Key Points
-
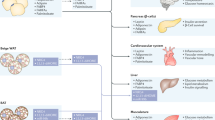
The heart is included in the network of endocrine organs that regulate energy usage and metabolism
-
The cardiac natriuretic peptide system and the sympathetic nervous system should be considered dual regulators of energy metabolism
-
Improved understanding of energy expenditure regulation by neurohormonal signals in adipose tissue might aid in finding ways to manipulate these processes
-
Improved understanding of receptors for the natriuretic peptides at the molecular level is needed, because these receptors are dynamically regulated and dictate responses to these peptides
-
Environmental factors (ambient temperature, diet and physical activity) coordinate metabolic fuel management in adipose tissue and muscle through both sympathetic nervous system tone and cardiac sensing of blood pressure
Abstract
Almost 20 years ago, the protein encoded by the ob locus in mice was identified as an adipocyte-secreted hormone, now termed leptin, which functions as a peripheral signal to communicate the organism's energy reserve—and thereby protects against starvation due to insufficient caloric resources. Additional peripheral factors have since been identified that coordinate interorgan crosstalk to manage energy resources. The heart is included in this network through its regulated release of natriuretic peptides A and B—cardiac hormones originally identified as important in blood pressure control. Emerging evidence that natriuretic peptide receptors are expressed in adipose tissue, and that circulating levels of these peptides are decreased in animals and humans with obesity, could imply that natriuretic peptides are also involved in the regulation of energy metabolism. The natriuretic peptides stimulate triglyceride lipolysis in adipocytes, a process also regulated by the sympathetic nervous system. In addition, these two pathways promote uncoupling of mitochondrial respiration and thermogenesis in brown adipocytes. This Review focuses on the roles of the natriuretic peptides and the sympathetic nervous system in regulating adipocyte metabolism. The potential for manipulating the natriuretic peptide pathway to increase energy expenditure in obesity and manage the complications of cardiometabolic disease is also discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Caterson, I. D. et al. Maintained intentional weight loss reduces cardiovascular outcomes: results from the Sibutramine Cardiovascular OUTcomes (SCOUT) trial. Diabetes Obes. Metab. 14, 523–530 (2012).
Lepor, N. E., Fouchia, D. D. & McCullough, P. A. New vistas for the treatment of obesity: turning the tide against the leading cause of morbidity and cardiovascular mortality in the developed world. Rev. Cardiovasc. Med. 14, 20–39; quiz 40 (2013).
Moro, C. & Lafontan, M. Natriuretic peptides and cGMP signaling control of energy homeostasis. Am. J. Physiol. Heart Circ. Physiol. 304, H358–H368 (2013).
Sarzani, R., Dessi-Fulgheri, P., Paci, V. M., Espinosa, E. & Rappelli, A. Expression of natriuretic peptide receptors in human adipose and other tissues. J. Endocrinol. Invest. 19, 581–585 (1996).
Dessi-Fulgheri, P. et al. Plasma atrial natriuretic peptide and natriuretic peptide receptor gene expression in adipose tissue of normotensive and hypertensive obese patients. J. Hypertens. 15, 1695–1699 (1997).
Wang, T. J. et al. Impact of obesity on plasma natriuretic peptide levels. Circulation 109, 594–600 (2004).
Dessi-Fulgheri, P., Sarzani, R. & Rappelli, A. The natriuretic peptide system in obesity-related hypertension: new pathophysiological aspects. J. Nephrol. 11, 296–299 (1998).
Morton, N. M. et al. A stratified transcriptomics analysis of polygenic fat and lean mouse adipose tissues identifies novel candidate obesity genes. PLoS ONE 6, e23944 (2011).
Sengenès, C., Berlan, M., De Glisezinski, I., Lafontan, M. & Galitzky, J. Natriuretic peptides: a new lipolytic pathway in human adipocytes. FASEB J. 14, 1345–1351 (2000).
Birkenfeld, A. L. et al. Lipid mobilization with physiological atrial natriuretic peptide concentrations in humans. J. Clin. Endocrinol. Metab. 90, 3622–3628 (2005).
Birkenfeld, A. L. et al. β-adrenergic and atrial natriuretic peptide interactions on human cardiovascular and metabolic regulation. J. Clin. Endocrinol. Metab. 91, 5069–5075 (2006).
Birkenfeld, A. L. et al. Atrial natriuretic peptide induces postprandial lipid oxidation in humans. Diabetes 57, 3199–3204 (2008).
Bordicchia, M. et al. Cardiac natriuretic peptides act via p38 MAPK to induce the brown fat thermogenic program in mouse and human adipocytes. J. Clin. Invest. 122, 1022–1036 (2012).
Engeli, S. et al. Natriuretic peptides enhance the oxidative capacity of human skeletal muscle. J. Clin. Invest. 122, 4675–4679 (2012).
Yehuda-Shnaidman, E., Buehrer, B., Pi, J., Kumar, N. & Collins, S. Acute stimulation of white adipocyte respiration by PKA-induced lipolysis. Diabetes 59, 2474–2483 (2010).
Zurlo, F., Larson, K., Bogardus, C. & Ravussin, E. Skeletal muscle metabolism is a major determinant of resting energy expenditure. J. Clin. Invest. 86, 1423–1427 (1990).
Bal, N. C. et al. Sarcolipin is a newly identified regulator of muscle-based thermogenesis in mammals. Nat. Med. 18, 1575–1579 (2012).
Sheldon, E. F. The so-called hibernating gland in mammals: A form of adipose tissue. Anat. Rec. 28, 331–347 (1924).
Aherne, W. & Hull, D. The site of heat production in the newborn infant. Proc. R. Soc. Med. 57, 1172–1173 (1964).
Rothwell, N. J. & Stock, M. J. A role for brown adipose tissue in diet-induced thermogenesis. Nature 281, 31–35 (1979).
Heaton, J. M. The distribution of brown adipose tissue in the human. J. Anat. 112, 35–39 (1972).
Lee, P., Swarbrick, M. M. & Ho, K. K. Brown adipose tissue in adult humans: a metabolic renaissance. Endocr. Rev. 34, 413–438 (2013).
Nedergaard, J., Bengtsson, T. & Cannon, B. Unexpected evidence for active brown adipose tissue in adult humans. Am. J. Physiol. Endocrinol. Metab. 293, E444–E452 (2007).
van Marken Lichtenbelt, W. D. et al. Cold-activated brown adipose tissue in healthy men. N. Engl. J. Med. 360, 1500–1508 (2009).
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
Virtanen, K. A. et al. Functional brown adipose tissue in healthy adults. N. Engl. J. Med. 360, 1518–1525 (2009).
Zingaretti, M. C. et al. The presence of UCP1 demonstrates that metabolically active adipose tissue in the neck of adult humans truly represents brown adipose tissue. FASEB J. 23, 3113–3120 (2009).
Saito, M. et al. High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 58, 1526–1531 (2009).
Ouellet, V. et al. Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J. Clin. Invest. 122, 545–552 (2012).
Orava, J. et al. Blunted metabolic responses to cold and insulin stimulation in brown adipose tissue of obese humans. Obesity (Silver Spring) http://dx.doi.org/10.1002/oby.20456.
Matthias, A. et al. Thermogenic responses in brown fat cells are fully UCP1-dependent. UCP2 or UCP3 do not substitute for UCP1 in adrenergically or fatty acid-induced thermogenesis. J. Biol. Chem. 275, 25073–25081 (2000).
Cannon, B. & Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 (2004).
Wang, T., Zang, Y., Ling, W., Corkey, B. E. & Guo, W. Metabolic partitioning of endogenous fatty acid in adipocytes. Obes. Res. 11, 880–887 (2003).
Maassen, J. A., Romijn, J. A. & Heine, R. J. Fatty acid-induced mitochondrial uncoupling in adipocytes as a key protective factor against insulin resistance and beta cell dysfunction: do adipocytes consume sufficient amounts of oxygen to oxidise fatty acids? Diabetologia 51, 907–908 (2008).
Stenson, B. M. et al. Activation of liver X receptor (LXR) regulates substrate oxidation in white adipocytes. Endocrinology 150, 4103–4113 (2009).
Wilson-Fritch, L. et al. Mitochondrial remodeling in adipose tissue associated with obesity and treatment with rosiglitazone. J. Clin. Invest. 114, 1281–1289 (2004).
Kusminski, C. M. & Scherer, P. E. Mitochondrial dysfunction in white adipose tissue. Trends Endocrinol. Metab. 23, 435–443 (2012).
Böttcher, H. & Fürst, P. Decreased white fat cell thermogenesis in obese individuals. Int. J. Obes. Relat. Metab. Disord. 21, 439–444 (1997).
Landsberg, L. & Young, J. B. The role of the sympathoadrenal system in modulating energy expenditure. J. Clin. Endocrinol. Metab. 13, 475–499 (1984).
Cousin, B. et al. Occurrence of brown adipocytes in rat white adipose tissue: molecular and morphological characterization. J. Cell Sci. 103, 931–942 (1992).
Wu, J. et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 150, 366–376 (2012).
Cypess, A. M. et al. Anatomical localization, gene expression profiling and functional characterization of adult human neck brown fat. Nat. Med. 19, 635–639 (2013).
Jespersen, N. Z. et al. A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans. Cell Metab. 17, 798–805 (2013).
Cao, W. et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell. Biol. 24, 3057–3067 (2004).
Fregly, M. J., Kikta, D. C., Threatte, R. M., Torres, J. L. & Barney, C. C. Development of hypertension in rats during chronic exposure to cold. J. Appl. Physiol. 66, 741–749 (1989).
Yuan, K. et al. Modification of atrial natriuretic peptide system in cold-induced hypertensive rats. Regul. Pept. 154, 112–120 (2009).
Sun, Z., Cade, J. R., Fregly, M. J. & Rowland, N. E. Effect of chronic treatment with propranolol on the cardiovascular responses to chronic cold exposure. Physiol. Behav. 62, 379–384 (1997).
Granneman, J. G., Lahners, K. N. & Chaudhry, A. Molecular cloning and expression of the rat β3-adrenergic receptor. Mol. Pharmacol. 40, 895–899 (1991).
Nahmias, C. et al. Molecular characterization of the mouse β3-adrenergic receptor: relationship with the atypical receptor of adipocytes. EMBO J. 10, 3721–3727 (1991).
Nakatsuji, H. et al. Reciprocal regulation of natriuretic peptide receptors by insulin in adipose cells. Biochem. Biophys. Res. Commun. 392, 100–105 (2010).
Pivovarova, O. et al. Insulin up-regulates natriuretic peptide clearance receptor expression in the subcutaneous fat depot in obese subjects: a missing link between CVD risk and obesity? J. Clin. Endocrinol. Metab. 97, E731–E739 (2012).
Potter, L. R. & Hunter, T. Guanylyl cyclase-linked natriuretic peptide receptors: structure and regulation. J. Biol. Chem. 276, 6057–6060 (2001).
Kuhn, M. Structure, regulation, and function of mammalian membrane guanylyl cyclase receptors, with a focus on guanylyl cyclase-A. Circ. Res. 93, 700–709 (2003).
Sengenes, C. et al. Natriuretic peptide-dependent lipolysis in fat cells is a primate specificity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 283, R257–R265 (2002).
Shibasaki, I. et al. Greater expression of inflammatory cytokines, adrenomedullin, and natriuretic peptide receptor-C in epicardial adipose tissue in coronary artery disease. Regul. Pept. 165, 210–217 (2010).
Fitzgibbons, T. P. et al. Similarity of mouse perivascular and brown adipose tissues and their resistance to diet-induced inflammation. Am. J. Physiol. Heart Circ. Physiol. 301, H1425–H1437 (2011).
Kiefer, F. W., Cohen, P. & Plutzky, J. Fifty shades of brown: perivascular fat, thermogenesis, and atherosclerosis. Circulation 126, 1012–1015 (2012).
Payne, G. A., Kohr, M. C. & Tune, J. D. Epicardial perivascular adipose tissue as a therapeutic target in obesity-related coronary artery disease. Br. J. Pharmacol. 165, 659–669 (2012).
Chang, L. et al. Loss of perivascular adipose tissue on peroxisome proliferator-activated receptor-γ deletion in smooth muscle cells impairs intravascular thermoregulation and enhances atherosclerosis. Circulation 126, 1067–1078 (2012).
Anker, S. D. et al. Prognostic importance of weight loss in chronic heart failure and the effect of treatment with angiotensin-converting-enzyme inhibitors: an observational study. Lancet 361, 1077–1083 (2003).
Anker, S. D. & Sharma, R. The syndrome of cardiac cachexia. Int. J. Cardiol. 85, 51–66 (2002).
Pureza, V. & Florea, V. G. Mechanisms for cachexia in heart failure. Curr. Heart Fail. Rep. http://dx.doi.org/10.1007/s11897-013-0153-9.
Melenovsky, V. et al. Relationships between right ventricular function, body composition and prognosis in advanced heart failure. J. Am. Coll. Cardiol. 62, 1660–1670 (2013).
Christensen, H. M. et al. Prevalence of cachexia in chronic heart failure and characteristics of body composition and metabolic status. Endocrine 43, 626–634 (2013).
Szabó, T. et al. Increased catabolic activity in adipose tissue of patients with chronic heart failure. Eur. J. Heart Fail. 15, 1131–1137 (2013).
Polak, J. et al. Lipolytic effects of B-type natriuretic peptide 1–32 in adipose tissue of heart failure patients compared with healthy controls. J. Am. Coll. Cardiol. 58, 1119–1125 (2011).
Welsh, P. & McMurray, J. J. B-type natriuretic peptide and glycaemia: an emerging cardiometabolic pathway? Diabetologia 55, 1240–1243 (2012).
Vijgen, G. H. et al. Increase in brown adipose tissue activity after weight loss in morbidly obese subjects. J. Clin. Endocrinol. Metab. 97, E1229–E1233 (2012).
Batal, O. et al. Effect of obesity on B-type natriuretic peptide levels in patients with pulmonary arterial hypertension. Am. J. Cardiol. 110, 909–914 (2012).
Lippi, G., Schena, F. & Guidi, G. C. Baseline and post-exercise N-terminal pro-B-type natriuretic peptide values are associated with body mass index. Am. J. Cardiol. 109, 303–304 (2012).
Magnusson, M. et al. Low plasma level of atrial natriuretic peptide predicts development of diabetes: the prospective Malmo Diet and Cancer study. J. Clin. Endocrinol. Metab. 97, 638–645 (2012).
Translational Research Institute for Metabolism and Diabetes Research Studies: BNP [online] (2013).
Luft, F. C. et al. Atriopeptin III kinetics and pharmacodynamics in normal and anephric rats. J. Pharmacol. Exp. Ther. 236, 416–418 (1986).
Nakao, K. et al. The pharmacokinetics of α-human atrial natriuretic polypeptide in healthy subjects. Eur. J. Clin. Pharmacol. 31, 101–103 (1986).
Yandle, T. G. et al. Metabolic clearance rate and plasma half life of α-human atrial natriuretic peptide in man. Life Sci. 38, 1827–1833 (1986).
Richards, A. M. et al. Brain natriuretic peptide: natriuretic and endocrine effects in essential hypertension. J. Hypertens. 11, 163–170 (1993).
Zakeri, R. & Burnett, J. C. Designer natriuretic peptides: a vision for the future of heart failure therapeutics. Can. J. Physiol. Pharmacol. 89, 593–601 (2011).
de Bold, M. K. et al. Characterization of a long-acting recombinant human serum albumin-atrial natriuretic factor (ANF) expressed in Pichia pastoris. Regul. Pept. 175, 7–10 (2012).
Wang, T. J. The natriuretic peptides and fat metabolism. N. Engl. J. Med. 367, 377–378 (2012).
Cypess, A. M. et al. Cold but not sympathomimetics activates human brown adipose tissue in vivo. Proc. Natl Acad. Sci. USA 109, 10001–10005 (2012).
Vosselman, M. J. et al. Systemic β-adrenergic stimulation of thermogenesis is not accompanied by brown adipose tissue activity in humans. Diabetes 61, 3106–3113 (2012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Collins, S. A heart–adipose tissue connection in the regulation of energy metabolism. Nat Rev Endocrinol 10, 157–163 (2014). https://doi.org/10.1038/nrendo.2013.234
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2013.234
This article is cited by
-
Effect of various weight loss interventions on serum NT-proBNP concentration in severe obese subjects without clinical manifest heart failure
Scientific Reports (2021)
-
Cellular and molecular pathobiology of heart failure with preserved ejection fraction
Nature Reviews Cardiology (2021)
-
Cardiac natriuretic peptides
Nature Reviews Cardiology (2020)
-
Collaborative Activities of Noradrenaline and Natriuretic Peptide for Glucose Utilization in Patients with Acute Coronary Syndrome
Scientific Reports (2019)
-
Association Between Cardiac Natriuretic Peptides and Lipid Profile: a Systematic Review and Meta-Analysis
Scientific Reports (2019)