Abstract
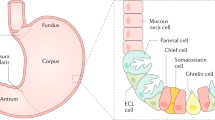
Autoimmune gastritis is a chronic progressive inflammatory condition that results in the replacement of the parietal cell mass by atrophic and metaplastic mucosa. A complex interaction of autoantibodies against the parietal cell proton pump and sensitized T cells progressively destroy the parietal cells, inducing hypochlorhydria and then achlorhydria, while autoantibodies against the intrinsic factor impair the absorption of vitamin B12. The resulting cobalamin deficiency manifests with megaloblastic anaemia and neurological and systemic signs and symptoms collectively known as pernicious anaemia. Previously believed to be predominantly a disease of elderly women of Northern European ancestry, autoimmune gastritis has now been recognized in all populations and ethnic groups, but because of the complexity of the diagnosis no reliable prevalence data are available. For similar reasons, as well as the frequent and often unknown overlap with Helicobacter pylori infection, the risk of gastric cancer has not been adequately assessed in these patients. This Review summarizes the epidemiology, pathogenesis and pathological aspects of autoimmune metaplastic atrophic gastritis. We also provide practical advice for the diagnosis and management of patients with this disease.
Key Points
-
Atrophic gastritis can be associated with long-standing Helicobacter pylori infection (multifocal atrophic gastritis) and with an autoimmune process that progressively destroys the oxyntic mucosa (autoimmune atrophic gastritis)
-
Both types of atrophic gastritis are underdiagnosed, in part because of inadequate biopsy sampling
-
Autoimmune atrophic gastritis progresses from a mild chronic inflammation of the gastric corpus to an advanced stage associated with a severe form of vitamin B12 deficiency anaemia known as pernicious anaemia
-
Traditionally, autoimmune atrophic gastritis has been viewed as a disease affecting predominantly elderly women of Northern European descent, but growing evidence suggests that there might be no racial specificity
-
The diagnosis of autoimmune gastritis rests on the demonstration of its characteristic histopathological features and the demonstration of autoantibodies against intrinsic factor and parietal cells
-
Management of the early stages of autoimmune atrophic gastritis is focused on the prevention of vitamin B12, folate and iron deficiencies
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Rugge, M. et al. Gastric mucosal atrophy: interobserver consistency using new criteria for classification and grading. Aliment. Pharmacol. Ther. 16, 1249–1259 (2002).
Coombs, C. F. A Note on the Cardiac Symptoms of Pernicious Anaemia. Br. Med. J. 2, 185 (1926).
Addison, T. Disease: Chronic Suprarenal Insufficiency, Usually due to Tuberculosis of Suprarenal Capsule. London Medical Gazette 43, 517–518 (1849).
Flint, A. A clinical lecture on anemia. American Medical Times 1, 181–186 (1860).
Biermer, M. A. Über eine eigentümliche Form von progressiver, perniciöser Anaemie [German]. Correspondenz-Blatt für Schweizer Aerzte 2, 15–17 (1872).
Mapel, D., Roberts, M., Overhiser, A. & Mason, A. The epidemiology, diagnosis, and cost of dyspepsia and Helicobacter pylori gastritis: a case–control analysis in the Southwestern United States. Helicobacter 18, 54–65 (2013).
Goh, K. L., Chan, W. K., Shiota, S. & Yamaoka, Y. Epidemiology of Helicobacter pylori infection and public health implications. Helicobacter 16 (Suppl. 1), 1–9 (2011).
Tan, H. J. & Goh, K. L. Changing epidemiology of Helicobacter pylori in Asia. J. Dig. Dis. 9, 186–189 (2008).
Bruce, M. G. & Maaroos, H. I. Epidemiology of Helicobacter pylori infection. Helicobacter 13 (Suppl. 1), 1–6 (2008).
Malaty, H. M. Epidemiology of Helicobacter pylori infection. Best Pract. Res. Clin. Gastroenterol. 21, 205–214 (2007).
Graham, D. Y. The changing epidemiology of GERD: geography and Helicobacter pylori. Am. J. Gastroenterol. 98, 1462–1470 (2003).
Brown, L. M. Helicobacter pylori: epidemiology and routes of transmission. Epidemiol. Rev. 22, 283–297 (2000).
Rook, G. A. & Brunet, L. R. Microbes, immunoregulation, and the gut. Gut 54, 317–320 (2005).
Straumann, A. Eosinophilic esophagitis: rapidly emerging disorder. Swiss Med. Wkly 142, w13513 (2012).
Arnold, I. C. et al. Helicobacter pylori infection prevents allergic asthma in mouse models through the induction of regulatory T cells. J. Clin. Invest. 121, 3088–3093 (2011).
Chen, Y. & Blaser, M. J. Inverse associations of Helicobacter pylori with asthma and allergy. Arch. Intern. Med. 167, 821–827 (2007).
Rugge, M. et al. Autoimmune gastritis: histology phenotype and OLGA staging. Aliment. Pharmacol. Ther. 35, 1460–1466 (2012).
Levine, S. & Ladd, W. S. Pernicious anemia: a clinical study of one-hundred and fifty consecutive cases with special reference to gastric anacidity. Johns Hopkins Hosp. Bull. 32, 254–266 (1921).
Friedlander, R. D. The racial factor in pernicious anemia: a study of five hundred cases. Am. J. Med. Sci. 187, 634–642 (1934).
Park, J. Y., Cornish, T. C., Lam-Himlin, D., Shi, C. & Montgomery, E. Gastric lesions in patients with autoimmune metaplastic atrophic gastritis (AMAG) in a tertiary care setting. Am. J. Surg. Pathol. 34, 1591–1598 (2010).
Bordi, C. et al. Endocrine cell growths in atrophic body gastritis. Critical evaluation of a histological classification. J. Pathol. 182, 339–346 (1997).
Hershko, C. et al. Variable hematologic presentation of autoimmune gastritis: age-related progression from iron deficiency to cobalamin depletion. Blood 107, 1673–1679 (2006).
Lahner, E. et al. Occurrence and risk factors for autoimmune thyroid disease in patients with atrophic body gastritis. Am. J. Med. 121, 136–141 (2008).
Carmel, R. Prevalence of undiagnosed pernicious anemia in the elderly. Arch. Intern. Med. 156, 1097–1100 (1996).
Eidt, S., Oberhuber, G., Schneider, A. & Stolte, M. The histopathological spectrum of type A gastritis. Pathol. Res. Pract. 192, 101–106 (1996).
Carmel, R. & Johnson, C. S. Racial patterns in pernicious anemia. Early age at onset and increased frequency of intrinsic-factor antibody in black women. N. Engl. J. Med. 298, 647–650 (1978).
Carmel, R., Johnson, C. S. & Weiner, J. M. Pernicious anemia in Latin Americans is not a disease of the elderly. Arch. Intern. Med. 147, 1995–1996 (1987).
Andres, E. et al. Clinical aspects of cobalamin deficiency in elderly patients. Epidemiology, causes, clinical manifestations, and treatment with special focus on oral cobalamin therapy. Eur. J. Intern. Med. 18, 456–462 (2007).
Andres, E. et al. Vitamin B12 (cobalamin) deficiency in elderly patients. CMAJ 171, 251–259 (2004).
Chan, J. C. et al. Longitudinal study of Chinese patients with pernicious anaemia. Postgrad. Med. J. 84, 644–650 (2008).
Chuke, P. O., Traub, N. E. & Patel, I. C. Pernicious anaemia in a Zambian. East Afr. Med. J. 49, 681–683 (1972).
Kocak, R. & Paydas, S. Pernicious anemia in Turkey. Int. J. Hematol. 55, 117–119 (1992).
Maktouf, C. et al. Megaloblastic anemia in North Africa. Haematologica 91, 990–991 (2006).
Schlemper, R. J., van der Werf, S. D., Vandenbroucke, J. P., Biemond, I. & Lamers, C. B. Seroepidemiology of gastritis in Japanese and Dutch working populations: evidence for the development of atrophic gastritis that is not related to Helicobacter pylori. Gut 37, 199–204 (1995).
Weck, M. N. & Brenner, H. Prevalence of chronic atrophic gastritis in different parts of the world. Cancer Epidemiol. Biomarkers Prev. 15, 1083–1094 (2006).
Wun Chan, J. C. et al. Pernicious anemia in Chinese: a study of 181 patients in a Hong Kong hospital. Medicine (Baltimore) 85, 129–138 (2006).
Claeys, D., Faller, G., Appelmelk, B. J., Negrini, R. & Kirchner, T. The gastric H+,K+-ATPase is a major autoantigen in chronic Helicobacter pylori gastritis with body mucosa atrophy. Gastroenterology 115, 340–347 (1998).
Negrini, R. et al. Helicobacter pylori infection induces antibodies cross-reacting with human gastric mucosa. Gastroenterology 101, 437–445 (1991).
Negrini, R. et al. Antigenic mimicry between Helicobacter pylori and gastric mucosa in the pathogenesis of body atrophic gastritis. Gastroenterology 111, 655–665 (1996).
Faller, G. et al. Antigastric autoantibodies in Helicobacter pylori gastritis: prevalence, in-situ binding sites and clues for clinical relevance. Virchows Arch. 427, 483–486 (1996).
Faller, G. et al. Antigastric autoantibodies in Helicobacter pylori infection: implications of histological and clinical parameters of gastritis. Gut 41, 619–623 (1997).
Appelmelk, B. J., Faller, G., Claeys, D., Kirchner, T. & Vandenbroucke-Grauls, C. M. Bugs on trial: the case of Helicobacter pylori and autoimmunity. Immunol. Today 19, 296–299 (1998).
Bergman, M. P. et al. The story so far: Helicobacter pylori and gastric autoimmunity. Int. Rev. Immunol. 24, 63–91 (2005).
D'Elios, M. M., Appelmelk, B. J., Amedei, A., Bergman, M. P. & Del, P. G. Gastric autoimmunity: the role of Helicobacter pylori and molecular mimicry. Trends Mol. Med. 10, 316–323 (2004).
D'Elios, M. M., Bergman, M. P., Amedei, A., Appelmelk, B. J. & Del, P. G. Helicobacter pylori and gastric autoimmunity. Microbes Infect. 6, 1395–1401 (2004).
Agreus, L. et al. Rationale in diagnosis and screening of atrophic gastritis with stomach-specific plasma biomarkers. Scand. J. Gastroenterol. 47, 136–147 (2012).
Stolte, M. et al. Active autoimmune gastritis without total atrophy of the glands. Z. Gastroenterol. 30, 729–735 (1992).
Stolte, M., Meier, E. & Meining, A. Cure of autoimmune gastritis by Helicobacter pylori eradication in a 21-year-old male. Z. Gastroenterol. 36, 641–643 (1998).
Muller, H., Rappel, S., Wundisch, T., Bayerdorffer, E. & Stolte, M. Healing of active, non-atrophic autoimmune gastritis by, H. pylori eradication. Digestion 64, 30–39 (2001).
Bergman, M. P. & D'Elios, M. M. Cytotoxic T cells in, H. pylori-related gastric autoimmunity and gastric lymphoma. J. Biomed. Biotechnol. 2010, 104918 (2010).
Negrini, R., Savio, A. & Appelmelk, B. J. Autoantibodies to gastric mucosa in Helicobacter pylori infection. Helicobacter 2 (Suppl. 1), S13–S16 (1997).
D'Elios, M. M. et al. Molecular specificity and functional properties of autoreactive T-cell response in human gastric autoimmunity. Int. Rev. Immunol. 24, 111–122 (2005).
Bergman, M., Del, P. G., van Kooyk, Y. & Appelmelk, B. Helicobacter pylori phase variation, immune modulation and gastric autoimmunity. Nat. Rev. Microbiol. 4, 151–159 (2006).
Oksanen, A. et al. Atrophic gastritis and Helicobacter pylori infection in outpatients referred for gastroscopy. Gut 46, 460–463 (2000).
Veijola, L. I., Oksanen, A. M., Sipponen, P. I. & Rautelin, H. I. Association of autoimmune type atrophic corpus gastritis with Helicobacter pylori infection. World J. Gastroenterol. 16, 83–88 (2010).
Bedeir, A. S., Lash, R. H., Lash, J. G. & Ray, M. B. Significant increase in IgG4+ plasma cells in gastric biopsy specimens from patients with pernicious anaemia. J. Clin. Pathol. 63, 999–1001 (2010).
Gershwin, M. E. & Shoenfeld, Y. Cutting-edge issues in organ-specific autoimmunity. Clin. Rev. Allergy Immunol. 41, 123–125 (2011).
Banka, S., Ryan, K., Thomson, W. & Newman, W. G. Pernicious anemia—genetic insights. Autoimmun. Rev. 10, 455–459 (2011).
Ohana, M. et al. Inhibitory effects of Helicobacter pylori infection on murine autoimmune gastritis. Gut 52, 1102–1110 (2003).
Cruchaud, A. & Juditz, E. An analysis of gastric parietal cell antibodies and thyroid cell antibodies in patients with pernicious anaemia and thyroid disorders. Clin. Exp. Immunol. 3, 771–781 (1968).
Doniach, D., Roitt, I. M. & Taylor, K. B. Autoimmune phenomena in pernicious anaemia. Serological overlap with thyroiditis, thyrotoxicosis, and systemic lupus erythematosus. Br. Med. J. 1, 1374–1379 (1963).
Doniach, D., Grant, D. & Newns, G. H. Multi-endocrine autoimmune disorder in mother and son. Proc. R. Soc. Med. 65, 488–490 (1972).
Irvine, W. J., Sumerling, M. D. & Davies, S. H. Immunological aspects of pernicious anaemia. Bibl. Haematol. 23, 73–78 (1965).
Irvine, W. J., Cullen, D. R. & Mawhinney, H. Natural history of autoimmune achlorhydric atrophic gastritis. A 1–15-year follow-up study. Lancet 2, 482–485 (1974).
Singer, W. & Sahay, B. M. Myasthenia Gravis, Hashimoto's Thyroiditis, and Pernicious Anaemia. Br. Med. J. 1, 904 (1966).
Markson, J. L. & Moore, J. M. Thyroid auto-antibodies in pernicious anaemia. Br. Med. J. 2, 1352–1355 (1962).
Feldt-Rasmussen, U. et al. Autoantibodies, immune complexes and HLA-D in thyrogastric autoimmunity. Tissue Antigens 22, 342–347 (1983).
Ottesen, M., Feldt-Rasmussen, U., Andersen, J., Hippe, E. & Schouboe, A. Thyroid function and autoimmunity in pernicious anemia before and during cyanocobalamin treatment. J. Endocrinol. Invest. 18, 91–97 (1995).
Chanarin, I. Pernicious anaemia as an autoimmune disease. Br. J. Haematol. 23 (Suppl.), 101–107 (1972).
Perros, P., Singh, R. K., Ludlam, C. A. & Frier, B. M. Prevalence of pernicious anaemia in patients with Type 1 diabetes mellitus and autoimmune thyroid disease. Diabet. Med. 17, 749–751 (2000).
Giannouli, S., Anagnostou, D., Soliotis, F. & Voulgarelis, M. Autoimmune manifestations in common variable immunodeficiency. Clin. Rheumatol. 23, 449–452 (2004).
Hashizume, T. Good's syndrome and pernicious anemia. Intern. Med. 41, 1062–1064 (2002).
Koskinen, S. Long-term follow-up of health in blood donors with primary selective IgA deficiency. J. Clin. Immunol. 16, 165–170 (1996).
Lackmann, G. M., Wahn, V., Poremba, C. & Niehues, T. A teenager with X-linked agammaglobulinemia and vitamin B12 deficiency anemia. J. Pediatr. Gastroenterol. Nutr. 41, 360–362 (2005).
Callender, S. T. & Denborough, M. A. A family study of pernicious anaemia. Br. J. Haematol. 3, 88–106 (1957).
Toh, B. H., van Driel, I. R. & Gleeson, P. A. Pernicious anemia. N. Engl. J. Med. 337, 1441–1448 (1997).
Delva, P. L., MacDonell, J. E. & Macintosh, O. C. Megaloblastic anemia occurring simultaneously in white female monozygotic twins. Can. Med. Assoc. J. 92, 1129–1131 (1965).
Masnou, H. et al. Pernicious anaemia in triplets. A case report and literature review. Gastroenterol. Hepatol. 30, 580–582 (2007).
Correa, P., Piazuelo, M. B. & Wilson, K. T. Pathology of gastric intestinal metaplasia: clinical implications. Am. J. Gastroenterol. 105, 493–498 (2010).
Goldenring, J. R., Nam, K. T. & Mills, J. C. The origin of pre-neoplastic metaplasia in the stomach: chief cells emerge from the Mist. Exp. Cell Res. 317, 2759–2764 (2011).
Jhala, N. C. et al. Pancreatic acinar cell metaplasia in autoimmune gastritis. Arch. Pathol. Lab. Med. 127, 854–857 (2003).
Bettington, M. & Brown, I. Autoimmune gastritis: novel clues to histological diagnosis. Pathology 45, 145–149 (2013).
Solcia, E., Rindi, G., Silini, E. & Villani, L. Enterochromaffin-like (ECL) cells and their growths: relationships to gastrin, reduced acid secretion and gastritis. Baillieres Clin. Gastroenterol. 7, 149–165 (1993).
Solcia, E. et al. Morphology and pathogenesis of endocrine hyperplasias, precarcinoid lesions, and carcinoids arising in chronic atrophic gastritis. Scand. J. Gastroenterol. Suppl. 180, 146–159 (1991).
Solcia, E. et al. Distinct patterns of chronic gastritis associated with carcinoid and cancer and their role in tumorigenesis. Yale J. Biol. Med. 65, 793–804 (1992).
Torbenson, M., Abraham, S. C., Boitnott, J., Yardley, J. H. & Wu, T. T. Autoimmune gastritis: distinct histological and immunohistochemical findings before complete loss of oxyntic glands. Mod. Pathol. 15, 102–109 (2002).
Junca J., de Soria, P. L., Granada, M. L., Flores, A. & Marquez, E. Detection of early abnormalities in gastric function in first-degree relatives of patients with pernicious anemia. Eur. J. Haematol. 77, 518–522 (2006).
Itsuno, M. et al. Multiple carcinoids and endocrine cell micronests in type A gastritis. Their morphology, histogenesis, and natural history. Cancer 63, 881–890 (1989).
Muller, J., Kirchner, T. & Muller-Hermelink, H. K. Gastric endocrine cell hyperplasia and carcinoid tumors in atrophic gastritis type A. Am. J. Surg. Pathol. 11, 909–917 (1987).
Rugge, M. et al. OLGA staging for gastritis: a tutorial. Dig. Liver Dis. 40, 650–658 (2008).
Rugge, M. & Genta, R. M. Staging and grading of chronic gastritis. Hum. Pathol. 36, 228–233 (2005).
Rugge, M, et al. Gastritis OLGA-staging and gastric cancer risk: a twelve-year clinico-pathological follow-up study. Aliment. Pharmacol. Ther. 31, 1104–1111 (2010).
Satoh, K. et al. Assessment of atrophic gastritis using the OLGA system. Helicobacter 13, 225–229 (2008).
Jevremovic, D., Torbenson, M., Murray, J. A., Burgart, L. J. & Abraham, S. C. Atrophic autoimmune pangastritis: A distinctive form of antral and fundic gastritis associated with systemic autoimmune disease. Am. J. Surg. Pathol. 30, 1412–1419 (2006).
Solcia, E. et al. Natural history, clinicopathologic classification and prognosis of gastric ECL cell tumors. Yale J. Biol. Med. 71, 285–290 (1998).
Cockburn, A. N., Morgan, C. J. & Genta, R. M. Neuroendocrine proliferations of the stomach: a pragmatic approach for the perplexed pathologist. Adv. Anat. Pathol. 20, 148–157 (2013).
Solcia, E. et al. in WHO Classification of Tumours of the Digestive System 2nd edn (eds Bosman, F. T., Carneiro, F., Hruban, R. H. & Theise, N. D.) 64–68 (IARC, 2010).
Carmack, S. W., Genta, R. M., Graham, D. Y. & Lauwers, G. Y. Management of gastric polyps: a pathology-based guide for gastroenterologists. Nat. Rev. Gastroenterol. Hepatol. 6, 331–341 (2009).
Shaib, Y. H., Rugge, M., Graham, D. Y. & Genta, R. M. Management of gastric polyps: an endoscopy based approach. Clin. Gastroenterol. Hepatol. http://dx.doi.org/10.1016/j.cgh.2013.03.019.
Abraham, S. C., Singh, V. K., Yardley, J. H. & Wu, T. T. Hyperplastic polyps of the stomach: associations with histologic patterns of gastritis and gastric atrophy. Am. J. Surg. Pathol. 25, 500–507 (2001).
Haruma, K., Sumii, K., Yoshihara, M., Watanabe, C. & Kajiyama, G. Gastric mucosa in female patients with fundic glandular polyps. J. Clin. Gastroenterol. 13, 565–569 (1991).
Haruma, K. et al. Pernicious anemia and Helicobacter pylori infection in Japan: evaluationin a country with a high prevalence of infection. Am. J. Gastroenterol. 90, 1107–1110 (1995).
Laxen, F. Gastric carcinoma and pernicious anaemia in long-term endoscopic follow-up of subjects with gastric polyps. Scand. J. Gastroenterol. 19, 535–540 (1984).
Laxen, F., Kekki, M., Sipponen, P. & Siurala, M. The gastric mucosa in stomachs with polyps: morphologic and dynamic evaluation. Scand. J. Gastroenterol. 18, 503–511 (1983).
Varis, K., Stenman, U. H., Lehtola, J. & Siurala, M. Gastric lesion and pernicious anemia: a family study. Acta Hepatogastroenterol. (Stuttg.) 25, 62–67 (1978).
Krasinskas, A. M., Abraham, S. C., Metz, D. C. & Furth, E. E. Oxyntic mucosa pseudopolyps: a presentation of atrophic autoimmune gastritis. Am. J. Surg. Pathol. 27, 236–241 (2003).
Vieth, M., Kushima, R., Borchard, F. & Stolte, M. Pyloric gland adenoma: a clinico-pathological analysis of 90 cases. Virchows Arch. 442, 317–321 (2003).
Chen, Z. M., Scudiere, J. R., Abraham, S. C. & Montgomery, E. Pyloric gland adenoma: an entity distinct from gastric foveolar type adenoma. Am. J. Surg. Pathol. 33, 186–193 (2009).
Abraham, S. C., Montgomery, E. A., Singh, V. K., Yardley, J. H. & Wu, T. T. Gastric adenomas: intestinal-type and gastric-type adenomas differ in the risk of adenocarcinoma and presence of background mucosal pathology. Am. J. Surg. Pathol. 26, 1276–1285 (2002).
Matsubara, A. et al. Frequent GNAS and KRAS mutations in pyloric gland adenoma of the stomach and duodenum. J. Pathol. 229, 579–587 (2013).
Strickland, R. G. & Mackay, I. R. A reappraisal of the nature and significance of chronic atrophic gastritis. Am. J. Dig. Dis. 18, 426–440 (1973).
Vannella, L. et al. Risk factors for progression to gastric neoplastic lesions in patients with atrophic gastritis. Aliment. Pharmacol. Ther. 31, 1042–1050 (2010).
Vannella, L., Lahner, E. & Annibale, B. Risk for gastric neoplasias in patients with chronic atrophic gastritis: a critical reappraisal. World J. Gastroenterol. 18, 1279–1285 (2012).
Vannella, L., Lahner, E., Osborn, J. & Annibale, B. Systematic review: gastric cancer incidence in pernicious anaemia. Aliment. Pharmacol. Ther. 37, 375–382 (2012).
Borch, K. Epidemiologic, clinicopathologic, and economic aspects of gastroscopic screening of patients with pernicious anemia. Scand. J. Gastroenterol. 21, 21–30 (1986).
Bresky, G. et al. Endoscopic findings in a biennial follow-up program in patients with pernicious anemia. Hepatogastroenterology 50, 2264–2266 (2003).
Elsborg, L., Andersen, D. & Bastrup-Madsen, P. Gastrocamera screening in pernicious anaemia. With special reference to the occurrence of gastric polyps and cancer. Scand. J. Gastroenterol. 8, 5–8 (1973).
Kokkola, A. et al. The risk of gastric carcinoma and carcinoid tumours in patients with pernicious anaemia. A prospective follow-up study. Scand. J. Gastroenterol. 33, 88–92 (1998).
Sjoblom, S. M., Sipponen, P. & Jarvinen, H. Gastroscopic follow up of pernicious anaemia patients. Gut 34, 28–32 (1993).
Annibale, B. et al. Two-thirds of atrophic body gastritis patients have evidence of Helicobacter pylori infection. Helicobacter 6, 225–233 (2001).
Lahner, E. et al. First endoscopic-histologic follow-up in patients with body-predominant atrophic gastritis: when should it be done? Gastrointest. Endosc. 53, 443–448 (2001).
Ruiz, B. et al. Morphometric assessment of gastric antral atrophy: comparison with visual evaluation. Histopathology 39, 235–242 (2001).
Genta, R. M. Review article: Gastric atrophy and atrophic gastritis—nebulous concepts in search of a definition. Aliment. Pharmacol. Ther. 12 (Suppl. 1), 17–23 (1998).
Dixon, M. F., Genta, R. M., Yardley, J. H. & Correa, P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am. J. Surg. Pathol. 20, 1161–1181 (1996).
Vannella, L. et al. Development of type I gastric carcinoid in patients with chronic atrophic gastritis. Aliment. Pharmacol. Ther. 33, 1361–1369 (2011).
Annibale, B. et al. Atrophic body gastritis patients with enterochromaffin-like cell dysplasia are at increased risk for the development of type I gastric carcinoid. Eur. J. Gastroenterol. Hepatol. 13, 1449–1456 (2001).
El-Zimaity, H. M., Ota, H., Graham, D. Y., Akamatsu, T. & Katsuyama, T. Patterns of gastric atrophy in intestinal type gastric carcinoma. Cancer 94, 1428–1436 (2002).
Genta, R. M. & Pusztaszeri, M. The gastric mucosa in gastric cancer patients in a low-incidence area. Eur. J. Gastroenterol. Hepatol. 18, 1085–1093 (2006).
Stabler, S. P. & Allen, R. H. Vitamin B12 deficiency as a worldwide problem. Annu. Rev. Nutr. 24, 299–326 (2004).
Annibale, B., Lahner, E. & Fave, G. D. Diagnosis and management of pernicious anemia. Curr. Gastroenterol. Rep. 13, 518–524 (2011).
Stabler, S. P. Clinical practice. Vitamin B12 deficiency. N. Engl. J. Med. 368, 149–160 (2013).
Zuckier, L. S. & Chervu, L. R. Schilling evaluation of pernicious anemia: current status. J. Nucl. Med. 25, 1032–1039 (1984).
Krynyckyi, B. R. & Zuckier, L. S. Accuracy of measurement of dual-isotope Schilling test urine samples: a multicenter study [corrected]. J. Nucl. Med. 36, 1659–1665 (1995).
Di Mario, F. et al. 'Serological biopsy' in first-degree relatives of patients with gastric cancer affected by Helicobacter pylori infection. Scand. J. Gastroenterol. 38, 1223–1227 (2003).
Di Mario, F. et al. Usefulness of serum pepsinogens in Helicobacter pylori chronic gastritis: relationship with inflammation, activity, and density of the bacterium. Dig. Dis. Sci. 51, 1791–1795 (2006).
Germana, B. et al. Clinical usefulness of serum pepsinogens I and II, gastrin-17 and anti-Helicobacter pylori antibodies in the management of dyspeptic patients in primary care. Dig. Liver Dis. 37, 501–508 (2005).
Guariso, G. et al. GastroPanel: evaluation of the usefulness in the diagnosis of gastro-duodenal mucosal alterations in children. Clin. Chim. Acta 402, 54–60 (2009).
Iijima, K. et al. Serum biomarker tests are useful in delineating between patients with gastric atrophy and normal, healthy stomach. World J. Gastroenterol. 15, 853–859 (2009).
Peitz, U. et al. Correlation of serum pepsinogens and gastrin-17 with atrophic gastritis in gastroesophageal reflux patients: a matched-pairs study. J. Gastroenterol. Hepatol. 26, 82–89 (2011).
Antico, A. et al. Clinical usefulness of the serological gastric biopsy for the diagnosis of chronic autoimmune gastritis. Clin. Dev. Immunol. 2012, 520970 (2012).
Faber, K. K. Achylia Gastrica mit Anamie [German]. Medizinische Klinik 5, 1310–1325 (1909).
Hershko, C. et al. Role of autoimmune gastritis, Helicobacter pylori and celiac disease in refractory or unexplained iron deficiency anemia. Haematologica 90, 585–595 (2005).
Genta, R. M. One more dysplasia. Eur. J. Gastroenterol. Hepatol. 13, 1411–1414 (2001).
Alonso, N. et al. A. Serum pepsinogen I: an early marker of pernicious anemia in patients with type 1 diabetes. J. Clin. Endocrinol. Metab. 90, 5254–5258 (2005).
De Block, C. E., De Leeuw, I. H. & Van Gaal, L. F. Autoimmune gastritis in type 1 diabetes: a clinically oriented review. J. Clin. Endocrinol. Metab. 93, 363–371 (2008).
De Block, C. E. et al. Autoimmune gastropathy in type 1 diabetic patients with parietal cell antibodies: histological and clinical findings. Diabetes Care 26, 82–88 (2003).
Lam-Tse, W. K. et al. The association between autoimmune thyroiditis, autoimmune gastritis and type 1 diabetes. Pediatr. Endocrinol. Rev. 1, 22–37 (2003).
Broussais, F. J. V. Histoire des phlegmasies ou inflammations chroniques: fondée sur de nouvelles observations de clinique et d'anatomie pathologique [French]. (Gabon & Crochard, 1822).
Cruveilhier, J. Académie Royale de Médecine. Rapport fait à cette Académie dans la séance du 22 Cctobre 1839 sur les pièces pathologiques modelées en relief et publiées par le Dr Félix Thibert [French]. (Impr. de F. Didot Frères, 1839).
Author information
Authors and Affiliations
Contributions
R. M. Genta and W. L. Neumann contributed to researching data for the article, discussion of content, writing, and reviewing/editing the manuscript before submission. E. Coss and M. Rugge researched data for the article, made substantial contributions to discussion of content and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
William L. Neumann is employed by Pathologists Bio-Medical Labs, Dallas, TX, a private, hospital-based pathology practice. At the time of writing, Dr Neumann was a Gastrointestinal Pathology Fellow at the University of Texas Southwestern, Dallas; the fellowship was partially funded by Miraca Life Sciences, where he was based at the time. Robert M. Genta is an employee of Miraca Life Sciences, a private pathology referral laboratory based in Irving, Texas. Dr Genta is also a professor of Pathology and Medicine (Division of Gastroenterology) at the University of Texas Southwestern Medical Center at Dallas. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Neumann, W., Coss, E., Rugge, M. et al. Autoimmune atrophic gastritis—pathogenesis, pathology and management. Nat Rev Gastroenterol Hepatol 10, 529–541 (2013). https://doi.org/10.1038/nrgastro.2013.101
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2013.101
This article is cited by
-
Marked intestinal trans-differentiation by autoimmune gastritis along with ectopic pancreatic and pulmonary trans-differentiation
Journal of Gastroenterology (2024)
-
Prevalence of Histological Gastritis in a Community Population and Association with Epigastric Pain
Digestive Diseases and Sciences (2024)
-
Chronic gastritis may predict risk of cerebral small vessel disease
BMC Gastroenterology (2023)
-
Gastric bacteria as potential biomarkers for the diagnosis of atrophic gastritis
Molecular Biology Reports (2023)
-
Neutrophil (dys)function due to altered immuno-metabolic axis in type 2 diabetes: implications in combating infections
Human Cell (2023)