Key Points
-
Hypercaloric diets lead to the dysregulation of multiple lipid metabolic pathways, which contributes to the onset and progression of metabolic disease
-
Lipidomic studies are starting to decipher the dysregulation of lipid metabolism associated with metabolic disease
-
Lipid metabolic pathways represent potential therapeutic targets to prevent or delay the onset and progression of metabolic disease
-
Further animal studies and clinical trials are required to define the stage of disease at which modulation of lipid metabolism will have maximal efficacy
-
Whether intervention into a single metabolic pathway or multiple pathways will produce optimal results remains to be determined
Abstract
Obesity, insulin resistance, type 2 diabetes mellitus and cardiovascular disease form a metabolic disease continuum that has seen a dramatic increase in prevalence in developed and developing countries over the past two decades. Dyslipidaemia resulting from hypercaloric diets is a major contributor to the pathogenesis of metabolic disease, and lipid-lowering therapies are the main therapeutic option for this group of disorders. However, the fact that dysfunctional lipid metabolism extends far beyond cholesterol and triglycerides is becoming increasingly clear. Lipidomic studies and mouse models are helping to explain the complex interactions between diet, lipid metabolism and metabolic disease. These studies are not only improving our understanding of this complex biology, but are also identifying potential therapeutic avenues to combat this growing epidemic. This Review examines what is currently known about phospholipid and sphingolipid metabolism in the setting of obesity and how metabolic pathways are being modulated for therapeutic effect.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Danaei, G. et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet 378, 31–40 (2011).
International Diabetes Federation. IDF Diabetes Atlas 6th edn (International Diabetes Federation, 2013).
Ridker, P. M., Mora, S., Rose, L. & Jupiter Trial Study Group. Percent reduction in LDL cholesterol following high-intensity statin therapy: potential implications for guidelines and for the prescription of emerging lipid-lowering agents. Eur. Heart J. 37, 1373–1379 (2016).
Hegele, R. A. et al. Nonstatin low-density lipoprotein-lowering therapy and cardiovascular risk reduction-statement from ATVB council. Arterioscler. Thromb. Vasc. Biol. 35, 2269–2280 (2015).
Naci, H., Brugts, J. & Ades, T. Comparative tolerability and harms of individual statins: a study-level network meta-analysis of 246 955 participants from 135 randomized, controlled trials. Circ. Cardiovasc. Qual. Outcomes 6, 390–399 (2013).
Navarese, E. P. et al. Meta-analysis of impact of different types and doses of statins on new-onset diabetes mellitus. Am. J. Cardiol. 111, 1123–1130 (2013).
Dobbins, R. L. et al. The composition of dietary fat directly influences glucose-stimulated insulin secretion in rats. Diabetes 51, 1825–1833 (2002).
Stein, D. T. et al. The insulinotropic potency of fatty acids is influenced profoundly by their chain length and degree of saturation. J. Clin. Invest. 100, 398–403 (1997).
Brown, M. S. & Goldstein, J. L. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab. 7, 95–96 (2008).
Hyötyläinen, T. & Orešicˇ, M. Optimizing the lipidomics workflow for clinical studies — practical considerations. Anal. Bioanal. Chem. 407, 4973–4993 (2015).
Wang, C., Wang, M. & Han, X. Applications of mass spectrometry for cellular lipid analysis. Mol. Biosyst. 11, 698–713 (2015).
Zhao, Y. Y., Wu, S. P., Liu, S., Zhang, Y. & Lin, R. C. Ultra-performance liquid chromatography-mass spectrometry as a sensitive and powerful technology in lipidomic applications. Chem. Biol. Interact. 220, 181–192 (2014).
Meikle, P. J., Wong, G., Barlow, C. K. & Kingwell, B. A. Lipidomics: potential role in risk prediction and therapeutic monitoring for diabetes and cardiovascular disease. Pharmacol. Ther. 143, 12–23 (2014).
Pietiläinen, K. H. et al. Acquired obesity is associated with changes in the serum lipidomic profile independent of genetic effects — a monozygotic twin study. PLoS ONE 2, e218 (2007).
Weir, J. M. et al. Plasma lipid profiling in a large population-based cohort. J. Lipid Res. 54, 2898–2908 (2013).
Meikle, P. J. et al. Plasma lipid profiling shows similar associations with prediabetes and type 2 diabetes. PLoS ONE 8, e74341 (2013). This study represents the most detailed lipidomic analysis of plasma lipids associated with diabetes mellitus.
Meikle, P. J. et al. Statin action favors normalization of the plasma lipidome in the atherogenic mixed dyslipidemia of MetS: potential relevance to statin-associated dysglycemia. J. Lipid Res. 56, 2381–2392 (2015).
Jove, M. et al. Plasma lipidomics discloses metabolic syndrome with a specific HDL phenotype. FASEB J. 28, 5163–5171 (2014).
Holcˇapek, M. et al. Lipidomic analysis of plasma, erythrocytes and lipoprotein fractions of cardiovascular disease patients using UHPLC/MS, MALDI-MS and multivariate data analysis. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 990, 52–63 (2015).
Camont, L. et al. Small, dense high-density lipoprotein-3 particles are enriched in negatively charged phospholipids: relevance to cellular cholesterol efflux, antioxidative, antithrombotic, anti-inflammatory, and antiapoptotic functionalities. Arterioscler. Thromb. Vasc. Biol. 33, 2715–2723 (2013).
Rached, F. et al. Defective functionality of small, dense HDL3 subpopulations in ST segment elevation myocardial infarction: relevance of enrichment in lysophosphatidylcholine, phosphatidic acid and serum amyloid A. Biochim. Biophys. Acta 1851, 1254–1261 (2015).
Rasmiena, A. A., Barlow, C. K., Ng, T. W., Tull, D. & Meikle, P. J. High density lipoprotein efficiently accepts surface but not internal oxidised lipids from oxidised low density lipoprotein. Biochim. Biophys. Acta 1861, 69–77 (2016).
Prentki, M. & Madiraju, S. R. Glycerolipid metabolism and signaling in health and disease. Endocr. Rev. 29, 647–676 (2008).
Coleman, R. A. & Lee, D. P. Enzymes of triacylglycerol synthesis and their regulation. Prog. Lipid Res. 43, 134–176 (2004).
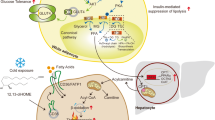
Perry, R. J., Samuel, V. T., Petersen, K. F. & Shulman, G. I. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature 510, 84–91 (2014).
Finck, B. N. & Hall, A. M. Does diacylglycerol accumulation in fatty liver disease cause hepatic insulin resistance? Biomed. Res. Int. 2015, 104132 (2015).
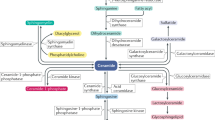
Cole, L. K., Vance, J. E. & Vance, D. E. Phosphatidylcholine biosynthesis and lipoprotein metabolism. Biochim. Biophys. Acta 1821, 754–761 (2012).
Vance, J. E. & Vance, D. E. Phospholipid biosynthesis in mammalian cells. Biochem. Cell Biol. 82, 113–128 (2004).
Steenbergen, R. et al. Disruption of the phosphatidylserine decarboxylase gene in mice causes embryonic lethality and mitochondrial defects. J. Biol. Chem. 280, 40032–40040 (2005).
Selathurai, A. et al. The CDP–ethanolamine pathway regulates skeletal muscle diacylglycerol content and mitochondrial biogenesis without altering insulin sensitivity. Cell Metab. 21, 718–730 (2015).
Arendt, B. M. et al. Nonalcoholic fatty liver disease is associated with lower hepatic and erythrocyte ratios of phosphatidylcholine to phosphatidylethanolamine. Appl. Physiol. Nutr. Metab. 38, 334–340 (2013).
Fu, S. et al. Aberrant lipid metabolism disrupts calcium homeostasis causing liver endoplasmic reticulum stress in obesity. Nature 473, 528–531 (2011). A mechanistic study examining how obesity can lead to an increased ratio of phosphatidylcholine:phosphatidylethanolamine and subsequent endoplasmic reticulum stress.
Li, Z. et al. The ratio of phosphatidylcholine to phosphatidylethanolamine influences membrane integrity and steatohepatitis. Cell Metab. 3, 321–331 (2006).
Martínez-Uña, M. et al. Excess S-adenosylmethionine reroutes phosphatidylethanolamine towards phosphatidylcholine and triglyceride synthesis. Hepatology 58, 1296–1305 (2013). An elegant study demonstrating an important role of S -adenosylmethionine in phospholipid metabolism.
Song, J. et al. Polymorphism of the PEMT gene and susceptibility to nonalcoholic fatty liver disease (NAFLD). FASEB J. 19, 1266–1271 (2005).
Dong, H. et al. The phosphatidylethanolamine N-methyltransferase gene V175M single nucleotide polymorphism confers the susceptibility to NASH in Japanese population. J. Hepatol. 46, 915–920 (2007).
Hebbard, L. & George, J. Animal models of nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 8, 35–44 (2011).
Jacobs, R. L., Devlin, C., Tabas, I. & Vance, D. E. Targeted deletion of hepatic CTP:phosphocholine cytidylyltransferase α in mice decreases plasma high density and very low density lipoproteins. J. Biol. Chem. 279, 47402–47410 (2004).
Walker, A. K. et al. A conserved SREBP-1/phosphatidylcholine feedback circuit regulates lipogenesis in metazoans. Cell 147, 840–852 (2011).
Jacobs, R. L., van der Veen, J. N. & Vance, D. E. Finding the balance: the role of S-adenosylmethionine and phosphatidylcholine metabolism in development of nonalcoholic fatty liver disease. Hepatology 58, 1207–1209 (2013).
Lessig, J. & Fuchs, B. Plasmalogens in biological systems: their role in oxidative processes in biological membranes, their contribution to pathological processes and aging and plasmalogen analysis. Curr. Med. Chem. 16, 2021–2041 (2009).
Wallner, S. & Schmitz, G. Plasmalogens the neglected regulatory and scavenging lipid species. Chem. Phys. Lipids 164, 573–589 (2011).
Wu, L. C. et al. Purification, identification, and cloning of lysoplasmalogenase, the enzyme that catalyzes hydrolysis of the vinyl ether bond of lysoplasmalogen. J. Biol. Chem. 286, 24916–24930 (2011).
Meikle, P. J. et al. Plasma lipidomic analysis of stable and unstable coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 31, 2723–2732 (2011).
Goodenowe, D. B. et al. Peripheral ethanolamine plasmalogen deficiency: a logical causative factor in Alzheimer's disease and dementia. J. Lipid Res. 48, 2485–2498 (2007).
Wood, P. Circulating plasmalogen levels and Alzheimer Disease Assessment Scale–Cognitive scores in Alzheimer patients. J. Psychiatry Neurosci. 35, 59–62 (2010).
Mankidy, R. et al. Membrane plasmalogen composition and cellular cholesterol regulation: a structure activity study. Lipids Health Dis. 9, 62 (2010).
Maeba, R. et al. Plasmalogens in human serum positively correlate with high-density lipoprotein and decrease with aging. J. Atheroscler. Thromb. 14, 12–18 (2007).
Maeba, R. et al. Myo-inositol treatment increases serum plasmalogens and decreases small dense LDL, particularly in hyperlipidemic subjects with metabolic syndrome. J. Nutr. Sci. Vitaminol (Tokyo) 54, 196–202 (2008).
Jürgens, G., Fell, A., Ledinski, G., Chen, Q. & Paltauf, F. Delay of copper-catalyzed oxidation of low density lipoprotein by in vitro enrichment with choline or ethanolamine plasmalogens. Chem. Phys. Lipids 77, 25–31 (1995).
Hajimoradi, M., Hassan, Z. M., Pourfathollah, A. A., Daneshmandi, S. & Pakravan, N. The effect of shark liver oil on the tumor infiltrating lymphocytes and cytokine pattern in mice. J. Ethnopharmacol. 126, 565–570 (2009).
Wentworth, J. M. et al. GM3 ganglioside and phosphatidylethanolamine-containing lipids are adipose tissue markers of insulin resistance in obese women. Int. J. Obes. (Lond.) 40, 706–713 (2016).
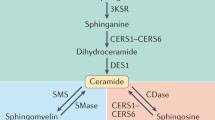
Holland, W. L. & Summers, S. A. Sphingolipids, insulin resistance, and metabolic disease: new insights from in vivo manipulation of sphingolipid metabolism. Endocr. Rev. 29, 381–402 (2008).
Holland, W. L. et al. Inhibition of ceramide synthesis ameliorates glucocorticoid-, saturated-fat-, and obesity-induced insulin resistance. Cell Metab. 5, 167–179 (2007). One of the first studies to characterize the relationship between ceramides, saturated fats and insulin resistance, and identify the enzymes involved as potential therapeutic targets.
Park, T. S. et al. Modulation of lipoprotein metabolism by inhibition of sphingomyelin synthesis in ApoE knockout mice. Atherosclerosis 189, 264–272 (2006).
Hojjati, M. R. et al. Effect of myriocin on plasma sphingolipid metabolism and atherosclerosis in apoE-deficient mice. J. Biol. Chem. 280, 10284–10289 (2005).
Park, T. S. et al. Ceramide is a cardiotoxin in lipotoxic cardiomyopathy. J. Lipid Res. 49, 2101–2112 (2008).
Eichler, F. S. et al. Overexpression of the wild-type SPT1 subunit lowers desoxysphingolipid levels and rescues the phenotype of HSAN1. J. Neurosci. 29, 14646–14651 (2009).
Bertea, M. et al. Deoxysphingoid bases as plasma markers in diabetes mellitus. Lipids Health Dis. 9, 84 (2010).
Othman, A. et al. Plasma deoxysphingolipids: a novel class of biomarkers for the metabolic syndrome? Diabetologia 55, 421–431 (2012).
Zuellig, R. A. et al. Deoxysphingolipids, novel biomarkers for type 2 diabetes, are cytotoxic for insulin-producing cells. Diabetes 63, 1326–1339 (2014). An important study providing a mechainistic framework for the role of deoxysphingolipids in type 2 diabetes mellitus.
Othman, A. et al. Fenofibrate lowers atypical sphingolipids in plasma of dyslipidemic patients: a novel approach for treating diabetic neuropathy? J. Clin. Lipidol. 9, 568–575 (2015).
Chaurasia, B. & Summers, S. A. Ceramides — lipotoxic inducers of metabolic disorders. Trends Endocrinol. Metab. 26, 538–550 (2015).
Chavez, J. A. et al. A role for ceramide, but not diacylglycerol, in the antagonism of insulin signal transduction by saturated fatty acids. J. Biol. Chem. 278, 10297–10303 (2003).
Stratford, S., DeWald, D. B. & Summers, S. A. Ceramide dissociates 3′-phosphoinositide production from pleckstrin homology domain translocation. Biochem. J. 354, 359–368 (2001).
Summers, S. A., Garza, L. A., Zhou, H. & Birnbaum, M. J. Regulation of insulin-stimulated glucose transporter GLUT4 translocation and Akt kinase activity by ceramide. Mol. Cell. Biol. 18, 5457–5464 (1998).
Chavez, J. A. et al. Ceramides and glucosylceramides are independent antagonists of insulin signaling. J. Biol. Chem. 289, 723–734 (2014).
Zhang, Q. J. et al. Ceramide mediates vascular dysfunction in diet-induced obesity by PP2A-mediated dephosphorylation of the eNOS–Akt complex. Diabetes 61, 1848–1859 (2012).
Bikman, B. T. et al. Fenretinide prevents lipid-induced insulin resistance by blocking ceramide biosynthesis. J. Biol. Chem. 287, 17426–17437 (2012). A study identifying Des1 as a potential therapeutic target for glucose homeostasis.
Chun, L. et al. Inhibition of ceramide synthesis reverses endothelial dysfunction and atherosclerosis in streptozotocin-induced diabetic rats. Diabetes Res. Clin. Pract. 93, 77–85 (2011).
Park, T. S., Rosebury, W., Kindt, E. K., Kowala, M. C. & Panek, R. L. Serine palmitoyltransferase inhibitor myriocin induces the regression of atherosclerotic plaques in hyperlipidemic ApoE-deficient mice. Pharmacol. Res. 58, 45–51 (2008).
Glaros, E. N. et al. Myriocin slows the progression of established atherosclerotic lesions in apolipoprotein E gene knockout mice. J. Lipid Res. 49, 324–331 (2008).
Glaros, E. N. et al. Inhibition of atherosclerosis by the serine palmitoyl transferase inhibitor myriocin is associated with reduced plasma glycosphingolipid concentration. Biochem. Pharmacol. 73, 1340–1346 (2007).
Park, T. S. & Goldberg, I. J. Sphingolipids, lipotoxic cardiomyopathy, and cardiac failure. Heart Fail. Clin. 8, 633–641 (2012).
Jiang, X. C., Goldberg, I. J. & Park, T. S. Sphingolipids and cardiovascular diseases: lipoprotein metabolism, atherosclerosis and cardiomyopathy. Adv. Exp. Med. Biol. 721, 19–39 (2011).
Holland, W. L. et al. An FGF21–adiponectin–ceramide axis controls energy expenditure and insulin action in mice. Cell Metab. 17, 790–797 (2013).
Holland, W. L. et al. Receptor-mediated activation of ceramidase activity initiates the pleiotropic actions of adiponectin. Nat. Med. 17, 55–63 (2011).
Lopez, X., Goldfine, A. B., Holland, W. L., Gordillo, R. & Scherer, P. E. Plasma ceramides are elevated in female children and adolescents with type 2 diabetes. J. Pediatr. Endocrinol. Metab. 26, 995–998 (2013).
Haus, J. M. et al. Plasma ceramides are elevated in obese subjects with type 2 diabetes and correlate with the severity of insulin resistance. Diabetes 58, 337–343 (2009).
Warshauer, J. T. et al. Effect of pioglitazone on plasma ceramides in adults with metabolic syndrome. Diabetes Metab. Res. Rev. 31, 734–744 (2015).
Bergman, B. C. et al. Serum sphingolipids: relationships to insulin sensitivity and changes with exercise in humans. Am. J. Physiol. Endocrinol. Metab. 309, E398–E408 (2015).
Amati, F. et al. Skeletal muscle triglycerides, diacylglycerols, and ceramides in insulin resistance: another paradox in endurance-trained athletes? Diabetes 60, 2588–2597 (2011).
Coen, P. M. et al. Reduced skeletal muscle oxidative capacity and elevated ceramide but not diacylglycerol content in severe obesity. Obesity (Silver Spring) 21, 2362–2371 (2013).
de la Maza, M. P. et al. Skeletal muscle ceramide species in men with abdominal obesity. J. Nutr. Health Aging 19, 389–396 (2015).
Coen, P. M. et al. Insulin resistance is associated with higher intramyocellular triglycerides in type I but not type II myocytes concomitant with higher ceramide content. Diabetes 59, 80–88 (2010).
Adams, J. M. et al. Ceramide content is increased in skeletal muscle from obese insulin-resistant humans. Diabetes 53, 25–31 (2004).
Skovbro, M. et al. Human skeletal muscle ceramide content is not a major factor in muscle insulin sensitivity. Diabetologia 51, 1253–1260 (2008).
Petersen, M. C. & Jurczak, M. J. CrossTalk opposing view: intramyocellular ceramide accumulation does not modulate insulin resistance. J. Physiol. 594, 3171–3174 (2016).
Summers, S. A. & Goodpaster, B. H. CrossTalk proposal: intramyocellular ceramide accumulation does modulate insulin resistance. J. Physiol. 594, 3167–3170 (2016).
Turpin, S. M. et al. Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab. 20, 678–686 (2014). One of a series of studies to differentiate the roles of CerS isoforms in insulin resistance.
Raichur, S. et al. CerS2 haploinsufficiency inhibits β-oxidation and confers susceptibility to diet-induced steatohepatitis and insulin resistance. Cell Metab. 20, 687–695 (2014). The second in a series of papers in this edition to address the specific role of CerS isoforms in insulin resistance.
Hla, T. & Kolesnick, R. C16:0-ceramide signals insulin resistance. Cell Metab. 20, 703–705 (2014). An editorial covering the two previous articles and highlighting the specific role of C16:0 ceramide in insulin resistance.
Gosejacob, D. et al. Ceramide synthase 5 is essential to maintain C16:0-ceramide pools and contributes to the development of diet-induced obesity. J. Biol. Chem. 291, 6989–7003 (2016).
Luukkonen, P. K. et al. Hepatic ceramides dissociate steatosis and insulin resistance in patients with non-alcoholic fatty liver disease. J. Hepatol. 64, 1167–1175 (2016).
Bergman, B. C. et al. Muscle sphingolipids during rest and exercise: a C18:0 signature for insulin resistance in humans. Diabetologia 59, 785–798 (2016). An important study that characterises the specific role of C18:0 ceramide in insulin resistance in skeletal muscle.
Hanamatsu, H. et al. Altered levels of serum sphingomyelin and ceramide containing distinct acyl chains in young obese adults. Nutr. Diabetes 4, e141 (2014).
Martinez-Ramirez, M. et al. HDL-sphingomyelin reduction after weight loss by an energy-restricted diet is associated with the improvement of lipid profile, blood pressure, and decrease of insulin resistance in overweight/obese patients. Clin. Chim. Acta 454, 77–81 (2016). An important human study that links alterations in sphingolipid metabolism resulting from a dietary intervention to a decrease in insulin resistance.
Li, Z. et al. Reducing plasma membrane sphingomyelin increases insulin sensitivity. Mol. Cell. Biol. 31, 4205–4218 (2011).
Sugimoto, M. et al. Characterization of the role of sphingomyelin synthase 2 in glucose metabolism in whole-body and peripheral tissues in mice. Biochim. Biophys. Acta 1861, 688–702 (2016).
Fan, Y. et al. Selective reduction in the sphingomyelin content of atherogenic lipoproteins inhibits their retention in murine aortas and the subsequent development of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 30, 2114–2120 (2010).
Aerts, J. M. et al. Pharmacological inhibition of glucosylceramide synthase enhances insulin sensitivity. Diabetes 56, 1341–1349 (2007).
Zhao, H. et al. Inhibiting glycosphingolipid synthesis improves glycemic control and insulin sensitivity in animal models of type 2 diabetes. Diabetes 56, 1210–1218 (2007).
Zhao, H. et al. Inhibiting glycosphingolipid synthesis ameliorates hepatic steatosis in obese mice. Hepatology 50, 85–93 (2009).
Chatterjee, S. et al. Inhibition of glycosphingolipid synthesis ameliorates atherosclerosis and arterial stiffness in apolipoprotein E−/− mice and rabbits fed a high-fat and -cholesterol diet. Circulation 129, 2403–2413 (2014). A study demonstrating the therapeutic potential of the regulation of glycosphingolipid synthesis to attenuate atherosclerosis.
Bietrix, F. et al. Inhibition of glycosphingolipid synthesis induces a profound reduction of plasma cholesterol and inhibits atherosclerosis development in APOE*3 Leiden and low-density lipoprotein receptor−/− mice. Arterioscler. Thromb. Vasc. Biol. 30, 931–937 (2010).
Glaros, E. N., Kim, W. S., Rye, K. A., Shayman, J. A. & Garner, B. Reduction of plasma glycosphingolipid levels has no impact on atherosclerosis in apolipoprotein E-null mice. J. Lipid Res. 49, 1677–1681 (2008).
Inokuchi, J. Insulin resistance as a membrane microdomain disorder. Yakugaku Zasshi 127, 579–586 (2007).
Tagami, S. et al. Ganglioside GM3 participates in the pathological conditions of insulin resistance. J. Biol. Chem. 277, 3085–3092 (2002).
Yamashita, T. et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl Acad. Sci. USA 100, 3445–3449 (2003).
Ussher, J. R. et al. Inhibition of de novo ceramide synthesis reverses diet-induced insulin resistance and enhances whole-body oxygen consumption. Diabetes 59, 2453–2464 (2010).
Murphy, M. P. Mitochondrial dysfunction indirectly elevates ROS production by the endoplasmic reticulum. Cell Metab. 18, 145–146 (2013).
de Mello, V. D. et al. Link between plasma ceramides, inflammation and insulin resistance: association with serum IL-6 concentration in patients with coronary heart disease. Diabetologia 52, 2612–2615 (2009).
Majumdar, I. & Mastrandrea, L. D. Serum sphingolipids and inflammatory mediators in adolescents at risk for metabolic syndrome. Endocrine 41, 442–449 (2012).
Caballero, F. et al. Specific contribution of methionine and choline in nutritional nonalcoholic steatohepatitis: impact on mitochondrial S-adenosyl-l-methionine and glutathione. J. Biol. Chem. 285, 18528–18536 (2010).
Mato, J. M. & Lu, S. C. Role of S-adenosyl-l-methionine in liver health and injury. Hepatology 45, 1306–1312 (2007).
Cano, A. et al. Methionine adenosyltransferase 1A gene deletion disrupts hepatic very low-density lipoprotein assembly in mice. Hepatology 54, 1975–1986 (2011).
Moylan, C. A. et al. Hepatic gene expression profiles differentiate presymptomatic patients with mild versus severe nonalcoholic fatty liver disease. Hepatology 59, 471–482 (2014).
Brites, P. et al. Alkyl-glycerol rescues plasmalogen levels and pathology of ether-phospholipid deficient mice. PLoS ONE 6, e28539 (2011).
Das, A. K., Holmes, R. D., Wilson, G. N. & Hajra, A. K. Dietary ether lipid incorporation into tissue plasmalogens of humans and rodents. Lipids 27, 401–405 (1992).
Marigny, K. et al. Modulation of endothelial permeability by 1-O-alkylglycerols. Acta Physiol. Scand. 176, 263–268 (2002).
Pédrono, F., Saïag, B., Moulinoux, J. P. & Legrand, A. B. 1-O-alkylglycerols reduce the stimulating effects of bFGF on endothelial cell proliferation in vitro. Cancer Lett. 251, 317–322 (2007).
Zhang, M., Sun, S., Tang, N., Cai, W. & Qian, L. Oral administration of alkylglycerols differentially modulates high-fat diet-induced obesity and insulin resistance in mice. Evid. Based Complement. Alternat. Med. 2013, 834027 (2013).
Rasmiena, A. A. et al. Plasmalogen modulation attenuates atherosclerosis in ApoE- and ApoE/GPx1-deficient mice. Atherosclerosis 243, 598–608 (2015). The first and only demonstration of plasmalogen modulation as a therapetic strategy in atherosclerosis.
Iannitti, T. & Palmieri, B. An update on the therapeutic role of alkylglycerols. Mar. Drugs 8, 2267–2300 (2010).
Wood, P. L., Khan, M. A., Mankidy, R., Smith, T. & Goodenowe, D. B. in Alzheimer's Disease Pathogenesis-Core Concepts, Shifting Paradigms and Therapeutic Targets Ch. 24 (ed. De La Monte, S.) 561–588 (InTech, 2011).
Wood, P. L. et al. Oral bioavailability of the ether lipid plasmalogen precursor, PPI-1011, in the rabbit: a new therapeutic strategy for Alzheimer's disease. Lipids Health Dis. 10, 227 (2011).
Grégoire, L. et al. Plasmalogen precursor analog treatment reduces levodopa-induced dyskinesias in parkinsonian monkeys. Behav. Brain Res. 286, 328–337 (2015).
Miville-Godbout, E. et al. Plasmalogen augmentation reverses striatal dopamine loss in MPTP mice. PLoS ONE 11, e0151020 (2016).
Kien, C. L. et al. A lipidomics analysis of the relationship between dietary fatty acid composition and insulin sensitivity in young adults. Diabetes 62, 1054–1063 (2013).
Lankinen, M. et al. A healthy nordic diet alters the plasma lipidomic profile in adults with features of metabolic syndrome in a multicenter randomized dietary intervention. J. Nutr. 146, 662–672 (2016).
Coen, P. M. et al. Exercise and weight loss improve muscle mitochondrial respiration, lipid partitioning, and insulin sensitivity after gastric bypass surgery. Diabetes 64, 3737–3750 (2015).
Bikman, B. T. & Summers, S. A. Ceramides as modulators of cellular and whole-body metabolism. J. Clin. Invest. 121, 4222–4230 (2011).
Chavez, J. A. & Summers, S. A. A ceramide-centric view of insulin resistance. Cell Metab. 15, 585–594 (2012).
Wang, S. et al. Simultaneous determination of nucleosides, myriocin, and carbohydrates in Cordyceps by HPLC coupled with diode array detection and evaporative light scattering detection. J. Sep. Sci. 32, 4069–4076 (2009).
Yu, J., Xu, H., Mo, Z., Zhu, H. & Mao, X. Determination of myriocin in natural and cultured Cordyceps cicadae using 9-fluorenylmethyl chloroformate derivatization and high-performance liquid chromatography with UV-detection. Anal. Sci. 25, 855–859 (2009).
Yu, S. H. et al. Hypoglycemic activity through a novel combination of fruiting body and mycelia of Cordyceps militaris in high-fat diet-induced type 2 diabetes mellitus mice. J. Diabetes Res. 2015, 723190 (2015).
Dong, Y. et al. Studies on the antidiabetic activities of Cordyceps militaris extract in diet-streptozotocin-induced diabetic Sprague-Dawley rats. Biomed. Res. Int. 2014, 160980 (2014).
Zhang, H. W. et al. Cordyceps sinensis (a traditional Chinese medicine) for treating chronic kidney disease. Cochrane Database Syst. Rev. 12, CD008353 (2014).
Mody, N. & McIlroy, G. D. The mechanisms of fenretinide-mediated anti-cancer activity and prevention of obesity and type-2 diabetes. Biochem. Pharmacol. 91, 277–286 (2014).
Siddique, M. M., Li, Y., Chaurasia, B., Kaddai, V. A. & Summers, S. A. Dihydroceramides: from bit players to lead actors. J. Biol. Chem. 290, 15371–15379 (2015).
Veronesi, U. et al. Fifteen-year results of a randomized phase III trial of fenretinide to prevent second breast cancer. Ann. Oncol. 17, 1065–1071 (2006).
Zanardi, S. et al. Clinical trials with retinoids for breast cancer chemoprevention. Endocr. Relat. Cancer 13, 51–68 (2006).
Sabichi, A. L. et al. Phase III prevention trial of fenretinide in patients with resected non-muscle-invasive bladder cancer. Clin. Cancer Res. 14, 224–229 (2008).
Aerts, J. M. et al. Glycosphingolipids and insulin resistance. Adv. Exp. Med. Biol. 721, 99–119 (2011).
Bennett, L. L. & Turcotte, K. Eliglustat tartrate for the treatment of adults with type 1 Gaucher disease. Drug Des. Devel. Ther. 9, 4639–4647 (2015).
Shayman, J. A. Developing novel chemical entities for the treatment of lysosomal storage disorders: an academic perspective. Am. J. Physiol. Renal Physiol. 309, F996–F999 (2015).
Shayman, J. A. The design and clinical development of inhibitors of glycosphingolipid synthesis: will invention be the mother of necessity? Trans. Am. Clin. Climatol. Assoc. 124, 46–60 (2013).
Yang, G. et al. Central role of ceramide biosynthesis in body weight regulation, energy metabolism, and the metabolic syndrome. Am. J. Physiol. Endocrinol. Metab. 297, E211–E224 (2009).
Kurek, K. et al. Inhibition of ceramide de novo synthesis ameliorates diet induced skeletal muscles insulin resistance. J. Diabetes Res. 2015, 154762 (2015).
Dekker, M. J. et al. Inhibition of sphingolipid synthesis improves dyslipidemia in the diet-induced hamster model of insulin resistance: evidence for the role of sphingosine and sphinganine in hepatic VLDL-apoB100 overproduction. Atherosclerosis 228, 98–109 (2013).
Chakraborty, M. et al. Myeloid cell-specific serine palmitoyltransferase subunit 2 haploinsufficiency reduces murine atherosclerosis. J. Clin. Invest. 123, 1784–1797 (2013).
Kasumov, T. et al. Ceramide as a mediator of non-alcoholic fatty liver disease and associated atherosclerosis. PLoS ONE 10, e0126910 (2015).
Kurek, K. et al. Inhibition of ceramide de novo synthesis reduces liver lipid accumulation in rats with nonalcoholic fatty liver disease. Liver Int. 34, 1074–1083 (2014).
Acknowledgements
P.J.M. is supported by a Senior Research Fellowship from the National Health and Medical Research Council of Australia.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to the researching data for the article, discussion of the content, writing the article and reviewing and/or editing the article before submission.
Corresponding author
Ethics declarations
Competing interests
P.J.M. declares no competing interests. S.A.S. is co-founder and scientific adviser of Centaurus Therapeutics Inc.
Rights and permissions
About this article
Cite this article
Meikle, P., Summers, S. Sphingolipids and phospholipids in insulin resistance and related metabolic disorders. Nat Rev Endocrinol 13, 79–91 (2017). https://doi.org/10.1038/nrendo.2016.169
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2016.169
This article is cited by
-
Infection with SARS-CoV-2 can cause pancreatic impairment
Signal Transduction and Targeted Therapy (2024)
-
Mitochondria-derived peptide MOTS-c: effects and mechanisms related to stress, metabolism and aging
Journal of Translational Medicine (2023)
-
The relationships between the plasma metabolome and orthostatic blood pressure responses
Scientific Reports (2023)
-
Serum ceramides in early pregnancy as predictors of gestational diabetes
Scientific Reports (2023)
-
A transcriptional network governing ceramide homeostasis establishes a cytokine-dependent developmental process
Nature Communications (2023)