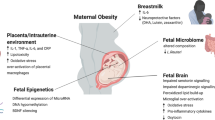
Abstract
Experimental and animal data suggest that maternal obesity during pregnancy adversely affects offspring health in the short-term and the long-term. Whether these effects occur in humans and influence population health is less clear. This Review explores evidence from intervention studies and observational studies that have used designs (such as family-based comparisons and Mendelian randomization) that might help improve understanding of the causal effects of maternal obesity in humans. Collectively, human studies provide evidence that maternal overweight and obesity is causally related to pregnancy complications, increased offspring weight and adiposity at birth, and the difficulties associated with delivery of large-for-gestational-age infants. The underlying mechanisms for these effects probably involve maternal and fetal dysregulation of glucose, insulin, lipid and amino acid metabolism. Some evidence exists that extreme maternal obesity (BMI ≥40 kg/m2) is causally related to a long-term increase in offspring adiposity, but further exploration of this relationship is needed. High gestational weight gain may result in a long-term increase in offspring adiposity if women are already overweight or have obesity at the start of pregnancy. To date, little high-quality human evidence exists that any of these effects are mediated by epigenetic mechanisms, but approaches to appropriately test this possibility are being developed.
Key Points
-
Maternal obesity is associated with considerable maternal and fetal metabolic perturbation
-
Prospective studies, including those with outcomes reported for first and second pregnancies and before and after bariatric surgery, suggest a causal association of maternal obesity with adverse pregnancy and perinatal outcomes
-
Evidence also exists that extreme maternal obesity (BMI ≥40 kg/m2) is causally related to increased adiposity of offspring during childhood and adulthood
-
A dose–response effect of maternal BMI (across the whole distribution of maternal BMI) on the adiposity of offspring during childhood and adulthood is not supported by current evidence
-
High gestational weight gain is weakly associated with adverse perinatal and long-term offspring outcomes; causal effects may be restricted to subgroups of the population
-
Nutritional experiences related to maternal adiposity in utero can have effects that persist into childhood, and these effects may be mediated by epigenetic modifications, but robust causal evidence is needed
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Kanagalingam, M. G., Forouhi, N. G., Greer, I. A. & Sattar, N. Changes in booking body mass index over a decade: retrospective analysis from a Glasgow Maternity Hospital. BJOG 112, 1431–1433 (2005).
American College of Obstetricians and Gynecologists. ACOG committee opinion number 315, September 2005. Obesity in pregnancy. Obstet. Gynecol. 106, 671–675 (2005).
Heslehurst, N. et al. Trends in maternal obesity incidence rates, demographic predictors, and health inequalities in 36,821 women over a 15-year period. BJOG 114, 187–194 (2007).
Heslehurst, N. et al. The impact of maternal BMI status on pregnancy outcomes with immediate short-term obstetric resource implications: a meta-analysis. Obes. Rev. 9, 635–683 (2008).
Nelson, S. M., Matthews, P., & Poston, L. Maternal metabolism and obesity: modifiable determinants of pregnancy outcome. Hum. Reprod. Update 16, 255–275 (2010).
Symonds, M. E., Sebert, S. P., Hyatt, M. A. & Budge, H. Nutritional programming of the metabolic syndrome. Nat. Rev. Endocrinol. 5, 604–610 (2009).
Barnes, S. K. & Ozanne, S. E. Pathways linking the early environment to long-term health and lifespan. Prog. Biophys. Mol. Biol. 106, 323–336 (2011).
Poston, L. Developmental programming and diabetes—the human experience and insight from animal models. Best Pract. Res. Clin. Endocrinol. Metab. 24, 541–552 (2010).
Remmers, F. & Delemarre-van de Waal, H. A. Developmental programming of energy balance and its hypothalamic regulation. Endocr. Rev. 32, 272–311 (2011).
Djelantik, A. A., Kunst A. E, van der Wal, M. F., Smit, H. A. & Vrijkotte, T. G. Contribution of overweight and obesity to the occurrence of adverse pregnancy outcomes in a multi-ethnic cohort: population attributive fractions for Amsterdam. BJOG 119, 283–290 (2012).
Villamor, E. & Cnattingius, S. Interpregnancy weight change and risk of adverse pregnancy outcomes: a population-based study. Lancet 368, 1164–1170 (2006).
Whiteman, V. E. et al. Interpregnancy body mass index changes and risk of stillbirth. Gynecol. Obstet. Invest. 72, 192–195 (2011).
Whiteman, V. E., McIntosh, C., Rao, K., Mbah, A. K. & Salihu, H. M. Interpregnancy BMI change and risk of primary caesarean delivery. J. Obstet. Gynaecol. 31, 589–593 (2011).
Maggard, M. A. et al. Pregnancy and fertility following bariatric surgery: a systematic review. JAMA 300, 2286–2296 (2008).
Smith, J. et al. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J. Clin. Endocrinol. Metab. 94, 4275–4283 (2009).
Aricha-Tamir, B., Weintraub, A. Y., Levi, I. & Sheiner, E. Downsizing pregnancy complications: a study of paired pregnancy outcomes before and after bariatric surgery. Surg. Obes. Relat. Dis. 8, 434–439 (2012).
Catalano, P. M. et al. The hyperglycemia and adverse pregnancy outcome study: associations of GDM and obesity with pregnancy outcomes. Diabetes Care 35, 780–786 (2012).
Hauguel, S., Desmaizieres, V. & Challier, J. C. Glucose uptake, utilization, and transfer by the human placenta as functions of maternal glucose concentration. Pediatr. Res. 20, 269–273 (1986).
Harmon, K. A. et al. Continuous glucose profiles in obese and normal-weight pregnant women on a controlled diet: metabolic determinants of fetal growth. Diabetes Care 34, 2198–2204 (2011).
Brizzi, P. et al. Lipoprotein metabolism during normal pregnancy. Am. J. Obstet. Gynecol. 181, 430–434 (1999).
Sattar, N. et al. Lipoprotein subfraction changes in normal pregnancy: threshold effect of plasma triglyceride on appearance of small dense low density lipoprotein. J. Clin. Endocrin. Metab. 82, 2483–2491 (1997).
Ramsay, J. E., et al. Maternal obesity is associated with dysregulation of metabolic, vascular, and inflammatory pathways. J. Clin. Endocrinol. Metab. 87, 4231–4237 (2002).
Robertson, A. F, Sprecher H. A review of human placental lipid metabolism and transport. Acta. Paediatr. Scand. 57 (Suppl. 183), 3–18 (1968).
de Benoist, B., Jackson, A. A., Hall, J. S. & Persaud, C. Whole-body protein turnover in Jamaican women during normal pregnancy. Hum. Nutr. Clin. Nutr. 39, 167–179 (1985).
Jackson, A. A. Measurement of protein turnover during pregnancy. Hum. Nutr. Clin. Nutr. 41, 497–498 (1987).
Thompson, G. N. & Halliday, D. Protein turnover in pregnancy. Eur. J. Clin. Nutr. 46, 411–417 (1992).
Willommet, L., Schutz, Y., Whitehead, R., Jéquier, E. & Fern, E. B. Whole body protein metabolism and resting energy expenditure in pregnant Gambian women. Am. J. Physiol. 263 E624–E631 (1992).
Chevalier, S., Marliss, E. B., Morais, J. A., Lamarche, M. & Gougeon, R. Whole-body protein anabolic response is resistant to the action of insulin in obese women. Am. J. Clin. Nutr. 82, 355–365 (2005).
Chevalier, S. et al. The greater contribution of gluconeogenesis to glucose production in obesity is related to increased whole-body protein catabolism. Diabetes 55, 675–681 (2006).
Duggleby, S. L. & Jackson, A. A. Relationship of maternal protein turnover and lean body mass during pregnancy and birth length. Clin. Sci. (Lond.) 101, 65–72 (2001).
Farley, D. M. et al. Placental amino acid transport and placental leptin resistance in pregnancies complicated by maternal obesity. Placenta 31, 718–724 (2010).
Jones, H. N., Jansson, T. & Powell, T. L. Full-length adiponectin attenuates insulin signaling and inhibits insulin-stimulated amino acid transport in human primary trophoblast cells. Diabetes 59, 1161–1170 (2010).
Wallace, J. M., Horgan, G. W. & Bhattacharya, S. Placental weight and efficiency in relation to maternal body mass index and the risk of pregnancy complications in women delivering singleton babies. Placenta 33, 611–618 (2012).
Garn, S. M., Hoff, K. & McCabe, K. D. Maternal fatness and placental size. Am. J. Clin. Nutr. 32, 277–279 (1979).
Roberts, K. A. et al. Placental structure and inflammation in pregnancies associated with obesity. Placenta 32, 247–254 (2011).
Challier, J. C et al. Obesity in pregnancy stimulates macrophage accumulation and inflammation in the placenta. Placenta 29, 274–281 (2008).
Basu, S. et al. Molecular phenotype of monocytes at the maternal–fetal interface. Am. J. Obstet. Gynecol. 205, 265 e1–e8 (2011).
Inskip, H. M. et al. Women's compliance with nutrition and lifestyle recommendations before pregnancy: general population cohort study. BMJ 338, b481 (2009).
Viswanathan, M. et al. Outcomes of maternal weight gain. Evidence Report/Technology Assessment No. 168 [online]. AHRQ Publication No. 08-E009. Rockville, MD: Agency for Healthcare Research and Quality. http://www.ahrq.gov/downloads/pub/evidence/pdf/admaternal/admaternal.pdf (2008).
Committee to reexamine IOM pregnancy weight guidelines, Food and Nutrition Board and Board on Children, Youth and Families. Weight gain during pregnancy: reexamining the guidelines (eds Rasmussen, K. M. & Yaktine A. L), (The National Academies Press, Washington, DC, 2009).
Thangaratinam, S. et al. Effects of interventions in pregnancy on maternal weight and obstetric outcomes: meta-analysis of randomised evidence. BMJ 344, e2088 (2012).
Crowther, C. A. et al. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N. Engl. J. Med. 352, 2477–2486 (2005).
Landon, M. B. et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N. Engl. J. Med. 361, 1339–1348 (2009).
Oteng-Ntim, E., Varma, R., Croker, H., Poston, L. & Doyle, P. Lifestyle interventions for overweight and obese pregnant women to improve pregnancy outcome: systematic review and meta-analysis. BMC Med. 10, 47 (2012).
Pettitt, D. J. & Knowler, W. C. Diabetes and obesity in the Pima Indians: a cross-generational vicious cycle. J. Obes. Weight Regul. 7, 61–75 (1988).
Ebbeling, C. B., Pawlak, D. B. & Ludwig, D. S. Childhood obesity: public-health crisis, common sense cure. Lancet 360, 473–482 (2002).
Taylor, P. D. & Poston, L. Developmental programming of obesity in mammals. Exp. Physiol. 92, 287–298 (2007).
Gillman, M. W. in A Life Course Approach to Chronic Disease Epidemiology 2nd edn Ch. 8 (eds Kuh, D. & Ben-Shlomo, Y.) 189–217 (Oxford University Press, Oxford, 2004).
Lawlor, D. A. The Society for Social Medicine John Pemberton Lecture 2011: developmental overnutrition—an old hypothesis with new importance? Int. J. Epidemiol. (in press).
Davey Smith, G., Leary, S., Ness, A. & Lawlor, D. A. Challenges and novel approaches in the epidemiological study of early life influences on later disease. Adv. Exp. Med. Biol. 646, 1–14 (2009).
Lawlor, D. A., Harbord, R. M., Sterne, J. A. C., Timpson, N. J. & Davey Smith, G. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat. Med. 27, 1133–1163 (2008).
Dabelea, D. et al. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes 49, 2208–2211 (2000).
Lawlor, D. A., Lichtenstein, P. & Långström, N. Association of maternal diabetes mellitus in pregnancy with offspring adiposity into early adulthood: sibling study in a prospective cohort of 280,866 men from 248,293 families. Circulation 123, 258–265 (2011).
Kral, J. G. et al. Large maternal weight loss from obesity surgery prevents transmission of obesity to children who were followed for 2 to 18 years. Pediatrics 118, e1644–e1649 (2006).
Whitaker, R. C., Wright, J. A., Pepe, M. S., Seidel, K. D. & Dietz, W. H. Predicting obesity in young adulthood from childhood and parental obesity. N. Engl. J. Med. 337, 869–873 (1997).
Lawlor, D. A. et al. Epidemiologic evidence for the fetal overnutrition hypothesis: findings from the Mater-university study of pregnancy and its outcomes. Am. J. Epidemiol. 165, 418–424 (2007).
Lawlor, D. A. et al. Exploring the developmental overnutrition hypothesis using parental–offspring associations and the FTO gene as an instrumental variable for maternal adiposity. PloS Med. 5, e33 (2008).
Lake, J. K., Power, C., Cole, T. J. Child to adult body mass index in the 1958 British birth cohort: associations with parental obesity. Arch. Dis. Child. 77, 376–381 (1997).
Patel, R. et al. Familial associations of adiposity: findings from a cross-sectional study of 12,181 parental–offspring trios from Belarus. PLoS ONE 6, e14607 (2011).
Subramanian, S. V., Ackerson, L. K. & Davey Smith, G. Parental BMI and childhood undernutrition in India: an assessment of intrauterine influence. Pediatrics 126, e663–e671 (2010).
Fleten, C. et al. Parental–offspring body mass index associations in the Norwegian Mother and Child Cohort Study: A family-based approach to study the role of the intrauterine environment in childhood adiposity. Am. J. Epidemiol. 176, 83–92 (2012).
Oken, E., Taveras, E. M., Kleinman, K. P., Rich-Edwards, J. W. & Gillman, M. W. Gestational weight gain and child adiposity at age 3 years. Am. J. Obstet. Gynecol. 196, 322.e1–322.e8 (2007).
Moreira, P., Padez, C., Mourão-Carvalhal, I. & Rosado, V. Maternal weight gain during pregnancy and overweight in Portuguese children. Int. J. Obes (Lond.) 31, 608–614 (2007).
Wrotniak, B. H., Shults, J., Butts, S. & Stettler, N. Gestational weight gain and risk of overweight in the offspring at age 7 y in a multicenter, multiethnic cohort study. Am. J. Clin. Nutr. 87, 1818–1824 (2008).
Fraser, A. et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 121, 2557–2564 (2010).
Oken, E., Rifas-Shiman, S. L., Field, A. E., Frazier, A. L. & Gillman, M. W. Maternal gestational weight gain and offspring weight in adolescence. Obstet. Gynecol. 112, 999–1006 (2008).
Mamun, A. A. et al. Associations of gestational weight gain with offspring body mass index and blood pressure at 21 years of age: evidence from a birth cohort study. Circulation 119, 1720–1727 (2009).
Hochner, H. et al. Associations of maternal prepregnancy body mass index and gestational weight gain with adult offspring cardiometabolic risk factors: the Jerusalem Perinatal Family Follow-up Study. Circulation 125, 1381–1389 (2012).
Lawlor, D. A. et al. Maternal and offspring adiposity-related genetic variants and gestational weight gain. Am. J. Clin. Nutr. 94, 149–155 (2011).
Lawlor, D. A., Lichtenstein, P., Fraser, A. & Långström, N. Does maternal weight gain in pregnancy have long-term effects on offspring adiposity? A sibling study in a prospective cohort of 146,894 men from 136,050 families. Am. J. Clin. Nutr. 94, 142–148 (2011).
Pitkin, R. M. Nutritional support in obstetrics and gynecology. Clin. Obstet. Gynecol. 19, 489–513 (1976).
Gillman, M. W. Gestational weight gain: now and the future. Circulation 125, 1339–1340 (2012).
Wen, X. et al. Components of gestational weight gain and postpartum weight retention [abstract 60-LB-P]. In The 29th Annual Scientific Meeting of The Obesity Society. Late-Breaking Abstracts (online). (2011).
Alfaradhi, M. Z. & Ozanne, S. E. Developmental programming in response to maternal overnutrition. Front. Genet. 2, 27 (2011).
Rogers, I. S. et al. Associations of size at birth and dual-energy X-ray absorptiometry measures of lean and fat mass at 9 to 10 y of age. Am. J. Clin. Nutr. 84, 739–747 (2006).
Mathers, J. C., Strathdee, G. & Relton, C. L. Induction of epigenetic alterations by dietary and other environmental factors. Adv. Genet. 71, 3–39 (2010).
Seki, Y., Williams, L., Vuguin, P. M. & Charron, M. J. Minireview: epigenetic programming of diabetes and obesity: animal models. Endocrinology 153, 1031–1038 (2012).
Tobi, E. W. et al. DNA methylation differences after exposure to prenatal famine are common and timing- and sex-specific. Hum. Mol. Genet. 18, 4046–4053 (2009).
Waterland, R. A. et al. Season of conception in rural Gambia affects DNA methylation at putative human metastable epialleles. PLoS Genet. 6, e1001252 (2010).
Bouchard, L. et al. Leptin gene epigenetic adaptation to impaired glucose metabolism during pregnancy. Diabetes Care 33, 2436–2441 (2010).
Godfrey, K. M. et al. Epigenetic gene promoter methylation at birth is associated with child's later adiposity. Diabetes 60, 1528–1534 (2011).
Relton, C. L. et al. DNA methylation patterns in cord blood DNA and body size in childhood. PLoS ONE 7, e31821 (2012).
Relton, C. L. & Davey Smith, G. Two-step epigenetic Mendelian randomization: a strategy for establishing the causal role of epigenetic processes in pathways to disease. Int. J. Epidemiol. 41, 161–176 (2012).
Groom, A. et al. Postnatal growth and DNA methylation are associated with differential gene expression of the TACSTD2 gene and childhood fat mass. Diabetes 61, 391–400 (2012).
Relton, C. L. & Davey Smith, G. Is epidemiology ready for epigenetics? Int. J. Epidemiol. 41, 5–9 (2012).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article and reviewed and/or edited the manuscript before submission. D. A. Lawlor and S. M. Nelson provided a substantial contribution to discussion of the content and D. A. Lawlor wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lawlor, D., Relton, C., Sattar, N. et al. Maternal adiposity—a determinant of perinatal and offspring outcomes?. Nat Rev Endocrinol 8, 679–688 (2012). https://doi.org/10.1038/nrendo.2012.176
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2012.176
This article is cited by
-
Association of maternal pre-pregnancy body mass index with birth weight and preterm birth among singletons conceived after frozen-thawed embryo transfer
Reproductive Biology and Endocrinology (2022)
-
Maternal diet, nutritional status and infant birth weight in Malaysia: a scoping review
BMC Pregnancy and Childbirth (2022)
-
Epigenetic regulation of pediatric and neonatal immune responses
Pediatric Research (2022)
-
Higher maternal adiposity reduces offspring birthweight if associated with a metabolically favourable profile
Diabetologia (2021)
-
Associations between maternal serum HDL-c concentrations during pregnancy and neonatal birth weight: a population-based cohort study
Lipids in Health and Disease (2020)