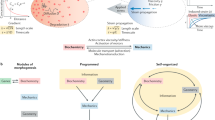
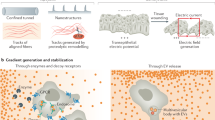
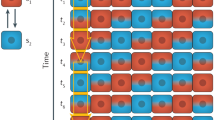
Abstract
Nearly 60 years ago, Alan Turing showed theoretically how two chemical species, termed morphogens, diffusing and reacting with each other can generate spatial patterns. Diffusion plays a crucial part in transporting chemical signals through space to establish the length scale of the pattern. When coupled to chemical reactions, mechanical processes — forces and flows generated by motor proteins — can also define length scales and provide a mechanochemical basis for morphogenesis. forces and flows generated by motor proteins — can also define length scales and provide a mechanochemical basis for morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Turing, A. M. The chemical basis of morphogenesis. Proc. R. Soc. Lond. B Biol. Sci. 237, 37–72 (1952).
Gierer, A. & Meinhardt, H. A theory of biological pattern formation. Kybernetik 12, 30–39 (1972).
Wolpert, L. Positional information and the spatial pattern of cellular differentiation. J. Theor. Biol. 25, 1–47 (1969).
Frohnhöfer, H. N. C. & Nüsslein-Volhard, C. Organization of the anterior pattern in the Drosophila embryo by the maternal gene bicoid. Nature 324, 120–125 (1986).
Kondo, S. & Miura, T. Reaction-diffusion model as a framework for understanding biological pattern formation. Science 329, 1616–1620 (2010).
Huxley, A. F. & Niedergerke, R. Structural changes in muscle during contraction. Interference microscopy of living cells. Nature 173, 971–976 (1954).
Huxley, H. E. & Hanson, J. Changes in the cross-striations of muscle during contraction and stretch and their structural interpretion. Nature 173, 973–976 (1954).
Gibbons, I. R. & Rowe, A. J. Dynein: a protein with adenosine triphosphatase activity from cilia. Science 149, 424–426 (1965).
Brady, S. T. A novel brain ATPase with properties expected for the fast axonal transport motor. Nature 317, 73–75 (1985).
Vale, R. D., Reese, T. S. & Sheetz, M. P. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 42, 39–50 (1985).
Crick, F. Diffusion in embryogenesis. Nature 225, 420–422 (1970).
Howard, J. Mechanics of Motor Proteins and the Cytoskeleton (Sinauer Associates, Sunderland, Massachusetts, 2001).
Luby-Phelps, K. Cytoarchitecture and physical properties of cytoplasm: volume, viscosity, diffusion, intracellular surface area. Int. Rev. Cytol. 192, 189–221 (2000).
Helenius, J., Brouhard, G., Kalaidzidis, Y., Diez, S. & Howard, J. The depolymerizing kinesin MCAK uses lattice diffusion to rapidly target microtubule ends. Nature 441, 115–119 (2006).
Gutzeit, H. O. & Koppa, R. Time-lapse film analysis of cytoplasmic streaming during late oogenesis of Drosophila. J. Embryol. Exp. Morphol. 67, 101–111 (1982).
Goldstein, R. E., Tuval, I. & van de Meent, J.-W. Microfluidics of cytoplasmic streaming and its implications for intracellular transport. Proc. Natl Acad. Sci. USA 105, 3663–3667 (2008).
Short, M. B. et al. Flows driven by flagella of multicellular organisms enhance long-range molecular transport. Proc. Natl Acad. Sci. USA 103, 8315–8319 (2006).
Baas, P. W., Deitch, J. S., Black, M. M. & Banker, G. A. Polarity orientation of microtubules in hippocampal neurons: uniformity in the axon and nonuniformity in the dendrite. Proc. Natl Acad. Sci. USA 85, 8335–8339 (1988).
Snider, J. et al. Intracellular actin-based transport: how far you go depends on how often you switch. Proc. Natl Acad. Sci. USA 101, 13204–13209 (2004).
Zimyanin, V. L. et al. In vivo imaging of oskar mRNA transport reveals the mechanism of posterior localization. Cell 134, 843–853 (2008).
Wilhelm, C. Out-of-equilibrium microrheology inside living cells. Phys. Rev. Lett. 101, 028101 (2008).
Engler, A. J., Sen, S., Sweeney, H. L. & Discher, D. E. Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 (2006).
Mayer, M., Depken, M., Bois, J. S., Jülicher, F. & Grill, S. W. Anisotropies in cortical tension reveal the physical basis of polarizing cortical flows. Nature 467, 617–621 (2010).
Rauzi, M., Lenne, P.-F. & Lecuit, T. Planar polarized actomyosin contractile flows control epithelial junction remodelling. Nature 468, 1110–1114 (2010).
Zerzour, R., Kroeger, J. & Geitmann, A. Polar growth in pollen tubes is associated with spatially confined dynamic changes in cell mechanical properties. Dev. Biol. 334, 437–446 (2009).
Howard, J. Mechanical signaling in networks of motor and cytoskeletal proteins. Annu. Rev. Biophys. 38, 217–234 (2009).
Vogel, V. & Sheetz, M. Local force and geometry sensing regulate cell functions. Nature Rev. Mol. Cell Biol. 7, 265–275 (2006).
Brown, G. C. & Kholodenko, B. N. Spatial gradients of cellular phospho-proteins. FEBS Lett. 457, 452–454 (1999).
Wartlick, O., Kicheva, A. & González-Gaitán, M. Morphogen gradient formation. Cold Spring Harb. Perspect. Biol. 1, a001255 (2009).
Kalab, P., Weis, K. & Heald, R. Visualization of a Ran-GTP gradient in interphase and mitotic Xenopus egg extracts. Science 295, 2452–2456 (2002).
Caudron, M., Bunt, G., Bastiaens, P. & Karsenti, E. Spatial coordination of spindle assembly by chromosome-mediated signaling gradients. Science 309, 1373–1376 (2005).
Kicheva, A. et al. Kinetics of morphogen gradient formation. Science 315, 521–525 (2007).
Belenkaya, T. Y. et al. Drosophila Dpp morphogen movement is independent of dynamin-mediated endocytosis but regulated by the glypican members of heparan sulfate proteoglycans. Cell 119, 231–244 (2004).
Yu, S. R. et al. Fgf8 morphogen gradient forms by a source-sink mechanism with freely diffusing molecules. Nature 461, 533–536 (2009).
Driever, W. & Nüsslein-Volhard, C. A gradient of bicoid protein in Drosophila embryos. Cell 54, 83–93 (1988).
Gregor, T., Bialek, W., de Ruyter van Steveninck, R. R., Tank, D. W. & Wieschaus, E. F. Diffusion and scaling during early embryonic pattern formation. Proc. Natl Acad. Sci. USA 102, 18403–18407 (2005).
Gregor, T., Wieschaus, E. F., McGregor, A. P., Bialek, W. & Tank, D. W. Stability and nuclear dynamics of the bicoid morphogen gradient. Cell 130, 141–152 (2007).
Spirov, A. et al. Formation of the bicoid morphogen gradient: an mRNA gradient dictates the protein gradient. Development 136, 605–614 (2009).
Abu-Arish, A., Porcher, A., Czerwonka, A., Dostatni, N. & Fradin, C. High mobility of Bicoid captured by fluorescence correlation spectroscopy: implication for the rapid establishment of its gradient. Biophys. J. 99, L33–L35 (2010).
Meinhardt, H. The Algorithmic Beauty of Sea Shells (Springer, Berlin; London, 2009).
Nakamasu, A., Takahashi, G., Kanbe, A. & Kondo, S. Interactions between zebrafish pigment cells responsible for the generation of Turing patterns. Proc. Natl Acad. Sci. USA 106, 8429–8434 (2009).
Naoz, M., Manor, U., Sakaguchi, H., Kachar, B. & Gov, N. S. Protein localization by actin treadmilling and molecular motors regulates stereocilia shape and treadmilling rate. Biophys. J. 95, 5706–5718 (2008).
Manor, U. et al. Regulation of stereocilia length by myosin XVa and whirlin depends on the actin-regulatory protein Eps8. Curr. Biol. 21, 167–172 (2011).
Varga, V. et al. Yeast Kinesin-8 depolymerizes microtubules in a length-dependent manner. Nature Cell Biol. 8, 957–962 (2006).
Varga, V., Leduc, C., Bormuth, V., Diez, S. & Howard, J. Kinesin-8 motors act cooperatively to mediate length-dependent microtubule depolymerization. Cell 138, 1174–1183 (2009).
Howard, J. & Hyman, A. A. Microtubule polymerases and depolymerases. Curr. Opin. Cell Biol. 19, 31–35 (2007).
Mayr, M. et al. The human kinesin Kif18A is a motile microtubule depolymerase essential for chromosome congression. Curr. Biol. 17, 488–498 (2007).
Stumpff, J., Vondassow, G., Wagenbach, M., Asbury, C. & Wordeman, L. The Kinesin-8 motor Kif18A suppresses kinetochore movements to control mitotic chromosome alignment. Dev. Cell 14, 252–262 (2008).
Wargacki, M. M., Tay, J. C., Muller, E. G., Asbury, C. L. & Davis, T. N. Kip3, the yeast Kinesin-8, is required for clustering of kinetochores at metaphase. Cell Cycle 9, 2581–2588 (2010).
Salbreux, G., Prost, J. & Joanny, J.-F. Hydrodynamics of cellular cortical flows and the formation of contractile rings. Phys. Rev. Lett. 103, 058102 (2009).
Bois, J., Jülicher, F. & Grill, S. Pattern formation in active fluids. Phys. Rev. Lett. 106, 028103 (2011).
Munro, E., Nance, J. & Priess, J. R. Cortical flows powered by asymmetrical contraction transport PAR proteins to establish and maintain anterior-posterior polarity in the early C. elegans embryo. Dev. Cell 7, 413–424 (2004).
David, D. J. V., Tishkina, A. & Harris, T. J. C. The PAR complex regulates pulsed actomyosin contractions during amnioserosa apical constriction in Drosophila. Development 137, 1645–1655 (2010).
Riedel-Kruse, I. H., Hilfinger, A., Howard, J. & Jülicher, F. How molecular motors shape the flagellar beat. HFSP J. 1, 192 (2007).
Aigouy, B. et al. Cell flow reorients the axis of planar polarity in the wing epithelium of Drosophila. Cell 142, 773–786 (2010).
Hamant, O. et al. Developmental patterning by mechanical signals in Arabidopsis. Science 322, 1650–1655 (2008).
Schneider, M. E., Dosé, A. C., Salles, F. T., Chang, W. et al. A new compartment at stereocilia tips defined by spatial and temporal patterns of myosin IIIa expression. J. Neurosci. 26, 10243–10252 (2006).
Acknowledgements
We thank N. Goehring, M. Mayer and F. Jülicher for discussions, and I. Tolic-Norrelykke for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Glossary
- Active diffusion
-
Random motion caused by randomly directed active forces, such as those generated by motor proteins.
- Advection
-
Directed transport driven by motor proteins or bulk fluid flow.
- Diffusion
-
The randomly directed motion of a molecule or particle that causes both mixing and the flux of particles from regions of high concentration to low concentration. Diffusion can be caused by thermal forces — that is, collisions with molecules in solution — or by randomly directed active forces, such as those generated by motor proteins that randomly change their direction.
- Diffusion coefficient
-
The constant of proportionality between the flux and the concentration gradient for a diffusing particle. Diffusion can be thermal or active.
- Friction coefficient
-
The constant or proportionality between a stress gradient and velocity.
- Length constant
-
The distance over which a quantity such as concentration decreases e-fold.
- Morphogens
-
Substances, such as proteins or small molecules, that are non-uniformly distributed in space and can influence cell growth or differentiation.
- Patterning
-
The establishment of features that are much larger than those of the individual molecular components, and which are stereotyped from one cell to another or one organism to another.
- Reaction–diffusion mechanism
-
A patterning process in which a diffusing morphogen undergoing chemical reactions (such as degradation or synthesis) forms a well-defined spatial distribution.
- Stress
-
Force per unit area.
- Viscoelastic material
-
A material that is both elastic (it can be stretched but returns to its original shape) and viscous (it deforms at a finite speed determined by the viscosity and the applied stress).
- Viscosity
-
The constant of proportionality between rates of stress and strain (the relative deformation of a solid body due to a stress).
Rights and permissions
About this article
Cite this article
Howard, J., Grill, S. & Bois, J. Turing's next steps: the mechanochemical basis of morphogenesis. Nat Rev Mol Cell Biol 12, 392–398 (2011). https://doi.org/10.1038/nrm3120
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3120
This article is cited by
-
Talking with force at cell–cell adhesions
Nature Cell Biology (2024)
-
The reaction-diffusion basis of animated patterns in eukaryotic flagella
Nature Communications (2023)
-
Morphogenesis in space offers challenges and opportunities for soft matter and biophysics
Communications Physics (2023)
-
Morphogenesis beyond in vivo
Nature Reviews Physics (2023)
-
A deep learning framework for quantitative analysis of actin microridges
npj Systems Biology and Applications (2023)