental biofilm prevention and control: A review. Int. J. Biol. Macromol. 79, 761–778 (2015).
Fleming, D., Chahin, L. & Rumbaugh, K. Glycoside hydrolases degrade polymicrobial bacterial biofilms in wounds. Antimicrob. Agents Chemother. 61, e01998-16 (2016).
Iwase, T. et al. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature 465, 346–349 (2010).
Schmelcher, M. et al. Evolutionarily distinct bacteriophage endolysins featuring conserved peptidoglycan cleavage sites protect mice from MRSA infection. J. Antimicrob. Chemother. 70, 1453–1465 (2015).
Becker, S. C. et al. Triple-acting lytic enzyme treatment of drug-resistant and intracellular Staphylococcus aureus. Sci. Rep. 6, 25063 (2016).
Baker, P. et al. Exopolysaccharide biosynthetic glycoside hydrolases can be utilized to disrupt and prevent Pseudomonas aeruginosa biofilms. Sci. Adv. 2, e1501632 (2016).
Okshevsky, M., Regina, V. R. & Meyer, R. L. Extracellular DNA as a target for biofilm control. Curr. Opin. Biotechnol. 33, 73–80 (2015).
Kaplan, J. B. et al. Low levels of β-lactam antibiotics induce extracellular DNA release and biofilm formation in Staphylococcus aureus. mBio 3, e00198-12 (2012).
Hymes, S. R., Randis, T. M., Sun, T. Y. & Ratner, A. J. DNase inhibits Gardnerella vaginalis biofilms in vitro and in vivo. J. Infect. Dis. 207, 1491–1497 (2013).
Manzenreiter, R. et al. Ultrastructural characterization of cystic fibrosis sputum using atomic force and scanning electron microscopy. J. Cyst. Fibros. 11, 84–92 (2012).
Konstan, M. W. & Ratjen, F. Effect of dornase alfa on inflammation and lung function: potential role in the early treatment of cystic fibrosis. J. Cyst. Fibros. 11, 78–83 (2012).
Thornton, R. B. et al. Neutrophil extracellular traps and bacterial biofilms in middle ear effusion of children with recurrent acute otitis media—a potential treatment target. PLoS ONE 8, e53837 (2013).
Telethon Kids Institute. Dissolving the glue in glue ear. Telethon Kids Institute https://www.telethonkids.org.au/our-research/early-environment/infection-and-vaccines/vaccine-trials-group/dissolving-the-glue-in-glue-ear/ (2017).
Baelo, A. et al. Disassembling bacterial extracellular matrix with DNase-coated nanoparticles to enhance antibiotic delivery in biofilm infections. J. Control Release 209, 150–158 (2015).
Liu, Y. et al. Topical delivery of low-cost protein drug candidates made in chloroplasts for biofilm disruption and uptake by oral epithelial cells. Biomaterials 105, 156–166 (2016).
Bhattacharya, M., Wozniak, D. J., Stoodley, P. & Hall-Stoodley, L. Prevention and treatment of Staphylococcus aureus biofilms. Expert Rev. Anti Infect. Ther. 13, 1499–1516 (2015).
DiGiandomenico, A. et al. Identification of broadly protective human antibodies to Pseudomonas aeruginosa exopolysaccharide Psl by phenotypic screening. J. Exp. Med. 209, 1273–1287 (2012).
Flores-Mireles, A. L., Pinkner, J. S., Caparon, M. G. & Hultgren, S. J. EbpA vaccine antibodies block binding of Enterococcus faecalis to fibrinogen to prevent catheter-associated bladder infection in mice. Sci. Transl Med. 6, 254ra127 (2014). This study shows that implant-associated biofilms can bind to host components on the implant surface and that abrogating microbial binding to host fibrinogen inhibits biofilm formation and catheter-associated bladder infection, and underscores the utility of using host–microbial infection models.
Brady, R. A. et al. Resolution of Staphylococcus aureus biofilm infection using vaccination and antibiotic treatment. Infect. Immun. 79, 1797–1803 (2011).
Goodman, S. D. et al. Biofilms can be dispersed by focusing the immune system on a common family of bacterial nucleoid-associated proteins. Mucosal Immunol. 4, 625–637 (2011). This study shows that the DNABII family of proteins can disrupt the eDNA component of the EPS scaffold as a combinatorial approach to biofilm infection in an animal model of otitis media.
Rocco, C. J., Davey, M. E., Bakaletz, L. O. & Goodman, S. D. Natural antigenic differences in the functionally equivalent extracellular DNABII proteins of bacterial biofilms provide a means for targeted biofilm therapeutics. Mol. Oral Microbiol. 32, 118–130 (2016).
Devaraj, A., Justice, S. S., Bakaletz, L. O. & Goodman, S. D. DNABII proteins play a central role in UPEC biofilm structure. Mol. Microbiol. 96, 1119–1135 (2015).
Novotny, L. A., Jurcisek, J. A., Goodman, S. D. & Bakaletz, L. O. Monoclonal antibodies against DNA-binding tips of DNABII proteins disrupt biofilms in vitro and induce bacterial clearance in vivo. EBioMedicine 10, 33–44 (2016).
Estelles, A. et al. A high-affinity native human antibody disrupts biofilm from Staphylococcus aureus bacteria and potentiates antibiotic efficacy in a mouse implant infection model. Antimicrob. Agents Chemother. 60, 2292–2301 (2016).
Freire, M. O. et al. A bacterial-biofilm-induced oral osteolytic infection can be successfully treated by immuno-targeting an extracellular nucleoid-associated protein. Mol. Oral Microbiol. 32, 74–88 (2017).
Novotny, L. A. et al. Antibodies against the majority subunit of type IV pili disperse nontypeable Haemophilus influenzae biofilms in a LuxS-dependent manner and confer therapeutic resolution of experimental otitis media. Mol. Microbiol. 96, 276–292 (2015).
McDougald, D., Rice, S. A., Barraud, N., Steinberg, P. D. & Kjelleberg, S. Should we stay or should we go: mechanisms and ecological consequences for biofilm dispersal. Nat. Rev. Microbiol. 10, 39–50 (2011).
Romling, U. & Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 272, 541–561 (2012).
Christensen, L. D. et al. Clearance of Pseudomonas aeruginosa foreign-body biofilm infections through reduction of the cyclic di-GMP level in the bacteria. Infect. Immun. 81, 2705–2713 (2013).
Burdette, D. L. et al. STING is a direct innate immune sensor of cyclic di-GMP. Nature 478, 515–518 (2011).
Barraud, N. et al. Nitric oxide signaling in Pseudomonas aeruginosa biofilms mediates phosphodiesterase activity, decreased cyclic di-GMP levels, and enhanced dispersal. J. Bacteriol. 191, 7333–7342 (2009). This study demonstrates the first link between NO and c-di-GMP signalling in triggering dispersal of P. aeruginosa biofilms.
Barraud, N., Kelso, M. J., Rice, S. A. & Kjelleberg, S. Nitric oxide: a key mediator of biofilm dispersal with applications in infectious diseases. Curr. Pharm. Des. 21, 31–42 (2015).
Howlin, R. P. et al. Low dose nitric oxide as targeted anti-biofilm adjunctive therapy to treat chronic Pseudomonas aeruginosa infection in cystic fibrosis. Mol. Ther. 25, 2104–2116 (2017). This proof-of-concept double-blind clinical trial demonstrates that NO, when administered as an adjuvant, decreases P. aeruginosa biofilm aggregates and enhances antibiotic efficacy in patients with cystic fibrosis.
Deppisch, C. et al. Gaseous nitric oxide to treat antibiotic resistant bacterial and fungal lung infections in patients with cystic fibrosis: a phase I clinical study. Infection 44, 513–520 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02498535 (2017).
Barraud, N. et al. Cephalosporin-3′-diazeniumdiolates: targeted NO-donor prodrugs for dispersing bacterial biofilms. Angew. Chem. Int. Ed Engl. 51, 9057–9060 (2012).
Collins, S. A. et al. Cephalosporin-3′-diazeniumdiolate NO-donor prodrug PYRRO-C3D enhances azithromycin susceptibility of Non-typeable Haemophilus influenzae biofilms. Antimicrob. Agents Chemother. 61, e02086-16 (2016).
Walker, W. T. et al. Primary ciliary dyskinesia ciliated airway cells show increased susceptibility to Haemophilus influenzae biofilm formation. Eur. Respir. J. 50, 1700612 (2017).
de la Fuente-Nunez, C., Reffuveille, F., Fairfull-Smith, K. E. & Hancock, R. E. Effect of nitroxides on swarming motility and biofilm formation, multicellular behaviors in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 57, 4877–4881 (2013).
Reffuveille, F., Fuente-Nunez Cde, L., Fairfull-Smith, K. E. & Hancock, R. E. Potentiation of ciprofloxacin action against Gram-negative bacterial biofilms by a nitroxide. Pathog. Dis. 73, ftv016 (2015).
Verderosa, A. D., Mansour, S. C., de la Fuente-Nunez, C., Hancock, R. E. & Fairfull-Smith, K. E. Synthesis and evaluation of ciprofloxacin–nitroxide conjugates as anti-biofilm agents. Molecules 21, E841 (2016).
Kutty, S. K. et al. Design, synthesis, and evaluation of fimbrolide–nitric oxide donor hybrids as antimicrobial agents. J. Med. Chem. 56, 9517–9529 (2013).
Anderson, J. K. et al. Chemorepulsion from the quorum signal autoinducer-2 promotes Helicobacter pylori biofilm dispersal. mBio 6, e00379 (2015).
Lauderdale, K. J., Malone, C. L., Boles, B. R., Morcuende, J. & Horswill, A. R. Biofilm dispersal of community-associated methicillin-resistant Staphylococcus aureus on orthopedic implant material. J. Orthop. Res. 28, 55–61 (2010).
Simonetti, O. et al. RNAIII-inhibiting peptide enhances healing of wounds infected with methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 52, 2205–2211 (2008).
Starkey, M. et al. Identification of anti-virulence compounds that disrupt quorum-sensing regulated acute and persistent pathogenicity. PLoS Pathog. 10, e1004321 (2014). This study identifies QSIs that inhibit P. aeruginosa cell-to-cell communication in vivo , and also decrease the formation of persister populations.
Brackman, G., Cos, P., Maes, L., Nelis, H. J. & Coenye, T. Quorum sensing inhibitors increase the susceptibility of bacterial biofilms to antibiotics in vitro and in vivo. Antimicrob. Agents Chemother. 55, 2655–2661 (2011).
Nascimento, M. M. et al. The effect of arginine on oral biofilm communities. Mol. Oral Microbiol. 29, 45–54 (2014).
He, J. et al. L-Arginine modifies the exopolysaccharide matrix and thwarts Streptococcus mutans outgrowth within mixed-species oral biofilms. J. Bacteriol. 198, 2651–2661 (2016).
Jakubovics, N. S. et al. Critical roles of arginine in growth and biofilm development by Streptococcus gordonii. Mol. Microbiol. 97, 281–300 (2015).
Kolderman, E. et al. L-Arginine destabilizes oral multi-species biofilm communities developed in human saliva. PLoS ONE 10, e0121835 (2015).
Gnanadhas, D. P., Elango, M., Datey, A. & Chakravortty, D. Chronic lung infection by Pseudomonas aeruginosa biofilm is cured by l-methionine in combination with antibiotic therapy. Sci. Rep. 5, 16043 (2015).
Banin, E., Vasil, M. L. & Greenberg, E. P. Iron and Pseudomonas aeruginosa biofilm formation. Proc. Natl Acad. Sci. USA 102, 11076–11081 (2005).
Oglesby-Sherrouse, A. G., Djapgne, L., Nguyen, A. T., Vasil, A. I. & Vasil, M. L. The complex interplay of iron, biofilm formation, and mucoidy affecting antimicrobial resistance of Pseudomonas aeruginosa. Pathog. Dis. 70, 307–320 (2014).
Lin, M. H., Shu, J. C., Huang, H. Y. & Cheng, Y. C. Involvement of iron in biofilm formation by Staphylococcus aureus. PLoS ONE 7, e34388 (2012).
Garcia, C. A., Alcaraz, E. S., Franco, M. A. & Passerini de Rossi, B. N. Iron is a signal for Stenotrophomonas maltophilia biofilm formation, oxidative stress response, OMPs expression, and virulence. Front. Microbiol. 6, 926 (2015).
Moreau-Marquis, S. et al. The ΔF508-CFTR mutation results in increased biofilm formation by Pseudomonas aeruginosa by increasing iron availability. Am. J. Physiol. Lung Cell. Mol. Physiol. 295, L25–L37 (2008).
Kaneko, Y., Thoendel, M., Olakanmi, O., Britigan, B. E. & Singh, P. K. The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity. J. Clin. Invest. 117, 877–888 (2007). This study uses two different models to explore the host–biofilm interactions that contribute to chronic infection and a novel antimicrobial approach to abrogate P. aeruginosa biofilm infection by exploiting a Pseudomonas 'Achilles heel'.
Moreau-Marquis, S., O'Toole, G. A. & Stanton, B. A. Tobramycin and FDA-approved iron chelators eliminate Pseudomonas aeruginosa biofilms on cystic fibrosis cells. Am. J. Respir. Cell. Mol. Biol. 41, 305–313 (2009). One of the first studies to use a human primary epithelial cell co-culture model to study relevant host–biofilm interactions. It shows P. aeruginosa biofilm formation characterized by antibiotic tolerance on airway cells with a common CFTR mutation and that iron metabolism has a key role.
Hunter, R. C. et al. Ferrous iron is a significant component of bioavailable iron in cystic fibrosis airways. mBio 4, e00557-13 (2013).
Lu, M. & Hansen, E. N. Hydrogen peroxide wound irrigation in orthopaedic surgery. J. Bone Joint Infect. 2, 3–9 (2017).
Ordinola-Zapata, R., Bramante, C., Aprecio, R., Handysides, R. & Jaramillo, D. Biofilm removal by 6% sodium hypochlorite activated by different irrigation techniques. Int. Endod. J. 47, 659–666 (2014).
Liu, H., Wei, X., Ling, J., Wang, W. & Huang, X. Biofilm formation capability of Enterococcus faecalis cells in starvation phase and its susceptibility to sodium hypochlorite. J. Endod. 36, 630–635 (2010).
von Ohle, C. et al. Real-time microsensor measurement of local metabolic activities in ex vivo dental biofilms exposed to sucrose and treated with chlorhexidine. Appl. Environ. Microbiol. 76, 2326–2334 (2010).
Mihailescu, R. et al. High activity of fosfomycin and rifampin against methicillin-resistant Staphylococcus aureus biofilm in vitro and in an experimental foreign-body infection model. Antimicrob. Agents Chemother. 58, 2547–2553 (2014).
Conlon, B. P. et al. Activated ClpP kills persisters and eradicates a chronic biofilm infection. Nature 503, 365–370 (2013).
Pletzer, D., Coleman, S. R. & Hancock, R. E. Anti-biofilm peptides as a new weapon in antimicrobial warfare. Curr. Opin. Microbiol. 33, 35–40 (2016).
Batoni, G., Maisetta, G. & Esin, S. Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim. Biophys. Acta 1858, 1044–1060 (2016).
Guo, L. et al. Precision-guided antimicrobial peptide as a targeted modulator of human microbial ecology. Proc. Natl Acad. Sci. USA 112, 7569–7574 (2015).
de la Fuente-Nunez, C. et al. D-Enantiomeric peptides that eradicate wild-type and multidrug-resistant biofilms and protect against lethal Pseudomonas aeruginosa infections. Chem. Biol. 22, 196–205 (2015).
Jones, E. A., McGillivary, G. & Bakaletz, L. O. Extracellular DNA within a nontypeable Haemophilus influenzae-induced biofilm binds human beta defensin-3 and reduces its antimicrobial activity. J. Innate Immun. 5, 24–38 (2013).
Yu, K. et al. Anti-adhesive antimicrobial peptide coating prevents catheter associated infection in a mouse urinary infection model. Biomaterials 116, 69–81 (2017).
Lam, S. J. et al. Combating multidrug-resistant Gram-negative bacteria with structurally nanoengineered antimicrobial peptide polymers. Nat. Microbiol. 1, 16162 (2016).
Scott, R. W. & Tew, G. N. Mimics of host defense proteins; strategies for translation to therapeutic applications. Curr. Top. Med. Chem. 17, 576–589 (2017).
Busscher, H. J. et al. Biomaterial-associated infection: locating the finish line in the race for the surface. Sci. Transl Med. 4, 153rv110 (2012).
De Jong, W. H. et al. Systemic and immunotoxicity of silver nanoparticles in an intravenous 28 days repeated dose toxicity study in rats. Biomaterials 34, 8333–8343 (2013).
Swartjes, J. J. et al. Current developments in antimicrobial surface coatings for biomedical applications. Curr. Med. Chem. 22, 2116–2129 (2015).
Schumacher, J. F. et al. Species-specific engineered antifouling topographies: correlations between the settlement of algal zoospores and barnacle cyprids. Biofouling 23, 307–317 (2007).
May, R. M. et al. An engineered micropattern to reduce bacterial colonization, platelet adhesion and fibrin sheath formation for improved biocompatibility of central venous catheters. Clin. Transl Med. 4, 9 (2015).
Falde, E. J., Yohe, S. T., Colson, Y. L. & Grinstaff, M. W. Superhydrophobic materials for biomedical applications. Biomaterials 104, 87–103 (2016).
Damodaran, V. B. & Murthy, N. S. Bio-inspired strategies for designing antifouling biomaterials. Biomaterials Res. 20, 18 (2016).
Zeng, G., Ogaki, R. & Meyer, R. L. Non-proteinaceous bacterial adhesins challenge the antifouling properties of polymer brush coatings. Acta Biomater. 24, 64–73 (2015).
Wen, L., Tian, Y. & Jiang, L. Bioinspired super-wettability from fundamental research to practical applications. Angew. Chem. Int. Ed Engl. 54, 3387–3399 (2015).
Bai, X., Xue, C. H. & Jia, S. T. Surfaces with sustainable superhydrophobicity upon mechanical abrasion. ACS Appl. Mater. Interfaces 8, 28171–28179 (2016).
Fadeeva, E. et al. Bacterial retention on superhydrophobic titanium surfaces fabricated by femtosecond laser ablation. Langmuir 27, 3012–3019 (2011).
Gilabert-Porres, J. et al. Design of a nanostructured active surface against Gram-positive and Gram-negative bacteria through plasma activation and in situ silver reduction. ACS Appl. Mater. Interfaces 8, 64–73 (2016).
Paula, A. J. & Koo, H. Nanosized building blocks for customizing novel antibiofilm approaches. J. Dent. Res. 96, 128–136 (2017).
Bayramov, D. F. & Neff, J. A. Beyond conventional antibiotics — new directions for combination products to combat biofilm. Adv. Drug Deliv. Rev. 112, 48–60 (2016).
Junter, G.-A., Thébault, P. & Lebrun, L. Polysaccharide-based antibiofilm surfaces. Acta Biomater. 30, 13–25 (2016).
Palumbo, F. S. et al. A polycarboxylic/amino functionalized hyaluronic acid derivative for the production of pH sensible hydrogels in the prevention of bacterial adhesion on biomedical surfaces. Int. J. Pharm. 478, 70–77 (2015).
Ciofu, O., Tolker-Nielsen, T., Jensen, P. O., Wang, H. & Hoiby, N. Antimicrobial resistance, respiratory tract infections and role of biofilms in lung infections in cystic fibrosis patients. Adv. Drug Deliv. Rev. 85, 7–23 (2015).
Liu, W. et al. Synthesis of TiO2 nanotubes with ZnO nanoparticles to achieve antibacterial properties and stem cell compatibility. Nanoscale 6, 9050–9062 (2014).
Jia, Z. et al. Bioinspired anchoring AgNPs onto micro-nanoporous TiO2 orthopedic coatings: trap-killing of bacteria, surface-regulated osteoblast functions and host responses. Biomaterials 75, 203–222 (2016).
Ashbaugh, A. G. et al. Polymeric nanofiber coating with tunable combinatorial antibiotic delivery prevents biofilm-associated infection in vivo. Proc. Natl Acad. Sci. USA 113, E6919–E6928 (2016). This article shows that combinations of antibiotic- loaded coatings are effective in preventing infection of the bone and joint tissue and implant biofilm formation in a mouse model of orthopaedic implant infection.
Min, J. et al. Designer dual therapy nanolayered implant coatings eradicate biofilms and accelerate bone tissue repair. ACS Nano 10, 4441–4450 (2016). This study demonstrates multi-layered implant nanocoatings that sequentially deliver an antibiotic and an osteoinductive growth factor to eliminate biofilms and promote osteointegration with host tissue.
Holzapfel, B. M. et al. How smart do biomaterials need to be? A translational science and clinical point of view. Adv. Drug Deliv. Rev. 65, 581–603 (2013).
Zhuang, J., Gordon, M. R., Ventura, J., Li, L. & Thayumanavan, S. Multi-stimuli responsive macromolecules and their assemblies. Chem. Soc. Rev. 42, 7421–7435 (2013).
Shchukin, D. & Mohwald, H. Materials science. A coat of many functions. Science 341, 1458–1459 (2013).
Schütz, C. A., Juillerat-Jeanneret, L., Mueller, H., Lynch, I. & Riediker, M. Therapeutic nanoparticles in clinics and under clinical evaluation. Nanomedicine 8, 449–467 (2013).
Natan, M. & Banin, E. From nano to micro: using nanotechnology to combat microorganisms and their multidrug resistance. FEMS Microbiol. Rev. 41, 302–322 (2017).
Rukavina, Z. & Vanic´, Ž. Current trends in development of liposomes for targeting bacterial biofilms. Pharmaceutics 8, E18 (2016).
Forier, K. et al. Lipid and polymer nanoparticles for drug delivery to bacterial biofilms. J. Control. Release 190, 607–623 (2014).
Alipour, M., Suntres, Z. E., Halwani, M., Azghani, A. O. & Omri, A. Activity and interactions of liposomal antibiotics in presence of polyanions and sputum of patients with cystic fibrosis. PLoS ONE 4, e5724 (2009).
Zazo, H., Colino, C. I. & Lanao, J. M. Current applications of nanoparticles in infectious diseases. J. Control. Release 224, 86–102 (2016).
Cipolla, D., Blanchard, J. & Gonda, I. Development of liposomal ciprofloxacin to treat lung infections. Pharmaceutics 8, 6 (2016).
Clancy, J. P. et al. Phase II studies of nebulised Arikace in CF patients with Pseudomonas aeruginosa infection. Thorax 68, 818–825 (2013). This study demonstrates good host tolerance, safety and efficacy of liposomal amikacin in a phase II double-blind placebo-controlled study of 105 patients with cystic fibrosis.
Jardeleza, C. et al. An in vivo safety and efficacy demonstration of a topical liposomal nitric oxide donor treatment for Staphylococcus aureus biofilm-associated rhinosinusitis. Transl Res. 166, 683–692 (2015).
Gao, L. et al. Nanocatalysts promote Streptococcus mutans biofilm matrix degradation and enhance bacterial killing to suppress dental caries in vivo. Biomaterials 101, 272–284 (2016).
Benoit, D. S. & Koo, H. Targeted, triggered drug delivery to tumor and biofilm microenvironments. Nanomedicine 11, 873–879 (2016).
Liu, Y. et al. Surface-adaptive, antimicrobially loaded, micellar nanocarriers with enhanced penetration and killing efficiency in staphylococcal biofilms. ACS Nano 10, 4779–4789 (2016).
Horev, B. et al. pH-activated nanoparticles for controlled topical delivery of farnesol to disrupt oral biofilm virulence. ACS Nano 9, 2390–2404 (2015). This study shows the biofilm-targeting efficacy of polymeric nanocarriers that release an antibacterial agent when triggered by acidic pH to disrupt the onset and severity of an oral disease (tooth decay) in vivo.
Radovic-Moreno, A. F. et al. Surface charge-switching polymeric nanoparticles for bacterial cell wall-targeted delivery of antibiotics. ACS Nano 6, 4279–4287 (2012).
Deacon, J. et al. Antimicrobial efficacy of tobramycin polymeric nanoparticles for Pseudomonas aeruginosa infections in cystic fibrosis: formulation, characterisation and functionalisation with dornase alfa (DNase). J. Control. Release 198, 55–61 (2015). This study shows the synthesis of polymeric nanoparticles for the delivery of tobramycin functionalized with dornase alfa to improve penetration into sputum from patients with cystic fibrosis.
Ahmadi, M. S. et al. Sustained nitric oxide-releasing nanoparticles induce cell death in Candida albicans yeast and hyphal cells, preventing biofilm formation in vitro and in a rodent central venous catheter model. Antimicrob. Agents Chemother. 60, 2185–2194 (2016).
Mihu, M. R. et al. Sustained nitric oxide-releasing nanoparticles interfere with methicillin-resistant Staphylococcus aureus adhesion and biofilm formation in a rat central venous catheter model. Antimicrob. Agents Chemother. 61, e02020-16 (2017).
Jo, H. & Ban, C. Aptamer–nanoparticle complexes as powerful diagnostic and therapeutic tools. Exp. Mol. Med. 48, e230 (2016).
Watters, C., Everett, J. A., Haley, C., Clinton, A. & Rumbaugh, K. P. Insulin treatment modulates the host immune system to enhance Pseudomonas aeruginosa wound biofilms. Infect. Immun. 82, 92–100 (2014).
Hajishengallis, G. et al. Complement inhibition in pre-clinical models of periodontitis and prospects for clinical application. Semin. Immunol. 28, 285–291 (2016).
Hall-Stoodley, L., Costerton, J. W. & Stoodley, P. Bacterial biofilms: from the natural environment to infectious diseases. Nat. Rev. Microbiol. 2, 95–108 (2004).
Bjarnsholt, T. et al. The in vivo biofilm. Trends Microbiol. 21, 466–474 (2013).
Papenfort, K. & Bassler, B. L. Quorum sensing signal–response systems in Gram-negative bacteria. Nat. Rev. Microbiol. 14, 576–588 (2016).
Strebhardt, K. & Ullrich, A. Paul Ehrlich's magic bullet concept: 100 years of progress. Nat. Rev. Cancer 8, 473–480 (2008).
Isaacs, A. & Lindenmann, J. Virus interference. I. The interferon. Proc. R. Soc. Lond. B Biol. Sci. 147, 258–267 (1957).
Varmus, H. Cancers: a “constellation” of diseases. NIH MedlinePlus https://medlineplus.gov/magazine/issues/winter13/articles/winter13pg2-3.html (2013).
Brauner, A., Fridman, O., Gefen, O. & Balaban, N. Q. Distinguishing between resistance, tolerance and persistence to antibiotic treatment. Nat. Rev. Microbiol. 14, 320–330 (2016).
Conlon, B. P., Rowe, S. E. & Lewis, K. Persister cells in biofilm associated infections. Adv. Exp. Med. Biol. 831, 1–9 (2015).
Stewart, P. S. et al. Contribution of stress responses to antibiotic tolerance in Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 59, 3838–3847 (2015).
Acknowledgements
Work in the authors' laboratory is supported, in part, by the US National Institute for Dental and Craniofacial Research grants DE018023, DE025220 and DE025848 (H.K.); The Ohio State University Infectious Disease Discovery Theme- Public Health Preparedness for Infectious Disease Transdisciplinary Team Grant (P.S.). The authors also thank the helpful comments of the reviewers. In addition, the authors regret that several important studies could only indirectly be acknowledged through review articles owing to space and reference number limitations.
Author information
Authors and Affiliations
Contributions
P.S. and L.H.-S. conceptualized the original outline of the article and managed its content and production. H.K., R.N.A., R.P.H., L.H.-S. and P.S. researched data for the article, made substantial contributions to discussions of the content, wrote the article and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
P.S. has received research funding from and/or has consulted from Philips Oral Healthcare, Smith & Nephew, Biocomposites Ltd., Zimmer-Biomet, Colgate-Palmolive. H.K. has received funding from Johnson & Johnson, Colgate–Palmolive and Dentsply. R.P.H. has consulted for Biocomposites Ltd. R.N.A and L.H.-S. declare no competing interests.
Supplementary information
Supplementary information S1 (table)
Overview of Current and Prospective Anti-Biofilm Strategies (DOCX 75 kb)
Glossary
- Extracellular polymeric substance
-
(EPS). The EPS can contain exopolysaccharides, fibrous and globular proteins (including extracellular enzymes), lipids and nucleic acids (eDNA). These components form a matrix that can be surface-associated or secreted locally, or deposited on abiotic and biotic surfaces. The EPS matrix functions as a 'multifunctional scaffold' that supports and protects embedded bacteria.
- Antimicrobial chemotherapy
-
The clinical treatment of microbial infections with antimicrobial agents.
- Lock therapy
-
An approach whereby high concentrations of antibiotics are injected into the catheter lumen for an extended period of time to eradicate bacteria. Catheter locks have been used to treat sepsis since the 1980s; however, with the understanding that infecting microorganisms are present as biofilms on medical device materials, this approach is now specifically tailored to improve efficacy.
- Adhesins
-
Bacterial or fungal surface-associated determinants that mediate adherence to living cells or attachment to abiotic surfaces and can promote virulence.
- Mannosides
-
A mannose glycoside consisting of a carbohydrate bound to the hydroxyl group of another compound by O-, N-, S- or C-glycosidic bonds, each with different susceptibilities to hydrolysis.
- Curli
-
A class of bacterial amyloid (aggregates of proteins that form insoluble fibres) produced by many members of the Enterobacteriaceae and a major component of the extracellular matrix, promoting surface adhesion, cell aggregation and biofilm formation.
- Type I pili
-
Filamentous surface structures that have a FimH adhesin at the pilus tip, mediating adherence to host cells and uropathogenic Escherichia coli invasion of bladder epithelial cells.
- Antimicrobial peptides
-
A subset of host defence peptides with antibiotic activity. Peptides such as LL-37 (cathelicidin) and human β-defensins are rapidly acting, small-molecule effectors that are part of the innate immune response of the host.
- Nitric oxide
-
(NO). A ubiquitous signalling molecule found in both prokaryotic and eukaryotic systems. NO is toxic in the millimolar range, but in the picomolar and nanomolar range it can be used to form reactive oxidative and nitrosative species that interact with proteins, DNA and metabolic enzymes. As NO is labile, the optimal concentration to disperse biofilms is difficult to measure; however, NO microelectrodes are highly sensitive and may provide excellent spatial and temporal resolution in tissues or body fluids.
- Cystic fibrosis transmembrane conductance regulator
-
(CFTR). A transmembrane protein and ion transport channel that regulates epithelial fluid homeostasis central to airway mucociliary clearance and defence against inhaled pathogens.
- Biguanides
-
Class of organic compounds (C2H7N5) used as oral antihyperglycaemic drugs. Derivatives of this compound with bactericidal activity are commonly used as antiseptic and disinfecting agents such as chlorhexidine.
- Nanoparticles
-
Structures with a size range between 1–1000 nm. They can be classified as organic or inorganic and can exhibit antibacterial properties or can be used as drug delivery systems.
- Surfactants
-
Compounds that lower the surface tension between liquids and solids. Surfactants are used as cleaning detergents, and some biofilm bacteria produce their own surfactants in order to disperse from a surface.
- Topographic surface patterns
-
Patterns, including protruding squares, cone-shapes, wrinkle and ridge-like patterning or nanopores, that disrupt bacterial adhesion.
- Biofouling
-
The unwanted accumulation of microorganisms and macroorganisms on surfaces. Microbial biofilms are often considered 'biofouling', particularly in the context of manmade industrial surfaces.
- Super-hydrophobic surfaces
-
Surfaces that maintain air at the solid–liquid interface when hydrated. This leads to improved functionality through water repellency or reduced drag.
- Smart surfaces
-
Surfaces that elicit their effect only upon contact with certain physiological, physical or physiochemical cues to provide targeted application, thus increasing therapeutic precision and reducing the risk of cytotoxity.
Rights and permissions
About this article
Cite this article
Koo, H., Allan, R., Howlin, R. et al. Targeting microbial biofilms: current and prospective therapeutic strategies. Nat Rev Microbiol 15, 740–755 (2017). https://doi.org/10.1038/nrmicro.2017.99
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro.2017.99
This article is cited by
-
Tetrahedral framework nucleic acids for improving wound healing
Journal of Nanobiotechnology (2024)
-
Oligoribonuclease mediates high adaptability of P. aeruginosa through metabolic conversion
BMC Microbiology (2024)
-
How probiotics, prebiotics, synbiotics, and postbiotics prevent dental caries: an oral microbiota perspective
npj Biofilms and Microbiomes (2024)
-
Programmed microalgae-gel promotes chronic wound healing in diabetes
Nature Communications (2024)
-
Current Strategies for Combating Biofilm-Forming Pathogens in Clinical Healthcare-Associated Infections
Indian Journal of Microbiology (2024)