Abstract
Chronic alcohol use has been linked to chronic pancreatitis for over a century, but it has not been until the last decade that the role of alcohol in chronic pancreatitis has been elucidated in animals and, only in recent years, in human populations. Although a dose-dependent association between alcohol consumption and chronic pancreatitis may exist, a staistical association has been shown only with the consumption of ≥5 alcoholic drinks per day. Smoking also confers a strong, independent and dose-dependent risk of pancreatitis that may be additive or multiplicative when combined with alcohol. Alcohol increases the risk of acute pancreatitis in several ways and, most importantly, changes the immune response to injury. Genetic factors are also important and further studies are needed to clarify the role of gene–environment interactions in pancreatitis. In humans, aggressive interventional counseling against alcohol use may reduce the frequency of recurrent attacks of disease and smoking cessation may help to slow the progression of acute to chronic pancreatitis.
Key Points
-
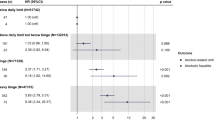
Studies suggest that a threshold of approximately 5 alcoholic drinks per day must be exceeded before the risk of chronic pancreatitis in individuals who drink alcohol exceeds that of the nondrinking population
-
Only a minority of patients with pancreatitis have a significant history of alcohol consumption, suggesting that most patients have a complex genetic disorder that is similar to other chronic inflammatory diseases
-
Smoking is a dose-dependent risk factor for pancreatitis, and its effects are additive or multiplicative when combined with alcohol
-
Chronic alcohol use at high daily doses (that is, ≥5 drinks daily) increases susceptibility to, and severity of, acute pancreatitis and accelerates the progression of chronic pancreatitis
-
Aggressive counseling leading to reduced alcohol consumption may reduce the likelihood of recurrent acute pancreatitis
-
Smoking cessation may slow the progression of chronic pancreatitis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Whitcomb, D. C. Acute pancreatitis. N. Engl. J. Med. 354, 2142–2150 (2006).
Katz, M., Carangelo, R., Miller, L. J. & Gorelick, F. Effect of ethanol on cholecystokinin-stimulated zymogen conversion in pancreatic acinar cells. Am. J. Physiol. 270, G171–G175 (1996).
Lu, Z., Karne, S., Kolodecik, T. & Gorelick, F. S. Alcohols enhance caerulein-induced zymogen activation in pancreatic acinar cells. Am. J. Physiol. Gastrointest. Liver Physiol. 282, G501–G507 (2002).
Pandol, S. J. et al. Ethanol diet increases the sensitivity of rats to pancreatitis induced by cholecystokinin octapeptide. Gastroenterology 117, 706–716 (1999).
Papachristou, G. I., Papachristou, D. J., Morinville, V. D., Slivka, A. & Whitcomb, D. C. Chronic alcohol consumption is a major risk factor for pancreatic necrosis in acute pancreatitis. Am. J. Gastroenterol. 101, 2605–2610 (2006).
Etemad, B. & Whitcomb, D. C. Chronic pancreatitis: diagnosis, classification, and new genetic developments. Gastroenterology 120, 682–707 (2001).
Friedreich, N. in Cyclopedia of the Practice of Medicine (William Wood, New York, 1878).
Whitcomb, D. C. et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat. Genet. 14, 141–145 (1996).
Whitcomb, D. C. Mechanisms of disease: Advances in understanding the mechanisms leading to chronic pancreatitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 1, 46–52 (2004).
Whitcomb, D. C. Hereditary pancreatitis: new insights into acute and chronic pancreatitis. Gut 45, 317–322 (1999).
Kloppel, G. & Maillet, B. Pseudocysts in chronic pancreatitis: a morphological analysis of 57 resection specimens and 9 autopsy pancreata. Pancreas 6, 266–274 (1991).
Kloppel, G. & Maillet, B. The morphological basis for the evolution of acute pancreatitis into chronic pancreatitis. Virchows Arch. A Pathol. Anat. Histopathol. 420, 1–4 (1992).
Bordalo, O. et al. Newer concept for the pathogenesis of chronic alcoholic pancreatitis. Am. J. Gastroenterol. 68, 278–285 (1977).
Braganza, J. M. Pancreatic disease: a casualty of hepatic “detoxification”? Lancet 2, 1000–1003 (1983).
McKim, S. E., Uesugi, T., Raleigh, J. A., McClain, C. J. & Arteel, G. E. Chronic intragastric alcohol exposure causes hypoxia and oxidative stress in the rat pancreas. Arch. Biochem. Biophys. 417, 34–43 (2003).
Schneider, A. & Whitcomb, D. C. Hereditary pancreatitis: a model for inflammatory diseases of the pancreas. Best Pract. Res. Clin. Gastroenterol. 16, 347–363 (2002).
Deng, X. et al. Chronic alcohol consumption accelerates fibrosis in response to cerulein-induced pancreatitis in rats. Am. J. Pathol. 166, 93–106 (2005).
Lankisch, P. G. et al. Natural history of acute pancreatitis: a long-term population-based study. Am. J. Gastroenterol. 104, 2797–2806 (2009).
Gukovsky, I. et al. A rat model reproducing key pathological responses of alcoholic chronic pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 294, G68–G79 (2008).
Whitcomb, D. C. & Barmada, M. M. A systems biology approach to genetic studies of pancreatitis and other complex diseases. Cell. Mol. Life Sci. 64, 1763–1777 (2007).
Friedman, S. L. Mechanisms of hepatic fibrogenesis. Gastroenterology 134, 1655–1669 (2008).
Xavier, R. J. & Podolsky, D. K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 448, 427–434 (2007).
Sarles, H. et al. The pancreatitis classification of Marseilles, Rome 1988. Scand. J. Gastroenterol. 24, 641 (1989).
Sarles, H. Proposal adopted unanimously by the participants of the Symposium, Marseilles 1963. Bibliotheca Gastroenterologica 7, 7–8 (1965).
Shrikhande, S. V. et al. Comparison of histological features and inflammatory cell reaction in alcoholic, idiopathic and tropical chronic pancreatitis. Br. J. Surg. 90, 1565–1572 (2003).
Stevens, T., Conwell, D. L. & Zuccaro, G. Pathogenesis of chronic pancreatitis: an evidence-based review of past theories and recent developments. Am. J. Gastroenterol. 99, 2256–2270 (2004).
Ammann, R. W., Heitz, P. U. & Kloppel, G. Course of alcoholic chronic pancreatitis: a prospective clinicomorphological long-term study. Gastroenterology 111, 224–231 (1996).
Takeyama, Y. Long-term prognosis of acute pancreatitis in Japan. Clin. Gastroenterol. Hepatol. 7, S15–S17 (2009).
Birgisson, H. et al. Acute pancreatitis: a prospective study of its incidence, aetiology, severity, and mortality in Iceland. Eur. J. Surg. 168, 278–282 (2002).
Eland, I. A., Sturkenboom, M. J., Wilson, J. H. & Stricker, B. H. Incidence and mortality of acute pancreatitis between 1985 and 1995. Scand. J. Gastroenterol. 35, 1110–1116 (2000).
Frey, C. F., Zhou, H., Harvey, D. J. & White, R. H. The incidence and case-fatality rates of acute biliary, alcoholic, and idiopathic pancreatitis in California, 1994–2001. Pancreas 33, 336–344 (2006).
Gislason, H. et al. Acute pancreatitis in Bergen, Norway. A study on incidence, etiology and severity. Scand. J. Surg. 93, 29–33 (2004).
Lindkvist, B., Appelros, S., Manjer, J. & Borgstrom, A. Trends in incidence of acute pancreatitis in a Swedish population: is there really an increase? Clin. Gastroenterol. Hepatol. 2, 831–837 (2004).
O'Farrell, A., Allwright, S., Toomey, D., Bedford, D. & Conlon, K. Hospital admission for acute pancreatitis in the Irish population, 1997–2004: could the increase be due to an increase in alcohol-related pancreatitis? J. Public Health (Oxf.) 29, 398–404 (2007).
Roberts, S. E., Williams, J. G., Meddings, D. & Goldacre, M. J. Incidence and case fatality for acute pancreatitis in England: geographical variation, social deprivation, alcohol consumption and aetiology—a record linkage study. Aliment. Pharmacol. Ther. 28, 931–941 (2008).
Tinto, A. et al. Acute and chronic pancreatitis—diseases on the rise: a study of hospital admissions in England 1989/1990–1999/2000. Aliment. Pharmacol. Ther. 16, 2097–2105 (2002).
Lankisch, P. G., Karimi, M., Bruns, A., Maisonneuve, P. & Lowenfels, A. B. Temporal trends in incidence and severity of acute pancreatitis in Luneburg County, Germany: a population-based study. Pancreatology 9, 420–426 (2009).
Yadav, D. & Lowenfels, A. B. Trends in the epidemiology of the first attack of acute pancreatitis: a systematic review. Pancreas 33, 323–330 (2006).
Sand, J., Valikoski, A. & Nordback, I. Alcohol consumption in the country and hospitalizations for acute alcohol pancreatitis and liver cirrhosis during a 20-year period. Alcohol Alcohol. 44, 321–325 (2009).
Dite, P. et al. Incidence of chronic pancreatitis in the Czech Republic. Eur. J. Gastroenterol. Hepatol. 13, 749–750 (2001).
Lankisch, P. G., Assmus, C., Maisonneuve, P. & Lowenfels, A. B. Epidemiology of pancreatic diseases in Luneburg County. A study in a defined german population. Pancreatology 2, 469–477 (2002).
Lin, Y. et al. Nationwide epidemiological survey of chronic pancreatitis in Japan. J. Gastroenterol. 35, 136–141 (2000).
Spanier, B. W., Dijkgraaf, M. G. & Bruno, M. J. Trends and forecasts of hospital admissions for acute and chronic pancreatitis in the Netherlands. Eur. J. Gastroenterol. Hepatol. 20, 653–658 (2008).
Yang, A. L., Vadhavkar, S., Singh, G. & Omary, M. B. Epidemiology of alcohol-related liver and pancreatic disease in the United States. Arch. Intern. Med. 168, 649–656 (2008).
Yadav, D. et al. Alcohol consumption, cigarette smoking, and the risk of recurrent acute and chronic pancreatitis. Arch. Intern. Med. 169, 1035–1045 (2009).
Layer, P. et al. The different courses of early- and late-onset idiopathic and alcoholic chronic pancreatitis. Gastroenterology 107, 1481–1487 (1994).
Lowenfels, A. B. et al. Racial factors and the risk of chronic pancreatitis. Am. J. Gastroenterol. 94, 790–794 (1999).
Morton, C., Klastsky, A. L. & Udaltsova, N. Smoking, coffee and pancreatitis. Am. J. Gastroenterol. 99, 731–738 (2004).
Frulloni, L. et al. Chronic pancreatitis: report from a multicenter Italian survey (PanCroInfAISP) on 893 patients. Dig. Liver Dis. 41, 311–317 (2009).
Ammann, R. W., Akovbiantz, A., Largiader, F. & Schueler, G. Course and outcome of chronic pancreatitis. Longitudinal study of a mixed medical-surgical series of 245 patients. Gastroenterology 86, 820–828 (1984).
Andersen, B. N., Pedersen, N. T., Scheel, J. & Worning, H. Incidence of alcoholic chronic pancreatitis in Copenhagen. Scand. J. Gastroenterol. 17, 247–252 (1982).
Marks, I. N., Bank, S. & Louw, J. H. Chronic pancreatitis in the Western Cape. Digestion 9, 447–453 (1973).
Robles-Diaz, G., Vargas, F., Uscanga, L. & Fernandez-del Castillo, C. Chronic pancreatitis in Mexico City. Pancreas 5, 479–483 (1990).
Talamini, G. et al. Alcohol and smoking as risk factors in chronic pancreatitis and pancreatic cancer. Dig. Dis. Sci. 44, 1301–1311 (1999).
Whitcomb, D. C. et al. Multicenter approach to recurrent acute and chronic pancreatitis in the United States: the North American Pancreatitis Study 2 (NAPS2). Pancreatology 8, 520–531 (2008).
Witt, H., Apte, M. V., Keim, V. & Wilson, J. S. Chronic pancreatitis: challenges and advances in pathogenesis, genetics, diagnosis, and therapy. Gastroenterology 132, 1557–1573 (2007).
Chari, S. T. et al. Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin. Gastroenterol. Hepatol. 4, 1010–1016 (2006).
Raina, A. et al. Evaluation and management of autoimmune pancreatitis: experience at a large US center. Am. J. Gastroenterol. 104, 2295–2306 (2009).
Corrao, G., Bagnardi, V., Zambon, A. & Arico, S. Exploring the dose-response relationship between alcohol consumption and the risk of several alcohol-related conditions: a meta-analysis. Addiction 94, 1551–1573 (1999).
Kristiansen, L., Gronbaek, M., Becker, U. & Tolstrup, J. S. Risk of pancreatitis according to alcohol drinking habits: a population-based cohort study. Am. J. Epidemiol. 168, 932–937 (2008).
Irving, H. M., Samokhvalov, A. V. & Rehm, J. Alcohol as a risk factor for pancreatitis. A systematic review and meta-analysis. JOP 10, 387–392 (2009).
Lankisch, P. G., Lowenfels, A. B. & Maisonneuve, P. What is the risk of alcoholic pancreatitis in heavy drinkers? Pancreas 25, 411–412 (2002).
Yadav, D., Eigenbrodt, M. L., Briggs, M. J., Williams, D. K. & Wiseman, E. J. Pancreatitis: prevalence and risk factors among male veterans in a detoxification program. Pancreas 34, 390–398 (2007).
Szabo, G., Mandrekar, P., Oak, S. & Mayerle, J. Effect of ethanol on inflammatory responses. Implications for pancreatitis. Pancreatology 7, 115–123 (2007).
Bell, R. L., Rodd, Z. A., Lumeng, L., Murphy, J. M. & McBride, W. J. The alcohol-preferring P rat and animal models of excessive alcohol drinking. Addict. Biol. 11, 270–288 (2006).
Lieber, C. S. & DeCarli, L. M. The feeding of ethanol in liquid diets. Alcohol Clin. Exp. Res. 10, 550–553 (1986).
Lieber, C. S. & DeCarli, L. M. Liquid diet technique of ethanol administration: 1989 update. Alcohol Alcohol. 24, 197–211 (1989).
Tsukamoto, H. et al. Severe and progressive steatosis and focal necrosis in rat liver induced by continuous intragastric infusion of ethanol and low fat diet. Hepatology 5, 224–232 (1985).
Tsukamoto, H., Towner, S. J., Yu, G. S. & French, S. W. Potentiation of ethanol-induced pancreatic injury by dietary fat. Induction of chronic pancreatitis by alcohol in rats. Am. J. Pathol. 131, 246–257 (1988).
Deng, X., Wood, P. G., Eagon, P. K. & Whitcomb, D. C. Rapid adaptation of pancreatic exocrine function to short term alcohol feeding in rats. Pancreatology 5, 183–195 (2005).
Deng, X., Wood, P. G., Eagon, P. K. & Whitcomb, D. C. Chronic alcohol-induced alterations in the pancreatic secretory control mechanisms. Dig. Dis. Sci. 49, 805–819 (2004).
Deng, X. & Whitcomb, D. C. Neurohormonal control of the exocrine pancreas. Curr. Opin. Gastroenterol. 14, 362–368 (1998).
Deng, X. et al. PYY inhibits CCK stimulated pancreatic secretion through the area postrema in unanesthetized rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 281, R645–R653 (2001).
Nordback, I., Pelli, H., Lappalainen-Lehto, R. & Sand, J. Is it long-term continuous drinking or the post-drinking withdrawal period that triggers the first acute alcoholic pancreatitis? Scand. J. Gastroenterol. 40, 1235–1239 (2005).
Wilson, J. S. & Apte, M. V. Role of alcohol metabolism in alcoholic pancreatitis. Pancreas 27, 311–315 (2003).
Criddle, D. N. et al. Ethanol toxicity in pancreatic acinar cells: mediation by nonoxidative fatty acid metabolites. Proc. Natl Acad. Sci. USA 101, 10738–10743 (2004).
Lieber, C. S. ALCOHOL: its metabolism and interaction with nutrients. Annu. Rev. Nutr. 20, 395–430 (2000).
Apte, M. V. & Wilson, J. S. Stellate cell activation in alcoholic pancreatitis. Pancreas 27, 316–320 (2003).
Nordback, I. H., Olson, J. L., Chacko, V. P. & Cameron, J. L. Detailed characterization of experimental acute alcoholic pancreatitis. Surgery 117, 41–49 (1995).
Laposata, E. A. & Lange, L. G. Presence of nonoxidative ethanol metabolism in human organs commonly damaged by ethanol abuse. Science 231, 497–499 (1986).
Pfutzer, R. H. et al. Pancreatic cholesterol esterase, ES-10, and fatty acid ethyl ester synthase III gene expression are increased in the pancreas and liver but not in the brain or heart with long-term ethanol feeding in rats. Pancreas 25, 101–106 (2002).
Criddle, D. N. et al. Fatty acid ethyl esters cause pancreatic calcium toxicity via inositol trisphosphate receptors and loss of ATP synthesis. Gastroenterology 130, 781–793 (2006).
Fortunato, F. et al. Pancreatic response to endotoxin after chronic alcohol exposure: switch from apoptosis to necrosis? Am. J. Physiol. Gastrointest. Liver Physiol. 290, G232–G241 (2005).
Li, H. S. et al. Rat mitochondrial ATP synthase ATP5G3: cloning and upregulation in pancreas after chronic ethanol feeding. Physiol. Genomics 6, 91–98 (2001).
Gullo, L., Pezzilli, R. & Ventrucci, M. Diagnostic value of the amino acid consumption test in pancreatic diseases. Pancreas 12, 64–67 (1996).
Mariani, A. et al. Accuracy of the plasma amino acid-consumption test in detecting pancreatic diseases is due to different methods. Pancreas 18, 203–211 (1999).
Kubisch, C. H. et al. Long-term ethanol consumption alters pancreatic gene expression in rats: a possible connection to pancreatic injury. Pancreas 33, 68–76 (2006).
Yuan, J. et al. Protein kinase D1 mediates NF-kappaB activation induced by cholecystokinin and cholinergic signaling in pancreatic acinar cells. Am. J. Physiol. Gastrointest. Liver Physiol. 295, G1190–G1201 (2008).
Gorelick, F. S. & Thrower, E. The acinar cell and early pancreatitis responses. Clin. Gastroenterol. Hepatol. 7, S10–S14 (2009).
Whitcomb, D. C. & Lowe, M. E. Human pancreatic digestive enzymes. Dig. Dis. Sci. 52, 1–17 (2007).
Frick, T. W., Fernandez-del Castillo, C., Bimmler, D. & Warshaw, A. L. Elevated calcium and activation of trypsinogen in rat pancreatic acini. Gut 41, 339–343 (1997).
Sutton, R. et al. Signal transduction, calcium and acute pancreatitis. Pancreatology 3, 497–505 (2003).
Kruger, B., Albrecht, E. & Lerch, M. M. The role of intracellular calcium signaling in premature protease activation and the onset of pancreatitis. Am. J. Pathol. 157, 43–50 (2000).
Rao, R. Endotoxemia and gut barrier dysfunction in alcoholic liver disease. Hepatology 50, 638–644 (2009).
Li, Y. Y. et al. Regulation of Hsp60 and the role of MK2 in a new model of severe experimental pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 297, G981–G989 (2009).
Singh, M., LaSure, M. M. & Bockman, D. E. Pancreatic acinar cell function and morphology in rats chronically fed an ethanol diet. Gastroenterology 82, 425–434 (1982).
Van Laethem, J., Robberecht, P., Resibois, A. & Deviere, J. Transforming growth factor beta promotes development of fibrosis after repeated courses of acute pancreatitis in mice. Gastroenterology 110, 576–582 (1996).
Muller-Pillasch, F. et al. TGFbeta and the extracellular matrix in pancreatitis. Hepatogastroenterology 46, 2751–2756 (1999).
Perides, G., Tao, X., West, N., Sharma, A. & Steer, M. L. A mouse model of ethanol dependent pancreatic fibrosis. Gut 54, 1461–1467 (2005).
Buchholz, M. et al. Transcriptome analysis of human hepatic and pancreatic stellate cells: organ-specific variations of a common transcriptional phenotype. J. Mol. Med. 83, 795–805 (2005).
Omary, M. B., Lugea, A., Lowe, A. W. & Pandol, S. J. The pancreatic stellate cell: a star on the rise in pancreatic diseases. J. Clin. Invest. 117, 50–59 (2007).
Bachem, M. G., Zhou, Z., Zhou, S. & Siech, M. Role of stellate cells in pancreatic fibrogenesis associated with acute and chronic pancreatitis. J. Gastroenterol. Hepatol. 21 (Suppl. 3), S92–S96 (2006).
Apte, M. V., Pirola, R. C. & Wilson, J. S. Battle-scarred pancreas: Role of alcohol and pancreatic stellate cells in pancreatic fibrosis. J. Gastroenterol. Hepatol. 21 (Suppl. 3), S97–S101 (2006).
Apte, M. V. et al. Does alcohol directly stimulate pancreatic fibrogenesis? Studies with rat pancreatic stellate cells. Gastroenterology 118, 780–794 (2000).
Li, J. et al. Does chronic ethanol intake cause chronic pancreatitis?: evidence and mechanism. Pancreas 37, 189–195 (2008).
Wittel, U. A. et al. Chronic pancreatic inflammation induced by environmental tobacco smoke inhalation in rats. Am. J. Gastroenterol. 101, 148–159 (2006).
Feick, P., Gerloff, A. & Singer, M. V. Effect of non-alcoholic compounds of alcoholic drinks on the pancreas. Pancreatology 7, 124–130 (2007).
Bode, C., Bode, J. C., Erhardt, J. G., French, B. A. & French, S. W. Effect of the type of beverage and meat consumed by alcoholics with alcoholic liver disease. Alcohol Clin. Exp. Res. 22, 1803–1805 (1998).
Santhosh, S. et al. A loss of function polymorphism (G191R) of anionic trypsinogen (PRSS2) confers protection against chronic pancreatitis. Pancreas 36, 317–320 (2008).
Witt, H. et al. A degradation-sensitive anionic trypsinogen (PRSS2) variant protects against chronic pancreatitis. Nat. Genet. 38, 668–673 (2006).
Witt, H. et al. Mutations in the gene encoding the serine protease inhibitor, Kazal type 1 are associated with chronic pancreatitis. Nat. Genet. 25, 213–216 (2000).
Pfutzer, R. H. et al. SPINK1/PSTI polymorphisms act as disease modifiers in familial and idiopathic chronic pancreatitis. Gastroenterology 119, 615–623 (2000).
Sharer, N. et al. Mutations of the cystic fibrosis gene in patients with chronic pancreatitis. N. Engl. J. Med. 339, 645–652 (1998).
Cohn, J. A. et al. Relation between mutations of the cystic fibrosis gene and idiopathic pancreatitis. N. Engl. J. Med. 339, 653–658 (1998).
Masson, E., Chen, J. M., Scotet, V., Le Marechal, C. & Ferec, C. Association of rare chymotrypsinogen C (CTRC) gene variations in patients with idiopathic chronic pancreatitis. Hum. Genet. 123, 83–91 (2008).
Muddana, V. et al. Association between calcium sensing receptor gene polymorphisms and chronic pancreatitis in a US population: role of serine protease inhibitor Kazal 1 type and alcohol. World J. Gastroenterol. 14, 4486–4491 (2008).
Felderbauer, P. et al. A novel mutation of the calcium sensing receptor gene is associated with chronic pancreatitis in a family with heterozygous SPINK1 mutations. BMC Gastroenterol. 3, 34 (2003).
Rosendahl, J. et al. Chymotrypsin C (CTRC) variants that diminish activity or secretion are associated with chronic pancreatitis. Nat. Genet. 40, 78–82 (2008).
Witt, H. et al. Mutation in the SPINK1 trypsin inhibitor gene, alcohol use, and chronic pancreatitis. JAMA 285, 2716–2717 (2001).
Schneider, A. et al. Limited contribution of the SPINK1 N34S mutation to the risk and severity of alcoholic chronic pancreatitis – a preliminary report from the United States. Dig. Dis. Sci. 48, 1110–1115 (2003).
Aoun, E. et al. Pathways to injury in chronic pancreatitis: decoding the role of the high-risk SPINK1 N34S haplotype using meta-analysis. PLoS ONE 3, e2003 (2008).
Chen, J. M., Mercier, B., Audrezet, M. P. & Ferec, C. Mutational analysis of the human pancreatic secretory trypsin inhibitor (PSTI) gene in hereditary and sporadic chronic pancreatitis. J. Med. Genet. 37, 67–69 (2000).
Pidasheva, S., D' Souza-Li, L., Canaff, L., Cole, D. E. & Hendy, G. N. CASRdb: calcium-sensing receptor locus-specific database for mutations causing familial (benign) hypocalciuric hypercalcemia, neonatal severe hyperparathyroidism, and autosomal dominant hypocalcemia. Hum. Mutat. 24, 107–111 (2004).
Yen, S., Hsieh, C. C. & MacMahon, B. Consumption of alcohol and tobacco and other risk factors for pancreatitis. Am. J. Epidemiol. 116, 407–414 (1982).
Bourliere, M., Barthet, M., Berthezene, P., Durbec, J. P. & Sarles, H. Is tobacco a risk factor for chronic pancreatitis and alcoholic cirrhosis? Gut 32, 1392–1395 (1991).
Lin, Y., Tamakoshi, A., Hayakawa, T., Ogawa, M. & Ohno, Y. Cigarette smoking as a risk factor for chronic pancreatitis: a case-control study in Japan. Research Committee on Intractable Pancreatic Diseases. Pancreas 21, 109–114 (2000).
Lowenfels, A. B., Zwemer, F. L., Jhangiani, S. & Pitchumoni, C. S. Pancreatitis in a native American Indian population. Pancreas 2, 694–697 (1987).
Tolstrup, J. S., Kristiansen, L., Becker, U. & Gronbaek, M. Smoking and risk of acute and chronic pancreatitis among women and men: a population-based cohort study. Arch. Intern. Med. 169, 603–609 (2009).
Lindkvist, B., Appelros, S., Manjer, J., Berglund, G. & Borgstrom, A. A prospective cohort study of smoking in acute pancreatitis. Pancreatology 8, 63–70 (2008).
Maisonneuve, P. et al. Cigarette smoking accelerates progression of alcoholic chronic pancreatitis. Gut 54, 510–514 (2005).
Maisonneuve, P. et al. Impact of smoking on patients with idiopathic chronic pancreatitis. Pancreas 33, 163–168 (2006).
Talamini, G. et al. Smoking cessation at the clinical onset of chronic pancreatitis and risk of pancreatic calcifications. Pancreas 35, 320–326 (2007).
Szabo, G. & Mandrekar, P. A recent perspective on alcohol, immunity, and host defense. Alcohol Clin. Exp. Res. 33, 220–232 (2009).
Angelini, G. et al. Association of chronic alcoholic liver and pancreatic disease: a prospective study. Am. J. Gastroenterol. 80, 998–1003 (1985).
Pelli, H., Sand, J., Laippala, P. & Nordback, I. Long-term follow-up after the first episode of acute alcoholic pancreatitis: time course and risk factors for recurrence. Scand. J. Gastroenterol. 35, 552–555 (2000).
Pelli, H., Lappalainen-Lehto, R., Piironen, A., Sand, J. & Nordback, I. Risk factors for recurrent acute alcohol-associated pancreatitis: a prospective analysis. Scand. J. Gastroenterol. 43, 614–621 (2008).
Gorry, M. C. et al. Mutations in the cationic trypsinogen gene are associated with recurrent acute and chronic pancreatitis. Gastroenterology 113, 1063–1068 (1997).
Ammann, R. W. & Muellhaupt, B. Progression of alcoholic acute to chronic pancreatitis. Gut 35, 552–556 (1994).
Nordback, I. et al. The recurrence of acute alcohol-associated pancreatitis can be reduced: a randomized controlled trial. Gastroenterology 136, 848–855 (2009).
Singer, M. V., Pfutzer, R. H. & Kiefer, F. Striving for abstinence in alcoholic pancreatitis: act of humanity, economic necessity, or flogging a dead horse after all? Gastroenterology 136, 757–760 (2009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Yadav, D., Whitcomb, D. The role of alcohol and smoking in pancreatitis. Nat Rev Gastroenterol Hepatol 7, 131–145 (2010). https://doi.org/10.1038/nrgastro.2010.6
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2010.6
This article is cited by
-
Radiomics analysis of contrast-enhanced T1W MRI: predicting the recurrence of acute pancreatitis
Scientific Reports (2023)
-
Pancreatic steatosis and metabolic pancreatic disease: a new entity?
Internal and Emergency Medicine (2023)
-
Activation of pancreatic stellate cells attenuates intracellular Ca2+ signals due to downregulation of TRPA1 and protects against cell death induced by alcohol metabolites
Cell Death & Disease (2022)
-
Metabolic implications of pancreatic fat accumulation
Nature Reviews Endocrinology (2022)
-
Repeated Stimulation of Toll-Like Receptor 2 and Dectin-1 Induces Chronic Pancreatitis in Mice Through the Participation of Acquired Immunity
Digestive Diseases and Sciences (2022)