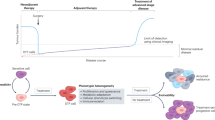
Key Points
-
Many bacterial infections persist in the host for long periods of time despite antibiotic treatment.
-
This persistence is an important medical concern, as it leads to the overuse of antibiotics and therefore contributes to antimicrobial resistance.
-
The role of antibiotic-tolerant persister cells in the recalcitrance and relapse of bacterial infections has gained recognition in recent years.
-
Persisters are often growth-arrested bacteria with a reduced metabolism that are able to restart growth after a stress.
-
The stresses that bacteria encounter during the infection of a host are triggers for the formation of persisters.
-
Toxin–antitoxin modules have an important role in the formation of growth-arrested persisters.
-
Understanding how growth-arrested persisters regrow is necessary to design better therapeutic strategies.
Abstract
Many bacteria can infect and persist inside their hosts for long periods of time. This can be due to immunosuppression of the host, immune evasion by the pathogen and/or ineffective killing by antibiotics. Bacteria can survive antibiotic treatment if they are resistant or tolerant to a drug. Persisters are a subpopulation of transiently antibiotic-tolerant bacterial cells that are often slow-growing or growth-arrested, and are able to resume growth after a lethal stress. The formation of persister cells establishes phenotypic heterogeneity within a bacterial population and has been hypothesized to be important for increasing the chances of successfully adapting to environmental change. The presence of persister cells can result in the recalcitrance and relapse of persistent bacterial infections, and it has been linked to an increase in the risk of the emergence of antibiotic resistance during treatment. If the mechanisms of the formation and regrowth of these antibiotic-tolerant cells were better understood, it could lead to the development of new approaches for the eradication of persistent bacterial infections. In this Review, we discuss recent developments in our understanding of bacterial persisters and their potential implications for the treatment of persistent infections.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Helaine, S. & Kugelberg, E. Bacterial persisters: formation, eradication, and experimental systems. Trends Microbiol. 22, 417–424 (2014).
Harms, A., Maisonneuve, E. & Gerdes, K. Mechanisms of bacterial persistence during stress and antibiotic exposure. Science 354, aaf4268 (2016).
Michiels, J. E., Van den Bergh, B., Verstraeten, N. & Michiels, J. Molecular mechanisms and clinical implications of bacterial persistence. Drug Resist. Updat. 29, 76–89 (2017).
Mechler, L. et al. A novel point mutation promotes growth phase-dependent daptomycin tolerance in Staphylococcus aureus. Antimicrob. Agents Chemother. 59, 5366–5376 (2015).
Van den Bergh, B. et al. Frequency of antibiotic application drives rapid evolutionary adaptation of Escherichia coli persistence. Nat. Microbiol. 1, 16020 (2016).
Cohen, N. R., Lobritz, M. A. & Collins, J. J. Microbial persistence and the road to drug resistance. Cell Host Microbe 13, 632–642 (2013).
Levin-Reisman, I. et al. Antibiotic tolerance facilitates the evolution of resistance. Science 355, 826–830 (2017).
Hobby, G. L., Meyer, K. & Chaffee, E. Observations on the mechanism of action of penicillin. Exp. Biol. Med. 50, 281–285 (1942).
Bigger, J. W. Treatment of staphylococcal infections with penicillin by intermittant sterilisation. Lancet 244, 497–500 (1944).
Balaban, N. Q., Merrin, J., Chait, R., Kowalik, L. & Leibler, S. Bacterial persistence as a phenotypic switch. Science 305, 1622–1625 (2004).
Adams, K. N. et al. Drug tolerance in replicating mycobacteria mediated by a macrophage-induced efflux mechanism. Cell 145, 39–53 (2011).
Wakamoto, Y. et al. Dynamic persistence of antibiotic-stressed mycobacteria. Science 339, 91–95 (2013).
Amato, S. et al. The role of metabolism in bacterial persistence. Front. Microbiol. 5, 70 (2014).
Helaine, S. et al. Internalization of Salmonella by macrophages induces formation of nonreplicating persisters. Science 343, 204–208 (2014).
Rhen, M., Eriksson, S., Clements, M., Bergström, S. & Normark, S. J. The basis of persistent bacterial infections. Trends Microbiol. 11, 80–86 (2003).
Grant, S. S. & Hung, D. T. Persistent bacterial infections, antibiotic tolerance, and the oxidative stress response. Virulence 4, 273–283 (2013).
Levine, M. M., Black, R. E. & Lanata, C. Precise estimation of the numbers of chronic carriers of Salmonella Typhi in Santiago, Chile, an endemic area. J. Infect. Dis. 146, 724–726 (1982).
Caygill, C. P. J., Hill, M. J., Braddick, M. & Sharp, J. C. M. Cancer mortality in chronic typhoid and paratyphoid carriers. Lancet 343, 83–84 (1994).
Bhan, M. K., Bahl, R. & Bhatnagar, S. Typhoid and paratyphoid fever. Lancet 366, 749–762 (2005).
Nomura, A., Stemmermann, G. N., Chyou, P.-H., Perez-Perez, G. I. & Blaser, M. J. Helicobacter pylori infection and the risk for duodenal and gastric ulceration. Ann. Intern. Med. 120, 977–981 (1994).
Wotherspoon, A. C., Ortiz-Hidalgo, C., Falzon, M. R. & Isaacson, P. G. Helicobacter pylori-associated gastritis and primary B-cell gastric lymphoma. Lancet 338, 1175–1176 (1991).
Eslick, G. D., Lim, L. L.-Y., Byles, J. E., Xia, H. H.-X. & Talley, N. J. Association of Helicobacter pylori infection with gastric carcinoma: a meta-analysis. Am. J. Gastroenterol. 94, 2373–2379 (1999).
Gomez, J. E. & McKinney, J. D. M. tuberculosis persistence, latency, and drug tolerance. Tuberculosis 84, 29–44 (2004).
Gotuzzo, E. et al. Association between specific plasmids and relapse in typhoid fever. J. Clin. Microbiol. 25, 1779–1781 (1987).
Foxman, B. Recurring urinary tract infection: incidence and risk factors. Am. J. Public Health 80, 331–333 (1990).
Österlund, A., Popa, R., Nikkilä, T., Scheynius, A. & Engstrand, L. Intracellular reservoir of Streptococcus pyogenes in vivo: a possible explanation for recurrent pharyngotonsillitis. Laryngoscope 107, 640–647 (1997).
Bryers, J. D. Medical biofilms. Biotechnol. Bioeng. 100, 1–18 (2008).
Lyczak, J. B., Cannon, C. L. & Pier, G. B. Lung infections associated with cystic fibrosis. Clin. Microbiol. Rev. 15, 194–222 (2002).
Jernberg, C., Löfmark, S., Edlund, C. & Jansson, J. K. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology 156, 3216–3223 (2010).
Norris, S. J. Antigenic variation with a twist — the Borrelia story. Mol. Microbiol. 60, 1319–1322 (2006).
Redpath, S., Ghazal, P. & Gascoigne, N. R. J. Hijacking and exploitation of IL-10 by intracellular pathogens. Trends Microbiol. 9, 86–92 (2001).
Scott, C. C., Botelho, R. J. & Grinstein, S. Phagosome maturation: a few bugs in the system. J. Membr. Biol. 193, 137–152 (2003).
Bayer-Santos, E. et al. The Salmonella effector SteD mediates MARCH8-dependent ubiquitination of MHC II molecules and inhibits T cell activation. Cell Host Microbe 20, 584–595 (2016).
Reddick, L. E. & Alto, N. M. Bacteria fighting back — how pathogens target and subvert the host innate immune system. Mol. Cell 54, 321–328 (2014).
Stewart, P. S. Mechanisms of antibiotic resistance in bacterial biofilms. Int. J. Med. Microbiol. 292, 107–113 (2002).
Monack, D. M., Mueller, A. & Falkow, S. Persistent bacterial infections: the interface of the pathogen and the host immune system. Nat. Rev. Microbiol. 2, 747–765 (2004).
Salama, N. R., Hartung, M. L. & Muller, A. Life in the human stomach: persistence strategies of the bacterial pathogen Helicobacter pylori. Nat. Rev. Microbiol. 11, 385–399 (2013).
Costerton, J. W., Stewart, P. S. & Greenberg, E. P. Bacterial biofilms: a common cause of persistent infections. Science 284, 1318–1322 (1999).
Jesaitis, A. J. et al. Compromised host defense on Pseudomonas aeruginosa biofilms: characterization of neutrophil and biofilm interactions. J. Immunol. 171, 4329–4339 (2003).
Domenech, M., Ramos-Sevillano, E., García, E., Moscoso, M. & Yuste, J. Biofilm formation avoids complement immunity and phagocytosis of Streptococcus pneumoniae. Infect. Immun. 81, 2606–2615 (2013).
Lawn, S. D., Butera, S. T. & Shinnick, T. M. Tuberculosis unleashed: the impact of human immunodeficiency virus infection on the host granulomatous response to Mycobacterium tuberculosis. Microbes Infect. 4, 635–646 (2002).
Kardas, P. Patient compliance with antibiotic treatment for respiratory tract infections. J. Antimicrob. Chemother. 49, 897–903 (2002).
Gonçalves-Pereira, J. & Póvoa, P. Antibiotics in critically ill patients: a systematic review of the pharmacokinetics of β-lactams. Crit. Care 15, R206 (2011).
Blair, J. M. A., Webber, M. A., Baylay, A. J., Ogbolu, D. O. & Piddock, L. J. V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 13, 42–51 (2015).
Brauner, A., Fridman, O., Gefen, O. & Balaban, N. Q. Distinguishing between resistance, tolerance and persistence to antibiotic treatment. Nat. Rev. Microbiol. 14, 320–330 (2016). This article explains the differences between antibiotic resistance, tolerance and persistence, and proposes a quantitative indicator of tolerance (the minimum duration for killing (MDK)) for use in the clinic.
Lewis, K. Persister cells. Annu. Rev. Microbiol. 64, 357–372 (2010).
Claudi, B. et al. Phenotypic variation of Salmonella in host tissues delays eradication by antimicrobial chemotherapy. Cell 158, 722–733 (2014).
Okoro, C. K. et al. High-resolution single nucleotide polymorphism analysis distinguishes recrudescence and reinfection in recurrent invasive nontyphoidal Salmonella typhimurium disease. Clin. Infect. Dis. 54, 955–963 (2012).
Russo, T. A., Stapleton, A., Wenderoth, S., Hooton, T. M. & Stamm, W. E. Chromosomal restriction fragment length polymorphism analysis of Escherichia coli strains causing recurrent urinary tract infections in young women. J. Infect. Dis. 172, 440–445 (1995).
Bingen, E. et al. DNA restriction fragment length polymorphism differentiates recurrence from relapse in treatment failures of Streptococcus pyogenes pharyngitis. J. Med. Microbiol. 37, 162–164 (1992).
Fridman, O., Goldberg, A., Ronin, I., Shoresh, N. & Balaban, N. Q. Optimization of lag time underlies antibiotic tolerance in evolved bacterial populations. Nature 513, 418–421 (2014). This evolutionary study uses the recently developed ScanLag technique to monitor bacterial regrowth after repeated antibiotic treatments and uncovers the importance of lag in the tolerance of bacterial populations.
Moyed, H. S. & Bertrand, K. P. hipA, a newly recognized gene of Escherichia coli K-12 that affects frequency of persistence after inhibition of murein synthesis. J. Bacteriol. 155, 768–775 (1983).
Mulcahy, L. R., Burns, J. L., Lory, S. & Lewis, K. Emergence of Pseudomonas aeruginosa strains producing high levels of persister cells in patients with cystic fibrosis. J. Bacteriol. 192, 6191–6199 (2010). This study uncovers a strong link between the formation of persisters and persistent bacterial infections.
Schumacher, M. A. et al. HipBA–promoter structures reveal the basis of heritable multidrug tolerance. Nature 524, 59–64 (2015). This study identifies how a mutation that was found in an E. coli UTI clinical isolate affects dimerization of the toxin HipA and, consequently, disrupts toxin–antitoxin–promoter interactions, which leads to an increase in toxicity and antibiotic tolerance.
LaFleur, M. D., Qi, Q. & Lewis, K. Patients with long-term oral carriage harbor high-persister mutants of Candida albicans. Antimicrob. Agents Chemother. 54, 39–44 (2010).
Mouton, J. M., Helaine, S., Holden, D. W. & Sampson, S. L. Elucidating population-wide mycobacterial replication dynamics at the single-cell level. Microbiology 162, 966–978 (2016).
Brooun, A., Liu, S. & Lewis, K. A. Dose–response study of antibiotic resistance in Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 44, 640–646 (2000).
Spoering, A. L. & Lewis, K. Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials. J. Bacteriol. 183, 6746–6751 (2001).
Keren, I., Kaldalu, N., Spoering, A., Wang, Y. & Lewis, K. Persister cells and tolerance to antimicrobials. FEMS Microbiol. Lett. 230, 13–18 (2004).
Maisonneuve, E., Castro-Camargo, M. & Gerdes, K. (p)ppGpp controls bacterial persistence by stochastic induction of toxin–antitoxin activity. Cell 154, 1140–1150 (2013). This study identifies a signalling pathway for the activation of toxin–antitoxin modules, leading to the formation of persisters.
Conlon, B. P. et al. Activated ClpP kills persisters and eradicates a chronic biofilm infection. Nature 503, 365–370 (2013). This study uses ADEP4 as a treatment to eradicate a chronic biofilm infection, which represents a great advance in the development of anti-persister therapies.
Burmølle, M. et al. Biofilms in chronic infections — a matter of opportunity — monospecies biofilms in multispecies infections. FEMS Immunol. Med. Microbiol. 59, 324–336 (2010).
Hunstad, D. A. & Justice, S. S. Intracellular lifestyles and immune evasion strategies of uropathogenic Escherichia coli. Annu. Rev. Microbiol. 64, 203–221 (2010).
Davies, J. C. Pseudomonas aeruginosa in cystic fibrosis: pathogenesis and persistence. Paediatr. Respir. Rev. 3, 128–134 (2002).
Oh, J. D., Karam, S. M. & Gordon, J. I. Intracellular Helicobacter pylori in gastric epithelial progenitors. Proc. Natl Acad. Sci. USA 102, 5186–5191 (2005).
Cammarota, G., Sanguinetti, M., Gallo, A. & Posteraro, B. Review article: biofilm formation by Helicobacter pylori as a target for eradication of resistant infection. Aliment. Pharmacol. Ther. 36, 222–230 (2012).
Prouty, A. M., Schwesinger, W. H. & Gunn, J. S. Biofilm formation and interaction with the surfaces of gallstones by Salmonella spp. Infect. Immun. 70, 2640–2649 (2002).
Amato, S. M., Orman, M. A. & Brynildsen, M. P. Metabolic control of persister formation in Escherichia coli. Mol. Cell 50, 475–487 (2013).
Dörr, T., Vulic´, M. & Lewis, K. Ciprofloxacin causes persister formation by inducing the TisB toxin in Escherichia coli. PLoS Biol. 8, e1000317 (2010).
Fasani, R. A. & Savageau, M. A. Molecular mechanisms of multiple toxin–antitoxin systems are coordinated to govern the persister phenotype. Proc. Natl Acad. Sci. USA 110, E2528–E2537 (2013).
Nguyen, D. et al. Active starvation responses mediate antibiotic tolerance in biofilms and nutrient-limited bacteria. Science 334, 982–986 (2011).
Ogura, T. & Hiraga, S. Mini-F plasmid genes that couple host cell division to plasmid proliferation. Proc. Natl Acad. Sci. USA 80, 4784–4788 (1983).
Gerdes, K., Rasmussen, P. B. & Molin, S. Unique type of plasmid maintenance function: postsegregational killing of plasmid-free cells. Proc. Natl Acad. Sci. USA 83, 3116–3120 (1986).
Pandey, D. P. & Gerdes, K. Toxin–antitoxin loci are highly abundant in free-living but lost from host-associated prokaryotes. Nucleic Acids Res. 33, 966–976 (2005).
Gerdes, K., Christensen, S. K. & Lobner-Olesen, A. Prokaryotic toxin–antitoxin stress response loci. Nat. Rev. Microbiol. 3, 371–382 (2005).
Yamaguchi, Y. & Inouye, M. Regulation of growth and death in Escherichia coli by toxin–antitoxin systems. Nat. Rev. Microbiol. 9, 779–790 (2011).
Rotem, E. et al. Regulation of phenotypic variability by a threshold-based mechanism underlies bacterial persistence. Proc. Natl Acad. Sci. USA 107, 12541–12546 (2010).
Cataudella, I., Trusina, A., Sneppen, K., Gerdes, K. & Mitarai, N. Conditional cooperativity in toxin–antitoxin regulation prevents random toxin activation and promotes fast translational recovery. Nucleic Acids Res. 40, 6424–6434 (2012).
Page, R. & Peti, W. Toxin–antitoxin systems in bacterial growth arrest and persistence. Nat. Chem. Biol. 12, 208–214 (2016). A comprehensive review of toxin–antitoxin systems in the context of persister formation.
Keren, I., Shah, D., Spoering, A., Kaldalu, N. & Lewis, K. Specialized persister cells and the mechanism of multidrug tolerance in Escherichia coli. J. Bacteriol. 186, 8172–8180 (2004).
Shah, D. et al. Persisters: a distinct physiological state of E. coli. BMC Microbiol. 6, 53 (2006).
Maisonneuve, E., Shakespeare, L. J., Jørgensen, M. G. & Gerdes, K. Bacterial persistence by RNA endonucleases. Proc. Natl Acad. Sci. USA 108, 13206–13211 (2011).
Verstraeten, N. et al. Obg and membrane depolarization are part of a microbial bet-hedging strategy that leads to antibiotic tolerance. Mol. Cell 59, 9–21 (2015). This study describes the regulation of the HokB–SokB toxin–antitoxin system by ObgE and (p)ppGpp in E. coli , and suggests an overlap in the regulation of both type I and type II toxin–antitoxin modules and central metabolism.
Maurizi, M. R. Proteases and protein degradation in Escherichia coli. Experientia 48, 178–201 (1992).
Kint, C., Verstraeten, N., Hofkens, J., Fauvart, M. & Michiels, J. Bacterial Obg proteins: GTPases at the nexus of protein and DNA synthesis. Crit. Rev. Microbiol. 40, 207–224 (2014).
Theodore, A., Lewis, K. & Vulic´, M. Tolerance of Escherichia coli to fluoroquinolone antibiotics depends on specific components of the SOS response pathway. Genetics 195, 1265–1276 (2013).
Shan, Y., Lazinski, D., Rowe, S., Camilli, A. & Lewis, K. Genetic basis of persister tolerance to aminoglycosides in Escherichia coli. mBio 6, e00078-15 (2015).
Mizusawa, S. & Gottesman, S. Protein degradation in Escherichia coli: the lon gene controls the stability of SulA protein. Proc. Natl Acad. Sci. USA 80, 358–362 (1983).
Germain, E., Castro-Roa, D., Zenkin, N. & Gerdes, K. Molecular mechanism of bacterial persistence by HipA. Mol. Cell 52, 248–254 (2013).
Pedersen, K. et al. The bacterial toxin RelE displays codon-specific cleavage of mRNAs in the ribosomal A site. Cell 112, 131–140 (2003).
Christensen, S. K. & Gerdes, K. RelE toxins from bacteria and Archaea cleave mRNAs on translating ribosomes, which are rescued by tmRNA. Mol. Microbiol. 48, 1389–1400 (2003).
Christensen-Dalsgaard, M., Jørgensen, M. G. & Gerdes, K. Three new RelE–homologous mRNA interferases of Escherichia coli differentially induced by environmental stresses. Mol. Microbiol. 75, 333–348 (2010).
Winther, K. S. & Gerdes, K. Enteric virulence associated protein VapC inhibits translation by cleavage of initiator tRNA. Proc. Natl Acad. Sci. USA 108, 7403–7407 (2011).
Winther, K., Tree, J. J., Tollervey, D. & Gerdes, K. VapCs of Mycobacterium tuberculosis cleave RNAs essential for translation. Nucleic Acids Res. 9860–9871 (2016).
Castro-Roa, D. et al. The Fic protein Doc uses an inverted substrate to phosphorylate and inactivate EF-Tu. Nat. Chem. Biol. 9, 811–817 (2013).
Bernard, P. et al. The F plasmid CcdB protein induces efficient ATP-dependent DNA cleavage by gyrase. J. Mol. Biol. 234, 534–541 (1993).
Jiang, Y., Pogliano, J., Helinski, D. R. & Konieczny, I. ParE toxin encoded by the broad-host-range plasmid RK2 is an inhibitor of Escherichia coli gyrase. Mol. Microbiol. 44, 971–979 (2002).
Harms, A. et al. Adenylylation of gyrase and Topo IV by FicT toxins disrupts bacterial DNA topology. Cell Rep. 12, 1497–1507 (2015).
Cheverton, A. M. et al. A Salmonella toxin promotes persister formation through acetylation of tRNA. Mol. Cell 63, 86–96 (2016).
Pedersen, K., Christensen, S. K. & Gerdes, K. Rapid induction and reversal of a bacteriostatic condition by controlled expression of toxins and antitoxins. Mol. Microbiol. 45, 501–510 (2002).
Harrison, J. J. et al. The chromosomal toxin gene yafQ is a determinant of multidrug tolerance for Escherichia coli growing in a biofilm. Antimicrob. Agents Chemother. 53, 2253–2258 (2009).
Lobato-Márquez, D., Moreno-Córdoba, I., Figueroa, V., Díaz-Orejas, R. & García-del Portillo, F. Distinct type I and type II toxin–antitoxin modules control Salmonella lifestyle inside eukaryotic cells. Sci. Rep. 5, 9374 (2015).
Kaspy, I. et al. HipA-mediated antibiotic persistence via phosphorylation of the glutamyl-tRNA-synthetase. Nat. Commun. 4, 3001 (2013).
Germain, E., Roghanian, M., Gerdes, K. & Maisonneuve, E. Stochastic induction of persister cells by HipA through (p)ppGpp-mediated activation of mRNA endonucleases. Proc. Natl Acad. Sci. USA 112, 5171–5176 (2015).
Conlon, B. P. et al. Persister formation in Staphylococcus aureus is associated with ATP depletion. Nat. Microbiol. 1, 16051 (2016).
Fu, Z., Tamber, S., Memmi, G., Donegan, N. P. & Cheung, A. L. Overexpression of MazFSa in Staphylococcus aureus induces bacteriostasis by selectively targeting mRNAs for cleavage. J. Bacteriol. 191, 2051–2059 (2009).
Donegan, N. P. & Cheung, A. L. Regulation of the mazEF toxin–antitoxin module in Staphylococcus aureus and its impact on sigB expression. J. Bacteriol. 191, 2795–2805 (2009).
Chowdhury, N., Kwan, B. W. & Wood, T. K. Persistence increases in the absence of the alarmone guanosine tetraphosphate by reducing cell growth. Sci. Rep. 6, 20519 (2016).
Baba, T. et al. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol. Syst. Biol. http://dx.doi.org/10.1038/msb4100050 (2006).
Hansen, S., Lewis, K. & Vulic´, M. Role of global regulators and nucleotide metabolism in antibiotic tolerance in Escherichia coli. Antimicrob. Agents Chemother. 52, 2718–2726 (2008).
Radzikowski, J. L., Schramke, H. & Heinemann, M. Bacterial persistence from a system-level perspective. Curr. Opin. Biotechnol. 46, 98–105 (2017).
Radzikowski, J. L. et al. Bacterial persistence is an active σS stress response to metabolic flux limitation. Mol. Syst. Biol. 12, 882 (2016).
Amato, S. M. & Brynildsen, M. P. Persister heterogeneity arising from a single metabolic stress. Curr. Biol. 25, 2090–2098 (2015).
Shan, Y. et al. ATP-dependent persister formation in Escherichia coli. mBio 8, e02267-16 (2017).
Pu, Y. et al. Enhanced efflux activity facilitates drug tolerance in dormant bacterial cells. Mol. Cell 62, 284–294 (2016).
Band, V. I. et al. Antibiotic failure mediated by a resistant subpopulation in Enterobacter cloacae. Nat. Microbiol. 1, 16053 (2016).
Pader, V. et al. Staphylococcus aureus inactivates daptomycin by releasing membrane phospholipids. Nat. Microbiol. 2, 16194 (2016).
Queck, S. Y. et al. RNAIII-independent target gene control by the agr quorum-sensing system: insight into the evolution of virulence regulation in Staphylococcus aureus. Mol. Cell 32, 150–158 (2008).
Painter, K. L., Krishna, A., Wigneshweraraj, S. & Edwards, A. M. What role does the quorum-sensing accessory gene regulator system play during Staphylococcus aureus bacteremia? Trends Microbiol. 22, 676–685 (2014).
Overgaard, M., Borch, J., Jørgensen, M. G. & Gerdes, K. Messenger RNA interferase RelE controls relBE transcription by conditional cooperativity. Mol. Microbiol. 69, 841–857 (2008).
Hall, A. M. J., Gollan, B. & Helaine, S. Toxin–antitoxin systems: reversible toxicity. Curr. Opin. Microbiol. 36, 102–110 (2017).
Christensen, S. K., Pedersen, K., Hansen, F. G. & Gerdes, K. Toxin–antitoxin loci as stress-response-elements: ChpAK/MazF and ChpBK cleave translated RNAs and are counteracted by tmRNA. J. Mol. Biol. 332, 809–819 (2003).
Allison, K. R., Brynildsen, M. P. & Collins, J. J. Metabolite-enabled eradication of bacterial persisters by aminoglycosides. Nature 473, 216–220 (2011). This was the first study to develop a method of persister eradication and test it in an animal model.
Pan, J., Bahar, A. A., Syed, H. & Ren, D. Reverting antibiotic tolerance of Pseudomonas aeruginosa PAO1 persister cells by (Z)-4-bromo-5-(bromomethylene)-3-methylfuran-2(5H)-one. PLoS ONE 7, e45778 (2012).
Harris, J. & Keane, J. How tumour necrosis factor blockers interfere with tuberculosis immunity. Clin. Exp. Immunol. 161, 1–9 (2010).
Belland, R. J. et al. Transcriptome analysis of chlamydial growth during IFNγ-mediated persistence and reactivation. Proc. Natl Acad. Sci. USA 100, 15971–15976 (2003).
Layton, J. C. & Foster, P. L. Error-prone DNA polymerase IV is controlled by the stress-response sigma factor, RpoS, in Escherichia coli. Mol. Microbiol. 50, 549–561 (2003).
Goodman, M. F. Error-prone repair DNA polymerases in prokaryotes and eukaryotes. Annu. Rev. Biochem. 71, 17–50 (2002).
Dörr, T., Lewis, K. & Vulic´, M. SOS response induces persistence to fluoroquinolones in Escherichia coli. PLOS Genet. 5, e1000760 (2009).
Beaber, J. W., Hochhut, B. & Waldor, M. K. SOS response promotes horizontal dissemination of antibiotic resistance genes. Nature 427, 72–74 (2004).
Völzing, K. G. & Brynildsen, M. P. Stationary-phase persisters to ofloxacin sustain DNA damage and require repair systems only during recovery. mBio 6, e00731-15 (2015).
Pearl Mizrahi, S., Gefen, O., Simon, I. & Balaban, N. Q. Persistence to anti-cancer treatments in the stationary to proliferating transition. Cell Cycle 15, 3442–3453 (2016).
Michiels, J. & Fauvart, M. (eds) Bacterial Persistence: Methods and Protocols (Springer New York, 2016).
Helaine, S. et al. Dynamics of intracellular bacterial replication at the single cell level. Proc. Natl Acad. Sci. USA 107, 3746–3751 (2010).
Manina, G., Dhar, N. & McKinney, J. D. Stress and host immunity amplify Mycobacterium tuberculosis phenotypic heterogeneity and induce nongrowing metabolically active forms. Cell Host Microbe 17, 32–46 (2015).
Wang, Z., Gerstein, M. & Snyder, M. RNA-Seq: a revolutionary tool for transcriptomics. Nat. Rev. Genet. 10, 57–63 (2009).
Levin-Reisman, I. et al. Automated imaging with ScanLag reveals previously undetectable bacterial growth phenotypes. Nat. Methods 7, 737–739 (2010).
Gurnev, P. A., Ortenberg, R., Dörr, T., Lewis, K. & Bezrukov, S. M. Persister-promoting bacterial toxin TisB produces anion-selective pores in planar lipid bilayers. FEBS Lett. 586, 2529–2534 (2012).
Hurdle, J. G., O'Neill, A. J., Chopra, I. & Lee, R. E. Targeting bacterial membrane function: an underexploited mechanism for treating persistent infections. Nat. Rev. Microbiol. 9, 62–75 (2011).
Chen, X., Zhang, M., Zhou, C., Kallenbach, N. R. & Ren, D. Control of bacterial persister cells by Trp/Arg-containing antimicrobial peptides. Appl. Environ. Microbiol. 77, 4878–4885 (2011).
Mukherjee, D., Zou, H., Liu, S., Beuerman, R. & Dick, T. Membrane-targeting AM-0016 kills mycobacterial persisters and shows low propensity for resistance development. Future Microbiol. 11, 643–650 (2016).
Kim, W. et al. NH125 kills methicillin-resistant Staphylococcus aureus persisters by lipid bilayer disruption. Future Med. Chem. 8, 257–269 (2016).
Schmidt, N. W. et al. Engineering persister-specific antibiotics with synergistic antimicrobial functions. ACS Nano 8, 8786–8793 (2014).
Lehar, S. M. et al. Novel antibody–antibiotic conjugate eliminates intracellular S. aureus. Nature 527, 323–328 (2015).
Yang, H., Yu, J. & Wei, H. Engineered bacteriophage lysins as novel anti-infectives. Front. Microbiol. 5, 542 (2014).
Briers, Y. et al. Art-175 is a highly efficient antibacterial against multidrug-resistant strains and persisters of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 58, 3774–3784 (2014).
De Soyza, A. et al. Lung transplantation for patients with cystic fibrosis and Burkholderia cepacia complex infection: a single-center experience. J. Heart Lung Transplant. 29, 1395–1404 (2010).
King, P. Haemophilus influenzae and the lung (Haemophilus and the lung). Clin. Transl Med. 1, 10 (2012).
Ficht, T. A. Intracellular survival of Brucella: defining the link with persistence. Vet. Microbiol. 92, 213–223 (2003).
Pawlowski, S. W., Warren, C. A. & Guerrant, R. Diagnosis and treatment of acute or persistent diarrhea. Gastroenterology 136, 1874–1886 (2009).
Cocanour, C. S. Best strategies in recurrent or persistent Clostridium difficile infection. Surg. Infect. (Larchmt) 12, 235–239 (2011).
Marzel, A. et al. Persistent infections by nontyphoidal Salmonella in humans: epidemiology and genetics. Clin. Infect. Dis. 62, 879–886 (2016).
Chong, Y. P. et al. Persistent Staphylococcus aureus bacteremia: a prospective analysis of risk factors, outcomes, and microbiologic and genotypic characteristics of isolates. Medicine (Baltimore) 92, 98–108 (2013).
Elwell, C., Mirrashidi, K. & Engel, J. Chlamydia cell biology and pathogenesis. Nat. Rev. Microbiol. 14, 385–400 (2016).
Radolf, J. D. et al. Treponema pallidum, the syphilis spirochete: making a living as a stealth pathogen. Nat. Rev. Microbiol. 14, 744–759 (2016).
Acknowledgements
The authors thank members of the Helaine laboratory for critical reading of the manuscript. R.A.F. is supported by a UK Medical Research Council (MRC) Centre for Molecular Bacteriology and Infection (CMBI) studentship (grant MR/J006874/1). S.H. and B.G. are supported by an MRC Career Development Award (grant MR/M009629/1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Nosocomial infections
-
Infections acquired when under medical care, also known as hospital-acquired infections (HAIs).
- Interleukin-10
-
(IL-10). A cytokine that suppresses the interferon (IFN)-mediated transcriptional response.
- Interferon-γ response
-
(IFNγ response). The transcriptional changes that take place in a cell due to the detection of the cytokine interferon-γ (IFNγ), some of these changes increase the antibacterial capabilities of phagocytes.
- Type III secretion system
-
(T3SS). A protein needle-like appendage that is used by bacteria to translocate effector proteins into eukaryotic cells to manipulate host cellular processes.
- MHC class II antigen presentation
-
The process of presenting antigen bound to MHC class II on the cell plasma membrane for recognition by cells of the adaptive immune system.
- Granulomas
-
Organized groupings of macrophages and other cell types around foreign material that the immune system is unable to eliminate.
- Bactericidal antibiotic
-
An antibiotic that kills bacteria, as opposed to a bacteriostatic antibiotic, which only inhibits growth.
- Salmonella-containing vacuole
-
(SCV). A membrane-bound compartment that resembles that of a modified late endosome, in which Salmonella spp. typically reside within host cells.
- Fluoroquinolones
-
A class of antibiotics that target DNA gyrase activity to induce the formation of lethal double-stranded breaks in bacterial DNA.
- Genovar
-
A classification used for differentiating between strains of the same serovar that differ substantially in their genetic content.
- Lag time
-
The time taken before resumption of the growth of growth-arrested bacteria.
- Diauxic shift
-
A shift in metabolism from one carbon source to another.
- Stochastic gene expression
-
The random (or noisy) fluctuations in the transcription of a particular gene.
- Stringent response
-
A global change in gene expression and protein regulation following amino acid starvation signalled by the alarmones guanosine tetraphosphate and guanosine pentaphosphate (collectively referred to as (p)ppGpp) in bacteria and plants, directing resources away from growth and towards amino acid synthesis to promote survival.
- Post-segregational killing
-
A mechanism of plasmid maintenance that is used by some low-copy-number plasmids through the action of toxin–antitoxin modules, whereby any progeny bacterial daughter cells produced after division that have not received a copy of the plasmid will be killed through unregulated toxin activity.
- SOS response
-
A global change in gene expression and protein regulation due to the degradation of the LexA repressor caused by the detection of DNA damage in a bacterial cell by the RecA recombinase.
- Alarmones
-
Intracellular signalling molecules that are produced in response to stress.
- Proton motive force
-
The movement of protons down an electrochemical gradient across a membrane to drive ATP synthesis and motility in bacteria.
- DNA gyrase
-
A topoisomerase enzyme that decreases the supercoiling of DNA during replication and transcription through cleaving, rotating and re-ligating the DNA double-strand.
- Trans-translation
-
A quality control mechanism in protein synthesis that uses transfer-messenger RNA (tmRNA) to rescue a ribosome that has stalled during translation.
- Isoniazid
-
An antibiotic that is commonly used for the treatment of tuberculosis. Isoniazid is a pro-drug that is converted into its active form by the catalase enzyme KatG inside bacterial cells, it is then able to inhibit the cytochrome P450 system, leading to the production of lethal free radicals.
- Antimicrobial peptides
-
(AMPs). Small peptides synthesized by plants and animals that have antimicrobial properties and often target the bacterial membrane.
- PhoPQ two-component system
-
Two-component systems comprise a sensor and a regulator that are able to detect environmental changes and mediate transcriptional changes in response. The PhoPQ system regulates the glycerophospholipid and protein content of the outer membrane in response to pH.
- Conditional cooperativity
-
The autoregulation of certain toxin–antitoxin modules, whereby the antitoxin and toxin are able to form a complex only at a certain stoichiometric ratio, which can then effectively repress transcription of the module.
- Transfer-messenger RNA
-
(tmRNA). A specialized form of RNA that mimics a tRNA with an mRNA-like coding element that is used during trans-translation to continue stalled protein synthesis and target the resultant aberrant protein for degradation.
- Deampylation
-
The removal of an adenylyl group.
Rights and permissions
About this article
Cite this article
Fisher, R., Gollan, B. & Helaine, S. Persistent bacterial infections and persister cells. Nat Rev Microbiol 15, 453–464 (2017). https://doi.org/10.1038/nrmicro.2017.42
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro.2017.42
This article is cited by
-
Bigger problems from smaller colonies: emergence of antibiotic-tolerant small colony variants of Mycobacterium avium complex in MAC-pulmonary disease patients
Annals of Clinical Microbiology and Antimicrobials (2024)
-
tRNA modification reprogramming contributes to artemisinin resistance in Plasmodium falciparum
Nature Microbiology (2024)
-
Counter-on-chip for bacterial cell quantification, growth, and live-dead estimations
Scientific Reports (2024)
-
TcrXY is an acid-sensing two-component transcriptional regulator of Mycobacterium tuberculosis required for persistent infection
Nature Communications (2024)
-
Modeling stress-induced responses: plasticity in continuous state space and gradual clonal evolution
Theory in Biosciences (2024)