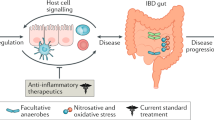
Abstract
The gastrointestinal microbiota has come to the fore in the search for the causes of IBD. This shift has largely been driven by the finding of genetic polymorphisms involved in gastrointestinal innate immunity (particularly polymorphisms in NOD2 and genes involved in autophagy) and alterations in the composition of the microbiota that might result in inflammation (so-called dysbiosis). Microbial diversity studies have continually demonstrated an expansion of the Proteobacteria phylum in patients with IBD. Individual Proteobacteria, in particular (adherent-invasive) Escherichia coli, Campylobacter concisus and enterohepatic Helicobacter, have all been associated with the pathogenesis of IBD. In this Review, we comprehensively describe the various associations of Proteobacteria and IBD. We also examine the importance of pattern recognition in the extracellular innate immune response of the host with particular reference to Proteobacteria, and postulate that Proteobacteria with adherent and invasive properties might exploit host defenses, drive proinflammatory change, alter the intestinal microbiota in favor of dysbiosis and ultimately lead to the development of IBD.
Key Points
-
The current paradigm of the pathogenesis of IBD involves a genetically and/or immunologically predisposed host responding inappropriately to an altered gastrointestinal microbiota, which leads to a protracted immune response and chronic inflammation
-
Proteobacteria are intricately involved in these luminal dysbiotic changes, wherein there is a breakdown in the balance of putative pathogenic bacteria and protective commensal bacteria
-
A subset of Proteobacteria with adherent-invasive capabilities potentially exploit the genetic defects of pathogen recognition and bacterial clearance, allowing them to perpetuate unchecked and providing the trigger that drives inflammation
-
The known genetic defects of pattern recognition receptors associated with susceptibility in developing IBD may paradoxically tilt the balance of the luminal microbiota towards pathogenic Gram-negative bacteria, in particular members of the Proteobacteria phylum
-
Studies correlating the genetic makeup of the host and the composition of the luminal microbiota will elucidate the relationship of Proteobacteria and the host immune system and the extent of its disruption in IBD
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hugot, J. P. et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 411, 599–603 (2001).
Hugot, J. P. et al. Mapping of a susceptibility locus for Crohn's disease on chromosome 16. Nature 379, 821–823 (1996).
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603–606 (2001).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nat. Genet. 39, 207–211 (2007).
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008).
Feller, M. et al. Mycobacterium avium subspecies paratuberculosis and Crohn's disease: a systematic review and meta-analysis. Lancet Infect. Dis. 7, 607–613 (2007).
Abubakar, I., Myhill, D., Aliyu, S. H. & Hunter, P. R. Detection of Mycobacterium avium subspecies paratuberculosis from patients with Crohn's disease using nucleic acid-based techniques: a systematic review and meta-analysis. Inflamm. Bowel Dis. 14, 401–410 (2008).
Mann, E. A. & Saeed, S. A. Gastrointestinal infection as a trigger for inflammatory bowel disease. Curr. Opin. Gastroenterol. 28, 24–29 (2012).
Willing, B. et al. Twin studies reveal specific imbalances in the mucosa-associated microbiota of patients with ileal Crohn's disease. Inflamm. Bowel Dis. 15, 653–660 (2009).
Sokol, H. et al. Low counts of Faecalibacterium prausnitzii in colitis microbiota. Inflamm. Bowel Dis. 15, 1183–1189 (2009).
Round, J. L. & Mazmanian, S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 9, 313–323 (2009).
Tamboli, C. P., Neut, C., Desreumaux, P. & Colombel, J. F. Dysbiosis in inflammatory bowel disease. Gut 53, 1–4 (2004).
Joossens, M. et al. Dysbiosis of the faecal microbiota in patients with Crohn's disease and their unaffected relatives. Gut 60, 631–637 (2011).
Lupp, C. et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2, 119–129 (2007).
Barnich, N., Denizot, J. & Darfeuille-Michaud, A. E. coli-mediated gut inflammation in genetically predisposed Crohn's disease patients. Pathol. Biol. (Paris) http://dx.doi.org/10.1016/j.patbio.2010.01.004.
Boudeau, J., Glasser, A. L., Masseret, E., Joly, B. & Darfeuille-Michaud, A. Invasive ability of an Escherichia coli strain isolated from the ileal mucosa of a patient with Crohn's disease. Infect. Immun. 67, 4499–4509 (1999).
Darfeuille-Michaud, A. et al. High prevalence of adherent-invasive Escherichia coli associated with ileal mucosa in Crohn's disease. Gastroenterology 127, 412–421 (2004).
Darfeuille-Michaud, A. et al. Presence of adherent Escherichia coli strains in ileal mucosa of patients with Crohn's disease. Gastroenterology 115, 1405–1413 (1998).
Mahendran, V. et al. Prevalence of Campylobacter species in adult Crohn's disease and the preferential colonization sites of Campylobacter species in the human intestine. PLoS ONE 6, e25417 (2011).
Man, S. M. et al. Host attachment, invasion, and stimulation of proinflammatory cytokines by Campylobacter concisus and other non-Campylobacter jejuni Campylobacter species. J. Infect. Dis. 202, 1855–1865 (2010).
Mukhopadhya, I. et al. Detection of Campylobacter concisus and other Campylobacter species in colonic biopsies from adults with ulcerative colitis. PLoS ONE 6, e21490 (2011).
Zhang, L. et al. Detection and isolation of Campylobacter species other than C. jejuni from children with Crohn's disease. J. Clin. Microbiol. 47, 453–455 (2009).
Kersters, K. et al. in The Prokaryotes (eds Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E. & Dworkin, M.) 3–37 (Springer, New York, 2006).
Stackebrandt, E., Murray, R. G. E. & Trüper, H. G. Proteobacteria classis nov., a name for the phylogenetic taxon that includes the “purple bacteria and their relatives”. Int. J. Syst. Bacteriol. 38, 321–325 (1988).
Mignot, T. The elusive engine in Myxococcus xanthus gliding motility. Cell. Mol. Life Sci. 64, 2733–2745 (2007).
Swan, B. K. et al. Potential for chemolithoautotrophy among ubiquitous bacteria lineages in the dark ocean. Science 333, 1296–1300 (2011).
Bryant, D. A. & Frigaard, N. U. Prokaryotic photosynthesis and phototrophy illuminated. Trends Microbiol. 14, 488–496 (2006).
Vesteinsdottir, H., Reynisdottir, D. B. & Orlygsson, J. Thiomonas islandica sp. nov., a moderately thermophilic, hydrogen- and sulfur-oxidizing betaproteobacterium isolated from a hot spring. Int. J. Syst. Evol. Microbiol. 61, 132–137 (2011).
Irgens, R. L., Gosink, J. J. & Staley, J. T. Polaromonas vacuolata gen. nov., sp. nov., a psychrophilic, marine, gas vacuolate bacterium from Antarctica. Int. J. Syst. Bacteriol. 46, 822–826 (1996).
Martin, W. & Muller, M. The hydrogen hypothesis for the first eukaryote. Nature 392, 37–41 (1998).
Singer, E. et al. Mariprofundus ferrooxydans PV-1 the first genome of a marine Fe(II) oxidizing Zetaproteobacterium. PLoS ONE 6, e25386 (2011).
Rehman, A. et al. Transcriptional activity of the dominant gut mucosal microbiota in chronic inflammatory bowel disease patients. J. Med. Microbiol. 59, 1114–1122 (2010).
Frank, D. N. et al. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl Acad. Sci. USA 104, 13780–13785 (2007).
Gophna, U., Sommerfeld, K., Gophna, S., Doolittle, W. F. & Veldhuyzen van Zanten, S. J. Differences between tissue-associated intestinal microfloras of patients with Crohn's disease and ulcerative colitis. J. Clin. Microbiol. 44, 4136–4141 (2006).
Dethlefsen, L., Huse, S., Sogin, M. L. & Relman, D. A. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 6, e280 (2008).
Khan, K. J. et al. Antibiotic therapy in inflammatory bowel disease: a systematic review and meta-analysis. Am. J. Gastroenterol. 106, 661–673 (2011).
Massei, F., Massimetti, M., Messina, F., Macchia, P. & Maggiore, G. Bartonella henselae and inflammatory bowel disease. Lancet 356, 1245–1246 (2000).
Ventura, A. et al. Systemic Bartonella henselae infection with hepatosplenic involvement. J. Pediatr. Gastroenterol. Nutr. 29, 52–56 (1999).
Baldwin, C. L. & Winter, A. J. Macrophages and Brucella. Immunol. Ser. 60, 363–380 (1994).
Musso, T. et al. Interaction of Bartonella henselae with the murine macrophage cell line J774: infection and proinflammatory response. Infect. Immun. 69, 5974–5980 (2001).
Sim, W. H. et al. Novel Burkholderiales 23S rRNA genes identified in ileal biopsy samples from children: preliminary evidence that a subtype is associated with perianal Crohn's disease. J. Clin. Microbiol. 48, 1939–1942 (2010).
Frank, D. N. et al. Disease phenotype and genotype are associated with shifts in intestinal-associated microbiota in inflammatory bowel diseases. Inflamm. Bowel Dis. 17, 179–184 (2011).
Walker, A. W. et al. High-throughput clone library analysis of the mucosa-associated microbiota reveals dysbiosis and differences between inflamed and non-inflamed regions of the intestine in inflammatory bowel disease. BMC Microbiol. 11, 7 (2011).
Shulman, S. T., Friedmann, H. C. & Sims, R. H. Theodor Escherich: the first pediatric infectious diseases physician? Clin. Infect. Dis. 45, 1025–1029 (2007).
Kaper, J. B., Nataro, J. P. & Mobley, H. L. Pathogenic Escherichia coli. Nat. Rev. Microbiol. 2, 123–140 (2004).
Rolhion, N. & Darfeuille-Michaud, A. Adherent-invasive Escherichia coli in inflammatory bowel disease. Inflamm. Bowel Dis. 13, 1277–1283 (2007).
Tabaqchali, S., O'Donoghue, D. P. & Bettelheim, K. A. Escherichia coli antibodies in patients with inflammatory bowel disease. Gut 19, 108–113 (1978).
Swidsinski, A. et al. Mucosal flora in inflammatory bowel disease. Gastroenterology 122, 44–54 (2002).
Martin, H. M. et al. Enhanced Escherichia coli adherence and invasion in Crohn's disease and colon cancer. Gastroenterology 127, 80–93 (2004).
Sepehri, S. et al. Characterization of Escherichia coli isolated from gut biopsies of newly diagnosed patients with inflammatory bowel disease. Inflamm. Bowel Dis. 17, 1451–1463 (2011).
Arnott, I. D. et al. Sero-reactivity to microbial components in Crohn's disease is associated with disease severity and progression, but not NOD2/CARD15 genotype. Am. J. Gastroenterol. 99, 2376–2384 (2004).
Mow, W. S. et al. High-level serum antibodies to bacterial antigens are associated with antibiotic-induced clinical remission in Crohn's disease: a pilot study. Dig. Dis. Sci. 49, 1280–1286 (2004).
Mow, W. S. et al. Association of antibody responses to microbial antigens and complications of small bowel Crohn's disease. Gastroenterology 126, 414–424 (2004).
Beaven, S. W. & Abreu, M. T. Biomarkers in inflammatory bowel disease. Curr. Opin. Gastroenterol. 20, 318–327 (2004).
Landers, C. J. et al. Selected loss of tolerance evidenced by Crohn's disease-associated immune responses to auto- and microbial antigens. Gastroenterology 123, 689–699 (2002).
Rolhion, N., Carvalho, F. A. & Darfeuille-Michaud, A. OmpC and the sigma(E) regulatory pathway are involved in adhesion and invasion of the Crohn's disease-associated Escherichia coli strain LF82. Mol. Microbiol. 63, 1684–1700 (2007).
Giaffer, M. H., Holdsworth, C. D. & Duerden, B. I. Virulence properties of Escherichia coli strains isolated from patients with inflammatory bowel disease. Gut 33, 646–650 (1992).
Liu, Y. et al. Immunocytochemical evidence of Listeria, Escherichia coli, and Streptococcus antigens in Crohn's disease. Gastroenterology 108, 1396–1404 (1995).
Boudeau, J., Barnich, N. & Darfeuille-Michaud, A. Type 1 pili-mediated adherence of Escherichia coli strain LF82 isolated from Crohn's disease is involved in bacterial invasion of intestinal epithelial cells. Mol. Microbiol. 39, 1272–1284 (2001).
Barnich, N., Boudeau, J., Claret, L. & Darfeuille-Michaud, A. Regulatory and functional co-operation of flagella and type 1 pili in adhesive and invasive abilities of AIEC strain LF82 isolated from a patient with Crohn's disease. Mol. Microbiol. 48, 781–794 (2003).
Barnich, N. et al. CEACAM6 acts as a receptor for adherent-invasive, E. coli, supporting ileal mucosa colonization in Crohn disease. J. Clin. Invest. 117, 1566–1574 (2007).
Kleessen, B., Kroesen, A. J., Buhr, H. J. & Blaut, M. Mucosal and invading bacteria in patients with inflammatory bowel disease compared with controls. Scand. J. Gastroenterol. 37, 1034–1041 (2002).
Glasser, A. L. et al. Adherent invasive Escherichia coli strains from patients with Crohn's disease survive and replicate within macrophages without inducing host cell death. Infect. Immun. 69, 5529–5537 (2001).
Chassaing, B. et al. Crohn disease--associated adherent-invasive E. coli bacteria target mouse and human Peyer's patches via long polar fimbriae. J. Clin. Invest. 121, 966–975 (2011).
Meconi, S. et al. Adherent-invasive Escherichia coli isolated from Crohn's disease patients induce granulomas in vitro. Cell. Microbiol. 9, 1252–1261 (2007).
Simpson, K. W. et al. Adherent and invasive Escherichia coli is associated with granulomatous colitis in boxer dogs. Infect. Immun. 74, 4778–4792 (2006).
Mansfield, C. S. et al. Remission of histiocytic ulcerative colitis in Boxer dogs correlates with eradication of invasive intramucosal Escherichia coli. J. Vet. Intern. Med. 23, 964–969 (2009).
Nash, J. H. et al. Genome sequence of adherent-invasive Escherichia coli and comparative genomic analysis with other, E. coli pathotypes. BMC Genomics 11, 667 (2010).
Miquel, S. et al. Complete genome sequence of Crohn's disease-associated adherent-invasive E. coli strain LF82. PLoS ONE 5, e12714 (2010).
Garrett, W. S. et al. Enterobacteriaceae act in concert with the gut microbiota to induce spontaneous and maternally transmitted colitis. Cell Host Microbe 8, 292–300 (2010).
Lee, I. A. & Kim, D. H. Klebsiella pneumoniae increases the risk of inflammation and colitis in a murine model of intestinal bowel disease. Scand. J. Gastroenterol. 46, 684–693 (2011).
Gutierrez, A. et al. Cytokine association with bacterial DNA in serum of patients with inflammatory bowel disease. Inflamm. Bowel Dis. 15, 508–514 (2009).
Kang, S. et al. Dysbiosis of fecal microbiota in Crohn's disease patients as revealed by a custom phylogenetic microarray. Inflamm. Bowel Dis. 16, 2034–2042 (2010).
Lamps, L. W. et al. Pathogenic Yersinia DNA is detected in bowel and mesenteric lymph nodes from patients with Crohn's disease. Am. J. Surg. Pathol. 27, 220–227 (2003).
Zippi, M. et al. Mesenteric adenitis caused by Yersinia pseudotubercolosis in a patient subsequently diagnosed with Crohn's disease of the terminal ileum. World J. Gastroenterol. 12, 3933–3935 (2006).
Saebo, A., Vik, E., Lange, O. J. & Matuszkiewicz, L. Inflammatory bowel disease associated with Yersinia enterocolitica O:3 infection. Eur. J. Intern. Med. 16, 176–182 (2005).
Safa, G., Loppin, M., Tisseau, L. & Lamoril, J. Cutaneous aseptic neutrophilic abscesses and Yersinia enterocolitica infection in a case subsequently diagnosed as Crohn's disease. Dermatology 217, 340–342 (2008).
Gradel, K. O. et al. Increased short- and long-term risk of inflammatory bowel disease after Salmonella or Campylobacter gastroenteritis. Gastroenterology 137, 495–501 (2009).
Jess, T. et al. Enteric Salmonella or Campylobacter infections and the risk of inflammatory bowel disease. Gut 60, 318–324 (2010).
Parent, K. & Mitchell, P. Pseudomonas-like group Va bacteria in Crohn's disease. Gastroenterology 75, 765 (1978).
Parent, K. & Mitchell, P. Cell wall-defective variants of Pseudomonas-like (group Va) bacteria in Crohn's disease. Gastroenterology 75, 368–372 (1978).
Graham, D. Y., Yoshimura, H. H. & Estes, M. K. DNA hybridization studies of the association of Pseudomonas maltophilia with inflammatory bowel diseases. J. Lab. Clin. Med. 101, 940–954 (1983).
Wagner, J. et al. Identification and characterisation of Pseudomonas 16S ribosomal DNA from ileal biopsies of children with Crohn's disease. PLoS ONE 3, e3578 (2008).
Wei, B. et al. Pseudomonas fluorescens encodes the Crohn's disease-associated I2 sequence and T-cell superantigen. Infect. Immun. 70, 6567–6575 (2002).
Sutton, C. L. et al. Identification of a novel bacterial sequence associated with Crohn's disease. Gastroenterology 119, 23–31 (2000).
Suchodolski, J. S., Xenoulis, P. G., Paddock, C. G., Steiner, J. M. & Jergens, A. E. Molecular analysis of the bacterial microbiota in duodenal biopsies from dogs with idiopathic inflammatory bowel disease. Vet. Microbiol. 142, 394–400 (2010).
Kangro, H. O., Chong, S. K., Hardiman, A., Heath, R. B. & Walker-Smith, J. A. A prospective study of viral and mycoplasma infections in chronic inflammatory bowel disease. Gastroenterology 98, 549–553 (1990).
Verma, R., Verma, A. K., Ahuja, V. & Paul, J. Real-time analysis of mucosal flora in patients with inflammatory bowel disease in India. J. Clin. Microbiol. 48, 4279–4282 (2010).
Duffy, M. et al. Sulfate-reducing bacteria colonize pouches formed for ulcerative colitis but not for familial adenomatous polyposis. Dis. Colon Rectum 45, 384–388 (2002).
Pitcher, M. C. & Cummings, J. H. Hydrogen sulphide: a bacterial toxin in ulcerative colitis? Gut 39, 1–4 (1996).
Goodman, M. J., Pearson, K. W., McGhie, D., Dutt, S. & Deodhar, S. G. Campylobacter and Giardia lamblia causing exacerbation of inflammatory bowel disease. Lancet 2, 1247 (1980).
Newman, A. & Lambert, J. R. Campylobacter jejuni causing flare-up in inflammatory bowel disease. Lancet 2, 919 (1980).
Kalischuk, L. D., Inglis, G. D. & Buret, A. G. Campylobacter jejuni induces transcellular translocation of commensal bacteria via lipid rafts. Gut Pathog. 1, 2 (2009).
Man, S. M. et al. Campylobacter concisus and other Campylobacter species in children with newly diagnosed Crohn's disease. Inflamm. Bowel Dis. 16, 1008–1016 (2010).
Zhang, L. et al. Isolation and detection of Campylobacter concisus from saliva of healthy individuals and patients with inflammatory bowel disease. J. Clin. Microbiol. 48, 2965–2967 (2010).
Istivan, T. S., Smith, S. C., Fry, B. N. & Coloe, P. J. Characterization of Campylobacter concisus hemolysins. FEMS Immunol. Med. Microbiol. 54, 224–235 (2008).
Istivan, T. S., Coloe, P. J., Fry, B. N., Ward, P. & Smith, S. C. Characterization of a haemolytic phospholipase A(2) activity in clinical isolates of Campylobacter concisus. J. Med. Microbiol. 53, 483–493 (2004).
Engberg, J. et al. Campylobacter concisus: an evaluation of certain phenotypic and genotypic characteristics. Clin. Microbiol. Infect. 11, 288–295 (2005).
Kaakoush, N. O. et al. The secretome of Campylobacter concisus. FEBS J. 277, 1606–1617 (2010).
Aabenhus, R., On, S. L., Siemer, B. L., Permin, H. & Andersen, L. P. Delineation of Campylobacter concisus genomospecies by amplified fragment length polymorphism analysis and correlation of results with clinical data. J. Clin. Microbiol. 43, 5091–5096 (2005).
Kalischuk, L. D. & Inglis, G. D. Comparative genotypic and pathogenic examination of Campylobacter concisus isolates from diarrheic and non-diarrheic humans. BMC Microbiol. 11, 53 (2011).
El-Omar, E. et al. Low prevalence of Helicobacter pylori in inflammatory bowel disease: association with sulphasalazine. Gut 35, 1385–1388 (1994).
Vare, P. O. et al. Seroprevalence of Helicobacter pylori infection in inflammatory bowel disease: is Helicobacter pylori infection a protective factor? Scand. J. Gastroenterol. 36, 1295–1300 (2001).
Feeney, M. A. et al. A case-control study of childhood environmental risk factors for the development of inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 14, 529–534 (2002).
Guslandi, M., Fanti, L. & Testoni, P. A. Helicobacter pylori seroprevalence in Crohn's disease: lack of influence by pharmacological treatment. Hepatogastroenterology 49, 1296–1297 (2002).
Luther, J., Dave, M., Higgins, P. D. & Kao, J. Y. Association between Helicobacter pylori infection and inflammatory bowel disease: a meta-analysis and systematic review of the literature. Inflamm. Bowel Dis. 16, 1077–1084 (2010).
Rad, R. et al. CD25+/Foxp3+ T cells regulate gastric inflammation and Helicobacter pylori colonization in vivo. Gastroenterology 131, 525–537 (2006).
Luther, J. et al. Helicobacter pylori DNA decreases pro-inflammatory cytokine production by dendritic cells and attenuates dextran sodium sulphate-induced colitis. Gut 60, 1479–1486 (2011).
Elliott, D. E., Urban, J. J., Argo, C. K. & Weinstock, J. V. Does the failure to acquire helminthic parasites predispose to Crohn's disease? FASEB J. 14, 1848–1855 (2000).
Mendall, M. A. et al. Childhood living conditions and Helicobacter pylori seropositivity in adult life. Lancet 339, 896–897 (1992).
Lemke, L. B. et al. Concurrent Helicobacter bilis infection in C57BL/6 mice attenuates proinflammatory, H. pylori-induced gastric pathology. Infect. Immun. 77, 2147–2158 (2009).
Hansen, R., Thomson, J. M., El-Omar, E. M. & Hold, G. L. The role of infection in the aetiology of inflammatory bowel disease. J. Gastroenterol. 45, 266–276 (2010).
Chalifoux, L. V. & Bronson, R. T. Colonic adenocarcinoma associated with chronic colitis in cotton top marmosets, Saguinus oedipus. Gastroenterology 80, 942–946 (1981).
Cahill, R. J. et al. Inflammatory bowel disease: an immunity-mediated condition triggered by bacterial infection with Helicobacter hepaticus. Infect. Immun. 65, 3126–3131 (1997).
Kullberg, M. C. et al. Helicobacter hepaticus-induced colitis in interleukin-10-deficient mice: cytokine requirements for the induction and maintenance of intestinal inflammation. Infect. Immun. 69, 4232–4241 (2001).
Jergens, A. E. et al. Helicobacter bilis triggers persistent immune reactivity to antigens derived from the commensal bacteria in gnotobiotic C3H/HeN mice. Gut 56, 934–940 (2007).
Bell, S. J., Chisholm, S. A., Owen, R. J., Borriello, S. P. & Kamm, M. A. Evaluation of Helicobacter species in inflammatory bowel disease. Aliment. Pharmacol. Ther. 18, 481–486 (2003).
Grehan, M., Danon, S., Lee, A., Daskalopoulos, G. & Mitchell, H. Absence of mucosa-associated colonic Helicobacters in an Australian urban population. J. Clin. Microbiol. 42, 874–876 (2004).
Bohr, U. R. et al. Identification of enterohepatic Helicobacter species in patients suffering from inflammatory bowel disease. J. Clin. Microbiol. 42, 2766–2768 (2004).
Thomson, J. M. et al. Enterohepatic Helicobacter in ulcerative colitis: potential pathogenic entities? PLoS ONE 6, e17184 (2011).
Zhang, L., Day, A., McKenzie, G. & Mitchell, H. Nongastric Helicobacter species detected in the intestinal tract of children. J. Clin. Microbiol. 44, 2276–2279 (2006).
Man, S. M., Zhang, L., Day, A. S., Leach, S. & Mitchell, H. Detection of enterohepatic and gastric Helicobacter species in fecal specimens of children with Crohn's disease. Helicobacter 13, 234–238 (2008).
Kaakoush, N. O. et al. Detection of Helicobacteraceae in intestinal biopsies of children with Crohn's disease. Helicobacter 15, 549–557 (2010).
Laharie, D. et al. Association between entero-hepatic Helicobacter species and Crohn's disease: a prospective cross-sectional study. Aliment. Pharmacol. Ther. 30, 283–293 (2009).
Keenan, J. I. et al. Helicobacter species in the human colon. Colorectal Dis. 12, 48–53 (2008).
Hansen, R., Thomson, J. M., Fox, J. G., El-Omar, E. M. & Hold, G. L. Could Helicobacter organisms cause inflammatory bowel disease? FEMS Immunol. Med. Microbiol. 61, 1–14 (2011).
Wells, J. M., Rossi, O., Meijerink, M. & van Baarlen, P. Epithelial crosstalk at the microbiota-mucosal interface. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4607–4614 (2011).
Hedayat, M., Netea, M. G. & Rezaei, N. Targeting of Toll-like receptors: a decade of progress in combating infectious diseases. Lancet Infect. Dis. 11, 702–712 (2011).
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Uematsu, S. & Fujimoto, K. The innate immune system in the intestine. Microbiol. Immunol. 54, 645–657 (2010).
Hold, G. L., Mukhopadhya, I. & Monie, T. P. Innate immune sensors and gastrointestinal bacterial infections. Clin. Dev. Immunol. 2011, 579650 (2011).
Cullen, T. W. & Trent, M. S. A link between the assembly of flagella and lipooligosaccharide of the Gram-negative bacterium Campylobacter jejuni. Proc. Natl Acad. Sci. USA 107, 5160–5165 (2010).
Hold, G. L. & Bryant, C. E. in Bacterial Lipopolysaccharides: Structure, Chemical Synthesis, Biogenesis and Interaction with Host Cells (eds Knirel, Y. A. & Valvano, M. A.) 371–388 (Springer, New York, 2011).
Shen, X. et al. The Toll-like receptor 4 D299G and T399I polymorphisms are associated with Crohn's disease and ulcerative colitis: a meta-analysis. Digestion 81, 69–77 (2006).
Rallabhandi, P. et al. Analysis of TLR4 polymorphic variants: new insights into TLR4/MD-2/CD14 stoichiometry, structure, and signaling. J. Immunol. 177, 322–332 (2006).
Arbour, N. C. et al. TLR4 mutations are associated with endotoxin hyporesponsiveness in humans. Nat. Genet. 25, 187–191 (2000).
Kiechl, S. et al. Toll-like receptor 4 polymorphisms and atherogenesis. N. Engl. J. Med. 347, 185–192 (2002).
Lorenz, E., Mira, J. P., Frees, K. L. & Schwartz, D. A. Relevance of mutations in the TLR4 receptor in patients with gram-negative septic shock. Arch. Intern. Med. 162, 1028–1032 (2002).
Eyking, A. et al. Toll-like receptor 4 variant D299G induces features of neoplastic progression in Caco-2 intestinal cells and is associated with advanced human colon cancer. Gastroenterology 141, 2154–2165 (2011).
Fuse, K., Katakura, K., Sakamoto, N. & Ohira, H. Toll-like receptor 9 gene mutations and polymorphisms in Japanese ulcerative colitis patients. World J. Gastroenterol. 16, 5815–5821 (2010).
Hong, J. et al. TLR2, TLR4 and TLR9 polymorphisms and Crohn's disease in a New Zealand Caucasian cohort. J. Gastroenterol. Hepatol. 22, 1760–1766 (2007).
Torok, H. P. et al. Crohn's disease is associated with a toll-like receptor-9 polymorphism. Gastroenterology 127, 365–366 (2004).
Eaves-Pyles, T. et al. Escherichia coli isolated from a Crohn's disease patient adheres, invades, and induces inflammatory responses in polarized intestinal epithelial cells. Int. J. Med. Microbiol. 298, 397–409 (2008).
Dasti, J. I., Tareen, A. M., Lugert, R., Zautner, A. E. & Gross, U. Campylobacter jejuni: a brief overview on pathogenicity-associated factors and disease-mediating mechanisms. Int. J. Med. Microbiol. 300, 205–211 (2010).
Ohl, M. E. & Miller, S. I. Salmonella: a model for bacterial pathogenesis. Annu. Rev. Med. 52, 259–274 (2001).
Kelly, D. P. & Wood, A. P. Reclassification of some species of Thiobacillus to the newly designated genera Acidithiobacillus gen. nov., Halothiobacillus gen. nov. and Thermithiobacillus gen. nov. Int. J. Syst. Evol. Microbiol. 50 (Pt 2), 511–516 (2000).
Willing, B. P. et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology 139, 1844–1854 e1 (2010).
Schwiertz, A. et al. Microbiota in pediatric inflammatory bowel disease. J. Pediatr. 157, 240–244e1 (2010).
Thomazini, C. M., Samegima, D. A., Rodrigues, M. A., Victoria, C. R. & Rodrigues, J. High prevalence of aggregative adherent Escherichia coli strains in the mucosa-associated microbiota of patients with inflammatory bowel diseases. Int. J. Med. Microbiol. 301, 475–479 (2011).
Mondot, S. et al. Highlighting new phylogenetic specificities of Crohn's disease microbiota. Inflamm. Bowel Dis. 17, 185–192 (2011).
Gibson, G. R., Cummings, J. H. & Macfarlane, G. T. Growth and activities of sulphate-reducing bacteria in gut contents of healthy subjects and patients with ulcerative colitis. FEMS Microbiol. Lett. 86, 103–111 (1991).
Ternhag, A., Torner, A., Svensson, A., Ekdahl, K. & Giesecke, J. Short- and long-term effects of bacterial gastrointestinal infections. Emerg. Infect. Dis. 14, 143–148 (2008).
Acknowledgements
Funding to Dr Hansen was provided by a Clinical Academic Training Fellowship from the Chief Scientist Office in Scotland (CAF/08/01). Other sources of funding for IBD research within the group include The Broad Medical Research Program, Crohn's in Childhood Research Association, NHS Grampian Hospital Endowments and Gastrointestinal Unit Hospital Research Funds.
Author information
Authors and Affiliations
Contributions
I. Mukhopadhya, R. Hansen and G. L. Hold all contributed to all aspects of this manuscript. E. M. El-Omar contributed to the discussion of content and review of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mukhopadhya, I., Hansen, R., El-Omar, E. et al. IBD—what role do Proteobacteria play?. Nat Rev Gastroenterol Hepatol 9, 219–230 (2012). https://doi.org/10.1038/nrgastro.2012.14
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2012.14
This article is cited by
-
Soil bacteria mediate organic carbon stability during alpine wetland biogeomorphic succession in the arid region of Central Asia
Plant and Soil (2024)
-
Strain-Specific Anti-Inflammatory Effects of Faecalibacterium prausnitzii Strain KBL1027 in Koreans
Probiotics and Antimicrobial Proteins (2024)
-
Assessment of cecal microbiota modulation from piglet dietary supplementation with copper
BMC Microbiology (2023)
-
The beneficial effects of commensal E. coli for colon epithelial cell recovery are related with Formyl peptide receptor 2 (Fpr2) in epithelial cells
Gut Pathogens (2023)
-
High prevalence of Mucosa-Associated extended-spectrum β-Lactamase-producing Escherichia coli and Klebsiella pneumoniae among Iranain patients with inflammatory bowel disease (IBD)
Annals of Clinical Microbiology and Antimicrobials (2023)