Key Points
-
Canonical histones H2A, H2B, H3 and H4 wrap DNA to form nucleosome particles that compact the genome. Histone variants have diverse additional roles in chromosome metabolism and can differ from canonical histones in stability, DNA wrapping and specialized domains.
-
The 'universal' variants, centromeric histone variant H3 (CenH3), H3.3, H2A.Z and H2A.X, appeared before the divergence of modern eukaryotes and function in common eukaryotic cellular processes such as histone replacement, chromosome segregation, DNA repair, and transcriptional regulation. Specialized histones have arisen in some lineages to perform additional tasks.
-
Centromeric nucleosomes contain CenH3 and form the essential foundation of the kinetochore. The subunit composition of the CenH3 histone core has been the subject of lively debate and it has been found to wrap DNA in a right-handed direction, opposite to that of ordinary nucleosomes.
-
H2A.Z has a conserved role in transcription initiation that might be descended from a hypothetical ancient mode of gene regulation by histone variants found in modern trypanosomes. Dynamic cellular processes might dramatically alter the stability of H2A.Z nucleosomes and thereby facilitate transcription initiation.
-
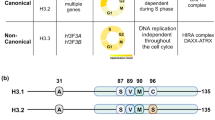
Chromatin must be remodelled in processes such as DNA repair and sex chromosome silencing. Variants H2A.X and H3.3 have prominent roles in remodelling, with specialized sperm histones and protamines mediating sperm packaging and decondensation.
-
Lineage-specific H2A variants have diverse carboxy-terminal tails that can wrap more DNA with additional oligopeptide motifs, or less DNA with a shorter docking domain. They sometimes have non-histone domains, which in macroH2As inhibit polyADP-ribosylation and contribute to conditional gene silencing.
Abstract
Histones wrap DNA to form nucleosome particles that compact eukaryotic genomes. Variant histones have evolved crucial roles in chromosome segregation, transcriptional regulation, DNA repair, sperm packaging and other processes. 'Universal' histone variants emerged early in eukaryotic evolution and were later displaced for bulk packaging roles by the canonical histones (H2A, H2B, H3 and H4), the synthesis of which is coupled to DNA replication. Further specializations of histone variants have evolved in some lineages to perform additional tasks. Differences among histone variants in their stability, DNA wrapping, specialized domains that regulate access to DNA, and post-translational modifications, underlie the diverse functions that histones have acquired in evolution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Luger, K., Mader, A. W., Richmond, R. K., Sargent, D. F. & Richmond, T. J. Crystal structure of the nucleosome core particle at 2.8 A resolution. Nature 389, 251–260 (1997).
Zlatanova, J., Bishop, T. C., Victor, J. M., Jackson, V. & van Holde, K. The nucleosome family: dynamic and growing. Structure 17, 160–171 (2009).
Allshire, R. C. & Karpen, G. H. Epigenetic regulation of centromeric chromatin: old dogs, new tricks? Nature Rev. Genet. 9, 923–937 (2008).
Malik, H. S. & Henikoff, S. Major evolutionary transitions in centromere complexity. Cell 138, 1067–1082 (2009).
Orsi, G. A., Couble, P. & Loppin, B. Epigenetic and replacement roles of histone variant H3.3 in reproduction and development. Int. J. Dev. Biol. 53, 231–243 (2009).
Zlatanova, J. & Thakar, A. H2A.Z: view from the top. Structure 16, 166–179 (2008).
Altaf, M., Auger, A., Covic, M. & Cote, J. Connection between histone H2A variants and chromatin remodeling complexes. Biochem. Cell Biol. 87, 35–50 (2009).
Ismail, I. H. & Hendzel, M. J. The γH2A.X: is it just a surrogate marker of double-strand breaks or much more? Environ. Mol. Mutagen. 49, 73–82 (2008).
Gonzalez-Romero, R., Mendez, J., Ausio, J. & Eirin-Lopez, J. M. Quickly evolving histones, nucleosome stability and chromatin folding: all about histone H2A.Bbd. Gene 413, 1–7 (2008).
Kouzarides, T. Chromatin modifications and their function. Cell 128, 693–705 (2007).
Park, Y. J. & Luger, K. Histone chaperones in nucleosome eviction and histone exchange. Curr. Opin. Struct. Biol. 18, 282–289 (2008).
Cairns, B. R. The logic of chromatin architecture and remodelling at promoters. Nature 461, 193–198 (2009).
Arents, G. & Moudrianakis, E. N. The histone fold: a ubiquitous architectural motif utilized in DNA compaction and protein dimerization. Proc. Natl Acad. Sci. USA 92, 11170–11174 (1995).
Sandman, K. & Reeve, J. N. Archaeal histones and the origin of the histone fold. Curr. Opin. Microbiol. 9, 520–525 (2006).
Pereira, S. L., Grayling, R. A., Lurz, R. & Reeve, J. N. Archaeal nucleosomes. Proc. Natl. Acad. Sci. USA 94, 12633–12637 (1997).
Marc, F., Sandman, K., Lurz, R. & Reeve, J. N. Archaeal histone tetramerization determines DNA affinity and the direction of DNA supercoiling. J. Biol. Chem. 277, 30879–30886 (2002).
Fahrner, R. L., Cascio, D., Lake, J. A. & Slesarev, A. An ancestral nuclear protein assembly: crystal structure of the Methanopyrus kandleri histone. Protein Sci. 10, 2002–2007 (2001).
Li, W. T., Sandman, K., Pereira, S. L. & Reeve, J. N. MJ1647, an open reading frame in the genome of the hyperthermophile Methanococcus jannaschii, encodes a very thermostable archaeal histone with a C-terminal extension. Extremophiles 4, 43–51 (2000).
Friedrich-Jahn, U., Aigner, J., Langst, G., Reeve, J. N. & Huber, H. Nanoarchaeal origin of histone H3? J. Bacteriol. 191, 1092–1096 (2009).
Alilat, M., Sivolob, A., Revet, B. & Prunell, A. Nucleosome dynamics. Protein and DNA contributions in the chiral transition of the tetrasome, the histone (H3-H4)2 tetramer-DNA particle. J. Mol. Biol. 291, 815–841 (1999).
Hackett, J. D. et al. Insights into a dinoflagellate genome through expressed sequence tag analysis. BMC Genomics 6, 80 (2005).
Malik, H. S. & Henikoff, S. Phylogenomics of the nucleosome. Nature Struct. Biol. 10, 882–891 (2003).
Santaguida, S. & Musacchio, A. The life and miracles of kinetochores. EMBO J. 28, 2511–2531 (2009).
Dawson, S. C., Sagolla, M. S. & Cande, W. Z. The CenH3 histone variant defines centromeres in Giardia intestinalis. Chromosoma 116, 175–184 (2007).
Talbert, P. B., Masuelli, R., Tyagi, A. P., Comai, L. & Henikoff, S. Centromeric localization and adaptive evolution of an Arabidopsis histone H3 variant. Plant Cell 14, 1053–1066 (2002).
Earnshaw, W. C. & Rothfield, N. Identification of a family of human centromere proteins using autoimmune sera from patients with scleroderma. Chromosoma 91, 313–321 (1985).
Malik, H. S., Vermaak, D. & Henikoff, S. Recurrent evolution of DNA-binding motifs in the Drosophila centromeric histone. Proc. Natl Acad. Sci. USA. 99, 1449–1454 (2002).
Churchill, M. E. & Suzuki, M. 'SPKK' motifs prefer to bind to DNA at A/T-rich sites. EMBO J. 8, 4189–4195 (1989).
Wieland, G., Orthaus, S., Ohndorf, S., Diekmann, S. & Hemmerich, P. Functional complementation of human centromere protein A (CENP-A) by Cse4p from Saccharomyces cerevisiae. Mol. Cell. Biol. 24, 6620–6630 (2004).
Dalal, Y., Wang, H., Lindsay, S. & Henikoff, S. Tetrameric structure of centromeric nucleosomes in interphase Drosophila cells. PLoS Biol. 5, e218 (2007).
Furuyama, T. & Henikoff, S. Centromeric nucleosomes induce positive DNA supercoils. Cell 138, 104–113 (2009).
Mizuguchi, G., Xiao, H., Wisniewski, J., Smith, M. M. & Wu, C. Nonhistone Scm3 and histones CenH3-H4 assemble the core of centromere-specific nucleosomes. Cell 129, 1153–1164 (2007).
Camahort, R. et al. Cse4 is part of an octameric nucleosome in budding yeast. Mol. Cell 35, 794–805 (2009). Together with references 30 and 32, this paper proposes mutually exclusive models for the CenH3 histone core; the correct model needs to accommodate the result of reference 31, which shows that CenH3 DNA wraps around the core in a right-handed direction.
Westermann, S. et al. Architecture of the budding yeast kinetochore reveals a conserved molecular core. J. Cell Biol. 163, 215–222 (2003).
Conde E Silva, N. et al. CENP-A-containing nucleosomes: easier disassembly versus exclusive centromeric localization. J. Mol. Biol. 370, 555–573 (2007).
Dalal, Y., Furuyama, T., Vermaak, D. & Henikoff, S. Structure, dynamics, and evolution of centromeric nucleosomes. Proc. Natl Acad. Sci. USA 104, 15974–15981 (2007).
Cui, B., Liu, Y. & Gorovsky, M. A. Deposition and function of histone H3 variants in Tetrahymena thermophila. Mol. Cell. Biol. 26, 7719–7730 (2006). Mutational analysis of canonical histone H3 and H3.3 shows that H3 is not essential in ciliate development, and H3.3 is not essential for transcription but is required in germline micronuclei.
Ahmad, K. & Henikoff, S. The histone variant H3.3 marks active chromatin by replication-independent nucleosome assembly. Mol. Cell 9, 1191–1200 (2002).
Tagami, H., Ray-Gallet, D., Almouzni, G. & Nakatani, Y. Histone H3.1 and H3.3 complexes mediate nucleosome assembly pathways dependent or independent of DNA synthesis. Cell 116, 51–61 (2004).
Polo, S. E., Roche, D. & Almouzni, G. New histone incorporation marks sites of UV repair in human cells. Cell 127, 481–493 (2006).
Mousson, F., Ochsenbein, F. & Mann, C. The histone chaperone Asf1 at the crossroads of chromatin and DNA checkpoint pathways. Chromosoma 116, 79–93 (2007).
Henikoff, S. Nucleosome destabilization in the epigenetic regulation of gene expression. Nature Rev. Genet. 9, 15–26 (2008).
Schwartz, B. E. & Ahmad, K. Transcriptional activation triggers deposition and removal of the histone variant H3.3. Genes Dev. 19, 804–814 (2005).
Sutcliffe, E. L. et al. Dynamic histone variant exchange accompanies gene induction in T cells. Mol. Cell. Biol. 29, 1972–1986 (2009).
Hake, S. B. & Allis, C. D. Histone H3 variants and their potential role in indexing mammalian genomes: the “H3 barcode hypothesis”. Proc. Natl Acad. Sci. USA 103, 6428–6435 (2006).
Loyola, A., Bonaldi, T., Roche, D., Imhof, A. & Almouzni, G. PTMs on H3 variants before chromatin assembly potentiate their final epigenetic state. Mol. Cell 24, 309–316 (2006).
Ng, R. K. & Gurdon, J. B. Epigenetic memory of an active gene state depends on histone H3.3 incorporation into chromatin in the absence of transcription. Nature Cell Biol. 10, 102–109 (2008). Using a Xenopus laevis nuclear transplantation assay, the authors show that epigenetic memory of a gene expression state is retained through 12 rounds of cell division without transcription and depends on the presence of wild-type H3.3 but not H3.
van der Heijden, G. W. et al. Chromosome-wide nucleosome replacement and H3.3 incorporation during mammalian meiotic sex chromosome inactivation. Nature Genet. 39, 251–258 (2007).
Ooi, S. L., Priess, J. R. & Henikoff, S. Histone H3.3 variant dynamics in the germline of Caenorhabditis elegans. PLoS Genet. 2, e97 (2006).
Jin, C. & Felsenfeld, G. Nucleosome stability mediated by histone variants H3.3 and H2A. Z. Genes Dev. 21, 1519–1529 (2007).
Hodl, M. & Basler, K. Transcription in the absence of histone H3.3. Curr. Biol. 19, 1221–1226 (2009).
Sakai, A., Schwartz, B. E., Goldstein, S. & Ahmad, K. Transcriptional and developmental functions of the H3.3 histone variant in Drosophila. Curr. Biol. 19, 1816–1820 (2009). References 51 and 52 show that H3.3 is dispensible for normal Drosophila development but is essential in the germ line, and reference 52 shows that the germline function does not require methylation of H3.3K4 or phosphorylation of Ser31.
Schulmeister, A., Schmid, M. & Thompson, E. M. Phosphorylation of the histone H3.3 variant in mitosis and meiosis of the urochordate Oikopleura dioica. Chromosome Res. 15, 189–201 (2007).
Wong, L. H. et al. Histone H3.3 incorporation provides a unique and functionally essential telomeric chromatin in embryonic stem cells. Genome Res. 19, 404–414 (2009).
Adam, M., Robert, F., Larochelle, M. & Gaudreau, L. H2A.Z is required for global chromatin integrity and for recruitment of RNA polymerase II under specific conditions. Mol. Cell. Biol. 21, 6270–6279 (2001).
Hardy, S. et al. The euchromatic and heterochromatic landscapes are shaped by antagonizing effects of transcription on H2A.Z deposition. PLoS Genet. 5, e1000687 (2009).
Zofall, M. et al. Histone H2A.Z cooperates with RNAi and heterochromatin factors to suppress antisense RNAs. Nature 461, 419–422 (2009).
Creyghton, M. P. et al. H2AZ is enriched at polycomb complex target genes in ES cells and is necessary for lineage commitment. Cell 135, 649–661 (2008). Reports a striking correlation between H2A.Z and Polycomb group protein locations in mouse embryonic stem cells but not in their differentiated descendants, suggesting that H2A.Z plays a key role in maintaining pluripotency.
Zilberman, D., Coleman-Derr, D., Ballinger, T. & Henikoff, S. Histone H2A.Z and DNA methylation are mutually antagonistic chromatin marks. Nature 456, 125–129 (2008). In A. thaliana , H2A.Z and DNA methylation are found to be quantitatively anti-correlated, and mutants in either one result in opposite changes in the other.
Thakar, A. et al. H2A.Z and H3.3 histone variants affect nucleosome structure: biochemical and biophysical studies. Biochemistry 48, 10852–10857 (2009). In contrast to the in vivo results of reference 68, these authors were unable to detect significant instability of H2A.Z and H3.3 nucleosomes in vitro.
Goldman, J. A., Garlick, J. D. & Kingston, R. E. Chromatin remodeling by imitation switch (ISWI) class ATP-dependent remodelers is stimulated by histone variant H2A. Z. J. Biol. Chem. 285, 4645–4651 (2009). This paper shows that H2A.Z nucleosomes are preferentially associated with nucleosome remodellers, with an enhanced activity of ISWI family remodellers that is dependent on the H2A.Z extended acidic patch.
Eirin-Lopez, J. M., Gonzalez-Romero, R., Dryhurst, D., Ishibashi, T. & Ausio, J. The evolutionary differentiation of two histone H2A.Z variants in chordates (H2A.Z-1 and H2A.Z-2) is mediated by a stepwise mutation process that affects three amino acid residues. BMC Evol. Biol. 9, 31 (2009).
Faast, R. et al. Histone variant H2A.Z is required for early mammalian development. Curr. Biol. 11, 1183–1187 (2001).
March-Diaz, R. et al. Histone H2A.Z and homologues of components of the SWR1 complex are required to control immunity in Arabidopsis. Plant J. 53, 475–487 (2008).
Ishibashi, T. et al. Acetylation of vertebrate H2A.Z and its effect on the structure of the nucleosome. Biochemistry 48, 5007–5017 (2009).
Viens, A. et al. Analysis of human histone H2AZ deposition in vivo argues against its direct role in epigenetic templating mechanisms. Mol. Cell. Biol. 26, 5325–5335 (2006).
Zhang, H., Roberts, D. N. & Cairns, B. R. Genome-wide dynamics of Htz1, a histone H2A variant that poises repressed/basal promoters for activation through histone loss. Cell 123, 219–231 (2005).
Jin, C. et al. H3.3/H2A.Z double variant-containing nucleosomes mark 'nucleosome-free regions' of active promoters and other regulatory regions. Nature Genet. 41, 941–945 (2009). Reports that nucleosomes containing both H3.3 and H2A.Z occupy promoters and insulator elements in vivo and are highly unstable.
Hartley, P. D. & Madhani, H. D. Mechanisms that specify promoter nucleosome location and identity. Cell 137, 445–458 (2009).
Mavrich, T. N. et al. Nucleosome organization in the Drosophila genome. Nature 453, 358–362 (2008). The authors mapped a large collection of Drosophila H2Av (H2A.Z) nucleosomes and found that where RNA polymerase II is paused just downstream of the transcriptional start site, the +1 H2A.Z nucleosome is positioned another 10 bp (1 rotational turn) further downstream, suggesting a role for H2A.Z in pausing polymerase.
Jiang, C. & Pugh, B. F. Nucleosome positioning and gene regulation: advances through genomics. Nature Rev. Genet. 10, 161–172 (2009).
Gevry, N., Chan, H. M., Laflamme, L., Livingston, D. M. & Gaudreau, L. p21 transcription is regulated by differential localization of histone H2A. Z. Genes Dev. 21, 1869–1881 (2007).
Gevry, N. et al. Histone H2A.Z is essential for estrogen receptor signaling. Genes Dev. 23, 1522–1533 (2009).
Choi, J., Heo, K. & An, W. Cooperative action of TIP48 and TIP49 in H2A.Z exchange catalyzed by acetylation of nucleosomal H2A. Nucleic Acids Res. 37, 5993–6007 (2009).
Fan, J. Y., Gordon, F., Luger, K., Hansen, J. C. & Tremethick, D. J. The essential histone variant H2A.Z regulates the equilibrium between different chromatin conformational states. Nature Struct. Biol. 9, 172–176 (2002).
Deal, R. B., Topp, C. N., McKinney, E. C. & Meagher, R. B. Repression of flowering in Arabidopsis requires activation of FLOWERING LOCUS C expression by the histone variant H2A. Z. Plant Cell 19, 74–83 (2007).
Ball, M. P. et al. Targeted and genome-scale strategies reveal gene-body methylation signatures in human cells. Nature Biotechnol. 27, 361–368 (2009).
Fan, J. Y., Rangasamy, D., Luger, K. & Tremethick, D. J. H2A.Z alters the nucleosome surface to promote HP1α-mediated chromatin fiber folding. Mol. Cell 16, 655–661 (2004).
Swaminathan, J., Baxter, E. M. & Corces, V. G. The role of histone H2Av variant replacement and histone H4 acetylation in the establishment of Drosophila heterochromatin. Genes Dev. 19, 65–76 (2005).
Hanai, K., Furuhashi, H., Yamamoto, T., Akasaka, K. & Hirose, S. RSF governs silent chromatin formation via histone H2Av replacement. PLoS Genet. 4, e1000011 (2008).
van Attikum, H. & Gasser, S. M. Crosstalk between histone modifications during the DNA damage response. Trends Cell Biol. 19, 207–217 (2009).
Shechter, D. et al. A distinct H2A.X isoform is enriched in Xenopus laevis eggs and early embryos and is phosphorylated in the absence of a checkpoint. Proc. Natl Acad. Sci. USA 106, 749–754 (2009).
Xiao, A. et al. WSTF regulates the H2A.X DNA damage response via a novel tyrosine kinase activity. Nature 457, 57–62 (2009).
Fernandez-Capetillo, O. et al. H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev. Cell. 4, 497–508 (2003).
Turner, J. M. et al. Silencing of unsynapsed meiotic chromosomes in the mouse. Nature Genet. 37, 41–47 (2005).
Van Doninck, K. et al. Phylogenomics of unusual histone H2A variants in Bdelloid rotifers. PLoS Genet. 5, e1000401 (2009). Shows that bdelloid rotifers, which periodically undergo severe dessication resulting in massive DNA breaks, have replaced H2A.X, which recruits DNA repair machinery in other eukaryotes, with novel H2A variants that might have evolved to facilitate DNA repair under dessicating conditions.
Marzluff, W. F., Gongidi, P., Woods, K. R., Jin, J. & Maltais, L. J. The human and mouse replication-dependent histone genes. Genomics 80, 487–498 (2002).
Marzluff, W. F., Sakallah, S. & Kelkar, H. The sea urchin histone gene complement. Dev. Biol. 300, 308–320 (2006).
Siegel, T. N. et al. Four histone variants mark the boundaries of polycistronic transcription units in Trypanosoma brucei. Genes Dev. 23, 1063–1076 (2009). Reports that trypanosomes have two versions of each of the four core histones, which form unique combinations at transcription initiation sites and termination sites. This suggests the existence of an ancestral mode of gene regulation based on histone variants and an ancient function for H2A.Z in marking promoters.
Bernhard, D. & Schlegel, M. Evolution of histone H4 and H3 genes in different ciliate lineages. J. Mol. Evol. 46, 344–354 (1998).
Katz, L. A., Bornstein, J. G., Lasek-Nesselquist, E. & Muse, S. V. Dramatic diversity of ciliate histone H4 genes revealed by comparisons of patterns of substitutions and paralog divergences among eukaryotes. Mol. Biol. Evol. 21, 555–562 (2004).
Gladyshev, E. & Meselson, M. Extreme resistance of bdelloid rotifers to ionizing radiation. Proc. Natl Acad. Sci. USA 105, 5139–5144 (2008).
Pehrson, J. R. & Fuji, R. N. Evolutionary conservation of histone macroH2A subtypes and domains. Nucleic Acids Res. 26, 2837–2842 (1998).
Chadwick, B. P. & Willard, H. F. Histone H2A variants and the inactive X chromosome: identification of a second macroH2A variant. Hum. Mol. Genet. 10, 1101–1113 (2001).
Costanzi, C. & Pehrson, J. R. MACROH2A2, a new member of the MARCOH2A core histone family. J. Biol. Chem. 276, 21776–21784 (2001).
Abbott, D. W., Chadwick, B. P., Thambirajah, A. A. & Ausio, J. Beyond the Xi: macroH2A chromatin distribution and post-translational modification in an avian system. J. Biol. Chem. 280, 16437–16445 (2005).
Doyen, C. M. et al. Mechanism of polymerase II transcription repression by the histone variant macroH2A. Mol. Cell. Biol. 26, 1156–1164 (2006).
Chakravarthy, S. & Luger, K. The histone variant macro-H2A preferentially forms “hybrid nucleosomes”. J. Biol. Chem. 281, 25522–25531 (2006).
Nusinow, D. A. et al. Poly(ADP-ribose) polymerase 1 is inhibited by a histone H2A variant, MacroH2A, and contributes to silencing of the inactive X chromosome. J. Biol. Chem. 282, 12851–12859 (2007).
Timinszky, G. et al. A macrodomain-containing histone rearranges chromatin upon sensing PARP1 activation. Nature Struct. Mol. Biol. 16, 923–929 (2009).
Ouararhni, K. et al. The histone variant mH2A1.1 interferes with transcription by down-regulating PARP-1 enzymatic activity. Genes Dev. 20, 3324–3336 (2006).
Buschbeck, M. et al. The histone variant macroH2A is an epigenetic regulator of key developmental genes. Nature Struct. Mol. Biol. (2009). This paper shows that mH2A serves as a repressive mark on autosomes, overlapping with Polycomb repressor complex 2 sites and contributing to regulation of homeobox genes during neuronal differentiation.
Eirin-Lopez, J. M., Ishibashi, T. & Ausio, J. H2A.Bbd: a quickly evolving hypervariable mammalian histone that destabilizes nucleosomes in an acetylation-independent way. FASEB J. 22, 316–326 (2008).
Chadwick, B. P. & Willard, H. F. A novel chromatin protein, distantly related to histone H2A, is largely excluded from the inactive X chromosome. J. Cell Biol. 152, 375–384 (2001).
Gautier, T. et al. Histone variant H2ABbd confers lower stability to the nucleosome. EMBO Rep. 5, 715–720 (2004).
Okuwaki, M., Kato, K., Shimahara, H., Tate, S. & Nagata, K. Assembly and disassembly of nucleosome core particles containing histone variants by human nucleosome assembly protein I. Mol. Cell. Biol. 25, 10639–10651 (2005).
Zhou, J., Fan, J. Y., Rangasamy, D. & Tremethick, D. J. The nucleosome surface regulates chromatin compaction and couples it with transcriptional repression. Nature Struct. Mol. Biol. 14, 1070–1076 (2007).
Yi, H. et al. Constitutive expression exposes functional redundancy between the Arabidopsis histone H2A gene HTA1 and other H2A gene family members. Plant Cell 18, 1575–1589 (2006).
Bergmuller, E., Gehrig, P. M. & Gruissem, W. Characterization of post-translational modifications of histone H2B-variants isolated from Arabidopsis thaliana. J. Proteome Res. 6, 3655–3668 (2007).
Lindsey, G. G., Orgeig, S., Thompson, P., Davies, N. & Maeder, D. L. Extended C-terminal tail of wheat histone H2A interacts with DNA of the “linker” region. J. Mol. Biol. 218, 805–813 (1991).
Green, G. R. Phosphorylation of histone variant regions in chromatin: unlocking the linker? Biochem. Cell Biol. 79, 275–287 (2001).
Eirin-Lopez, J. M. & Ausio, J. Origin and evolution of chromosomal sperm proteins. Bioessays 31, 1062–1070 (2009).
Hammoud, S. S. et al. Distinctive chromatin in human sperm packages genes for embryo development. Nature 460, 473–478 (2009).
Palmer, D. K., O'Day, K. & Margolis, R. L. The centromere specific histone CENP-A is selectively retained in discrete foci in mammalian sperm nuclei. Chromosoma 100, 32–36 (1990).
Gatewood, J. M., Cook, G. R., Balhorn, R., Schmid, C. W. & Bradbury, E. M. Isolation of four core histones from human sperm chromatin representing a minor subset of somatic histones. J. Biol. Chem. 265, 20662–20666 (1990).
van der Heijden, G. W. et al. Sperm-derived histones contribute to zygotic chromatin in humans. BMC Dev. Biol. 8, 34 (2008).
Ueda, K. et al. Unusual core histones specifically expressed in male gametic cells of Lilium longiflorum. Chromosoma 108, 491–500 (2000).
Ingouff, M., Hamamura, Y., Gourgues, M., Higashiyama, T. & Berger, F. Distinct dynamics of HISTONE3 variants between the two fertilization products in plants. Curr. Biol. 17, 1032–1037 (2007).
Talbert, P. B. & Henikoff, S. Chromatin-based transcriptional punctuation. Genes Dev. 23, 1037–1041 (2009).
Aggarwal, B. D. & Calvi, B. R. Chromatin regulates origin activity in Drosophila follicle cells. Nature 430, 372–376 (2004).
Miotto, B. & Struhl, K. HBO1 histone acetylase is a coactivator of the replication licensing factor Cdt1. Genes Dev. 22, 2633–2638 (2008).
Ren, Q. & Gorovsky, M. A. Histone H2A.Z acetylation modulates an essential charge patch. Mol. Cell 7, 1329–1335 (2001).
Millar, C. B., Xu, F., Zhang, K. & Grunstein, M. Acetylation of H2AZ Lys 14 is associated with genome-wide gene activity in yeast. Genes Dev. 20, 711–722 (2006).
Tanabe, M. et al. Activation of facultatively silenced Drosophila loci associates with increased acetylation of histone H2AvD. Genes Cells 13, 1279–1288 (2008).
Wan, Y. et al. Role of the histone variant H2A.Z/Htz1p in TBP recruitment, chromatin dynamics, and regulated expression of oleate-responsive genes. Mol. Cell. Biol. 29, 2346–2358 (2009).
de Napoles, M. et al. Polycomb group proteins Ring1A/B link ubiquitylation of histone H2A to heritable gene silencing and X inactivation. Dev. Cell. 7, 663–676 (2004).
Sarcinella, E., Zuzarte, P. C., Lau, P. N., Draker, R. & Cheung, P. Monoubiquitylation of H2A.Z distinguishes its association with euchromatin or facultative heterochromatin. Mol. Cell. Biol. 27, 6457–6468 (2007).
Stock, J. K. et al. Ring1-mediated ubiquitination of H2A restrains poised RNA polymerase II at bivalent genes in mouse ES cells. Nature Cell Biol. 9, 1428–1435 (2007).
Wang, H. et al. Role of histone H2A ubiquitination in Polycomb silencing. Nature 431, 873–878 (2004).
Sogin, M. L. & Silberman, J. D. Evolution of the protists and protistan parasites from the perspective of molecular systematics. Int. J. Parasitol. 28, 11–20 (1998).
Clayton, C. E. Life without transcriptional control? From fly to man and back again. EMBO J. 21, 1881–1888 (2002).
Martinez-Calvillo, S., Nguyen, D., Stuart, K. & Myler, P. J. Transcription initiation and termination on Leishmania major chromosome 3. Eukaryot. Cell. 3, 506–517 (2004).
Vanacova, S., Liston, D. R., Tachezy, J. & Johnson, P. J. Molecular biology of the amitochondriate parasites, Giardia intestinalis, Entamoeba histolytica and Trichomonas vaginalis. Int. J. Parasitol. 33, 235–255 (2003).
Lowell, J. E. & Cross, G. A. A variant histone H3 is enriched at telomeres in Trypanosoma brucei. J. Cell. Sci. 117, 5937–5947 (2004).
Ghosh, S. & Klobutcher, L. A. A development-specific histone H3 localizes to the developing macronucleus of Euplotes. Genesis 26, 179–188 (2000).
Cui, B. & Gorovsky, M. A. Centromeric histone H3 is essential for vegetative cell division and for DNA elimination during conjugation in Tetrahymena thermophila. Mol. Cell. Biol. 26, 4499–4510 (2006).
Zeitlin, S. G. et al. Double-strand DNA breaks recruit the centromeric histone CENP-A. Proc. Natl Acad. Sci. USA 106, 15762–15767 (2009).
Cervantes, M. D., Xi, X., Vermaak, D., Yao, M. C. & Malik, H. S. The CNA1 histone of the ciliate Tetrahymena thermophila is essential for chromosome segregation in the germline micronucleus. Mol. Biol. Cell 17, 485–497 (2006).
Iribarren, C., Morin, V., Puchi, M. & Imschenetzky, M. Sperm nucleosomes disassembly is a requirement for histones proteolysis during male pronucleus formation. J. Cell. Biochem. 103, 447–455 (2008).
Govin, J. et al. Pericentric heterochromatin reprogramming by new histone variants during mouse spermiogenesis. J. Cell Biol. 176, 283–294 (2007). Describes new H2A variants enriched in pericentric heterochromatin in spermatids that form sub-nucleosomal chromatin particles lacking H3 and H4.
Tachiwana, H., Osakabe, A., Kimura, H. & Kurumizaka, H. Nucleosome formation with the testis-specific histone H3 variant, H3t, by human nucleosome assembly proteins in vitro. Nucleic Acids Res. 36, 2208–2218 (2008).
Li, A. et al. Characterization of nucleosomes consisting of the human testis/sperm-specific histone H2B variant (hTSH2B). Biochemistry 44, 2529–2535 (2005).
Churikov, D. et al. Novel human testis-specific histone H2B encoded by the interrupted gene on the X chromosome. Genomics 84, 745–756 (2004).
Boulard, M. et al. The NH2 tail of the novel histone variant H2BFWT exhibits properties distinct from conventional H2B with respect to the assembly of mitotic chromosomes. Mol. Cell. Biol. 26, 1518–1526 (2006).
Aul, R. B. & Oko, R. J. The major subacrosomal occupant of bull spermatozoa is a novel histone H2B variant associated with the forming acrosome during spermiogenesis. Dev. Biol. 242, 376–387 (2002).
Syed, S. H. et al. The incorporation of the novel histone variant H2AL2 confers unusual structural and functional properties of the nucleosome. Nucleic Acids Res. 37, 4684–4695 (2009).
Gaucher, J. et al. From meiosis to postmeiotic events: the secrets of histone disappearance. FEBS J. 277, 509–604 (2009).
Huson, D. H. et al. Dendroscope: an interactive viewer for large phylogenetic trees. BMC Bioinformatics 8, 460 (2007).
Acknowledgements
We thank our colleagues for stimulating discussions and anonymous reviewers for their helpful comments. Our work has been funded by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Histone chaperone
-
An escort protein that performs a transfer reaction on a histone, such as deposition onto DNA, eviction from DNA, transfer to another chaperone or enzyme, or storage for later use.
- Supercoil
-
A contortion in DNA that occurs as a consequence to over- or under-twisting of the DNA helix. Supercoils can be introduced during DNA packaging and DNA–RNA synthesis. Topoisomerases sense supercoiling and can either generate or dissipate it by changing DNA topology.
- Centromere
-
The region of a chromosome that is attached to the spindle during nuclear division.
- Kinetochore
-
A large multiprotein complex that assembles onto the centromere of the chromosome and links it to the microtubules of the mitotic spindle. The kinetochore is also a signalling centre for many of the proteins that control the progression of mitosis.
- Epigenetic memory
-
An effect on gene expression or function that is not a result of DNA sequence changes and is heritable through cell division.
- Nuclease-hypersensitive site
-
A chromosomal site that shows increased sensitivity to nucleases such as DNase I and that are correlated to regions of reduced nucleosome density and gene regulatory sites.
- Heterochromatin
-
A highly condensed form of chromatin with very low transcriptional activity. It occurs at defined sites, such as around centromeres or telomeres. Typically it is composed of repetitive sequences and transposons, with few genes present.
- Homeobox gene
-
One of a family of genes that encode homeodomain-containing transcription factors, which are involved in the patterning of the body during development.
- Protamine
-
A small, highly basic protein that tightly packages sperm DNA, replacing histones completely or to varying degrees in many animals.
Rights and permissions
About this article
Cite this article
Talbert, P., Henikoff, S. Histone variants — ancient wrap artists of the epigenome. Nat Rev Mol Cell Biol 11, 264–275 (2010). https://doi.org/10.1038/nrm2861
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm2861
This article is cited by
-
Epigenetic regulation of human-specific gene expression in the prefrontal cortex
BMC Biology (2023)
-
Bacterial histones unveiled
Nature Microbiology (2023)
-
Histone exchange sensors reveal variant specific dynamics in mouse embryonic stem cells
Nature Communications (2023)
-
Histone 3.3-related chromatinopathy: missense variants throughout H3-3A and H3-3B cause a range of functional consequences across species
Human Genetics (2023)
-
Epigenetic remodelling in human hepatocellular carcinoma
Journal of Experimental & Clinical Cancer Research (2022)