Abstract
Rabies is a life-threatening neglected tropical disease: tens of thousands of cases are reported annually in endemic countries (mainly in Africa and Asia), although the actual numbers are most likely underestimated. Rabies is a zoonotic disease that is caused by infection with viruses of the Lyssavirus genus, which are transmitted via the saliva of an infected animal. Dogs are the most important reservoir for rabies viruses, and dog bites account for >99% of human cases. The virus first infects peripheral motor neurons, and symptoms occur after the virus reaches the central nervous system. Once clinical disease develops, it is almost certainly fatal. Primary prevention involves dog vaccination campaigns to reduce the virus reservoir. If exposure occurs, timely post-exposure prophylaxis can prevent the progression to clinical disease and involves appropriate wound care, the administration of rabies immunoglobulin and vaccination. A multifaceted approach for human rabies eradication that involves government support, disease awareness, vaccination of at-risk human populations and, most importantly, dog rabies control is necessary to achieve the WHO goal of reducing the number of cases of dog-mediated human rabies to zero by 2030.
Similar content being viewed by others
Introduction
Rabies is a zoonotic disease caused by viruses of the Lyssavirus genus (in the family Rhabdoviridae of the order Mononegavirales) that was first described in the 4th century BC1. Rabies virus (RABV), the prototype virus of the Lyssavirus genus (Table 1), is by far the most common causative agent of rabies2 and is most readily transmitted by the bite of an infected mammal (Fig. 1). Dog-transmitted rabies causes >99% of the human cases reported. Both animal and human rabies are entirely preventable through vaccination, and the first efficacious rabies vaccines for human use were developed in the 19th century. However, in the 21st century, the virus is still enzootic (that is, endemic in animals) in many regions of the world, and human rabies remains one of the most serious and distressing diseases and an important threat to public health3. Indeed, when an individual with rabies develops symptoms, the disease is nearly always fatal4. Rabies is often considered a disease of poverty, ignorance and, in some circumstances, misinformation5.
a | The rabies virus (RABV) virion is bullet-shaped, with a plasma membrane covered in homotrimers of type 1 transmembrane glycoprotein with peplomers (glycoprotein spikes). The ∼12 kb negative-sense RNA genome of lyssaviruses encodes five viral proteins: the nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G) and large RNA polymerase protein (L). Within the virion, the genomic RNA is tightly encapsidated with N and P to form a ribonucleoprotein (RNP), which is closely associated with a viral RNA polymerase complex consisting of P and L. The RNP forms a helix that is encapsidated with M, which in turn connects the RNP and the lipid bilayer membrane envelope. b | Hippocampal neurons of a laboratory mouse infected with street RABV (that is, obtained from a naturally infected dog) that were isolated during the clinical phase of the disease. Despite clear neuronal infection (shown by the presence of N, labelled in red), the structural integrity of the neurons seems to be preserved. Green labelling denotes the neuronal marker tubulin β3 chain; blue labelling indicates cell nuclei. c | A cross section of the virion demonstrates the distinct G peplomers (arrow), plasma membrane (arrowhead) and RNP core (yellow arrow). d | Longitudinal view of the virion, highlighting the bullet shape. Here a virion is being engulfed by a cell, presumably through endocytic mechanisms. *Indicates the cell from which the virion is budding. Parts c and d are reproduced with permission from Kati Franzke.
RABV enters peripheral nerves at the synapse level at the site of the bite and is transported to neurons in the central nervous system (CNS); the virus then replicates and causes cerebral damage. Rabies can manifest in two classical forms (furious and paralytic) with a range of symptoms, but ultimately leads to coma and death. The priority for reducing the burden of human rabies is controlling dog rabies, especially in free-roaming community dogs6–9. Rabies elimination was achieved in domestic dogs in North America, Europe and some countries of Latin America as well as in wildlife species in Europe and Canada10. The subsequent substantial reduction in cases of human rabies in such regions advocates for the ‘One Health’ approach to rabies control (that is, a collaborative and transdisciplinary approach at the local, national and global level that aims to achieve optimal health outcomes by recognizing that the health of people is connected to the health of animals and the environment)11. With this in mind, the WHO, World Organisation for Animal Health (OIE) and Food and Agriculture Organization of the United Nations (FAO) have set the target to eliminate dog-transmitted human rabies in endemic countries by 2030 (Ref. 12). This Primer summarizes the epidemiology of RABV; global efforts to diagnose, control and eliminate the transmission of RABV by terrestrial animals; and the mechanisms and pathophysiology of RABV infection. Conundrums regarding treatment options and both existing and potential future prophylactic tools to combat infection of the CNS are discussed, along with initiatives aimed at the global elimination of this deadly zoonotic virus.
Epidemiology
Lyssaviruses and their hosts
Since the 1970s, methods of viral characterization have revealed the extensive diversity of the Lyssavirus genus13 (Table 1), and the taxonomy of this genus continues to evolve, owing to enhanced surveillance and sequencing technologies. Most Lyssavirus spp. are maintained by chiropteran hosts (the only mammals capable of true flight; that is, bats). The reservoir hosts and geographical range of some of these viruses, such as the European bat lyssaviruses (EBLVs) in Europe, are quite well documented, but similar information for many others is lacking, owing to the recovery of only small numbers of isolates14. By contrast, RABV is maintained in most parts of the world by mesocarnivores (animals whose diet consists of 50–70% meat), including dogs, foxes, raccoon dogs, raccoons, mongooses and skunks3. Exceptions include the maintenance of RABV in multiple bat species (only in the Americas) and the reported role of a herbivore, the kudu, as an RABV reservoir in Namibia15. For all Lyssavirus spp., other mammals, including humans, cattle and equids, represent cases of cross-species transmission (or spillover; that is, the ability of a virus to infect a member of a new host species)3. Although such spillover infections normally result in ‘dead-end’ infections with no further spread to other hosts, neuroinvasion leading to clinical disease can still occur.
RABV is estimated to be responsible for hundreds of thousands of cases of animal rabies annually, and the application of sequencing methods to this extensive pool of data has enabled the investigation of the epidemiology and evolution of RABV and has provided insights into the detailed phylogeography of disease outbreaks16. Such studies identify natural landscape features such as mountain ranges and waterways as frequent barriers to epizootic spread, whereas man-made roads and human-mediated movement of animals, often accompanying large-scale human migrations, can facilitate disease dispersion; such information is valuable to control programmes17.
RABV probably evolved from a bat-associated progenitor18, and several host-switching events yielded a complex pattern of RABV variants. Worldwide, seven main lineages of RABV are recognized13,19; within each lineage, multiple variants, each associated with a particular mammalian host and geographical range, have emerged. The most widespread lineage is found in several continents (Europe, the Americas, Africa and Asia), and it is maintained by dog populations in many countries and by several wildlife species (for example, foxes, raccoon dogs, skunks and jackals)20. Throughout the Americas, many bat-associated RABV variants persist21, including one harboured by the vampire bat, which is an important source of disease for humans and domestic livestock in Latin America.
Disease
Large-scale control programmes have eliminated dog rabies in Europe, the United States and Canada and substantially reduced its prevalence in Latin America, with a concomitant decrease in cases of human rabies22. However, as dog rabies is controlled, the existence of sympatric (occurring in the same area) wildlife rabies reservoirs often becomes more-evident, and sylvatic rabies (affecting wildlife) has been identified in countries that were historically considered free of rabies23. In Europe, oral vaccination campaigns have demonstrated the feasibility of eliminating rabies from wildlife reservoirs, but the continued circulation of Lyssavirus spp. in bat hosts with an aerial lifestyle will challenge future global rabies eradication efforts, owing to difficulties in the development and administration of vaccines to these highly mobile, poorly accessible species3.
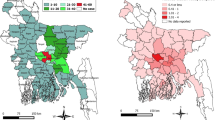
Globally, human rabies is considered a neglected zoonosis, especially in resource-limited countries in Africa and Asia where the disease burden falls disproportionately and mortality is substantial24 (Fig. 2). It has been estimated that someone dies from rabies in a rabies-endemic country every 10–20 minutes and that 40–50% of fatalities are in children <15 years of age3. Owing to their small size and frequent interactions with domestic or free-roaming dogs, children are at a higher risk of being bitten, not only on their extremities but also on the head, which leads to shorter incubation times. Thus, in endemic countries, rabies should be considered a neglected paediatric disease25.
Dog-transmitted human rabies caused by the rabies virus (RABV) is responsible for most human fatalities. The map does not reflect very low numbers of cases of human rabies resulting from exposure to RABV via wildlife reservoirs in areas where dog rabies has been eliminated (for example, North America). Figure reprinted from World Health Organization. Rabies: epidemiology and burden of disease. http://www.who.int/rabies/epidemiology/en/ (date accessed: 17/10/2017) (Ref. 174).
The estimated 60,000 human deaths annually worldwide3,26 do not capture the true burden of this disease. Predictions suggest that as many as 20,000 lives are lost annually in India alone26. Lack of surveillance27 due to limited health-care services, such as laboratory capabilities, qualified personnel and medical and veterinary infrastructures, results in under-reporting of human cases, which is further compounded by cultural, religious and social taboos28. Furthermore, in humans, rabies can present with symptoms that are common to other clinically indistinguishable conditions (for example, Guillain–Barré syndrome and cerebral malaria)29; consequently, without confirmation of the diagnosis using established laboratory protocols30, rabies might be misdiagnosed. These inaccuracies in collating empirical data of disease incidence can discourage policymakers from considering rabies as a high-priority disease26,31.
Mechanisms/pathophysiology
Host infection
The main mode of RABV transmission is through exposure of broken skin to the saliva of an infected animal. Exposures that can lead to infection can vary from severe bites, most often by a dog, to superficial skin lesions. These lesions are often described as cryptic infection because the exposure goes unrecognized and unreported32, and most frequently result from contact with bats. Other potential transmission routes include organ transplantation from donors with undiagnosed or misdiagnosed rabies, which caused several cases of rabies in transplant recipients33–36. Infection via the aerosol route has also been rarely reported37–39 and probably depends on efficient viral excretion in the saliva and exposed ocular or nasal mucosa37,40.
In vitro, lyssaviruses can infect most cell types, although the underlying molecular interactions are not completely understood. In the host, lyssaviruses generally infect peripheral nerves (sometimes also muscle cells) at the motor endplate of neuromuscular junctions or at other innervated tissues (Fig. 3). After receptor-mediated entry, the virus is contained in endosomal transport vesicles41,42 and retrogradely transported along axons via microtubules43–47. In the neuronal soma, the ribonucleoproteins (RNPs), which are complexes formed by the viral RNA, nucleoproteins and phosphoproteins, are released into the cytoplasm from the vesicles48 and primary transcription occurs, leading to the production of viral proteins. With the accumulation of viral proteins, cytoplasmic inclusion bodies are formed, which are sites of viral RNA synthesis (secondary transcription)49. From replication sites in inclusion bodies, newly generated RNPs are transported to postsynaptic membranes, where the new virions are assembled and trans-synaptically transmitted50 to next-order neurons in a viral glycoprotein-dependent manner51. This process includes budding in the synaptic cleft and subsequent receptor-mediated entry at presynaptic membranes (Fig. 3). The virus eventually reaches the brain; what brain area is infected is determined by what motor neurons innervate the site of entry. From the brain, the virus spreads to the salivary glands and is intermittently excreted in the saliva, ready to be transmitted to another host. Local symptoms start once the virus, after disseminating in the CNS, centrifugally spreads and reaches the dorsal root ganglia corresponding to the nerves at the site of entry; in the dorsal root ganglia, viral replication causes inflammation, which triggers pain, paraesthesia (a tingling sensation) and/or pruritus (itch)52. The virus could also reach peripheral nerves; inflammation in these nerves (caused by viral replication) could result in weakness in paralytic rabies. The mechanisms underlying the symptoms that manifest during the acute neurological phase are not completely understood.
At the motor endplate, after receptor-mediated endocytosis at the presynaptic membrane (step 1), the virion travels by retrograde axonal transport towards the neuron soma (step 2), probably in a dynein-dependent manner, as suggested by transport directionality43, measured transport velocities59 and co-transport with the dynein-motor transported tumour necrosis factor receptor superfamily member 16 (p75NTR)42. In the soma, the ribonucleoprotein (RNP) is released from the endocytic vesicle (step 3), and primary transcription produces 5′-capped and polyadenylated (AAA) viral mRNAs, a process driven by the virion-associated large RNA polymerase protein (L) and the phosphoprotein (P) (step 4). Viral proteins are translated on free ribosomes in the cytoplasm with the exception of the G protein, which is translated through the endoplasmic reticulum (ER) Golgi network (step 5). In later phases, the matrix protein (M) shifts the activity of the RNA polymerase complex (enzymatic subunit L and cofactor P) from transcriptase to replicase175,176 and replication of full-length RNA genomes (step 6) occurs. The negative-sense RNA genome is first transcribed in full-length, positive-sense RNA strands, which are subsequently transcribed into full-length, negative-sense RNA strands. Both replication products, full-length positive-sense and negative-sense RNA genomes, start to be assembled with the nucleoprotein (N) to form new RNPs while the replication process is ongoing and can either be used as templates for further RNA synthesis (full-length genome replication and mRNA transcription) or encapsidated into progeny virions. Assembly and budding of virions is essentially mediated by M177, but how M is recruited to budding sites is unknown. M is located in the cytoplasm, inclusion bodies (not shown) and the nucleus and accumulates at cellular membranes178. Oligomerization of M at membranes could increase the binding affinity of M for the lipid bilayer and could play a major part in membrane curvature, envelopment of RNPs and virus egress (step 7). A late domain (a protein interaction motif that has a role in the budding process) in M increases the efficiency of the virus egress179. The glycoprotein (G) on the surface of virions is required for receptor binding and subsequent pH-dependent membrane fusion of the endocytic vesicle180. G is transported to budding sites through the secretory pathway, which includes translation at the rough ER and transport through the Golgi apparatus. During surface transport, G is glycosylated181 and forms homotrimers that are incorporated into budding virions182. nAChR, nicotinic acetylcholine receptor; NCAM, neural cell adhesion molecule; RABV, rabies virus. Figure adapted with permission from Ref. 182, Macmillan Publishers Limited.
Receptors and neuroinvasion
The nicotinic acetylcholine receptor (nAChR), neuronal cell adhesion molecule (NCAM) and tumour necrosis factor receptor superfamily member 16 (TNFRSF16; also known as p75NTR) have been proposed to be involved, to a greater or lesser degree, in RABV cell entry53–55 and, therefore, are considered as RABV receptors (Fig. 3). However, there is conflicting evidence on the role of p75NTR56, and other potential receptors have also been suggested57. p75NTR co-internalization and subsequent retrograde axonal co-transport with RABV have been directly shown by live-cell imaging in sensory dorsal root ganglion neurons42. Nevertheless, how RABV enters neurons at peripheral inoculation sites has yet to be completely defined. Within neuromuscular junctions, the presence of nAChR at, or close to, the postsynaptic membranes of muscle cells might support the infection of muscle cells before neuronal infection. Virus amplification (due to replication) in muscle cells and the subsequent budding of progeny virions in the synaptic cleft could increase the efficiency of neuroinvasion through receptors at presynaptic axonal membranes. However, nAChR could also act as an attachment receptor that could concentrate extracellular virus in the synaptic cleft for presentation to receptors such as NCAM and p75NTR on the presynaptic membranes of motor neurons57 (Fig. 3). Finally, RABV could directly bind to neuronal receptors and infect the neuron independent of nAChR binding or muscle cell infection. In fact, whether one or more of these receptor-mediated pathways occur and whether infection of non-neuronal cells at peripheral sites contributes to the efficiency of neuroinvasion and disease development remains controversial. In addition to neuromuscular junctions, sensory neurons could also serve as entry routes, as RABV has been detected in sensory nerves58. Both retrograde and anterograde axonal transport have been observed in cultivated dorsal root ganglion neurons44,59 (Supplementary information S1 (video)), and in vivo anterograde trans-synaptic transfer in sensory neuronal circuits has been demonstrated60.
Immune response and host manipulation
RABV virulence resides in its ability to escape the host's immune response while retaining the viability of infected neurons. The concept of the CNS as an immune-privileged cellular environment has been challenged by the discovery that peripheral immune cells can cross the intact blood–brain barrier and that CNS neurons and glia can regulate macrophage and lymphocyte responses. Thus, it must be assumed that RABV replication in the CNS occurs in a fully immunocompetent cellular environment. However, in experimental models, attenuated and pathogenic RABV strains induce different inflammatory markers and other host parameters61,62, and virus clearance from the brain, which correlates with the level of B cell infiltration and antibody production in the CNS63,64, is reduced in models infected with pathogenic RABV strains. These findings support the hypothesis that adaptive immune responses are impaired in natural RABV infections.
Enhancing the permeability of the blood–brain barrier to the infiltration of immune cells is considered necessary for successful virus clearance, and enhanced permeability increased the clearance of attenuated RABV from the CNS in experimental models65. Consistent with the findings in the canine model that infected animals develop limited immune responses66, wild-type RABV infection does not increase the permeability of the blood–brain barrier67. The permeability of the blood–brain barrier remains unaltered in dogs and patients in both furious and paralytic rabies68 during early-stage clinical disease, as demonstrated by MRI (in humans and dogs)52,69 and diffusion tensor imaging (in dogs). However, the permeability of the blood–brain barrier is considered one of the main reasons for the rare survival of patients with clinical rabies and might be a potential target for future treatments70. Nevertheless, whether the diminished immune response in the brain of patients infected by wild-type lyssaviruses is the result of a reduced permeability of the blood–brain barrier or the virus-dependent inhibition of immune cell infiltration to the CNS remains to be investigated. In any case, virus-neutralizing antibodies (VNAs), which might be detected in the acute neurological phase of naturally acquired RABV infection in humans, are considered to be unable to prevent a fatal outcome of the infection.
Immune evasion. Numerous mechanisms have been proposed to explain how RABV can evade or counteract the host's defence strategies71,72, and RABV replication in the CNS seems to be characterized by limited pathogen recognition and immune reactions (Fig. 4). Type I interferon is the main mediator of antiviral innate immune responses, but RABVs seem to be poor inducers of type I interferon62, and such dampened type I interferon initial responses at the virus peripheral entry sites might only partially eliminate replication.
Viral infection triggers numerous signalling cascades, including the induction of the genes encoding type I interferons and chemoattractive and inflammatory responses, resulting in an antiviral environment and efficient innate immune responses182,183. However, the rabies virus (RABV) has developed several different mechanisms to counteract these innate signalling cascades184–188. Expression of antiviral cytokines is activated by the transcription factor nuclear factor-κB (NF-κB). NF-κB is a homodimeric or heterodimeric complex of various proteins, of which the heterodimeric complex composed of transcription factor p65 (p65) and NF-κB p50 subunit (p50) seems to be the most abundant. A variant of p65, p43, which stabilizes p50-containing dimers and promotes their activation, is inhibited by RABV matrix protein (M)189,190. The innate immune system recognizes intracellular RABV replication through internal receptors such as probable ATP-dependent RNA helicase DDX58 (also known as retinoic acid-inducible gene 1 protein (RIG-1))186,191,192, which promotes type I interferon regulator factor 3 (IRF3) phosphorylation and IFN transcription. RABV nucleoprotein (N) interferes with RIG-1 activation186, whereas RABV phosphoprotein (P) inhibits IFN induction by blocking IRF3 phosphorylation. The binding of interferon to its receptors triggers the dimerization, phosphorylation, association with interferon regulatory factor 9 (IRF9) and nuclear import of signal transducer and activator of transcription 1 (STAT1) and STAT2 heterodimers, which promote the transcription of interferon stimulated genes (ISGs). The nuclear import of STAT1-STAT2 is prevented by P193, and, within the nucleus, an N-terminal truncated phosphoprotein form of P (P3) interferes with STAT1- STAT2-dependent transcription193. P also inhibits type II IFN-γ-dependent signalling through STAT1 homodimers (not shown)193. IFNAR, IFN-α and IFN-β receptor; JAK1, tyrosine-protein kinase JAK1; ssRNA, single-stranded RNA; TYK2, nonreceptor tyrosine-protein kinase TYK2. Figure adapted with permission from Ref. 182, Macmillan Publishers Limited.
Several mechanisms could enable RABV to escape or manipulate adaptive immune surveillance in the nervous system. Upregulation of FASLG expression73 and subsequent activation of Fas ligand could induce apoptosis of antigen-activated T cells and contribute to the termination of the immune response. The expression of HLA class I histocompatibility antigen, α chain G (HLA-G), a nonclassical, immunosuppressive HLA74,75, and upregulation of the expression of programmed cell death 1 ligand 1 (PDL1; also known as B7H1) could inhibit T cell proliferation and apoptosis73,74,76.
Neuronal survival. Although the clinical manifestations of RABV infection can be severe, both macroscopic and histopathological changes are often quite mild. RABV replication is self-limited by suboptimal transcription signals and large RNA polymerase protein expression to avert cytopathogenic effects77, thereby supporting the maintenance of vital CNS functions and intraneuronal transport machineries. The viral load in the brain is greater in dogs that develop furious rabies than in dogs with paralytic rabies, and in dogs with paralytic rabies the viral intracellular spread is reduced69,78. In paralytic dog rabies, axonal integrity is also disrupted at the level of the brainstem, impairing the viral spread to each hemisphere78,79. Similar data for human infection are lacking.
Viral infections cause cellular stress responses that modulate the host's gene expression by affecting the regulation of mRNA translation, localization and degradation while promoting viral transcription, replication and translation. One stress response induced by RABV infection is the assembly of both viral and host mRNA complexes into dynamic cytoplasmic structures known as stress granules80. Although an antiviral role for stress granules has been suggested81, it is not clear whether the formation of stress granules is directly stimulated by RABV as a means to control the number of viral transcripts and limit cytopathogenic effects and consequently cellular damage80.
Furthermore, RABV could also directly modulate neuronal cell death, as it has been hypothesized that a PDZ-binding domain in the cytoplasmic tail of glycoprotein could be involved in the regulation of neuronal survival by binding to microtubule-associated serine/threonine protein kinase 2 (MAST2)82. The complex formed by MAST2 and phosphatidylinositol 3,4,5-trisphosphate 3-phosphatase and dual-specificity phosphatase and tensin homologue (PTEN) can prevent or reduce neurotrophin-dependent apoptosis inhibition. Thus, the binding of glycoprotein to MAST2 could impair the pro-apoptotic function of the MAST2–PTEN complex. In addition, a high affinity of glycoprotein to the PDZ-binding domain of tyrosine-protein phosphatase non-receptor type 4 (PTPN4) correlated with virulence attenuation83,84. Accordingly, a model has been proposed in which the binding of viral proteins to the PDZ-binding domains of host proteins could represent a decisive virulence marker82. This model is supported by the following observations: the strong binding of glycoprotein from an attenuated RABV to the PDZ-binding domain of PTPN4 increased cell death; pathogenicity and apoptosis induction by RABV inversely correlate85; and, in histopathological findings, neuronal apoptosis does not have an important role in human rabies-associated encephalitis86. Evidence of an autophagic response has been observed in wild-type RABV infection and this response might be influenced by matrix protein expression87,88. Indications of an autophagic response include minimal pathology with minimal or no inflammation89; signs (in immunohistochemistry tests) of mitochondrial outer membrane permeabilization (but not of apoptosis) in post-mortem brain examination90; and preserved brain and spinal cord function despite abundant viral particles, as in both furious and paralytic rabies coma occurs only at the pre-terminal phase of the disease, and in paralytic rabies electrophysiological and pathological measurements of peripheral nerves show either axonopathy or myelinopathy91. Apoptosis seems to be more-prevalent in infections with attenuated or fixed viruses (a fixed virus is a cultured strain in which the incubation period and virulence have been stabilized)92.
Diagnosis, screening and prevention
Clinical disease
Clinical descriptions have reported many manifestations of rabies93. It has been demonstrated that symptoms do not correlate with the site of viral replication in the brain69,89. Two classical forms of rabies are generally recognized: furious (also called encephalitic) and paralytic. The factors that determine the development of either form remain ill-defined69,94, but each form may be characterized by specific symptoms, although case definition can typically be established with certainty only when the disease reaches the acute neurological phase (Fig. 5). Incubation periods can vary considerably, although most patients develop symptoms 20–90 days after exposure3. The cause of this variation is probably multifactorial and can include the site of virus entry and the viral load, the species and strain of the infecting virus and the immunological competence of the host. Initial symptoms are triggered by the viral replication in the dorsal root ganglia and include pain, paraesthesia and/or pruritus52. After a short prodromal phase, the patient enters the acute neurological phase of the disease, which is characterized by clinical manifestations. Furious rabies typically presents during this phase with intermittent agitation, hypersalivation and hydrophobia (a flight response to fluids) (Fig. 5). By contrast, the paralytic form presents with muscle weakness and, eventually, paralysis and generally a longer acute neurological phase than furious rabies94. Both forms of rabies lead to coma and death.
The clinical presentation of rabies can vary substantially between patients, which makes diagnosis on the basis of observed symptoms problematic. In individuals with furious rabies, hyperactivity manifests during the prodromal phase and can alternate with confusion or agitation once the patient enters the acute neurological phase. In patients with paralytic rabies, during the prodromal phase the patient is fully alert, but confusion or drowsiness can intermittently occur during the acute neurological phase, although not as prominently as seen in the furious form of the disease. The course of clinical disease and some clinical manifestations can be observed in both paralytic and furious rabies; thus, it is generally not possible to differentiate between the two forms until the acute neurological phase. The average time from the onset of symptoms to death is 7.8 days in furious rabies and 11 days in paralytic rabies94.
Diagnostic evaluation
The specific clinical symptoms of hydrophobia or aerophobia (fear of drafts or fresh air) provide a strong indication of RABV infection. However, only the results of laboratory tests can provide a definitive diagnosis of rabies30, as other differential diagnoses are possible (Fig. 6). In fact, the absence of pathognomonic symptoms means that fluctuating consciousness, phobic or inspiratory spasms and autonomic stimulation can indicate infection with RABV. Some presentations can commonly occur in paralytic rabies or other atypical forms, caused by infection with dog or bat variants of RABV90. Although early diagnosis is important, the lack of tools to enable survival once the patient develops clinical disease means that, regardless of the timeliness of the diagnosis, the prognosis is extremely poor and will almost certainly result in death if RABV infection is confirmed29.
The diagnosis of rabies cannot be made solely on the basis of clinical presentation, as several other infectious or noninfectious aetiologies are possible. The geographical context can provide valuable information during the evaluation of a patient presenting with rabies-like symptoms. It is also important to establish whether the patient had contact with potentially infected animals and, if this is the case, what kind. Rarely, rabies can occur after interaction with less commonly reported animal hosts, such as rodents. The circumstance of the bite can provide information on the category of exposure that occurred. Several autoimmune encephalitides are thought to be quite common in rabies-endemic areas and, therefore, should be considered alongside other potential aetiologies, such as bacterial infections (for example, Mycobacterium tuberculosis, Clostridium tetani or Rickettsia spp.), other viral infections (for example, herpes simplex or enteroviruses) and Guillain–Barré syndrome. Parasitic infections (for example, Plasmodium spp.) are another possibility, as rabies could be misdiagnosed as cerebral malaria in the absence of laboratory evaluation. Importantly, the exclusion of other rabies mimics and confirmation of diagnosis can cause delay in management planning.
Ante-mortem diagnostic testing. In endemic settings, ante-mortem diagnostic tests are not routinely conducted, as facilities often lack the necessary resources. This situation is unfortunate, as ante-mortem confirmation of infection enables appropriate patient care and alerts caregivers to potential risks of RABV transmission. The most useful samples for ante-mortem diagnosis are obtained from nuchal (nape) skin biopsies and saliva (Table 2). Live virus has been occasionally isolated from human saliva93,95; in dog models, high viral titres can be intermittently detected in secretions (saliva) up to 10 days before the onset of clinical disease96. Skin specimens can be tested for the presence of virus antigen (that is, the nucleoprotein) in neurons in close proximity to hair follicles, and both skin and saliva samples can be tested for the presence of viral RNA by reverse transcription PCR (RT-PCR)97. Indeed, a number of RT-PCR-based and quantitative RT-PCR (RT-qPCR)-based assays have been proved rapid, highly sensitive and specific and are increasingly used for ante-mortem RABV testing98–101. Other biological samples that could be used for RT-PCR testing include urine and extracted hair follicles102; as detecting the virus, especially in the early stages of disease, is challenging, increasing the number of tested samples can improve the probability of identifying the virus, if present. Ante-mortem testing of corneal impressions is of limited value and is no longer recommended30, and other diagnostic methods such as viral culture are not sufficiently sensitive or timely for diagnostic application and require tissue culture facilities that are often unavailable. Although saliva can be obtained quite easily, because of the intermittent secretion of the virus into this fluid the diagnostic findings should be interpreted with caution, and multiple samples collected daily might be required to confirm virus presence30,97,102. However, it must be stressed that laboratory tests can only confirm infection: a negative ante-mortem test result does not necessarily rule out infection.
Humoral immune responses to RABV usually arise in the serum or the cerebral spinal fluid (CSF) at late stages of infection103. Nevertheless, although it is not directly considered a diagnostic test, the detection of virus-specific antibodies against viral antigens in the serum of unvaccinated individuals, or in the CSF of both vaccinated and unvaccinated individuals, can be of diagnostic value. VNAs in the CSF were detected in 20 (22.4%) of 89 clinically suspected cases of rabies in India104. By contrast, antibodies in the CSF were not detected in 31 Thai patients with rabies and 43 unvaccinated patients who were infected with dog RABV variants from Cambodia, Madagascar and Senegal52. Individuals who develop VNAs in the serum or CSF early during the disease course are considered potentially favourable candidates for aggressive management105. Of note, most documented survivors of rabies were diagnosed solely by the presence of VNAs in the CSF or serum, and no viral RNA or antigens were detected in biological samples (Table 3).
Post-mortem diagnostic testing. Brain tissue is the specimen of choice for post-mortem diagnosis29. Historically, the presence of Negri bodies (intracellular accumulations of RABV particles), first described in 1903, was considered indicative of rabies, but this test has now been replaced by more-sensitive and specific methods98,106. However, the detection of Negri bodies upon autopsy can warrant further testing for RABV infection. The fluorescent antibody test (FAT) to detect virus antigen in brain impressions is the diagnostic test recommended by both the WHO and the OIE, and it yields reliable results in fresh specimens in <4 hours107 (Table 2). The sensitivity of the FAT depends on the quality of the specimen and the degree of autolysis of the brain tissue. However, the FAT requires expensive equipment; thus, it is often unavailable in resource-limited settings. To overcome this limitation, a direct immunohistochemical test (dRIT) that uses a mixture of biotinylated antibodies and light microscopy has been generated; dRIT is cost-effective and can rapidly detect virus antigen in fresh brain material108 (Table 2). Extensive validation of dRIT, through evaluation against the gold-standard FAT and comparison with other molecular-based methods, has been completed, and this procedure, or variations thereof109, is expected to increase the availability of antigen detection methods to endemic regions and could enable reliable primary diagnosis in resource-limited areas27. Further antigen detection tests, including lateral flow devices (LFDs), have been developed (Table 2) but lack validation. Depending on the sensitivity and specificity of routine diagnostic tests (such as the FAT and RT-PCR), a confirmation of results could be required by a validated secondary method, such as the rabies tissue culture isolation test (RTCIT) for virus isolation or the mouse inoculation test (MIT), in which brain samples are inoculated intracranially in laboratory mice, in areas where tissue culture facilities for RTCIT are unavailable107 (Table 2).
Molecular diagnostic assays that can detect the RNA of many lyssaviruses are available110,111 (Table 2) and will replace the cumbersome existing secondary tests for rabies diagnosis in animals. Compared with the FAT, molecular methods are less prone to subjective interpretation and have the added benefit that they can identify the virus species, a useful tool in cases with undefined history of potential exposure to RABV. Molecular assays are now recognized by the OIE; however, these tests need to be performed in well-controlled environments to avoid false-positive results and, as a consequence, the standardization of the assays and use of appropriate controls are required before these tools can be used for routine post-mortem diagnosis of rabies in animals in resource-limited settings30. Most post-mortem diagnoses are currently based on the previously recommended techniques (the FAT, RTCIT and MIT): adoption of new reliable and validated tools (dRIT and RT-PCR) will improve diagnostic options for rabies in endemic areas where empirical data are lacking and the burden of the disease is disproportionately high.
Prevention
Primary prevention is the best defence against rabies. It includes: elimination of animal rabies through dog vaccination campaigns; promoting awareness of the disease and available prevention; and encouraging responsible dog ownership and vaccination, particularly in endemic areas, where existing cultural practices might need to be adapted to integrate these interventions27. Where possible, rabies pre-exposure prophylaxis (PrEP) via immunization is recommended for travellers to endemic regions, although such advice might not always be followed112. However, in the absence of infrastructure to support such initiatives, secondary prevention attempts to prevent the onset of clinical disease through post-exposure prophylaxis (PEP). Assessing the category of exposure as defined by the WHO (Box 1) determines the course of action required. The combination of PrEP and PEP can successfully prevent infection in virtually 100% of cases and, regardless of whether or not PrEP has been administered, appropriate wound care and application of PEP are almost invariably effective in preventing deaths30.
Rabies vaccines. Several inactivated preparations of RABV are available as vaccines to immunize humans and domestic animals. For wildlife vaccination, both live-attenuated and subunit vaccines are available. Human rabies vaccines are administered intramuscularly or intradermally, and the same vaccines that are used for PrEP can also be given as part of PEP. The goal of vaccination is to induce VNAs against glycoprotein that provide protection against RABV infection113, although the precise location and mechanisms by which VNAs inhibit viral replication are unknown. The availability of inactivated, lyophilized, cell culture-based vaccines with increased antigenicity has led to a reduction in the number of doses needed to elicit a sufficient VNA response and, hence, a reduction in the number of clinic visits required, which in turn has improved patient participation, in both PrEP and PEP113. The WHO has issued recommendations for inactivated rabies vaccines for human use114 and pre-qualified three vaccines (Table 4). All licenced vaccines have good safety records, and severe adverse events after vaccination are very rare. However, clinicians should be aware that, as with virtually all vaccines, minor adverse effects, such as mild fever and localized reactions to inoculations, are common.
Although the use of refrigerated reconstituted vaccines has not been sanctioned by any major organization or vaccine manufacturer, studies have demonstrated that it is safe, and such vaccine preparations can retain immunogenicity for weeks to months, as long as uninterrupted local electrical supply can support storage115,116. Vaccines are now locally manufactured in India, Thailand and China, and the WHO is developing methods to ensure the uniformity, safety and efficacy of these new products, in line with existing recommendations114,115,116,117,118,119. Research has focused on the development of new vaccines that could reduce the number of clinic visits required through overexpression of the virus glycoprotein. Live-attenuated RABV variants in which glycoprotein expression is twofold to threefold the physiological levels have proven to be effective as PrEP in murine and canine models120,121,122. An attenuated virus vaccine with three copies of the gene encoding glycoprotein induced RABV VNAs in the CSF of mice following intrathecal administration123 and could be of value as a human therapeutic.
Primary prevention: pre-exposure immunization. The near-100% case fatality rate and high case incidence of RABV infection in children <15 years of age have led to the proposal that PrEP should be considered as part of a recommended paediatric vaccination strategy in high-risk regions25,30,124. PrEP regimens currently consist of the administration of 4 vaccine doses, on days 0, 7, 14, and 21 or 28 (Ref. 30). Where PrEP is implemented, the assessment of serological positivity is important to ensure adequate protection and longevity of the antibody response. Furthermore, the administration of rabies vaccines as part of the Expanded Programme on Immunization initiative for children would increase the probability of preventing the development of clinical disease in exposed individuals in the absence of rabies immunoglobulin (RIG) treatment125. However, in endemic areas economic difficulties limit the availability of vaccines; thus, widespread PrEP coverage is unattainable and, as a result, PEP is the primary barrier to preventing deaths30. Population groups who are at increased risk of exposure to lyssaviruses, including veterinarians, scientists and bat workers, should also receive PrEP100,126. Travellers to endemic areas might be unaware of the need for vaccination, and even if they are, misperception of vaccine-associated risks might prevent them from seeking PrEP112.
Secondary prevention: wound care. Appropriate wound care (in combination with PEP regimens) can save lives; in the absence of wound care, the probability of successful PEP decreases127. Wounds should be irrigated with soapy water and scrubbed to remove foreign bodies, and the use of iodine-based antiseptics is recommended. Unless absolutely necessary, suturing the wounds should be avoided or delayed, certainly until after the administration of RIG, as neuronal trauma could allow direct viral access to nerve endings. The local treatment of the wound as well as PEP should be sought immediately after exposure.
Secondary prevention: post-exposure prophylaxis. If exposure is suspected, the WHO guidelines for risk assessment should be followed30 (Box 1) and medical advice should be sought to evaluate the need for PEP. PEP should be administered as soon as possible after a potential exposure to increase the probability of preventing clinical disease; of note, rare cases of very long incubation times in humans suggest that PEP could be of value even if the administration is delayed and should, therefore, always be provided in suspected cases if concerns are raised. Although the development of clinical disease is almost 100% preventable with prompt PEP, failures do occur, most often because of deviations in recommended PEP protocols101. Such deviations include delay in seeking PEP, improper wound care, lack of or incorrect RIG administration and inadequate vaccine doses. Very rarely, failures can result from the use of substandard black-market products128. Cases of patients who develop fatal rabies despite receiving adequate PEP (true PEP failures) are rare129 and can be attributed to short incubation periods that result from multiple bites, especially on highly innervated areas such as the face, neck or hands.
Rabies immunoglobulin and vaccination. PEP can include both the administration of RIG and vaccination, although individuals who received adequate PrEP should receive a booster vaccination, but not RIG. The application of RIG directly to the wound aims to neutralize any live virus in the immediate wound area and prevent the spread of the virus in the time that it takes to develop sufficient immunity in response to vaccination (up to 14 days103). Human RIG (HRIG) or, if not available, chromatography-purified, pepsin-digested equine RIG (ERIG) can be used. The WHO recommends a weight-based total dose calculation: 20 IU/kg for HRIG and 40 IU/kg for ERIG. If possible, all of the calculated dose of RIG should be infiltrated into wounds, and any residual RIG should be injected intramuscularly at a distant site30. However, owing to high manufacturing costs and difficulties in large-scale production, RIG is often scarce in endemic countries26. Although ERIG is safe, easier to produce than HRIG, effective and increasingly available in rabies-endemic countries, the development of new alternatives is warranted130. Studies have assessed the application of RIG to wounds alone, omitting the injection of the usually large residual volume of RIG to distant sites, in an effort to save RIG supplies for future patients131. This approach could enable resource-limited, rabies-endemic countries to provide PEP to all patients with category III exposure131. Nevertheless, a lack of knowledge regarding the potential tools available for PEP means that, even where RIG is available, it might not be used.
In addition to RIG administration, vaccination is advised at a distant site. For previously vaccinated patients, an intradermal or intramuscular booster regimen has been recommended by the WHO30,132 (Table 4). For unvaccinated individuals, several vaccine regimens are approved for PEP; as all vaccines are considered equally effective, the choice of the regimen is determined by the available vaccine and the local medical centre experience30 (Table 4). Immunocompromised patients and those with low CD4+ T cell counts might respond poorly or not at all to vaccination133. Such patients require intense PEP, including wound care with RIG infiltration and vaccination, to elicit an adequate immune response. Patients receiving haemodialysis could also have impaired immunological responsiveness to vaccines, although rabies vaccination is safe and effective if applied before the onset of symptoms134. Vaccination for PEP can be time consuming and costly, owing to a need for multiple separate clinic visits. Studies have attempted to shorten PEP vaccination regimens to 1 week135 to make PEP more-affordable and accessible136. Although the use of intradermal rabies vaccination schedules is expanding worldwide137,138, the WHO requirement to use reconstituted vaccine within 6 hours limits this practice to large clinics, which encounter several patients who need PEP each day.
Management
The management of furious and paralytic rabies does not substantially differ. Treatment approaches are mainly symptomatic and might differ on a case-by-case basis owing to the variety of rabies manifestations. Furthermore, the availability of therapies varies greatly across countries. Once the clinical signs and symptoms of rabies are evident, the prognosis is uniformly dismal, generally culminating in death within 5–11 days52, although survival can be prolonged by 1–3 months in some patients receiving critical care104.
Exposure assessment
In patients who present with encephalitis with undefined origin, the initial assessment should involve a thorough evaluation that should focus on looking for minor bites that might have been missed and the examination of existing wounds. Anamnesis (medical history of the patient) should include any complicating factors, such as underlying illnesses or (allergic) reactions to previous vaccinations or drugs. If exposure to RABV is suspected and domestic animals are implicated, the animal should be observed for clinical signs for 10 days30 and PEP should be initiated. Further treatment can be stopped if the suspect animal remains healthy after quarantine. If the animal shows clinical disease and is available for testing, rabies can be excluded only by a negative FAT result that is confirmed by another reference test (RTCIT or MIT), as recommended by the OIE. In areas where terrestrial rabies has been eliminated, bat rabies constitutes a small but not negligible human health threat and, therefore, this possibility should be considered139,140. If possible, a veterinary examination of the implicated animal should be arranged with necropsy when necessary.
Management of overt rabies. The hospitalization of patients with rabies-like clinical manifestations is required to confirm the diagnosis and prevent contamination or exposure of others to virus-containing biological fluids (Fig. 7). Regardless of whether the diagnosis of rabies has been confirmed, these patients should receive intensive care, if available, as recovery is possible (albeit very rarely); the duration of treatment should be determined on a case-by-case basis. However, in most cases palliative care is the only option, as there is, as yet, no therapy of proven efficacy for human rabies30, and the decision to provide aggressive intensive care is fraught with several medical, ethical, legal, social and economic challenges for physicians as well as caregivers of patients with rabies141,142. The data on in vitro efficacy of antiviral drugs have rarely translated to in vivo successes in experimental models and, therefore, these research findings have not translated to clinical settings143,144. Intravenous sedatives (for example, diazepam, midazolam or barbiturates) and analgesics (for example, morphine or ketamine) might be required to relieve agitation, phobic spasms, anxiety and other neurological symptoms in patients with furious rabies30,141. Antipsychotics (for example, haloperidol) can be beneficial; however, patients with rabies tend to require higher and more numerous doses than generally prescribed, which could increase the risk of developing neuroleptic malignant syndrome. In furious rabies, all of these symptomatic or palliative treatments aim to alleviate limbic manifestations caused by functional selective impairment via neurotransmitter dysregulation52. New approaches (such as experimental drugs or protocols) might be considered to treat a critically ill patient if the potential benefits outweigh the risks.
The treatment of patients with suspected or laboratory-confirmed rabies at present is purely symptomatic; sedation therapy should alleviate neurological symptoms but avoid compromising cardiopulmonary function. Future therapies should aim to eliminate the virus completely and be applied to alert patients whose nervous system functions are still not corrupted. However, before such therapies can be included in intensive care regimens, it is necessary to ensure that they do not to pose any harmful complications. The proposed therapeutics shown remain to be proven effective in experimental animal models. ICU, intensive care unit.
Palliative care is imperative even if critical care is not feasible because of medical, logistical or financial reasons. Unfortunately, few studies have addressed palliation in rabies141,145, and standard national guidelines for palliative care remain absent in endemic regions. Patients continue to be isolated, often physically restrained, for fear of aggression and transmission of infection to others.
Surviving rabies. Fewer than 20 adequately documented survivors of rabies have been reported worldwide105 (Table 3). Most reports are dated after 2000, which could reflect an increased awareness of the disease and greater access to better critical care facilities in rabies-endemic countries142. Although almost all described patients had poor functional outcomes that required lengthy rehabilitation and care, with long-term emotional and financial repercussions for family members, complete or good functional recovery is possible. This possibility should compel physicians to consider and manage rabies as a curable disease, which is not usually possible in under-resourced rabies-endemic countries146. Incremental triage of potential candidates for an intensive therapeutic approach should be implemented, and favourable factors that could indicate a potential candidate include young age and immunocompetence, prior vaccination, early appearance of VNAs in the CSF and serum, rabies due to a bat RABV variant and mild neurological illness at initiation of therapy144,147.
The difficulty in treating clinical rabies is clear. One therapeutic modality that was developed in the United States that seemed to show early promise of success was the ‘Milwaukee protocol’ (Ref. 148), which enabled the survival of an unvaccinated 15-year-old girl who was infected with a US bat RABV variant. The Milwaukee protocol focused on supportive critical care with the aim to evade fatal dysautonomia (dysfunction of the autonomic nervous system) during the first week of hospitalization and to presumably slow the normal metabolic processes, thereby enabling naturally developed immune responses to confer a sterilizing immunity until RABV was cleared from the brain. This goal was achieved by induction of a therapeutic coma (by administering GABA receptor agonists and NMDA (N-methyl-d-aspartic acid) receptor antagonists) and concurrent administration of antiviral drugs. The original protocol has since undergone several modifications, and induction of coma is no longer recommended. However, despite an intial success, the protocol remains controversial, owing to the associated risks and uncertain benefit149.
Quality of life
Globally, dog-mediated rabies is estimated to cause >3.7 million (95% CI 1.6–10.4 million) disability-adjusted life years (DALYs), resulting, for example, from adverse events of vaccination with nerve tissue vaccines (which are derived from animal brain tissue, for example, sheep, goat or mouse) or mortality due to rabies26. The annual global economic burden of dog-mediated rabies is US$8.6 billion (95% CI $2.9–21.5 billion); lost productivity due to premature deaths is a major component (55%), whereas direct costs of PEP (depending on the biologics used and their regimens) and indirect costs of seeking PEP (such as travel, accommodation, multiple clinic visits, lost income and so on) account for 20% and 15% of the economic burden, respectively26. Although difficult to quantify because of the lack of validated methods, anxiety associated with life-threatening bites from rabid animals can potentially contribute substantially (>10%) to the estimated DALYs26, and is often augmented by uncertainty about the availability and affordability of PEP in many dog rabies-endemic countries30. Livestock losses due to dog-mediated rabies account for a small proportion of the global economic burden (6%); however, they disproportionately affect communities that rely on livestock for subsistence and livelihoods26.
Outlook
In the 21st century, it is anticipated that human rabies will be confined to history: the goal is to eliminate the disease by preventing the transmission of the virus between terrestrial animals (principally dogs) and humans. For this vision to be realized, a combination of improved diagnostic tools and veterinary and medical infrastructures — to enable access to more-inexpensive and efficacious biologics (vaccines, new antivirals and HRIG) — as well as increased education and awareness of rabies in endemic countries is urgently required27.
Diagnosis
One of the first steps towards human rabies elimination is to ensure that all regions have the capability to accurately diagnose the infection using a network of dedicated laboratories with validated diagnostic tools. The development of molecular diagnostic techniques and next-generation sequencing has revolutionized high-throughput diagnostic testing and genetic characterization of microorganisms. However, these techniques are not available in all diagnostic laboratories and remain largely a research tool for pathogen discovery rather than pathogen diagnosis per se27. The future challenge is to combine molecular assays that can genetically characterize the RABV variant with point-of-care testing98. The ‘laboratory in a box’ concept, which consists of a toolbox of basic laboratory equipment that can be used for preliminary field diagnosis, will enable the rapid assessment of cases of suspected RABV infection and, as a result, to provide initial care early150, while awaiting confirmation of diagnosis from reference laboratories27. Studies are underway to assess such new technologies.
The WHO and OIE have clarified that both human and animal rabies should be considered notifiable diseases. However, in areas without the appropriate laboratory infrastructures, the timeliness of case reports is often inadequate. Thus, national governments should consider it a high priority to follow a step-wise approach towards dog rabies elimination151. Such approaches link achievable goals on a structured pathway to eliminate the disease152. The FAO in collaboration with the OIE and the WHO has proposed a Progressive Control Pathway towards rabies elimination, in which the final stage is maintaining both humans and animals free of rabies.
Vector control and disease awareness
The elimination of RABV from dogs (the principal reservoir) and, as a result, the prevention of human disease rely on pragmatic actions that can be adopted where the veterinary structures are adequate to support a sustained approach153. The costs required to eliminate dog-mediated rabies in all endemic countries have been estimated at $6.3 billion, whereas current spending is only $2.4 billion7. Most cases of human rabies would be prevented if dog populations in endemic regions were controlled (principally by immunization but also by reducing their numbers by sterilization or culling of sick animals) and dogs were licensed and vaccinated. Numerous studies have demonstrated that 70% vaccine coverage is required to stop rabies transmission in free-roaming dog communities154–156, and responsible dog ownership and vaccination are pivotal in preventing human rabies11. Targeted campaigns for vaccination of domestic dogs that were supported by adequate supplies and awareness campaigns have demonstrated that dog vaccination can be achieved to a level that prevents virus circulation. However, to be effective, vaccinations require follow-up visits for booster vaccinations; dog owners’ compliance with this requirement is scarce and, therefore, these campaigns need to be supported by a global emphasis on eliminating rabies. Oral vaccination of free-roaming dog populations has been proposed as a complementary measure to parenteral vaccination, but assessment of vaccine coverage using oral formulations is challenging157. The lessons learned from the oral vaccination of wildlife species have demonstrated difficulties in achieving adequate seroconversion with this formulation and, therefore, oral vaccination for dog populations might also be challenging158. Additionally, the development of vaccines against Lyssavirus spp. that are more-divergent from RABV has been proposed but is unlikely to gain support unless the burden of these viruses to human or animal populations is demonstrated126,153.
Although the actual burden of human rabies remains difficult to quantify, it is evident that rabies can be managed at a community level by increasing disease awareness in addition to the control of dog populations. Increasing disease awareness in children, and especially what actions to take if an exposure is suspected, could contribute to reducing deaths and could be achieved through the adoption of a few straightforward messages as part of an educational curriculum147,159. Nevertheless, cultural approaches to dog ownership and the multifactorial challenges associated with dog licensing and vaccination mean that any elimination campaign will require long-term investment7. The number of animal bites is often high in regions where rabies is endemic, and every bite should be considered a potential exposure160. It is estimated that PEP is given to >15 million individuals each year to prevent the establishment of disease161. However, owing to lack of awareness, the simplest actions required as part of PEP, including washing the wound and avoiding suturing it, are rarely implemented. Moreover, the biologics required for PEP present challenges, and alternative products are needed. Vaccines require multiple inoculations and are often unavailable because of the lack of a sustained supply across endemic regions. The use of a paediatric rabies vaccine as part of an Expanded Programme on Immunization initiative would increase the probability of exposed children not to develop rabies if they do not receive PEP125. Optimization of current vaccines to reduce production costs and the number of doses required and to develop non-injectable formulations could enable the inclusion of rabies vaccines into existing childhood immunization schemes. New approaches to overcome the scarcity of RIG, the high production costs and the problems associated with blood products include the development of murine monoclonal antibody cocktails162, camelid nanobodies (single-domain antibodies)163 and the generation of antibodies in planta164. However, all of these research efforts require substantial economic investments to reach commercial viability.
Management of clinical disease
The outlook for treating clinical rabies remains poor. Despite promising in vitro proof-of-principle data, none of the antivirals tested have demonstrated sufficient in vivo efficacy in experimental models to warrant further investigation. The development of antivirals for rabies remains an urgent priority, but it is a major challenge because of numerous viral factors (including replication in the CNS and multiple species) and host factors, such as various stages of presentation, exposure history and immune response. An effective antiviral that could be used therapeutically in symptomatic patients with rabies would have to cross the blood–brain barrier, interfere with RABV replication (without affecting the life cycle of the host's cells) and inhibit detrimental host responses to RABV infection. Strategies already attempted include mechanisms to cause temporary disruption of the blood–brain barrier, such as ultrasound and microbubbles, which could facilitate the entry of therapeutics to the optimal site for their action165. Small interfering RNA (siRNA) or multiple artificial microRNAs (miRNAs) targeting multiple RABV genome sequences have been shown to suppress viral replication in vitro166. Broad-spectrum antiviral molecules such as favipiravir (6-fluoro-3-hydroxy-2-pyrazinecarboxamide) have demonstrated an in vitro effect, and favipiravir was also effective as a PrEP in mice167,168. Appropriate drug delivery and targeting systems that have been developed for neurodegenerative diseases could be applied to rabies therapeutic tools. Transvascular delivery of therapeutic siRNA molecules to neuronal cells using short peptides derived from the RABV glycoprotein has been reported169. Other delivery methods that have been tested include stable nucleic acid lipid particles or nanoengineered particles with siRNAs or miRNAs170. If such therapeutics are demonstrated to be beneficial in vivo, then their administration to patients with confirmed or even suspected rabies with alert or mildly depressed sensorium could be an option. Certainly, it is only with a better understanding of the host factors and pathogenetic mechanisms of RABV that a safe, effective antiviral therapeutic protocol can be developed in the future.
As part of the Millennium Development Goals to reduce poverty and preventable childhood deaths from infectious diseases, especially in resource-limited regions of the world, dog-mediated rabies has been targeted for global eradication by 2030 within all endemic regions12. Rabies in wildlife will also need to be controlled to prevent repeated host-switching of the virus from wildlife reservoirs into domestic dogs16,171. Successful elimination of dog rabies from some regions across Latin America has demonstrated once again how targeted initiatives can be successful in clearing the virus3,172. However, the number of rabies cases across endemic regions and the knowledge gained from targeted initiatives suggest that the 2030 eradication target will require additional financial support to be achieved7,173, and the acknowledgement of rabies as a neglected tropical disease by the WHO will encourage investments172. Despite all of the challenges, the eradication goal should promote global initiatives to encourage pharmaceutical companies, international organizations, governments, charities and non-governmental bodies to work together to achieve human rabies elimination worldwide.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
How to cite this Primer
Fooks, A. R. et al. Rabies. Nat. Rev. Dis. Primers 3, 17091 (2017).
References
Neville, J. in Historical Perspective of Rabies in Europe and the Mediterranean Basin (eds King, A. A., Fooks, A. R., Aubert, M. & Wandeler, A. I. ) 1–12 (OIE, 2004).
Afonso, C. L. et al. Taxonomy of the order Mononegavirales: update 2016. Arch. Virol. 161, 2351–2360 (2016).
Fooks, A. R. et al. Current status of rabies and prospects for elimination. Lancet 384, 1389–1399 (2014).
Jackson, A. C. Rabies pathogenesis. J. Neurovirol. 8, 267–269 (2002).
Warrell, M. J. The dilemma of managing human rabies encephalitis. Trop. Med. Int. Health 21, 456–457 (2016).
Meslin, F. X. & Briggs, D. J. Eliminating canine rabies, the principal source of human infection: What will it take? Antiviral Res. 98, 291–296 (2013).
Wallace, R. M., Undurraga, E. A., Blanton, J. D., Cleaton, J. & Franka, R. Elimination of dog-mediated human rabies deaths by 2030: Needs assessment and alternatives for progress based on dog vaccination. Front. Vet. Sci. 4, 9 (2017).This article highlights the resources needed to achieve elimination of dog-mediated human rabies deaths by 2030, which will result in a concomitant reduction in human cases of rabies.
[No authors listed.] Aiming for elimination of dog-mediated human rabies cases by 2030. Vet. Rec. 178, 86–87 (2016).
Reece, J. F. Rabies in India: an ABC approach to combating the disease in street dogs. Vet. Rec. 161, 292–293 (2007).
Lankester, F. et al. Implementing Pasteur's vision for rabies elimination. Sci. Justice 345, 1562–1564 (2014).
Lavan, R. P., King, A., Sutton, D. J. & Tunceli, K. Rationale and support for a One Health program for canine vaccination as the most cost-effective means of controlling zoonotic rabies in endemic settings. Vaccine 35, 1668–1674 (2017).This article contains case studies from endemic, resource-poor regions illustrating how mass canine vaccination programmes that achieve a minimum 70% coverage are cost-effective in controlling zoonotic rabies and decrease the economic burden of rabies by reducing expenditures on post-exposure prophylaxis.
Abela-Ridder, B. et al. The beginning of the end of rabies? Lancet Glob. Health 4, e780–e781 (2016).
Nadin-Davis, S. A. in Rabies: Scientific Basis of the Disease and its Management (ed. Jackson, A. C. ) 123–177 (Academic Press, 2013).
Banyard, A. C. & Fooks, A. R. The impact of novel lyssavirus discovery. Microbiol. Australia 38, 18–21 (2017).
Scott, T. et al. Complete Genome and molecular epidemiological data infer the maintenance of rabies among kudu (Tragelaphus strepsiceros) in Namibia. PLoS ONE 8, e58739 (2013).
Brunker, K. et al. Elucidating the phylodynamics of endemic rabies virus in eastern Africa using whole-genome sequencing. Virus Evol. 1, 1–11 (2015).
Horton, D. et al. Complex epidemiology of a zoonotic disease in a culturally diverse region: phylogeography of rabies virus in the Middle East. PLoS Negl. Trop. Dis. 9, e0003569 (2015).
Badrane, H. & Tordo, N. Host switching in Lyssavirus history from the Chiroptera to the Carnivora orders. J. Virol. 75, 8096–8104 (2001).This is a pivotal manuscript showing that host-switching occurred in the history of lyssaviruses and that lyssaviruses evolved in chiropters long before the emergence of carnivoran rabies, very likely following spillovers from bats.
Bourhy, H. et al. The origin and phylogeography of dog rabies virus. J. Gen. Virol. 89, 2673–2681 (2008).
Bernardi, F. et al. Antigenic and genetic characterization of rabies viruses isolated from domestic and wild animals of Brazil identifies the hoary fox as a rabies reservoir. J. Gen. Virol. 86, 3153–3162 (2005).
Banyard, A. C. et al. in Rabies: Scientific Basis of the Disease and its Management (ed. Jackson, A. C. ) 215–267 (Academic Press, 2013).
Vigilato, M. A. N. et al. Progress towards eliminating canine rabies: policies and perspectives from Latin America and the Caribbean. Philos. Trans R. Soc. Lond. B Biol Sci. 368, 20120143 (2013).
Chiou, H.-Y. et al. Molecular characteridation of cryptically circulating rabies virus from ferret badgers, Taiwan. Emerg. Infect. Dis. 20, 790–798 (2014).
Baxter, J. M. One in a million, or one in thousand: What is the morbidity of rabies in India? J. Glob. Health 2, 10303 (2012).
Fooks, A. R., Koraka, P., de Swart, R. L., Rupprecht, C. E. & Osterhaus, A. D. Development of a multivalent paediatric human vaccine for rabies virus in combination with Measles-Mumps-Rubella (MMR). Vaccine 32, 2020–2021 (2014).
Hampson, K. et al. Estimating the global burden of endemic canine rabies. PLoS Negl. Trop. Dis. 9, e0003709 (2015).
Banyard, A. C., Horton, D. L., Freuling, C., Muller, T. & Fooks, A. R. Control and prevention of canine rabies: the need for building laboratory-based surveillance capacity. Antiviral Res. 98, 357–364 (2013).
Dodet, B., Korejwo, J. & Briggs, D. J. Eliminating the scourge of dog-transmitted rabies. Vaccine 31, 1359 (2013).
Mallewa, M. et al. Rabies encephalitis in malaria-endemic area, Malawi, Africa. Emerg. Infect. Dis. 13, 136–139 (2007).This is a case study report from Malawi showing that rabies is regularly misdiagnosed if a clinical diagnosis is undertaken without laboratory confirmation of rabies.
World Health Organization. WHO Expert Consultation on Rabies, Second Report. WHO Technical Report Series, no. 982 (WHO, 2013).
Lembo, T. et al. The feasibility of canine rabies elimination in Africa: dispelling doubts with data. PLoS Negl. Trop. Dis. 4, e626 (2010).
Messenger, S. L., Smith, J. S. & Rupprecht, C. E. Emerging epidemiology of bat-associated cryptic cases of rabies in humans in the United States. Clin. Infect. Dis. 35, 738–747 (2002).
Srinivasan, A. et al. Transmission of rabies virus from an organ donor to four transplant recipients. N. Engl. J. Med. 352, 1103–1111 (2005).
Chen, S. et al. Rabies virus transmission in solid organ transplantation, China, 2015–2016. Emerg. Infect. Dis. 23, 1600–1602 (2017).
Maier, T. et al. Management and outcomes after multiple corneal and solid organ transplantations from a donor infected with rabies virus. Clin. Infect. Dis. 50, 1112–1119 (2010).
Vora, N. M. et al. Clinical management and humoral immune responses to rabies post-exposure prophylaxis among three patients who received solid organs from a donor with rabies. Transplant Infect. Dis. 17, 389–395 (2015).
Johnson, N., Phillpotts, R. & Fooks, A. R. Airborne transmission of lyssaviruses. J. Med. Microbiol. 55, 785–790 (2006).
Winkler, W. G., Baker, E. F. Jr & Hopkins, C. C. An outbreak of non-bite transmitted rabies in a laboratory animal colony. Am. J. Epidemiol. 95, 267–277 (1972).
Winkler, W. G. Airborne rabies virus isolation. Wildl. Dis. 4, 37–40 (1968).
Davis, A. D., Rudd, R. J. & Bowen, R. A. Effects of aerosolized rabies virus exposure on bats and mice. J. Infect. Dis. 195, 1144–1150 (2007).
Klingen, Y., Conzelmann, K. K. & Finke, S. Double-labeled rabies virus: live tracking of enveloped virus transport. J. Virol. 82, 237–245 (2008).
Gluska, S. et al. Rabies Virus Hijacks and accelerates the p75NTR retrograde axonal transport machinery. PLoS Pathog. 10, e1004348 (2014).
Tsiang, H. Evidence for an intraaxonal transport of fixed and street rabies virus. J. Neuropathol. Exp. Neurol. 38, 286–299 (1979).
Lycke, E. & Tsiang, H. Rabies virus infection of cultured rat sensory neurons. J. Virol. 61, 2733–2741 (1987).
Gillet, J. P., Derer, P. & Tsiang, H. Axonal transport of rabies virus in the central nervous system of the rat. J. Neuropathol. Exp. Neurol. 45, 619–634 (1986).
Kucera, P., Dolivo, M., Coulon, P. & Flamand, A. Pathways of the early propagation of virulent and avirulent rabies strains from the eye to the brain. J. Virol. 55, 158–162 (1985).
Ceccaldi, P. E., Gillet, J. P. & Tsiang, H. Inhibition of the transport of rabies virus in the central nervous system. J. Neuropathol. Exp. Neurol. 48, 620–630 (1989).
Piccinotti, S. & Whelan, S. P. Rabies internalizes into primary peripheral neurons via clathrin coated pits and requires fusion at the cell body. PLoS Pathog. 12, e1005753 (2016).
Lahaye, X. et al. Functional characterization of Negri bodies (NBs) in rabies virus-infected cells: Evidence that NBs are sites of viral transcription and replication. J. Virol. 83, 7948–7958 (2009).
Charlton, K. M. & Casey, G. A. Experimental rabies in skunks: immunofluorescence light and electron microscopic studies. Lab. Invest. 41, 36–44 (1979).
Etessami, R. et al. Spread and pathogenic characteristics of a G-deficient rabies virus recombinant: an in vitro and in vivo study. J. Gen. Virol. 81, 2147–2153 (2000).
Hemachudha, T. et al. Human rabies: neuropathogenesis, diagnosis, and management. Lancet Neurol. 12, 498–513 (2013).
Thoulouze, M. I. et al. The neural cell adhesion molecule is a receptor for rabies virus. J. Virol. 72, 7181–7190 (1998).
Tuffereau, C., Benejean, J., Blondel, D., Kieffer, B. & Flamand, A. Low-affinity nerve-growth factor receptor (P75NTR) can serve as a receptor for rabies virus. EMBO J. 17, 7250–7259 (1998).
Lentz, T. L., Burrage, T. G., Smith, A. L., Crick, J. & Tignor, G. H. Is the acetylcholine receptor a rabies virus receptor? Science 215, 182–184 (1982).The data in this paper indicate that acetylcholine receptors serve as receptors for the rabies virus.
Tuffereau, C. et al. The rabies virus glycoprotein receptor p75NTR is not essential for rabies virus infection. J. Virol. 81, 13622–13630 (2007).
Lafon, M. Rabies virus receptors. J. Neurovirol. 11, 82–87 (2005).
Velandia-Romero, M. L., Castellanos, J. E. & Martínez-Gutiérrez, M. In vivo differential susceptibility of sensory neurons to rabies virus infection. J. Neurovirol. 19, 367–375 (2013).
Bauer, A. et al. Anterograde glycoprotein-dependent transport of newly generated rabies virus in dorsal root ganglion neurons. J. Virol. 88, 14172–14183 (2014).
Zampieri, N., Jessell, T. M. & Murray, A. J. Mapping sensory circuits by anterograde transsynaptic transfer of recombinant rabies virus. Neuron 81, 766–778 (2014).
Zhang, D. et al. Genome-wide transcriptional profiling reveals two distinct outcomes in central nervous system infections of rabies virus. Front. Microbiol. 7, 751 (2016).
Wang, Z. W. et al. Attenuated rabies virus activates, while pathogenic rabies virus evades, the host innate immune responses in the central nervous system. J. Virol. 79, 12554–12565 (2005).
Hooper, D. C., Roy, A., Barkhouse, D. A., Li, J. & Kean, R. B. Rabies virus clearance from the central nervous system. Adv. Virus Res. 79, 55–71 (2011).
Hooper, D. C., Phares, T. W., Fabis, M. J. & Roy, A. The production of antibody by invading B cells is required for the clearance of rabies virus from the central nervous system. PLoS Negl. Trop. Dis. 3, e535 (2009).The data in this paper show that the production of rabies-virus-specific antibodies by infiltrating B cells across the blood–brain barrier is crucial for the elimination of rabies virus.
Gnanadurai, C. W. & Fu, Z. F. CXCL10 and blood-brain barrier modulation in rabies virus infection. Oncotarget 7, 10694–10695 (2016).
Wang, L., Cao, Y., Tang, Q. & Liang, G. Role of the blood-brain barrier in rabies virus infection and protection. Protein Cell 4, 901–903 (2013).
Roy, A., Phares, T. W., Koprowski, H. & Hooper, D. C. Failure to open the blood-brain barrier and deliver immune effectors to central nervous system tissues leads to the lethal outcome of silver-haired bat rabies virus infection. J. Virol. 81, 1110–1118 (2007).
Laothamatas, J., Hemachudha, T., Mitrabhakdi, E., Wannakrairot, P. & Tulayadaechanont, S. MR imaging in human rabies. AJNR Am. J. Neuroradiol. 24, 1102–1109 (2003).
Laothamatas, J. et al. Furious and paralytic rabies of canine origin: neuroimaging with virological and cytokine studies. J. Neurovirol. 14, 119–129 (2008).
Hooper, D. C., Roy, A., Kean, R. B., Phares, T. W. & Barkhouse, D. A. Therapeutic immune clearance of rabies virus from the CNS. Future Virol. 6, 387–397 (2011).
Johnson, N., Cunningham, A. F. & Fooks, A. R. The immune response to rabies virus infection and vaccination. Vaccine 28, 3896–3901 (2010).
Lafon, M. Evasive strategies in rabies virus infection. Adv. Virus Res. 79, 33–53 (2011).
Baloul, L., Camelo, S. & Lafon, M. Up-regulation of Fas ligand (FasL) in the central nervous system: a mechanism of immune evasion by rabies virus. J. Neurovirol. 10, 372–382 (2004).
Kojima, D. et al. Pathology of the spinal cord of C57BL/6J mice infected with rabies virus (CVS-11 strain). J. Vet. Med. Sci. 71, 319–324 (2009).
Kojima, D. et al. Lesions of the central nervous system induced by intracerebral inoculation of BALB/c mice with rabies virus (CVS-11). J. Vet. Med. Sci. 72, 1011–1016 (2010).
Lafon, M. et al. Detrimental contribution of the immuno-inhibitor B7-H1 to rabies virus encephalitis. J. Immunol. 180, 7506–7515 (2008).
Finke, S., Cox, J. H. & Conzelmann, K. K. Differential transcription attenuation of rabies virus genes by intergenic regions: generation of recombinant viruses overexpressing the polymerase gene. J. Virol. 74, 7261–7269 (2000).
Shuangshoti, S. et al. Intracellular spread of rabies virus is reduced in the paralytic form of canine rabies compared to the furious form. PLoS Negl. Trop. Dis. 10, e0004748 (2016).
Shuangshoti, S. et al. Reduced viral burden in paralytic compared to furious canine rabies is associated with prominent inflammation at the brainstem level. BMC Vet. Res. 9, 31 (2013).
Nikolic, J., Civas, A., Lama, Z., Lagaudriere-Gesbert, C. & Blondel, D. Rabies virus infection induces the formation of stress granules closely connected to the viral factories. PLoS Pathog. 12, e1005942 (2016).
Onomoto, K., Yoneyama, M., Fung, G., Kato, H. & Fujita, T. Antiviral innate immunity and stress granule responses. Trends Immunol. 35, 420–428 (2014).
Caillet-Saguy, C. et al. Strategies to interfere with PDZ-mediated interactions in neurons: What we can learn from the rabies virus. Prog. Biophys. Mol. Biol. 119, 53–59 (2015).
Prehaud, C. et al. Attenuation of rabies virulence: takeover by the cytoplasmic domain of its envelope protein. Sci. Signal. 3, ra5 (2010).
Babault, N. et al. Peptides targeting the PDZ domain of PTPN4 are efficient inducers of glioblastoma cell death. Structure 19, 1518–1524 (2011).
Prehaud, C., Lay, S., Dietzschold, B. & Lafon, M. Glycoprotein of nonpathogenic rabies viruses is a key determinant of human cell apoptosis. J. Virol. 77, 10537–10547 (2003).
Jackson, A. C., Randle, E., Lawrance, G. & Rossiter, J. P. Neuronal apoptosis does not play an important role in human rabies encephalitis. J. Neurovirol. 14, 368–375 (2008).
Peng, J. et al. Wild-type rabies virus induces autophagy in human and mouse neuroblastoma cell lines. Autophagy 12, 1704–1720 (2016).
Li, L. et al. Autophagy is highly targeted among host comparative proteomes during infection with different virulent RABV strains. Oncotarget 8, 21336–21350 (2017).
Tirawatnpong, S. et al. Regional distribution of rabies viral antigen in central nervous system of human encephalitic and paralytic rabies. J. Neurol. Sci. 92, 91–99 (1989).
Hemachudha, T., Laothamatas, J. & Rupprecht, C. E. Human rabies: a disease of complex neuropathogenetic mechanisms and diagnostic challenges. Lancet Neurol. 1, 101–109 (2002).
Mitrabhakdi, E. et al. Difference in neuropathogenetic mechanisms in human furious and paralytic rabies. J. Neurol. Sci. 238, 3–10 (2005).
Jackson, A. C. & Park, H. Apoptotic cell death in experimental rabies in suckling mice. Acta Neuropathol. 95, 159–164 (1998).
Warrell, M. J. & Warrell, D. A. Rabies and other lyssavirus diseases. Lancet 363, 959–969 (2004).
Hemachudha, T. et al. Pathophysiology of human paralytic rabies. J. Neurovirol. 11, 93–100 (2005).
Hunter, M. et al. Immunovirological correlates in human rabies treated with therapeutic coma. J. Med. Virol. 82, 1255–1265 (2010).
Fekadu, M., Shaddock, J. H. & Baer, G. M. Excretion of rabies virus in the saliva of dogs. J. Infect. Dis. 145, 715–719 (1982).
Dacheux, L. et al. A reliable diagnosis of human rabies based on analysis of skin biopsy specimens. Clin. Infect. Dis. 47, 1410–1417 (2008).
Fooks, A. R. et al. Emerging technologies for the detection of rabies virus: challenges and hopes in the 21st century. PLoS Negl. Trop. Dis. 3, e530 (2009).
Nadin-Davis, S. A., Sheen, M. & Wandeler, A. I. Development of real-time reverse transcriptase polymerase chain reaction methods for human rabies diagnosis. J. Med. Virol. 81, 1484–1497 (2009).
Fooks, A. R. Rabies - the need for a ‘one medicine’ approach. Vet. Rec. 161, 289–290 (2007).
Wilde, H. et al. Failure of postexposure treatment of rabies in children. Clin. Infect. Dis. 22, 228–232 (1996).
Wacharapluesadee, S. & Hemachudha, T. Ante- and post-mortem diagnosis of rabies using nucleic acid-amplification tests. Expert Rev. Mol. Diagn. 10, 207–218 (2010).
Rupprecht, C. E. & Plotkin, S. in Vaccines (eds Plotkin, S., Orenstein, W. & Offit, P. ) 646–668 (Elsevier, 2013).
Mani, R. S. & Anand, A. M. & Madhusudana, S. N. Human rabies in India: an audit from a rabies diagnostic laboratory. Trop. Med. Int. Health 21, 556–563 (2016).
Jackson, A. C. Human rabies: a 2016 update. Curr. Infect. Dis. Rep. 18, 38 (2016).
Meslin, F., Kaplan, M. & Koprowski, H. Laboratory Techniques in Rabies (World Health Organization, 1996).
Office International des Epizooties. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals (OIE, 2013).
Lembo, T. et al. Evaluation of a direct, rapid immunohistochemical test for rabies diagnosis. Emerg. Infect. Dis. 12, 310–313 (2006).
Coetzer, A., Sabeta, C., Markotter, W., Rupprecht, C. E. & Nel, L. H. Comparison of biotinylated monoclonal and polyclonal antibodies in an evaluation of a direct rapid immunohistochemcial test for the routine diagnosis of rabies in southern Africa. PLoS Negl. Trop. Diseases 8, e3189 (2014).
Freuling, C. M. et al. in Current Laboratory Techniques in Rabies diagnosis, Research and prevention Vol. 1 Ch. 7 (eds Rupprecht, C. & Nagarajan, T. ) 63–84 (Academic Press, 2015)
Wadhwa, A. et al. A pan-Lyssavirus Taqman real-time RT-PCR assay for the detection of highly variable rabies virus and other lyssaviruses. PLoS Negl. Trop. Dis. 11, e0005258 (2017).
Fooks, A. R., Johnson, N., Brookes, S. M., Parsons, G. & McElhinney, L. M. Risk factors associated with travel to rabies endemic countries. J. Appl. Microbiol. 94 (Suppl.), 31S–36S (2003).
Rupprecht, C. E., Nagarajan, T. & Ertl, H. Current status and development of vaccines and other biologics for human rabies prevention. Expert Rev. Vaccines, 15 731–749 (2016).
World Health Organization. Oral Vaccination of Dogs Against Rabies. Guidance for Research on Oral Rabies Vaccines and Field Application of Oral Vaccination of Dogs Against Rabies (ed. Meslin, F. ) (WHO, 2007).
Khawplod, P. et al. Use of rabies vaccines after reconstitution and storage. Clin. Infect. Dis. 34, 404–406 (2002).
Lodmell, D. L. & Ewalt, L. C. Rabies cell culture vaccines reconstituted and stored at 4 degrees C for 1 year prior to use protect mice against rabies virus. Vaccine 22, 3237–3239 (2004).
Khawplod, P. et al. Potency, sterility and immunogenicity of rabies tissue culture vaccine after reconstitution and refrigerated storage for 1 week. Vaccine 20, 2240–2242 (2002).
Kamoltham, T., Khawplod, P. & Wilde, H. Rabies intradermal post-exposure vaccination of humans using reconstituted and stored vaccine. Vaccine 20, 3272–3276 (2002).
Hu, R. L., Fooks, A. R., Zhang, S. F., Liu, Y. & Zhang, F. Inferior rabies vaccine quality and low immunization coverage in dogs (Canis familiaris) in China. Epidemiol. Infect. 136, 1556–1563 ( 2008 ).
Faber, M. et al. Overexpression of the rabies virus glycoprotein results in enhancement of apoptosis and antiviral immune response. J. Virol. 76, 3374–3381 (2002).
Faber, M. et al. Effective preexposure and postexposure prophylaxis of rabies with a highly attenuated recombinant rabies virus. Proc. Natl Acad. Sci. USA 106, 11300–11305 (2009).
Gnanadurai, C. W. et al. Differential host immune responses after infection with wild-type or lab-attenuated rabies viruses in dogs. PLoS Negl. Trop. Dis. 9, e0004023 (2015).
Li, J. et al. Postexposure treatment with the live-attenuated rabies virus (RV) vaccine TriGAS triggers the clearance of wild-type RV from the Central Nervous System (CNS) through the rapid induction of genes relevant to adaptive immunity in CNS tissues. J. Virol. 86, 3200–3210 (2012).
Kessels, J. A. et al. Pre-exposure rabies prophylaxis: a systematic review. Bull. World Health Organ. 95, 210–219C (2017).
World Health Organization. Rabies vaccines: WHO position paper-recommendations. Vaccine 28, 7140–7142 (2010).
Evans, J. S., Horton, D. L., Easton, A. J., Fooks, A. R. & Banyard, A. C. Rabies virus vaccines: is there a need for a pan-lyssavirus vaccine? Vaccine 30, 7447–7454 (2012).
Hanlon, C. A., Niezgoda, M., Morrill, P. A. & Rupprecht, C. E. The incurable wound revisited: progress in human rabies prevention? Vaccine 19, 2273–2279 (2001).
McLaughlin, K. Scandal clouds China's global vaccine ambitions. Science 352, 506 (2016).
Shantavasinkul, P. et al. Failure of rabies postexposure prophylaxis in patients presenting with unusual manifestations. Clin. Infect. Dis. 50, 77–79 (2010).
Both, L. et al. Passive immunity in the prevention of rabies. Lancet Infect. Dis. 12, 397–407 (2012).
Bharti, O. K. et al. Success story of a low cost intra-dermal rabies vaccination (IDRV) clinic-lessons learnt over five years of 12,000 patient vaccinations “without failure” at DDU Hospital Shimla, Himachal Pradesh, India — “Saving a drop of rabies vaccine and immunoglobulins” 12 innovations to make Himachal Pradesh rabies free state by 2020. World J. Vaccines 5, 129–139 (2015).
Shantavasinkul, p. et al. A 4-site, single-visit intradermal postexposure prophylaxis regimen for previously vaccinated patients: experiences with >5000 patients. Clin. Infect. Dis. 51, 1070–1072 (2010).
Wilde, H., Khawplod, P. & Hemachudha, T. Post-exposure rabies prophylaxis in patients with AIDS. Vaccine 27, 5726–5727 (2009).
Tanisaro, T. et al. Neutralizing antibody response after intradermal rabies vaccination in hemodialysis patients. Vaccine 28, 2385–2387 (2010).
Shantavasinkul, P. et al. Postexposure rabies prophylaxis completed in 1 week: preliminary study. Clin. Infect. Dis. 50, 56–60 (2010).
Wilde, H., Lumlertdacha, B., Meslin, F. X., Ghai, S. & Hemachudha, T. Worldwide rabies deaths prevention-A focus on the current inadequacies in postexposure prophylaxis of animal bite victims. Vaccine 34, 187–189 (2016).
Warrell, M. J. et al. A simplified 4-site economical intradermal post-exposure rabies vaccine regimen: a randomised controlled comparison with standard methods. PLoS Negl. Trop. Dis. 2, e224 (2008).
Brown, D. et al. Intradermal pre-exposure rabies vaccine elicits long lasting immunity. Vaccine 26, 3909–3912 (2008).
Simani, S. et al. Six fatal cases of classical rabies virus without biting incidents, Iran 1990–2010. J. Clin. Virol. 54, 251–254 (2012).
Dato, V. M., Campagnolo, E. R., Long, J. & Rupprecht, C. E. A. Systematic review of human bat rabies virus variant cases: evaluating unprotected physical contact with claws and teeth in support of accurate risk assessments. PLoS ONE 11, e0159443 (2016).
Tarantola, A. et al. Caring for patients with rabies in developing countries — the neglected importance of palliative care. Trop. Med. Int. Health 21, 564–567 (2016).
Mani, R. S. Human rabies survivors in India: an emerging paradox? PLoS Negl. Trop. Dis. 10, e0004774 (2016).
Jackson, A. C. Why does the prognosis remain so poor in human rabies? Expert Rev. Anti Infect. Ther. 8, 623–625 (2010).
Jackson, A. C. Current and future approaches to the therapy of human rabies Antiviral Res. 99, 61–67 (2013).
Marsden, S. C. & Cabanban, C. R. Rabies: a significant palliative care issue. Prog. Palliative Care 14, 62–67 (2006).
de Souza, A. & Madhusudana, S. N. Survival from rabies encephalitis. J. Neurol. Sci. 339, 8–14 (2014).
Sambo, M. et al. Knowledge, attitudes and practices (KAP) about rabies prevention and control: a community survey in Tanzania. PLoS Negl. Trop. Dis. 8, e3310 (2014).
Willoughby, R. E. Jr et al. Survival after treatment of rabies with induction of coma. N. Engl. J. Med. 352, 2508–2514 ( 2005 ).
Zeiler, F. A. & Jackson, A. C. Critical appraisal of the Milwaukee protocol for rabies: this failed approach should be abandoned. Can. J. Neurol. Sci. 43, 44–51 (2016).
Duong, V. et al. Laboratory diagnostics in dog-mediated rabies: an overview of performance and a proposed strategy for various settings. Int. J. Infect. Dis. 46, 107–114 (2016).
Coetzer, A. et al. The SARE tool for rabies control: current experience in Ethiopia. Antiviral Res. 135, 74–80 (2016).
Fahrion, A. S. et al. The road to dog rabies control and elimination — what keeps us from moving faster? Front. Public Health 5, 103 (2017).
Rupprecht, C., Kuzmin, I. & Meslin, F. Lyssaviruses and rabies: current conundrums, concerns, contradictions and controversies. F1000Res 6, 184 (2017).This is a fundamental review of rabies, providing a realistic plan to achieve success in eliminating this disease.
Coleman, P. G. & Dye, C. Immunization coverage required to prevent outbreaks of dog rabies. Vaccine 14, 185–186 (1996).
Hampson, K. et al. Transmission dynamics and prospects for the elimination of canine rabies. PLoS Biol. 7, e1000053 (2009).
Leung, T. & Davis, S. A. Rabies vaccination targets for stray dog populations. Front. Vet. Sci. 4, 52 (2017).
World Health Organization. Oral Vaccination of Dogs Against Rabies (ed Meslin, F. ) (World Health Organization, 2007).
Muller, T. et al. Analysis of vaccine-virus-associated rabies cases in red foxes (Vulpes vulpes) after oral rabies vaccination campaigns in Germany and Austria. Arch. Virol. 154, 1081–1091 (2009).
Lapiz, S. M. et al. Implementation of an intersectoral program to eliminate human and canine rabies: the Bohol Rabies Prevention and Elimination Project. PLoS Negl. Trop. Dis. 6, e1891 (2012).
Cleaveland, S., Fevre, E. M., Kaare, M. & Coleman, P. G. Estimating human rabies mortality in the United Republic of Tanzania from dog bite injuries. Bull. World Health Organ. 80, 304–310 (2002).
World Health Organization. WHO Expert Consultation on Rabies, First Report. WHO Technical Report Series, no. 931 (WHO, 2005).
Muller, T. et al. Development of a mouse monoclonal antibody cocktail for post-exposure rabies prophylaxis in humans. PLoS Negl. Trop. Dis. 3, e542 (2009).This paper presents results from an in vivo study in mice, proving that mouse monoclonal antibodies conferred protection against rabies that was comparable with that of human rabies immunoglobulin.
Terryn, S. et al. Protective effect of different anti-rabies virus VHH constructs against rabies disease in mice. PLoS ONE 9, e109367 (2014).
Both, L. et al. Production, characterization, and antigen specificity of recombinant 62-71-3, a candidate monoclonal antibody for rabies prophylaxis in humans. FASEB J. 27, 2055–2065 (2013).
Gabathuler, R. Approaches to transport therapeutic drugs across the blood-brain barrier to treat brain diseases. Neurobiol. Dis. 37, 48–57 (2010).
Israsena, N., Supavonwong, P., Ratanasetyuth, N., Khawplod, P. & Hemachudha, T. Inhibition of rabies virus replication by multiple artificial microRNAs. Antiviral Res. 84, 76–83 (2009).
Yamada, K., Noguchi, K., Komeno, T., Furuta, Y. & Nishizono, A. Efficacy of favipiravir (T-705) in rabies postexposure prophylaxis. J. Infect. Dis. 213, 1253–1261 (2016).
Virojanapirom, P., Lumlertdacha, B., Wipattanakitchareon, A. & Hemachudha, T. T-705 as a potential therapeutic agent for rabies. J. Infect. Dis. 213, 1253–1261 (2016).
Kumar, P. et al. Transvascular delivery of small interfering RNA to the central nervous system. Nature 448, 39–43 (2007).
Schnee, M. et al. An mRNA vaccine encoding rabies virus glycoprotein induces protection against lethal infection in mice and correlates of protection in adult and newborn pigs. PLoS Negl. Trop. Dis. 10, e0004746 (2016).
Elmore, S. A. et al. Management and modeling approaches for controlling raccoon rabies: the road to elimination. PLoS Negl. Trop. Dis. 11, e0005579 (2017).
Abela-Ridder, B. Rabies: 100 per cent fatal, 100 per cent preventable. Vet. Rec. 177, 148–149 (2015).
Shwiff, S., Anderson, A. & Hampson, K. Potential economic benefits of eliminating canine rabies. Antiviral Res. 98, 352–356 (2013).
World Health Organization. Rabies: epidemiology and burden of disease. WHOhttp://www.who.int/rabies/epidemiology/en/ (2017).
Finke, S., Mueller-Waldeck, R. & Conzelmann, K. K. Rabies virus matrix protein regulates the balance of virus transcription and replication. J. Gen. Virol. 84, 1613–1621 (2003).
Finke, S. & Conzelmann, K. K. Dissociation of rabies virus matrix protein functions in regulation of viral RNA synthesis and virus assembly. J. Virol. 77, 12074–12082 (2003).
Mebatsion, T., Weiland, F. & Conzelmann, K. K. Matrix protein of rabies virus is responsible for the assembly and budding of bullet-shaped particles and interacts with the transmembrane spike glycoprotein G. J. Virol. 73, 242–250 (1999).
Pollin, R., Granzow, H., Kollner, B., Conzelmann, K. K. & Finke, S. Membrane and inclusion body targeting of lyssavirus matrix proteins. Cell. Microbiol. 15, 200–212 (2013).
Wirblich, C. et al. PPEY motif within the rabies virus (RV) matrix protein is essential for efficient virion release and RV pathogenicity. J. Virol. 82, 9730–9738 (2008).
Roche, S. & Gaudin, Y. Evidence that rabies virus forms different kinds of fusion machines with different pH thresholds for fusion. J. Virol. 78, 8746–8752 (2004).
Anilionis, A., Wunner, W. H. & Curtis, P. J. Structure of the glycoprotein gene in rabies virus. Nature 294, 275–278 (1981).
Schnell, M. J., McGettigan, J. P., Wirblich, C. & Papaneri, A. The cell biology of rabies virus: using stealth to reach the brain. Nat. Rev. Microbiol. 8, 51–61 (2010).
Scott, T. P. & Nel, L. H. Subversion of the immune response by rabies virus. Viruses 231, 1–26 (2016).
Blondel, D., Maarifi, G., Nisole, S. & Chelbi-Alix, M. K. Resistance to Rhabdoviridae infection and subversion of antiviral responses. Viruses 7, 3675–3702 (2015).
Brzozka, K., Finke, S. & Conzelmann, K. K. Inhibition of interferon signaling by rabies virus phosphoprotein P: activation-dependent binding of STAT1 and STAT2. J. Virol. 80, 2675–2683 (2006).This report shows that the rabies virus phosphoprotein is responsible for preventing IFN-α/β-stimulated and IFN-γ-stimulated JAK-STAT signalling in rabies-virus-infected cells by the retention of activated STATs (signal transducer and activator of transcription) in the cytoplasm.
Masatani, T. et al. Rabies virus nucleoprotein functions to evade activation of the RIG-I-mediated antiviral response. J. Virol. 84, 4002–4012 (2010).
Vidy, A., Chelbi-Alix, M. & Blondel, D. Rabies virus P protein interacts with STAT1 and inhibits interferon signal transduction pathways. J. Virol. 79, 14411–14420 (2005).
Brzozka, K., Finke, S. & Conzelmann, K. K. Identification of the rabies virus alpha/beta interferon antagonist: phosphoprotein P interferes with phosphorylation of interferon regulatory factor 3. J. Virol. 79, 7673–7681 (2005).The data in this paper show that the rabies virus phosphoprotein is an interferon antagonist counteracting transcriptional activation of type I interferon.
Luco, S. et al. RelAp43, a member of the NF-κB family involved in innate immune response against Lyssavirus infection. PLoS Pathog. 8, e1003060 (2012).
Ben Khalifa, Y. et al. The matrix protein of rabies virus binds to RelAp43 to modulate NF-κB-dependent gene expression related to innate immunity. Sci. Rep. 6, 39420 (2016).
Hornung, V. et al. 5′-triphosphate RNA is the ligand for RIG-I. Science 314, 994–997 (2006).
Faul, E. J. et al. Rabies virus infection induces type I interferon production in an IPS-1 dependent manner while dendritic cell activation relies on IFNAR signaling. PLoS Pathog. 6, e1001016 (2010).
Vidy, A., El Bougrini, J., Chelbi-Alix, M. K. & Blondel, D. The nucleocytoplasmic rabies virus P protein counteracts interferon signaling by inhibiting both nuclear accumulation and DNA binding of STAT1. J. Virol. 81, 4255–4263 (2007).
Amarasinghe, G. K. et al. Taxonomy of the order Mononegavirales: update 2017. Arch. Virol. 162, 2493–2504 (2017).
Karande, S. et al. Atypical rabies encephalitis in a six-year-old boy: clinical, radiological, and laboratory findings. Int. J. Infect. Dis. 36, 1–3 (2015).
Manoj, S., Mukherjee, A., Johri, S. & Kumar, K. V. Recovery from rabies, a universally fatal disease. Mil. Med. Res. 3, 21 (2016).
Kumar, K. V., Ahmad, F. M. & Dutta, V. Pituitary cachexia after rabies encephalitis. Neurol. India 63, 255–256 (2015).
Weyer, J. et al. A case of human survival of rabies, South Africa. South Afr. J. Infect. Dis. 31, 66–68 (2016).
Wiedeman, J. et al. Recovery of a patient from clinical rabies — California, 2011. MMWR Morb. Mortal. Wkly Rep. 61, 61–65 (2012).
Netravathi, M. et al. Unique clinical and imaging findings in a first ever documented PCR positive rabies survival patient: a case report. J. Clin. Virol. 70, 83–88 (2015).
Karahocagil, M. K. et al. Complete recovery from clinical rabies: case report. Turkiye Klinikleri J. Med. Sci. 33, 547–552 (2013).
PromedMail. Rabies - Brazil (04): Pernambuco), Recovery. (PromedMail, 2008).
Madhusudana, S. N., Nagaraj, D., Uday, M., Ratnavalli, E. & Kumar, M. V. Partial recovery from rabies in a six-year-old girl. Int. J. Infect. Dis. 6, 85–86 (2002).
Alvarez, L. et al. Partial recovery from rabies in a nine-year-old boy. Pediatr. Infect. Dis. J. 13, 1154–1155 (1994).
Tillotson, J. R., Axelrod, D. & Lyman, D. O. Rabies in a laboratory worker — New York. MMWR Morb. Mortal. Wkly Rep. 26, 183–184 (1977).
Tillotson, J. R., Axelrod, D. & Lyman, D. O. Follow-up on rabies — New York. MMWR Morb. Mortal. Wkly Rep. 26, 249–250 (1977).
Porras, C. et al. Recovery from rabies in man. Ann. Intern. Med. 85, 44–48 (1976).
Hattwick, M. A., Weis, T. T., Stechschulte, C. J., Baer, G. M. & Gregg, M. B. Recovery from rabies. A case report. Ann. Intern. Med. 76, 931–942 (1972).
Acknowledgements
A.R.F. and A.C.B. were financially supported by the UK Department for Environment, Food and Rural Affairs (Defra), the Scottish Government and the Welsh Government (grant number SV3500).
Author information
Authors and Affiliations
Contributions
Introduction (A.R.F. and A.C.B.); Epidemiology (S.N.-D.); Mechanisms/pathophysiology (A.C.B., C.F., S.F., T.M. and T.H.); Diagnosis, screening and prevention (A.C.B., F.C. and E.P.-M.); Management (A.C.B., T.H., R.S.M. and H.W.); Quality of life (R.S.M.); Outlook (A.C.B. and A.R.F.); Overview of Primer (A.R.F.).
Corresponding author
Ethics declarations
Competing interests
T.M., C.F. and S.F. have a research cooperation and project (2014–2019) with a German vaccine company on oral vaccination of wildlife (for example, mechanisms of oral vaccination and immunity and development of a novel oral rabies virus vaccine). All other authors declare no competing interests.
Supplementary information
Supplementary information S1 (video)
Rat dorsal root ganglion neurons were cultivated in compartmentalized chambers and infected with rabies virus expressing a green fluorescent protein (GFP)-tagged phosphoprotein (Bauer A. et al. Anterograde Glycoprotein-Dependent Transport of Newly Generated Rabies Virus in Dorsal Root Ganglion Neurons. J. Virol. 88, 14172-14183 (2014)). At two days post-infection, the axonal transport of labelled particles was analyzed by live confocal laser scanning microscopy. The time-lapse images show anterograde (from left to right) and retrograde (from right to left) transport of GFP-labelled particles. Scale bar: 3 μm. (AVI 3048 kb)
Rights and permissions
About this article
Cite this article
Fooks, A., Cliquet, F., Finke, S. et al. Rabies. Nat Rev Dis Primers 3, 17091 (2017). https://doi.org/10.1038/nrdp.2017.91
Published:
DOI: https://doi.org/10.1038/nrdp.2017.91