Key Points
-
More than 30 mammalian genes comprise the TGFβ superfamily of growth factors. They have important roles in the regulation of cellular functions both in the adult organism and during embryogenesis.
-
Signal duration and signal magnitude are crucial determinants of the outcome of TGFβ superfamily signalling, in particular in early embryonic patterning, in which TGFβ superfamily ligands act as morphogens that elicit distinct responses in a dose-dependent fashion.
-
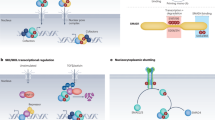
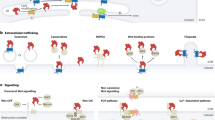
The canonical TGFβ signalling pathway involves ligand-dependent assembly of a heteromeric receptor complex, receptor-kinase activation and subsequent phosphorylation and activation of SMAD proteins, which are transcriptional regulators that consequently accumulate in the nucleus. Thus, SMADs both transmit the signal into the nucleus and execute the downstream effects by directly regulating target-gene transcription.
-
Signal duration and signal magnitude are relayed faithfully into the nucleus in a quantitative manner. Such continuous sensing of signal intensity might involve cycling of receptors through the endocytic pathway.
-
In addition, nuclear accumulation of active nuclear SMAD complexes is dynamically maintained in strict accordance with the degree of receptor activation at any time of signalling. SMAD phosphorylation by active receptors, and constitutive SMAD dephosphorylation by nuclear phosphatases, are coupled through nucleocytoplasmic shuttling of SMADs.
-
An in-depth understanding of the pleiotropic cellular functions that can be evoked by TGFβ superfamily signalling will depend on a more quantitative knowledge of temporal and spatial characteristics of the pathway.
Abstract
Ligands of the transforming growth factor-β (TGFβ) superfamily of growth factors initiate signal transduction through a bewildering complexity of ligand–receptor interactions. Signalling then converges to nuclear accumulation of transcriptionally active SMAD complexes and gives rise to a plethora of specific functional responses in both embryos and adult organisms. Current research is focused on the mechanisms that regulate SMAD activity to evoke cell-type-specific and context-dependent transcriptional programmes. An equally important challenge is understanding the functional role of signal strength and duration. How are these quantitative aspects of the extracellular signal regulated? How are they then sensed and interpreted, and how do they affect responses?
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Blobe, G. C., Schiemann, W. P. & Lodish, H. F. Role of transforming growth factor β in human disease. N. Engl. J. Med. 342, 1350–1358 (2000).
Massagué, J., Blain, S. W. & Lo, R. S. TGFβ signaling in growth control, cancer, and heritable disorders. Cell 103, 295–309 (2000).
Gray, A. M. & Mason, A. J. Requirement for activin A and transforming growth factor-β1 pro-regions in homodimer assembly. Science 247, 1328–1330 (1990).
Shimmi, O., Umulis, D., Othmer, H. & O'Connor, M. B. Facilitated transport of a Dpp/Scw heterodimer by Sog/Tsg leads to robust patterning of the Drosophila blastoderm embryo. Cell 120, 873–886 (2005). Evidence for an intriguing hypothesis explaining the biphasic signal required for specifying the two dorsal tissue types in D. melanogaster.
Dubois, C. M., Laprise, M. H., Blanchette, F., Gentry, L. E. & Leduc, R. Processing of transforming growth factor β1 precursor by human furin convertase. J. Biol. Chem. 270, 10618–10624 (1995).
Constam, D. B. & Robertson, E. J. Regulation of bone morphogenetic protein activity by pro domains and proprotein convertases. J. Cell Biol. 144, 139–149 (1999).
Ben-Haim, N. et al. The nodal precursor acting via activin receptors induces mesoderm by maintaining a source of its convertases and BMP4. Dev. Cell 11, 313–323 (2006).
Ge, G., Hopkins, D. R., Ho, W. B. & Greenspan, D. S. GDF11 forms a bone morphogenetic protein 1-activated latent complex that can modulate nerve growth factor-induced differentiation of PC12 cells. Mol. Cell. Biol. 25, 5846–5858 (2005).
Annes, J. P., Munger, J. S. & Rifkin, D. B. Making sense of latent TGFβ activation. J. Cell Sci. 116, 217–224 (2003).
Ge, G. & Greenspan, D. S. BMP1 controls TGFβ1 activation via cleavage of latent TGFβ-binding protein. J. Cell Biol. 175, 111–120 (2006).
Shi, Y. & Massagué, J. Mechanisms of TGF-β signaling from cell membrane to the nucleus. Cell 113, 685–700 (2003).
Yeo, C. & Whitman, M. Nodal signals to SMADs through Cripto-dependent and Cripto-independent mechanisms. Mol. Cell 7, 949–957 (2001).
Feng, X. H. & Derynck, R. Specificity and versatility in TGFβ signaling through SMADs. Annu. Rev. Cell Dev. Biol. 21, 659–693 (2005).
Allendorph, G. P., Vale, W. W. & Choe, S. Structure of the ternary signaling complex of a TGF-β superfamily member. Proc. Natl Acad. Sci. USA 103, 7643–7648 (2006). Insights into constraints for receptor assembly gained through a structure of a ternary ligand–type II–type I receptor complex.
Yagi, K. et al. Alternatively spliced variant of SMAD2 lacking exon 3. Comparison with wild-type SMAD2 and SMAD3. J. Biol. Chem. 274, 703–709 (1999).
Sapkota, G., Alarcon, C., Spagnoli, F. M., Brivanlou, A. H. & Massagué, J. Balancing BMP signaling through integrated inputs into the SMAD1 linker. Mol. Cell 25, 441–454 (2007).
Liu, F. SMAD3 phosphorylation by cyclin-dependent kinases. Cytokine Growth Factor Rev. 17, 9–17 (2006).
Ross, S. et al. SMADs orchestrate specific histone modifications and chromatin remodeling to activate transcription. EMBO J. 25, 4490–4502 (2006).
Moustakas, A. & Heldin, C. H. Non-SMAD TGF-β signals. J. Cell Sci. 118, 3573–3584 (2005).
Ozdamar, B. et al. Regulation of the polarity protein Par6 by TGFβ receptors controls epithelial cell plasticity. Science 307, 1603–1609 (2005).
Lee, M. K. et al. TGF-β activates Erk MAP kinase signalling through direct phosphorylation of ShcA. EMBO J. 26, 3957–3967 (2007).
Itoh, S. & ten Dijke, P. Negative regulation of TGF-β receptor/SMAD signal transduction. Curr. Opin. Cell Biol. 19, 176–184 (2007).
Hayashi, H. et al. The MAD-related protein SMAD7 associates with the TGFβ receptor and functions as an antagonist of TGFβ signaling. Cell 89, 1165–1173 (1997).
Kavsak, P. et al. SMAD7 binds to Smurf2 to form an E3 ubiquitin ligase that targets the TGF β receptor for degradation. Mol. Cell 6, 1365–1375 (2000).
Ebisawa, T. et al. Smurf1 interacts with transforming growth factor-β type I receptor through SMAD7 and induces receptor degradation. J. Biol. Chem. 276, 12477–12480 (2001).
Ogunjimi, A. A. et al. Regulation of Smurf2 ubiquitin ligase activity by anchoring the E2 to the HECT domain. Mol. Cell 19, 297–308 (2005).
Shi, W. et al. GADD34–PP1c recruited by SMAD7 dephosphorylates TGFβ type I receptor. J. Cell Biol. 164, 291–300 (2004).
Zhang, S. et al. SMAD7 antagonizes transforming growth factor β signaling in the nucleus by interfering with functional SMAD–DNA complex formation. Mol. Cell. Biol. 27, 4488–4499 (2007).
Macias-Silva, M., Hoodless, P. A., Tang, S. J., Buchwald, M. & Wrana, J. L. Specific activation of SMAD1 signaling pathways by the BMP7 type I receptor, ALK2. J. Biol. Chem. 273, 25628–25636 (1998).
Sakuma, R. et al. Inhibition of Nodal signalling by Lefty mediated through interaction with common receptors and efficient diffusion. Genes Cells 7, 401–412 (2002).
Greenwald, J. et al. The BMP7/ActRII extracellular domain complex provides new insights into the cooperative nature of receptor assembly. Mol. Cell 11, 605–617 (2003).
Rebbapragada, A., Benchabane, H., Wrana, J. L., Celeste, A. J. & Attisano, L. Myostatin signals through a transforming growth factor β-like signaling pathway to block adipogenesis. Mol. Cell. Biol. 23, 7230–7242 (2003).
Andersson, O., Reissmann, E. & Ibanez, C. F. Growth differentiation factor 11 signals through the transforming growth factor-β receptor ALK5 to regionalize the anterior–posterior axis. EMBO Rep. 7, 831–837 (2006).
Oh, S. P. et al. Activin type IIA and IIB receptors mediate Gdf11 signaling in axial vertebral patterning. Genes Dev. 16, 2749–2754 (2002).
Balemans, W. & Van Hul, W. Extracellular regulation of BMP signaling in vertebrates: a cocktail of modulators. Dev. Biol. 250, 231–250 (2002).
Piccolo, S. et al. The head inducer Cerberus is a multifunctional antagonist of Nodal, BMP and Wnt signals. Nature 397, 707–710 (1999).
Hemmati-Brivanlou, A., Kelly, O. G. & Melton, D. A. Follistatin, an antagonist of activin, is expressed in the Spemann organizer and displays direct neuralizing activity. Cell 77, 283–295 (1994).
Thompson, T. B., Lerch, T. F., Cook, R. W., Woodruff, T. K. & Jardetzky, T. S. The structure of the follistatin:activin complex reveals antagonism of both type I and type II receptor binding. Dev. Cell 9, 535–543 (2005).
Schier, A. F. Nodal signaling in vertebrate development. Annu. Rev. Cell Dev. Biol. 19, 589–621 (2003).
Cheng, S. K., Olale, F., Brivanlou, A. H. & Schier, A. F. Lefty blocks a subset of TGFβ signals by antagonizing EGF-CFC coreceptors. PLoS Biol. 2, e30 (2004).
Chen, Y. G. et al. Determinants of specificity in TGF-β signal transduction. Genes Dev. 12, 2144–2152 (1998).
Miyazawa, K., Shinozaki, M., Hara, T., Furuya, T. & Miyazono, K. Two major SMAD pathways in TGF-β superfamily signalling. Genes Cells 7, 1191–1204 (2002).
Gilchrist, R. B. et al. Molecular basis of oocyte–paracrine signalling that promotes granulosa cell proliferation. J. Cell Sci. 119, 3811–3821 (2006).
Goumans, M. J. et al. Activin receptor-like kinase (ALK)1 is an antagonistic mediator of lateral TGFβ/ALK5 signaling. Mol. Cell 12, 817–828 (2003). Evidence for the formation of receptor complexes that contain both ALK1 and ALK5 and activate both subclasses of R-SMADs in response to a single ligand, TGFβ.
Goumans, M. J. et al. Balancing the activation state of the endothelium via two distinct TGF-β type I receptors. EMBO J. 21, 1743–1753 (2002).
Byfield, S. D. & Roberts, A. B. Lateral signaling enhances TGF-β response complexity. Trends Cell Biol. 14, 107–111 (2004).
Batut, J., Howell, M. & Hill, C. S. Kinesin-mediated transport of SMAD2 is required for signaling in response to TGF-β ligands. Dev. Cell 12, 261–274 (2007). A requirement for intact microtubules and the motor protein kinesin-1 for efficient SMAD2 phosphorylation suggests a sophisticated mechanism that presents SMADs to receptors.
Shi, W. et al. Endofin acts as a SMAD anchor for receptor activation in BMP signaling. J. Cell Sci. 120, 1216–1224 (2007).
Chen, Y. G., Wang, Z., Ma, J., Zhang, L. & Lu, Z. Endofin, a FYVE domain protein, interacts with SMAD4 and facilitates transforming growth factor-β signaling. J. Biol. Chem. 282, 9688–9695 (2007).
Massagué, J., Seoane, J. & Wotton, D. SMAD transcription factors. Genes Dev. 19, 2783–2810 (2005).
Dennler, S. et al. Direct binding of SMAD3 and SMAD4 to critical TGF β-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Zawel, L. et al. Human SMAD3 and SMAD4 are sequence-specific transcription activators. Mol. Cell 1, 611–617 (1998).
Shi, Y. et al. Crystal structure of a SMAD MH1 domain bound to DNA: insights on DNA binding in TGF-β signaling. Cell 94, 585–594 (1998).
Kim, J., Johnson, K., Chen, H. J., Carroll, S. & Laughon, A. Drosophila Mad binds to DNA and directly mediates activation of vestigial by Decapentaplegic. Nature 388, 304–308 (1997). Identification of the first SBE.
Gao, S., Steffen, J. & Laughon, A. Dpp-responsive silencers are bound by a trimeric Mad–Medea complex. J. Biol. Chem. 280, 36158–36164 (2005).
Pyrowolakis, G., Hartmann, B., Muller, B., Basler, K. & Affolter, M. A simple molecular complex mediates widespread BMP-induced repression during Drosophila development. Dev. Cell 7, 229–240 (2004). Identification of the MAD–Medea–Schnurri repressor complex.
Yao, L. C. et al. Schnurri transcription factors from Drosophila and vertebrates can mediate Bmp signaling through a phylogenetically conserved mechanism. Development 133, 4025–4034 (2006). D. melanogaster Schnurri and its vertebrate homologue are functionally equivalent: both bind BMP-responsive elements together with SMAD1 and SMAD4 (MAD and Medea). Interestingly, in a D. melanogaster context, this causes target-gene repression, but causes target-gene activation in a vertebrate context.
Korchynskyi, O. & ten Dijke, P. Identification and functional characterization of distinct critically important bone morphogenetic protein-specific response elements in the Id1 promoter. J. Biol. Chem. 277, 4883–4891 (2002).
Stroschein, S. L., Wang, W., Zhou, S., Zhou, Q. & Luo, K. Negative feedback regulation of TGF-β signaling by the SnoN oncoprotein. Science 286, 771–774 (1999).
Levy, L. et al. Arkadia activates SMAD3/SMAD4-dependent transcription by triggering signal-induced SnoN degradation. Mol. Cell. Biol. 27, 6068–6083 (2007). The E3-ubiquitin ligase Arkadia is absolutely required for SMAD3-dependent gene activation from SNON-repressed promoters by inducing degradation of SNON in response to TGFβ.
Nagano, Y. et al. Arkadia induces degradation of SnoN and c-Ski to enhance TGF-β signaling. J. Biol. Chem. 282, 20492–20501 (2007).
Chen, X., Rubock, M. J. & Whitman, M. A transcriptional partner for MAD proteins in TGF-β signalling. Nature 383, 691–696 (1996).
Chen, X. et al. SMAD4 and FAST-1 in the assembly of activin-responsive factor. Nature 389, 85–89 (1997).
Germain, S., Howell, M., Esslemont, G. M. & Hill, C. S. Homeodomain and winged-helix transcription factors recruit activated SMADs to distinct promoter elements via a common SMAD interaction motif. Genes Dev. 14, 435–451 (2000).
Kunwar, P. S. et al. Mixer/Bon and FoxH1/Sur have overlapping and divergent roles in Nodal signaling and mesendoderm induction. Development 130, 5589–5599 (2003).
Randall, R. A., Germain, S., Inman, G. J., Bates, P. A. & Hill, C. S. Different SMAD2 partners bind a common hydrophobic pocket in SMAD2 via a defined proline-rich motif. EMBO J. 21, 145–156 (2002).
Kang, Y., Chen, C. R. & Massagué, J. A self-enabling TGFβ response coupled to stress signaling: SMAD engages stress response factor ATF3 for Id1 repression in epithelial cells. Mol. Cell 11, 915–926 (2003).
Cordenonsi, M. et al. Integration of TGF-β and Ras/MAPK signaling through p53 phosphorylation. Science 315, 840–843 (2007). p53 enables crosstalk between receptor tyrosine kinase signalling and TGFβ signalling.
He, W. et al. Hematopoiesis controlled by distinct TIF1γ and SMAD4 branches of the TGFβ pathway. Cell 125, 929–941 (2006). Proposes a mechanism by which ectodermin (also known as TIF1γ or TRIM33) can compete with SMAD4 for SMAD2/3 binding, thus forming an alternative transcriptionally active complex.
Dupont, S. et al. Germ-layer specification and control of cell growth by Ectodermin, a SMAD4 ubiquitin ligase. Cell 121, 87–99 (2005). Ectodermin is shown to restrict TGFβ signalling by acting as a RING-domain ubiquitin ligase that targets SMAD4 for degradation.
Gurdon, J. B., Dyson, S. & St Johnston, D. Cells' perception of position in a concentration gradient. Cell 95, 159–162 (1998).
Green, J. B. & Smith, J. C. Graded changes in dose of a Xenopus activin A homologue elicit stepwise transitions in embryonic cell fate. Nature 347, 391–394 (1990).
Dyson, S. & Gurdon, J. B. The interpretation of position in a morphogen gradient as revealed by occupancy of activin receptors. Cell 93, 557–568 (1998).
Inman, G. J. et al. SB-431542 is a potent and specific inhibitor of transforming growth factor-β superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 62, 65–74 (2002).
Jullien, J. & Gurdon, J. Morphogen gradient interpretation by a regulated trafficking step during ligand-receptor transduction. Genes Dev. 19, 2682–2694 (2005).
Raftery, L. A. & Sutherland, D. J. Gradients and thresholds: BMP response gradients unveiled in Drosophila embryos. Trends Genet. 19, 701–708 (2003).
Lee, M. A., Heasman, J. & Whitman, M. Timing of endogenous activin-like signals and regional specification of the Xenopus embryo. Development 128, 2939–2952 (2001).
Di Guglielmo, G. M., Le Roy, C., Goodfellow, A. F. & Wrana, J. L. Distinct endocytic pathways regulate TGF-β receptor signalling and turnover. Nature Cell Biol. 5, 410–421 (2003). Introduces the concept of two distinct competing endocytic routes for TGFβ receptors.
Le Roy, C. & Wrana, J. L. Clathrin- and non-clathrin-mediated endocytic regulation of cell signalling. Nature Rev. Mol. Cell Biol. 6, 112–126 (2005).
Inman, G. J., Nicolás, F. J. & Hill, C. S. Nucleocytoplasmic shuttling of SMADs 2, 3, and 4 permits sensing of TGF-β receptor activity. Mol. Cell 10, 283–294 (2002).
Nicolás, F. J., De Bosscher, K., Schmierer, B. & Hill, C. S. Analysis of SMAD nucleocytoplasmic shuttling in living cells. J. Cell Sci. 117, 4113–4125 (2004).
Schmierer, B. & Hill, C. S. Kinetic analysis of SMAD nucleocytoplasmic shuttling reveals a mechanism for transforming growth factor β-dependent nuclear accumulation of SMADs. Mol. Cell. Biol. 25, 9845–9858 (2005).
Lo, R. S. & Massagué, J. Ubiquitin-dependent degradation of TGF-β-activated SMAD2. Nature Cell Biol. 1, 472–478 (1999).
Xu, L. & Massagué, J. Nucleocytoplasmic shuttling of signal transducers. Nature Rev. Mol. Cell Biol. 5, 209–219 (2004).
Chen, H. B., Shen, J., Ip, Y. T. & Xu, L. Identification of phosphatases for SMAD in the BMP/DPP pathway. Genes Dev. 20, 648–653 (2006).
Lin, X. et al. PPM1A functions as a SMAD phosphatase to terminate TGFβ signaling. Cell 125, 915–928 (2006). Convincingly establishes PPM1A as an R-SMAD C-terminal phosphatase.
Duan, X., Liang, Y. Y., Feng, X. H. & Lin, X. Dephosphorylation of SMAD1 in the BMP signaling pathway by PPM1A. J. Biol. Chem. 281, 36526–36532 (2006).
Yoshizaki, T. et al. Protein phosphatase-2C α as a positive regulator of insulin sensitivity through direct activation of phosphatidylinositol 3-kinase in 3T3-L1 adipocytes. J. Biol. Chem. 279, 22715–22726 (2004).
Strovel, E. T., Wu, D. & Sussman, D. J. Protein phosphatase 2Cα dephosphorylates axin and activates LEF-1-dependent transcription. J. Biol. Chem. 275, 2399–2403 (2000).
Cheng, A., Kaldis, P. & Solomon, M. J. Dephosphorylation of human cyclin-dependent kinases by protein phosphatase type 2C α and β2 isoforms. J. Biol. Chem. 275, 34744–34749 (2000).
Pierreux, C. E., Nicolás, F. J. & Hill, C. S. Transforming growth factor β-independent shuttling of SMAD4 between the cytoplasm and nucleus. Mol. Cell. Biol. 20, 9041–9054 (2000).
Xu, L., Kang, Y., Col, S. & Massagué, J. SMAD2 nucleocytoplasmic shuttling by nucleoporins CAN/Nup214 and Nup153 feeds TGFβ signaling complexes in the cytoplasm and nucleus. Mol. Cell 10, 271–282 (2002).
Dudu, V. et al. Postsynaptic MAD signaling at the Drosophila neuromuscular junction. Curr. Biol. 16, 625–635 (2006).
Chen, H. B., Rud, J. G., Lin, K. & Xu, L. Nuclear targeting of transforming growth factor-β-activated SMAD complexes. J. Biol. Chem. 280, 21329–21336 (2005).
Kolch, W. Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem. J. 351, 289–305 (2000).
Schoeberl, B., Eichler-Jonsson, C., Gilles, E. D. & Muller, G. Computational modeling of the dynamics of the MAP kinase cascade activated by surface and internalized EGF receptors. Nature Biotechnol. 20, 370–375 (2002).
Podos, S. D., Hanson, K. K., Wang, Y. C. & Ferguson, E. L. The DSmurf ubiquitin-protein ligase restricts BMP signaling spatially and temporally during Drosophila embryogenesis. Dev. Cell 1, 567–578 (2001).
Turing, A. The chemical basis of morphogenesis. Phil. Trans. R. Soc. Lond. B 237, 37–72 (1952).
Meinhardt, H. Models for positional signalling with application to the dorsoventral patterning of insects and segregation into different cell types. Development 107 Suppl., 169–180 (1989).
Piccolo, S. et al. Cleavage of Chordin by Xolloid metalloprotease suggests a role for proteolytic processing in the regulation of Spemann organizer activity. Cell 91, 407–416 (1997).
Hopkins, D. R., Keles, S. & Greenspan, D. S. The bone morphogenetic protein 1/Tolloid-like metalloproteinases. Matrix Biol. 26, 508–523 (2007).
Lee, H. X., Ambrosio, A. L., Reversade, B. & De Robertis, E. M. Embryonic dorsal-ventral signaling: secreted frizzled-related proteins as inhibitors of tolloid proteinases. Cell 124, 147–159 (2006).
Muraoka, O. et al. Sizzled controls dorso-ventral polarity by repressing cleavage of the Chordin protein. Nature Cell Biol. 8, 329–338 (2006).
Reeves, G. T., Muratov, C. B., Schupbach, T. & Shvartsman, S. Y. Quantitative models of developmental pattern formation. Dev. Cell 11, 289–300 (2006).
Teleman, A. A., Strigini, M. & Cohen, S. M. Shaping morphogen gradients. Cell 105, 559–562 (2001).
Freeman, M. & Gurdon, J. B. Regulatory principles of developmental signaling. Annu. Rev. Cell Dev. Biol. 18, 515–539 (2002).
Lander, A. D. Morpheus unbound: reimagining the morphogen gradient. Cell 128, 245–256 (2007).
Ferguson, E. L. & Anderson, K. V. Decapentaplegic acts as a morphogen to organize dorsal–ventral pattern in the Drosophila embryo. Cell 71, 451–461 (1992).
Nellen, D., Burke, R., Struhl, G. & Basler, K. Direct and long-range action of a DPP morphogen gradient. Cell 85, 357–368 (1996).
Arora, K., Levine, M. S. & O'Connor, M. B. The screw gene encodes a ubiquitously expressed member of the TGF-β family required for specification of dorsal cell fates in the Drosophila embryo. Genes Dev. 8, 2588–2601 (1994).
Neul, J. L. & Ferguson, E. L. Spatially restricted activation of the SAX receptor by SCW modulates DPP/TKV signaling in Drosophila dorsal–ventral patterning. Cell 95, 483–494 (1998).
Sutherland, D. J., Li, M., Liu, X. Q., Stefancsik, R. & Raftery, L. A. Stepwise formation of a SMAD activity gradient during dorsal–ventral patterning of the Drosophila embryo. Development 130, 5705–5716 (2003).
Wang, Y. C. & Ferguson, E. L. Spatial bistability of Dpp-receptor interactions during Drosophila dorsal–ventral patterning. Nature 434, 229–234 (2005).
Blader, P., Rastegar, S., Fischer, N. & Strahle, U. Cleavage of the BMP-4 antagonist chordin by zebrafish tolloid. Science 278, 1937–1940 (1997).
Marques, G. et al. Production of a DPP activity gradient in the early Drosophila embryo through the opposing actions of the SOG and TLD proteins. Cell 91, 417–426 (1997).
Mullins, M. C. Holy Tolloido: Tolloid cleaves SOG/Chordin to free DPP/BMPs. Trends Genet. 14, 127–129 (1998).
Xu, L., Alarcon, C., Col, S. & Massagué, J. Distinct domain utilization by SMAD3 and SMAD4 for nucleoporin interaction and nuclear import. J. Biol. Chem. 278, 42569–42577 (2003).
Kurisaki, A., Kose, S., Yoneda, Y., Heldin, C. H. & Moustakas, A. Transforming growth factor-β induces nuclear import of SMAD3 in an importin-β1 and Ran-dependent manner. Mol. Biol. Cell 12, 1079–1091 (2001).
Xiao, Z., Liu, X. & Lodish, H. F. Importin β mediates nuclear translocation of SMAD 3. J. Biol. Chem. 275, 23425–23428 (2000).
Xiao, Z., Watson, N., Rodriguez, C. & Lodish, H. F. Nucleocytoplasmic shuttling of SMAD1 conferred by its nuclear localization and nuclear export signals. J. Biol. Chem. 276, 39404–39410 (2001).
Xiao, Z., Latek, R. & Lodish, H. F. An extended bipartite nuclear localization signal in SMAD4 is required for its nuclear import and transcriptional activity. Oncogene 22, 1057–1069 (2003).
Xu, L. et al. Msk is required for nuclear import of TGF-β/BMP-activated SMADs. J. Cell Biol. 178, 981–994 (2007).
Watanabe, M., Masuyama, N., Fukuda, M. & Nishida, E. Regulation of intracellular dynamics of SMAD4 by its leucine-rich nuclear export signal. EMBO Rep. 1, 176–182 (2000).
Masuyama, N., Hanafusa, H., Kusakabe, M., Shibuya, H. & Nishida, E. Identification of two SMAD4 proteins in Xenopus. Their common and distinct properties. J. Biol. Chem. 274, 12163–12170 (1999).
Kurisaki, A. et al. The mechanism of nuclear export of SMAD3 involves exportin 4 and Ran. Mol. Cell. Biol. 26, 1318–1332 (2006).
Ashe, H. L. & Briscoe, J. The interpretation of morphogen gradients. Development 133, 385–394 (2006).
Randall, R. A. et al. Recognition of phosphorylated-SMAD2-containing complexes by a novel SMAD interaction motif. Mol. Cell. Biol. 24, 1106–1121 (2004).
Wharton, S. J., Basu, S. P. & Ashe, H. L. SMAD affinity can direct distinct readouts of the embryonic extracellular Dpp gradient in Drosophila. Curr. Biol. 14, 1550–1558 (2004).
Saka, Y. & Smith, J. C. A mechanism for the sharp transition of morphogen gradient interpretation in Xenopus. BMC Dev. Biol. 7, 47 (2007).
Latinkic, B. V. et al. The Xenopus Brachyury promoter is activated by FGF and low concentrations of activin and suppressed by high concentrations of activin and by paired-type homeodomain proteins. Genes Dev. 11, 3265–3276 (1997).
Green, J. B., New, H. V. & Smith, J. C. Responses of embryonic Xenopus cells to activin and FGF are separated by multiple dose thresholds and correspond to distinct axes of the mesoderm. Cell 71, 731–739 (1992).
Ladher, R., Mohun, T. J., Smith, J. C. & Snape, A. M. Xom: a Xenopus homeobox gene that mediates the early effects of BMP-4. Development 122, 2385–2394 (1996).
Trindade, M., Tada, M. & Smith, J. C. DNA-binding specificity and embryological function of Xom (Xvent-2). Dev. Biol. 216, 442–456 (1999).
Messenger, N. J. et al. Functional specificity of the Xenopus T-domain protein Brachyury is conferred by its ability to interact with SMAD1. Dev. Cell 8, 599–610 (2005).
Artinger, M., Blitz, I., Inoue, K., Tran, U. & Cho, K. W. Interaction of goosecoid and brachyury in Xenopus mesoderm patterning. Mech. Dev. 65, 187–196 (1997).
Arora, K. et al. The Drosophila schnurri gene acts in the Dpp/TGF β signaling pathway and encodes a transcription factor homologous to the human MBP family. Cell 81, 781–790 (1995).
Grieder, N. C., Nellen, D., Burke, R., Basler, K. & Affolter, M. Schnurri is required for Drosophila Dpp signaling and encodes a zinc finger protein similar to the mammalian transcription factor PRDII-BF1. Cell 81, 791–800 (1995).
Muller, B., Hartmann, B., Pyrowolakis, G., Affolter, M. & Basler, K. Conversion of an extracellular Dpp/BMP morphogen gradient into an inverse transcriptional gradient. Cell 113, 221–233 (2003). Establishes the concept of a repressive Brinker gradient that is reciprocal to a DPP gradient.
Affolter, M., Marty, T., Vigano, M. A. & Jazwinska, A. Nuclear interpretation of Dpp signaling in Drosophila. EMBO J. 20, 3298–3305 (2001).
Barrio, R. & de Celis, J. F. Regulation of spalt expression in the Drosophila wing blade in response to the Decapentaplegic signaling pathway. Proc. Natl Acad. Sci. USA 101, 6021–6026 (2004).
Chenna, R. et al. Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res. 31, 3497–3500 (2003).
Acknowledgements
We would like to thank M. Howell, L. Raftery, E. Sahai, S. Leevers and members of the Hill laboratory for comments on the manuscript. Work in the Hill laboratory is funded by Cancer Research UK and the European Union (EU). B.S. was supported by an Erwin-Schrödinger Fellowship of the Austrian Science Foundation and an EU Marie Curie Fellowship.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Hereditary haemorrhagic telangiectasia
-
An autosomal dominant disorder that is characterized by multisystemic vascular dysplasia and recurrent haemorrhage, caused by mutations in the endothelium-specific type I receptor ALK1 or the co-receptor endoglin.
- Morphogen
-
A ligand that is secreted by cells and, by spreading to other cells, determines distinct cell fates in a dose-dependent manner.
- Activin
-
One of a class of actively signalling homo- or heterodimers that consist of two of several β-chains (βA, βB, βC or βE in mammals).
- Subtilisin-like proprotein convertase (SPC) family
-
A major family of endoproteolytic processing enzymes of the secretory pathway.
- MH1 and MH2 domains
-
Conserved N-terminal and C-terminal domains, respectively, that are found in SMADs. MH stands for MAD homology in reference to the founder member of the SMAD family, Drosophila mothers against decapentaplegic (MAD).
- FYVE domain
-
Named after the four first proteins that were found to contain this domain (Fab1, YOTB, Vac1 and EEA1). FYVE domains bind phosphatidylinositol-3-phosphate and target proteins to endosomal membranes.
- Schnurri
-
A zinc-finger transcription factor that mediates DPP-induced repression together with MAD–Medea in D. melanogaster, but acts as an activator in response to BMPs in mammals.
- EEA1-positive endosome
-
A vesicular body that is highly enriched in phosphatidylinositol-3-phosphate, which binds the FYVE-domain protein EEA1 (early endosomal antigen-1).
- Karyopherin
-
One of a group of structurally unrelated proteins that are divided into two functional subgroups: importins bind a cargo in the cytoplasm, transport it through the nuclear pore and release it in the nucleus on binding of the small GTPase Ran in its GTP-bound form. Exportins bind a cargo in the nucleus together with Ran-GTP, transport it into the cytoplasm and release it upon GTP hydrolysis.
- Nuclear localization signal
-
(NLS). A short basic stretch of amino acids, rich in Arg or Lys, that is recognized by importin-α–importin-β complexes.
- Exportin-1
-
The prototypic nuclear export receptor, transporting a plethora of substrates. Specifically inhibited by the drug leptomycin B.
- Nuclear export signal
-
(NES). A short, Leu- or Ile-rich motif recognized by exportin-1.
- BMP1 family
-
A small group of extracellular metalloproteases that process precursors to form mature functional proteins, which are necessary for extracellular matrix formation, such as procollagens. Members of this family are crucially involved in signalling because they cleave the BMP2–BMP4 inhibitor chordin and the latent precursors of TGFβ, GDF8 and GDF11.
Rights and permissions
About this article
Cite this article
Schmierer, B., Hill, C. TGFβ–SMAD signal transduction: molecular specificity and functional flexibility. Nat Rev Mol Cell Biol 8, 970–982 (2007). https://doi.org/10.1038/nrm2297
Issue Date:
DOI: https://doi.org/10.1038/nrm2297
This article is cited by
-
Mowat-Wilson syndrome: unraveling the complexities of diagnosis, treatment, and symptom management
Egyptian Journal of Medical Human Genetics (2024)
-
Unveiling the molecular Hallmarks of Peyronie’s disease: a comprehensive narrative review
International Journal of Impotence Research (2024)
-
Ketosis prevents abdominal aortic aneurysm rupture through C–C chemokine receptor type 2 downregulation and enhanced extracellular matrix balance
Scientific Reports (2024)
-
Recent advances and applications of CRISPR-Cas9 in cancer immunotherapy
Molecular Cancer (2023)
-
Inhibition of TGF-β signaling enables long-term proliferation of mouse primary epithelial stem/progenitor cells of the tympanic membrane and the middle ear mucosa
Scientific Reports (2023)