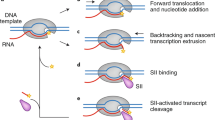
Key Points
-
RNA polymerase II (Pol II) elongation is a highly regulated process.
-
Regulation of transcription is often mediated at the level of promoter-proximal pausing of Pol II, in which Pol II is paused approximately 30–60 nucleotides downstream of the transcription start site (TSS) and awaits recruitment of kinase positive transcription elongation factor-b (P-TEFb).
-
P-TEFb is the main factor required to release paused Pol II from the promoter-proximal region, and can directly or indirectly be recruited by many factors, including bromodomain-containing protein 4 (BRD4) and the super elongation complex (SEC).
-
Elongation rates throughout the gene body are not uniform but vary between, and within genes, and can range from ∼1 to 6 kb per minute.
-
Transient slowdown of Pol II is observed up to 15 kb downstream of the TSS, at exons and near the poly(A) cleavage site.
-
Elongation rates can affect co-transcriptional RNA processes such as splicing and termination, as well as genome stability.
Abstract
Recent advances in sequencing techniques that measure nascent transcripts and that reveal the positioning of RNA polymerase II (Pol II) have shown that the pausing of Pol II in promoter-proximal regions and its release to initiate a phase of productive elongation are key steps in transcription regulation. Moreover, after the release of Pol II from the promoter-proximal region, elongation rates are highly dynamic throughout the transcription of a gene, and vary on a gene-by-gene basis. Interestingly, Pol II elongation rates affect co-transcriptional processes such as splicing, termination and genome stability. Increasing numbers of factors and regulatory mechanisms have been associated with the steps of transcription elongation by Pol II, revealing that elongation is a highly complex process. Elongation is thus now recognized as a key phase in the regulation of transcription by Pol II.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kwak, H. & Lis, J. T. Control of transcriptional elongation. Annu. Rev. Genet. 47, 483–508 (2013).
Adelman, K. & Lis, J. Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans. Nature Rev. Genet. 13, 720–731 (2012).
Kwak, H., Fuda, N. J., Core, L. J. & Lis, J. T. Precise maps of RNA polymerase reveal how promoters direct initiation and pausing. Science 339, 950–953 (2013). In this study, pausing was mapped at the genome-wide level with base-pair resolution, showing the dependency of strong promoter-proximal pausing on core promoter elements.
Li, J. & Gilmour, D. Distinct mechanisms of transcriptional pausing orchestrated by GAGA factor and M1BP, a novel transcription factor. EMBO J. 32, 1829–1841 (2013).
Weber, C. M., Ramachandran, S. & Henikoff, S. Nucleosomes are context-specific, H2A.Z-modulated barriers to RNA polymerase. Mol. Cell 53, 819–830 (2014).
Peterlin, B. & Price, D. Controlling the elongation phase of transcription with P-TEFb. Mol. Cell 23, 297–305 (2006).
Zhou, Q., Li, T. & Price, D. H. RNA polymerase II elongation control. Annu. Rev. Biochem. 81, 119–143 (2012).
Lis, J., Mason, P., Peng, J., Price, D. & Werner, J. P-TEFb kinase recruitment and function at heat shock loci. Genes Dev. 14, 792–803 (2000).
Jonkers, I., Kwak, H. & Lis, J. T. Genome-wide dynamics of Pol II elongation and its interplay with promoter proximal pausing, chromatin, and exons. eLife 3, e02407 (2014). This study measures elongation rates genome-wide and shows that the half-lives of paused Pol II complexes on 3,181 genes are uniformly long with an average of 7 minutes.
Danko, C. et al. Signaling pathways differentially affect RNA polymerase II initiation, pausing, and elongation rate in cells. Mol. Cell 50, 212–222 (2013).
Alexander, R., Innocente, S., Barrass, J. & Beggs, J. Splicing-dependent RNA polymerase pausing in yeast. Mol. Cell 40, 582–593 (2010).
Veloso, A. et al. Rate of elongation by RNA polymerase II is associated with specific gene features and epigenetic modifications. Genome Res. 24, 896–905 (2014).
Fuchs, G. et al. 4sUDRB-seq: measuring genomewide transcriptional elongation rates and initiation frequencies within cells. Genome Biol. 15, R69 (2014).
Saponaro, M. et al. RECQL5 controls transcript elongation and suppresses genome instability associated with transcription stress. Cell 157, 1037–1049 (2014). This manuscript documents that RECQL5 slows down transcript elongation and suppresses genome rearrangements at common fragile sites.
Dujardin, G. et al. How slow RNA polymerase II elongation favors alternative exon skipping. Mol. Cell 54, 683–690 (2014).
Schor, I., Fiszbein, A., Petrillo, E. & Kornblihtt, A. Intragenic epigenetic changes modulate NCAM alternative splicing in neuronal differentiation. EMBO J. 32, 2264–2274 (2013).
Mata, M. de la et al. A slow RNA polymerase II affects alternative splicing in vivo. Mol. Cell 12, 525–532 (2003).
Moehle, E. A., Braberg, H., Krogan, N. J. & Guthrie, C. Adventures in time and space: splicing efficiency and RNA polymerase II elongation rate. RNA Biol. 11, 313–319 (2014).
Plant, K., Dye, M., Lafaille, C. & Proudfoot, N. Strong polyadenylation and weak pausing combine to cause efficient termination of transcription in the human gamma-globin gene. Mol. Cell. Biol. 25, 3276–3285 (2005).
Gromak, N., West, S. & Proudfoot, N. Pause sites promote transcriptional termination of mammalian, RNA polymerase II. Mol. Cell. Biol. 26, 3986–3996 (2006).
Skourti-Stathaki, K., Kamieniarz-Gdula, K. & Proudfoot, N. J. R-loops induce repressive chromatin marks over mammalian gene terminators. Nature 516, 436–439 (2014).
Hazelbaker, D., Marquardt, S., Wlotzka, W. & Buratowski, S. Kinetic competition between RNA Polymerase II and Sen1-dependent transcription termination. Mol. Cell 49, 55–66 (2013).
Sainsbury, S., Bernecky, S. & Cramer, P. Structural basis of transcription initiation by RNA polymerase II. Nature Rev. Mol. Cell. Biol. 16, 129–143 (2015).
Porrua, O. & Libri, D. Transcription termination and the control of the transcriptome: why, where and how to stop. Nature Rev. Mol. Cell. Biol. 16, 190–202 (2015).
Venkatesh, S. S. & Workman, J. L. Histone exchange, chromatin structure and the regulation of transcription. Nature Rev. Mol. Cell. Biol. 16, 178–189 (2015).
Ehrensberger, A. H., Kelly, G. P. & Svejstrup, J. Q. Mechanistic interpretation of promoter-proximal peaks and RNAPII density maps. Cell 154, 713–715 (2013).
Venters, B. & Pugh, B. Genomic organization of human transcription initiation complexes. Nature 502, 53–58 (2013).
Core, L. J. et al. Defining the status of RNA polymerase at promoters. Cell Rep. 2, 1025–1035 (2012).
Rahl, P. et al. c-Myc regulates transcriptional pause release. Cell 141, 432–445 (2010).
Henriques, T. et al. Stable pausing by RNA polymerase II provides an opportunity to target and integrate regulatory signals. Mol. Cell 52, 517–528 (2013). This study reports that promoter-paused elongation complexes are highly stable, with half-lives of minutes in D. melanogaster.
Core, L. J., Waterfall, J. J. & Lis, J. T. Nascent RNA sequencing reveals widespread pausing and divergent initiation at human promoters. Science 322, 1845–1848 (2008).
Min, I. M. et al. Regulating RNA polymerase pausing and transcription elongation in embryonic stem cells. Genes Dev. 25, 742–754 (2011).
Nechaev, S. et al. Global analysis of short RNAs reveals widespread promoter-proximal stalling and arrest of Pol II in Drosophila. Science 327, 335–338 (2010).
Gilchrist, D. et al. Pausing of RNA polymerase II disrupts DNA-specified nucleosome organization to enable precise gene regulation. Cell 143, 540–551 (2010).
Lee, H., Kraus, K., Wolfner, M. & Lis, J. DNA sequence requirements for generating paused polymerase at the start of hsp70. Genes Dev. 6, 284–295 (1992).
Shopland, L., Hirayoshi, K., Fernandes, M. & Lis, J. HSF access to heat shock elements in vivo depends critically on promoter architecture defined by GAGA factor, TFIID, and RNA polymerase II binding sites. Genes Dev. 9, 2756–2769 (1995).
Kouzine, F. et al. Global regulation of promoter melting in naive lymphocytes. Cell 153, 988–999 (2013). This genome-wide analysis of resting lymphocytes identifies promoter melting as a third major rate-limiting step in transcription (following PIC formation and pause release).
Soutoglou, E. & Talianidis, I. Coordination of PIC assembly and chromatin remodeling during differentiation-induced gene activation. Science 295, 1901–1904 (2002).
Brannan, K. et al. mRNA decapping factors and the exonuclease Xrn2 function in widespread premature termination of RNA polymerase II transcription. Mol. Cell 46, 311–324 (2012).
Wagschal, A. et al. Microprocessor, Setx, Xrn2, and Rrp6 co-operate to induce premature termination of transcription by RNAPII. Cell 150, 1147–1157 (2012).
Cheng, B. et al. Functional association of Gdown1 with RNA polymerase II poised on human genes. Mol. Cell 45, 38–50 (2012).
Jishage, M. et al. Transcriptional regulation by Pol II(G.) Involving mediator and competitive interactions of Gdown1 and TFIIF with pol II. Mol. Cell 45, 51–63 (2012).
Davis, M., Guo, J., Price, D. & Luse, D. Functional interactions of the RNA polymerase II-interacting proteins Gdown1 and TFIIF. J. Biol. Chem. 289, 11143–11152 (2014).
Chen, F., Gao, X. & Shilatifard, A. Stably paused genes revealed through inhibition of transcription initiation by the TFIIH inhibitor triptolide. Genes Dev. 29, 39–47 (2015).
Buckley, M. S., Kwak, H., Zipfel, W. R. & Lis, J. T. Kinetics of promoter Pol II on Hsp70 reveal stable pausing and key insights into its regulation. Genes Dev. 28, 14–19 (2014).
Saunders, A., Core, L., Sutcliffe, C., Lis, J. & Ashe, H. Extensive polymerase pausing during Drosophila axis patterning enables high-level and pliable transcription. Genes Dev. 27, 1146–1158 (2013).
Rougvie, A. E. & Lis, J. T. The RNA polymerase II molecule at the 5′ end of the uninduced hsp70 gene of D. melanogaster is transcriptionally engaged. Cell 54, 795–804 (1988).
Guenther, M., Levine, S., Boyer, L., Jaenisch, R. & Young, R. A. Chromatin landmark and transcription initiation at most promoters in human cells. Cell 130, 77–88 (2007).
Zeitlinger, J. et al. RNA polymerase stalling at developmental control genes in the Drosophila melanogaster embryo. Nature Genet. 39, 1512–1516 (2007).
Lagha, M. et al. Paused pol II coordinates tissue morphogenesis in the Drosophila embryo. Cell 153, 976–987 (2013).
Kapanidis, A. N. et al. Initial transcription by RNA polymerase proceeds through a DNA-scrunching mechanism. Science 314, 1144–1147 (2006).
Pal, M., Ponticelli, A. & Luse, D. The role of the transcription bubble & TFIIB in promoter clearance by RNA polymerase II. Mol. Cell 19, 101–110 (2005).
Strobel, E. & Roberts, J. Regulation of promoter-proximal transcription elongation: enhanced DNA scrunching drives λQ antiterminator-dependent escape from a σ70-dependent pause. Nucleic Acids Res. 42, 5097–5108 (2014).
Hendrix, D. A., Hong, J.-W. W., Zeitlinger, J., Rokhsar, D. S. & Levine, M. S. Promoter elements associated with RNA pol II stalling in the Drosophila embryo. Proc. Natl Acad. Sci. USA 105, 7762–7767 (2008).
Core, L. et al. Analysis of nascent RNA identifies a unified architecture of initiation regions at mammalian promoters and enhancers. Nature Genet. 46, 1311–1320 (2014).
Li, J. et al. Kinetic competition between elongation rate and binding of NELF controls promoter-proximal pausing. Mol. Cell 50, 711–722 (2013).
Hargreaves, D., Horng, T. & Medzhitov, R. Control of inducible gene expression by signal-dependent transcriptional elongation. Cell 138, 129–145 (2009).
Heinz, S., Romanoski, C. E., Benner, C. & Glass, C. K. The selection and function of cell type-specific enhancers. Nature Rev. Mol. Cell. Biol. 16, 144–154 (2015).
Allen, B. L. & Taatjes, D. J. The Mediator complex: a central integrator of transcription. Nature Rev. Mol. Cell. Biol. 16, 155–166 (2015).
Takahashi, H. et al. Human mediator subunit MED26 functions as a docking site for transcription elongation factors. Cell 146, 92–104 (2011).
Ghavi-Helm, Y. et al. Enhancer loops appear stable during development and are associated with paused polymerase. Nature 512, 96–100 (2014).
Lee, C. et al. NELF and GAGA factor are linked to promoter-proximal pausing at many genes in Drosophila. Mol. Cell. Biol. 28, 3290–3300 (2008).
Farkas, G., Leibovitch, B. & Elgin, S. Chromatin organization and transcriptional control of gene expression in Drosophila. Gene 253, 117–136 (2000).
Chopra, V. S. et al. Transcriptional activation by GAGA factor is through its direct interaction with dmTAF3. Dev. Biol. 317, 660–670 (2008).
Blau, J. et al. Three functional classes of transcriptional activation domain. Mol. Cell. Biol. 16, 2044–2055 (1996).
Krumm, A., Hickey, L. B. & Groudine, M. Promoter-proximal pausing of RNA polymerase II defines a general rate-limiting step after transcription initiation. Genes Dev. 9, 559–572 (1995).
Bunch, H. et al. TRIM28 regulates RNA polymerase II promoter-proximal pausing and pause release. Nature Struct. Mol. Biol. 21, 876–883 (2014).
Jiang, L. et al. Polo-like kinase 1 inhibits the activity of positive transcription elongation factor of RNA Pol II b (P-TEFb). PloS ONE 8, e72289 (2013).
Smith, E., Lin, C. & Shilatifard, A. The super elongation complex (SEC) and MLL in development and disease. Genes Dev. 25, 661–672 (2011).
Itzen, F., Greifenberg, A., Bosken, C. & Geyer, M. Brd4 activates P-TEFb for RNA polymerase II CTD phosphorylation. Nucleic Acids Res. 42, 7577–7590 (2014).
Jang, M. et al. The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol. Cell 19, 523–534 (2005).
Zou, Z. et al. Brd4 maintains constitutively active NF-κB in cancer cells by binding to acetylated RelA. Oncogene 33, 2395–2404 (2014).
Huang, B., Yang, X.-D. D., Zhou, M.-M. M., Ozato, K. & Chen, L.-F. F. Brd4 coactivates transcriptional activation of NF-κB via specific binding to acetylated RelA. Mol. Cell. Biol. 29, 1375–1387 (2009).
Luo, Z. et al. The super elongation complex family of RNA polymerase II elongation factors: gene target specificity and transcriptional output. Mol. Cell. Biol. 32, 2608–2617 (2012).
Lin, C. et al. Dynamic transcriptional events in embryonic stem cells mediated by the super elongation complex (SEC). Genes Dev. 25, 1486–1498 (2011).
Smith, E. et al. The little elongation complex regulates small nuclear RNA transcription. Mol. Cell 44, 954–965 (2011).
Lin, C. et al. AFF4, a component of the ELL/P-TEFb elongation complex and a shared subunit of MLL chimeras, can link transcription elongation to leukemia. Mol. Cell 37, 429–437 (2010).
Luo, Z., Lin, C. & Shilatifard, A. The super elongation complex (SEC) family in transcriptional control. Nature Rev. Mol. Cell. Biol. 13, 543–547 (2012).
Gardini, A. et al. Integrator regulates transcriptional initiation and pause release following activation. Mol. Cell 56, 128–139 (2014).
Kim, J., Guermah, M. & Roeder, R. G. The human PAF1 complex acts in chromatin transcription elongation both independently and cooperatively with SII/TFIIS. Cell 140, 491–503 (2010).
Wier, A., Mayekar, M., Héroux, A., Arndt, K. & VanDemark, A. Structural basis for Spt5-mediated recruitment of the Paf1 complex to chromatin. Proc. Natl Acad. Sci. USA 110, 17290–17295 (2013).
He, N. et al. Human Polymerase-Associated Factor complex (PAFc) connects the Super Elongation Complex (SEC) to RNA polymerase II on chromatin. Proc. Natl Acad. Sci. USA 108, E636–645 (2011).
Flajollet, S. et al. The elongation complex components BRD4 and MLLT3/AF9 are transcriptional coactivators of nuclear retinoid receptors. PloS ONE 8, e64880 (2013).
Diamant, G. & Dikstein, R. Transcriptional control by NF-κB: elongation in focus. Biochim. Biophys. Acta 1829, 937–945 (2013).
Nowak, D. et al. RelA Ser276 phosphorylation is required for activation of a subset of NF-κB-dependent genes by recruiting cyclin-dependent kinase 9/cyclin T1 complexes. Mol. Cell. Biol. 28, 3623–3638 (2008).
Barboric, M., Nissen, R. M., Kanazawa, S., Jabrane-Ferrat, N. & Peterlin, B. M. NF-κB binds P-TEFb to stimulate transcriptional elongation by RNA polymerase II. Mol. Cell 8, 327–337 (2001).
Mertz, J. et al. Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc. Natl Acad. Sci. USA 108, 16669–16674 (2011).
Lovén, J. et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 153, 320–334 (2013). This study shows that exceptionally high levels of the co-activators Mediator and BRD4 are associated with super-enhancers that drive the expression of key oncogenes.
Delmore, J. et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146, 904–917 (2011).
Oeckinghaus, A., Hayden, M. & Ghosh, S. Crosstalk in NF-κB signaling pathways. Nature Immunol. 12, 695–708 (2011).
Fang, L. et al. ATM regulates NF-κB-dependent immediate-early genes via RelA Ser 276 phosphorylation coupled to CDK9 promoter recruitment. Nucleic Acids Res. 42, 8416–8432 (2014).
McNamara, R. P., McCann, J. L., Gudipaty, S. A. & D'Orso, I. Transcription factors mediate the enzymatic disassembly of promoter-bound 7SK snRNP to locally recruit P-TEFb for transcription elongation. Cell Rep. 5, 1256–1268 (2013).
Gilchrist, D. et al. Regulating the regulators: the pervasive effects of Pol II pausing on stimulus-responsive gene networks. Genes Dev. 26, 933–944 (2012).
Ji, X. et al. SR proteins collaborate with 7SK and promoter-associated nascent RNA to release paused polymerase. Cell 153, 855–868 (2013). This study implicates an RNA-binding protein that was traditionally thought to function in splicing in the regulated release of paused Pol II to productive elongation.
Barboric, M. et al. 7SK snRNP/P-TEFb couples transcription elongation with alternative splicing and is essential for vertebrate development. Proc. Natl Acad. Sci. USA 106, 7798–7803 (2009).
Hnisz, D. et al. Super-enhancers in the control of cell identity and disease. Cell 155, 934–947 (2013).
Whyte, W. et al. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Cell 153, 307–319 (2013).
Brown, J. D. et al. NF-κB directs dynamic super enhancer formation in inflammation and atherogenesis. Mol. Cell 56, 219–231 (2014).
Lai, F. et al. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature 494, 497–501 (2013).
Yamaguchi, Y., Inukai, N., Narita, T., Wada, T. & Handa, H. Evidence that negative elongation factor represses transcription elongation through binding to a DRB sensitivity-inducing factor/RNA polymerase, II complex and RNA. Mol. Cell. Biol. 22, 2918–2927 (2002).
Schaukowitch, K. et al. Enhancer RNA facilitates NELF release from immediate early genes. Mol. Cell 56, 29–42 (2014).
Schaaf, C. A. et al. Genome-wide control of RNA polymerase II activity by cohesin. PLoS Genet. 9, e1003382 (2013).
Thummel, C. S., Burtis, K. C. & Hogness, D. S. Spatial and temporal patterns of E74 transcription during Drosophila development. Cell 61, 101–111 (1990).
Heidemann, M., Hintermair, C., Voß, K. & Eick, D. Dynamic phosphorylation patterns of RNA polymerase II CTD during transcription. Biochim. Biophys. Acta 1829, 55–62 (2013).
Glover-Cutter, K., Kim, S., Espinosa, J. & Bentley, D. RNA polymerase II pauses and associates with pre-mRNA processing factors at both ends of genes. Nature Struct. Mol. Biol. 15, 71–78 (2007).
Martin, R., Rino, J., Carvalho, C., Kirchhausen, T. & Carmo-Fonseca, M. Live-cell visualization of pre-mRNA splicing with single-molecule sensitivity. Cell Rep. 4, 1144–1155 (2013).
Zentner, G. & Henikoff, S. Regulation of nucleosome dynamics by histone modifications. Nature Struct. Mol. Biol. 20, 259–266 (2013).
Bintu, L. et al. Nucleosomal elements that control the topography of the barrier to transcription. Cell 151, 738–749 (2012). In this study, optical tweezers were used to measure the movement of individual transcribing Pol II complexes through nucleosomes in real-time and thereby describes the energetic barriers in nucleosomes that could contribute to pausing.
Ardehali, M. B. et al. Spt6 enhances the elongation rate of RNA polymerase II in vivo. EMBO J. 28, 1067–1077 (2009).
Wu, L., Li, L., Zhou, B., Qin, Z. & Dou, Y. H2B ubiquitylation promotes RNA pol II processivity via PAF1 and pTEFb. Mol. Cell 54, 920–931 (2014).
Jung, I. et al. H2B monoubiquitylation is a 5′-enriched active transcription mark and correlates with exon-intron structure in human cells. Genome Res. 22, 1026–1035 (2012).
Shilatifard, A., Conaway, R. C. & Conaway, J. W. The RNA polymerase II elongation complex. Annu. Rev. Biochem. 72, 693–715 (2003).
Amit, M. et al. Differential GC content between exons and introns establishes distinct strategies of splice-site recognition. Cell Rep. 1, 543–556 (2012).
Tilgner, H. et al. Nucleosome positioning as a determinant of exon recognition. Nature Struct. Mol. Biol. 16, 996–1001 (2009).
Schwartz, S., Meshorer, E. & Ast, G. Chromatin organization marks exon-intron structure. Nature Struct. Mol. Biol. 16, 990–995 (2009).
Close, P. et al. DBIRD complex integrates alternative mRNA splicing with RNA polymerase II transcript elongation. Nature 484, 386–389 (2012).
Huff, J., Plocik, A., Guthrie, C. & Yamamoto, K. Reciprocal intronic and exonic histone modification regions in humans. Nature Struct. Mol. Biol. 17, 1495–1499 (2010).
Saint-André, V., Batsché, E., Rachez, C. & Muchardt, C. Histone H3 lysine 9 trimethylation and HP1γ favor inclusion of alternative exons. Nature Struct. Mol. Biol. 18, 337–344 (2011).
Schor, I. E., Rascovan, N., Pelisch, F., Alló, M. & Kornblihtt, A. R. Neuronal cell depolarization induces intragenic chromatin modifications affecting NCAM alternative splicing. Proc. Natl Acad. Sci. USA 106, 4325–4330 (2009).
Ip, J. et al. Global impact of RNA polymerase II elongation inhibition on alternative splicing regulation. Genome Res. 21, 390–401 (2011).
Hein, P. P. et al. RNA polymerase pausing and nascent-RNA structure formation are linked through clamp-domain movement. Nature Struct. Mol. Biol. 21, 794–802 (2014).
Skourti-Stathaki, K., Proudfoot, N. & Gromak, N. Human senataxin resolves RNA/DNA hybrids formed at transcriptional pause sites to promote Xrn2- dependent termination. Mol. Cell 42, 794–805 (2011).
Ozer, A., Pagano, J. M. & Lis, J. T. New technologies provide quantum changes in the scale, speed, and success of SELEX methods and aptamer characterization. Mol. Ther. Nucleic Acids 3, e183 (2014).
Lee, T. I. & Young, R. A. Transcriptional regulation and its misregulation in disease. Cell 152, 1237–1251 (2013).
Zuber, J. et al. RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 478, 524–528 (2011).
Dawson, M. et al. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 478, 529–533 (2011).
Singh, J. & Padgett, R. Rates of in situ transcription and splicing in large human genes. Nature Struct. Mol. Biol. 16, 1128–1133 (2009).
Churchman, L. & Weissman, J. Nascent transcript sequencing visualizes transcription at nucleotide resolution. Nature 469, 368–373 (2011).
Yao, J., Munson, K., Webb, W. & Lis, J. Dynamics of heat shock factor association with native gene loci in living cells. Nature 442, 1050–1053 (2006).
Boireau, S. et al. The transcriptional cycle of HIV-1 in real-time and live cells. J. Cell Biol. 179, 291–304 (2007).
Brody, Y. et al. The in vivo kinetics of RNA polymerase II elongation during co-transcriptional splicing. PLoS Biol. 9, e1000573 (2011).
Darzacq, X. et al. In vivo dynamics of RNA polymerase II transcription. Nature Struct. Mol. Biol. 14, 796–806 (2007).
Tennyson, C. N., Klamut, H. J. & Worton, R. G. The human dystrophin gene requires 16 hours to be transcribed and is cotranscriptionally spliced. Nature Genet. 9, 184–190 (1995).
Mason, P. & Struhl, K. Distinction & relationship between elongation rate & processivity of RNA polymerase II in vivo. Mol. Cell 17, 831–840 (2005).
Ameur, A. et al. Total RNA sequencing reveals nascent transcription and widespread co-transcriptional splicing in the human brain. Nature Struct. Mol. Biol. 18, 1435–1440 (2011).
Acknowledgements
The authors thank C. Danko, F. Duarte and D. Mahat for their critical evaluation of the manuscript. J.T.L. was supported by NIGMS (National Institute of General Medical Sciences) from the US National Institutes of Health under award GM25232. I.J. was supported by a European Research Council Advanced Grant (ERCadv-671274). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health or the European Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- DRB
-
A small drug that inhibits P-TEFb kinase activity. It is used to characterize pausing and elongation complexes, and to measure elongation rates genome-wide.
- Carboxy-terminal domain (CTD) of Pol II
-
The CTD of Pol II, which is positioned at the end of the largest Pol II subunit, is an unstructured, yet evolutionarily conserved, domain that comprises many tandem copies of the consensus heptapeptide YSPTSPS. Phosphorylation of these repeats is crucial for the regulation of Pol II function.
- +1 nucleosome
-
The first well-positioned nucleosome downstream of the transcription start site, which can form a barrier for elongating Pol II and might increase Pol II promoter-proximal pausing. The position of the +1 nucleosome depends on transcription, nucleosome remodelling, and DNA sequences.
- Pre-initiation complex
-
(PIC). A complex consisting of general transcription factors and Pol II that binds at the transcription start site, before DNA melting and transcription initiation.
- Open promoters
-
Promoters that are nucleosome-free and easily accessible to transcription factors and Pol II. These promoters are primed for, or undergo, active transcription.
- DNA melting
-
The process of unwinding and 'opening' double-stranded DNA at the transcription start site by general transcription factors to form a transcription bubble, which allows initiation of Pol II activity.
- General transcription factors
-
(GTFs). Factors that bind the core promoter region, facilitate DNA melting and transcription bubble formation, and position Pol II to initiate transcription and escape the promoter region.
- Enhancers
-
Regulatory regions that bind sequence-specific TFs and have potential transcription start sites and can interact with gene promoters three dimensionally to regulate gene expression.
- Mediator
-
A multisubunit co-activator complex that can interact with TFs, GTFs and Pol II and is essential for transcription. Mediator has been shown to mediate interaction between enhancers and gene promoters, for example at super-enhancers.
- Enhancer RNAs
-
(eRNAs). RNAs that derive from the transcription of enhancers. Some of these enhancer-derived RNAs contribute to enhancer function.
- Histone chaperones
-
Proteins that facilitate the movement of Pol II through chromatin by loosening the nucleosome–DNA interactions and then restoring these in the wake of Pol II.
- R-loops
-
An RNA–DNA hybrid structure formed during the transcription of a sequence with high GC-content that has the potential to pause Pol II. R-loops are associated with transcription termination and genome instability.
- Exon skipping
-
A form of alternative splicing, in which an exon is 'skipped' and removed as part of the flanking introns during transcription.
- MLL–ELL fusions
-
A fusion formed between the MLL gene (which encodes mixed-lineage leukaemia) and the ELL gene (which encodes eleven-nineteen Lys-rich leukaemia) that greatly increases the leukaemogenic potential of a cell.
Rights and permissions
About this article
Cite this article
Jonkers, I., Lis, J. Getting up to speed with transcription elongation by RNA polymerase II. Nat Rev Mol Cell Biol 16, 167–177 (2015). https://doi.org/10.1038/nrm3953
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3953
This article is cited by
-
The dynamics and functional mechanisms of H2B mono-ubiquitination
Crop Health (2024)
-
Metabolic response to a heterologous poly-3-hydroxybutyrate (PHB) pathway in Phaeodactylum tricornutum
Applied Microbiology and Biotechnology (2024)
-
Super-enhancers complexes zoom in transcription in cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
PRO-IP-seq tracks molecular modifications of engaged Pol II complexes at nucleotide resolution
Nature Communications (2023)
-
RNA polymerase II associates with active genes during DNA replication
Nature (2023)