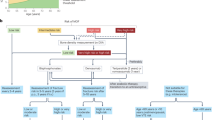
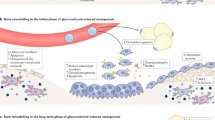
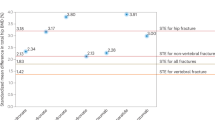
Abstract
Osteoporosis is a metabolic bone disorder that is characterized by low bone mass and micro-architectural deterioration of bone tissue. Fractures of the proximal femur, the vertebrae and the distal radius are the most frequent osteoporotic fractures, although most fractures in the elderly are probably at least partly related to bone fragility. The incidence of fractures varies greatly by country, but on average up to 50% of women >50 years of age are at risk of fractures. Fractures severely affect the quality of life of an individual and are becoming a major public health problem owing to the ageing population. Postmenopausal osteoporosis, resulting from oestrogen deficiency, is the most common type of osteoporosis. Oestrogen deficiency results in an increase in bone turnover owing to effects on all types of bone cells. The imbalance in bone formation and resorption has effects on trabecular bone (loss of connectivity) and cortical bone (cortical thinning and porosity). Osteoporosis is diagnosed using bone density measurements of the lumbar spine and proximal femur. Preventive strategies to improve bone health include diet, exercise and abstaining from smoking. Fractures may be prevented by reducing falls in high-risk populations. Several drugs are licensed to reduce fracture risk by slowing down bone resorption (such as bisphosphonates and denosumab) or by stimulating bone formation (such as teriparatide). Improved understanding of the cellular basis for osteoporosis has resulted in new drugs targeted to key pathways, which are under development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
[No authors listed.] Consensus development conference on osteoporosis. Hong Kong, April 1–2, 1993. Am. J. Med. 95, 1S–78S (1993).
Kanis, J. A. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. Osteoporos Int. 4, 368–381 (1994). This paper summarizes the key report from the WHO that defined osteoporosis on the basis of a bone density T-score.
Kanis, J. A. FRAX: WHO Fracture Risk Assessment Tool. QHOhttps://www.shef.ac.uk/FRAX/tool.jsp (2016).
Siris, E. S. et al. The clinical diagnosis of osteoporosis: a position statement from the National Bone Health Alliance Working Group. Osteoporos Int. 25, 1439–1443 (2014).
Eastell, R. Treatment of postmenopausal osteoporosis. N. Engl. J. Med. 338, 736–746 (1998).
Office of the Surgeon General (US). Bone Health and Osteoporosis: A Report of the Surgeon General (Office of the Surgeon General, 2004).
Hernlund, E. et al. Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos 8, 136–136 (2013).
Burge, R. et al. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J. Bone Miner. Res. 22, 465–475 (2007).
van Staa, T. P., Dennison, E. M., Leufkens, H. G. & Cooper, C. Epidemiology of fractures in England and Wales. Bone 29, 517–522 (2001).
Cheng, S. Y. et al. Geographic trends in incidence of hip fractures: a comprehensive literature review. Osteoporos Int. 22, 2575–2586 (2011).
Cauley, J. A., Chalhoub, D., Kassem, A. M. & Fuleihan, G. E.-H. Geographic and ethnic disparities in osteoporotic fractures. Nat. Rev. Endocrinol. 10, 338–351 (2014).
Johnell, O., Gullberg, B., Allander, E. & Kanis, J. A. The apparent incidence of hip fracture in Europe: a study of national register sources. Osteoporos Int. 2, 298–302 (1992).
Cooper, C. et al. Secular trends in the incidence of hip and other osteoporotic fractures. Osteoporos Int. 22, 1277–1288 (2011).
Morin, S. N., Lix, L. M., Majumdar, S. R. & Leslie, W. D. Temporal trends in the incidence of osteoporotic fractures. Curr. Osteoporos Rep. 11, 263–269 (2013).
Leslie, W. D. et al. Trends in hip fracture rates in Canada. JAMA 302, 883–889 (2009).
Xia, W. B. et al. Rapidly increasing rates of hip fracture in Beijing, China. J. Bone Miner. Res. 27, 125–129 (2012).
Kinsella, K. & Wan, H. U.S. Census Bureau, International Population Reports, P95/09-1, An Aging World: 2008 (U.S. Government Printing Office, 2009).
Cooper, C., Campion, G. & Melton, L. J. Hip fractures in the elderly: a world-wide projection. Osteoporos Int. 2, 285–289 (1992). This paper highlights for the first time the epidemic in hip fractures.
Pocock, N. A. et al. Genetic determinants of bone mass in adults. A twin study. J. Clin. Invest. 80, 706–710 (1987).
Hernandez-de Sosa, N. et al. Heritability of bone mineral density in a multivariate family-based study. Calcif. Tissue Int. 94, 590–596 (2014).
Michaëlsson, K., Melhus, H., Ferm, H., Ahlbom, A. & Pedersen, N. L. Genetic liability to fractures in the elderly. Arch. Intern. Med. 165, 1825–1830 (2005).
Ioannidis, J. P. A. et al. Differential genetic effects of ESR1 gene polymorphisms on osteoporosis outcomes. JAMA 292, 2105–2114 (2004).
Uitterlinden, A. G. et al. The association between common vitamin D receptor gene variations and osteoporosis: a participant-level meta-analysis. Ann. Intern. Med. 145, 255–264 (2006).
Langdahl, B. L. et al. Large-scale analysis of association between polymorphisms in the transforming growth factor β1 gene (TGFB1) and osteoporosis: the GENOMOS study. Bone 42, 969–981 (2008).
van Meurs, J. B. J. et al. Large-scale analysis of association between LRP5 and LRP6 variants and osteoporosis. JAMA 299, 1277–1290 (2008).
Ralston, S. H. et al. Large-scale evidence for the effect of the COLIA1 Sp1 polymorphism on osteoporosis outcomes: the GENOMOS study. PLoS Med. 3, e90 (2006).
Xie, W., Ji, L., Zhao, T. & Gao, P. Identification of transcriptional factors and key genes in primary osteoporosis by DNA microarray. Med. Sci. Monit. 21, 1333–1344 (2015).
Keupp, K. et al. Mutations in WNT1 cause different forms of bone fragility. Am. J. Hum. Genet. 92, 565–574 (2013).
Zheng, H.-F. et al. WNT16 influences bone mineral density, cortical bone thickness, bone strength, and osteoporotic fracture risk. PLoS Genet. 8, e1002745 (2012).
Richards, J. B. et al. Bone mineral density, osteoporosis, and osteoporotic fractures: a genome-wide association study. Lancet 371, 1505–1512 (2008).
Estrada, K. et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nat. Genet. 44, 491–501 (2012).
Garnero, P., Sornay-Rendu, E., Chapuy, M. C. & Delmas, P. D. Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis. J. Bone Miner. Res. 11, 337–349 (1996).
Sims, N. A. & Civitelli, R. Cell–cell signaling: broadening our view of the basic multicellular unit. Calcif. Tissue Int. 94, 2–3 (2014).
Khosla, S. New insights into androgen and estrogen receptor regulation of the male skeleton. J. Bone Miner. Res. 30, 1134–1137 (2015). This review describes the importance of oestrogen action on the male skeleton and its role on bone homeostasis.
Manolagas, S. C. From estrogen-centric to aging and oxidative stress: a revised perspective of the pathogenesis of osteoporosis. Endocr. Rev. 31, 266–300 (2010).
Seeman, E. & Delmas, P. D. Bone quality — the material and structural basis of bone strength and fragility. N. Engl. J. Med. 354, 2250–2261 (2006).
Manolagas, S. C., O'Brien, C. A. & Almeida, M. The role of estrogen and androgen receptors in bone health and disease. Nat. Rev. Endocrinol. 9, 699–712 (2013).
Clowes, J. A., Riggs, B. L. & Khosla, S. The role of the immune system in the pathophysiology of osteoporosis. Immunol. Rev. 208, 207–227 (2005).
Bonjour, J.-P. & Chevalley, T. Pubertal timing, bone acquisition, and risk of fracture throughout life. Endocr. Rev. 35, 820–847 (2014).
Szulc, P., Seeman, E., Duboeuf, F., Sornay-Rendu, E. & Delmas, P. D. Bone fragility: failure of periosteal apposition to compensate for increased endocortical resorption in postmenopausal women. J. Bone Miner. Res. 21, 1856–1863 (2006).
Nishiyama, K. K., Macdonald, H. M., Buie, H. R., Hanley, D. A. & Boyd, S. K. Postmenopausal women with osteopenia have higher cortical porosity and thinner cortices at the distal radius and tibia than women with normal aBMD: an in vivo HR-pQCT study. J. Bone Miner. Res. 25, 882–890 (2010).
Patsch, J. M. et al. Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J. Bone Miner. Res. 28, 313–324 (2013).
Gennari, L., Khosla, S. & Bilezikian, J. P. Estrogen and fracture risk in men. J. Bone Miner. Res. 23, 1548–1551 (2008).
Santen, R. J., Brodie, H., Simpson, E. R., Siiteri, P. K. & Brodie, A. History of aromatase: saga of an important biological mediator and therapeutic target. Endocr. Rev. 30, 343–375 (2009).
Khosla, S. Update on estrogens and the skeleton. J. Clin. Endocrinol. Metab. 95, 3569–3577 (2010).
Kovacs, C. S. Osteoporosis presenting in pregnancy, puerperium, and lactation. Curr. Opin. Endocrinol. Diabetes Obes. 21, 468–475 (2014).
Smith, E. P. et al. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N. Engl. J. Med. 331, 1056–1061 (1994).
Falahati-Nini, A. et al. Relative contributions of testosterone and estrogen in regulating bone resorption and formation in normal elderly men. J. Clin. Invest. 106, 1553–1560 (2000).
Weitzmann, M. N. & Pacifici, R. Estrogen deficiency and bone loss: an inflammatory tale. J. Clin. Invest. 116, 1186–1194 (2006).
Kousteni, S. et al. Nongenotropic, sex-nonspecific signaling through the estrogen or androgen receptors: dissociation from transcriptional activity. Cell 104, 719–730 (2001).
Syed, F. A. et al. Skeletal effects of estrogen are mediated by opposing actions of classical and nonclassical estrogen receptor pathways. J. Bone Miner. Res. 20, 1992–2001 (2005).
Dallas, S. L., Prideaux, M. & Bonewald, L. F. The osteocyte: an endocrine cell … and more. Endocr. Rev. 34, 658–690 (2013).
Rachner, T. D., Khosla, S. & Hofbauer, L. C. Osteoporosis: now and the future. Lancet 377, 1276–1287 (2011).
Chow, J., Tobias, J. H., Colston, K. W. & Chambers, T. J. Estrogen maintains trabecular bone volume in rats not only by suppression of bone resorption but also by stimulation of bone formation. J. Clin. Invest. 89, 74–78 (1992).
Oursler, M. J. Estrogen regulation of gene expression in osteoblasts and osteoclasts. Crit. Rev. Eukaryot. Gene Expr. 8, 125–140 (1998).
Khastgir, G. et al. Anabolic effect of estrogen replacement on bone in postmenopausal women with osteoporosis: histomorphometric evidence in a longitudinal study. J. Clin. Endocrinol. Metab. 86, 289–295 (2001).
Hofbauer, L. C. & Schoppet, M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA 292, 490–495 (2004).
Eghbali-Fatourechi, G. et al. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J. Clin. Invest. 111, 1221–1230 (2003).
Nakashima, T. et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 17, 1231–1234 (2011).
Mödder, U. I. et al. Regulation of circulating sclerostin levels by sex steroids in women and in men. J. Bone Miner. Res. 26, 27–34 (2011).
Fujita, K. et al. Effects of estrogen on bone mRNA levels of sclerostin and other genes relevant to bone metabolism in postmenopausal women. J. Clin. Endocrinol. Metab. 99, E81–E88 (2014).
Lee, K., Jessop, H., Suswillo, R., Zaman, G. & Lanyon, L. Endocrinology: bone adaptation requires oestrogen receptor-α. Nature 424, 389–389 (2003).
Almeida, M. et al. Estrogens attenuate oxidative stress and the differentiation and apoptosis of osteoblasts by DNA-binding-independent actions of the ERα. J. Bone Miner. Res. 25, 769–781 (2010).
Goettsch, C. et al. NADPH oxidase 4 limits bone mass by promoting osteoclastogenesis. J. Clin. Invest. 123, 4731–4738 (2013).
Kanis, J. A., Johnell, O., Oden, A., Johansson, H. & McCloskey, E. FRAX™ and the assessment of fracture probability in men and women from the UK. Osteoporos Int. 19, 385–397 (2008).
Nguyen, N. D., Frost, S. A., Center, J. R., Eisman, J. A. & Nguyen, T. V. Development of prognostic nomograms for individualizing 5-year and 10-year fracture risks. Osteoporos Int. 19, 1431–1444 (2008).
Hippisley-Cox, J. & Coupland, C. Predicting risk of osteoporotic fracture in men and women in England and Wales: prospective derivation and validation of QFractureScores. BMJ 339, b4229 (2009).
[No authors listed.] Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ. Techn. Rep. Ser. 843, 1–129 (1994).
Marshall, D., Johnell, O. & Wedel, H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ 312, 1254–1259 (1996).
Kanis, J. A. Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359, 1929–1936 (2002).
Vasikaran, S. et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int. 22, 391–420 (2011).
Diez-Perez, A. et al. Treatment failure in osteoporosis. Osteoporos Int. 23, 2769–2774 (2012).
Hernandez, C. J., Beaupré, G. S. & Carter, D. R. A theoretical analysis of the relative influences of peak BMD, age-related bone loss and menopause on the development of osteoporosis. Osteoporos Int. 14, 843–847 (2003).
Cadogan, J., Eastell, R., Jones, N. & Barker, M. E. Milk intake and bone mineral acquisition in adolescent girls: randomised, controlled intervention trial. BMJ 315, 1255–1260 (1997).
Chevalley, T., Rizzoli, R., Hans, D., Ferrari, S. & Bonjour, J.-P. Interaction between calcium intake and menarcheal age on bone mass gain: an eight-year follow-up study from prepuberty to postmenarche. J. Clin. Endocrinol. Metab. 90, 44–51 (2005).
Winzenberg, T., Shaw, K., Fryer, J. & Jones, G. Effects of calcium supplementation on bone density in healthy children: meta-analysis of randomised controlled trials. BMJ 333, 775–775 (2006).
Bischoff-Ferrari, H. A. et al. Dietary calcium and serum 25-hydroxyvitamin D status in relation to BMD among U. S. adults. J. Bone Miner. Res. 24, 935–942 (2009).
Shea, B. et al. Meta-analyses of therapies for postmenopausal osteoporosis. VII. Meta-analysis of calcium supplementation for the prevention of postmenopausal osteoporosis. Endocr. Rev. 23, 552–559 (2002).
Elders, P. J. et al. Long-term effect of calcium supplementation on bone loss in perimenopausal women. J. Bone Miner. Res. 9, 963–970 (1994).
Bischoff-Ferrari, H. A. et al. Calcium intake and hip fracture risk in men and women: a meta-analysis of prospective cohort studies and randomized controlled trials. Am. J. Clin. Nutr. 86, 1780–1790 (2007).
Bolland, M. J. et al. Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: meta-analysis. BMJ 341, c3691 (2010).
Wang, L., Manson, J. E., Song, Y. & Sesso, H. D. Systematic review: vitamin D and calcium supplementation in prevention of cardiovascular events. Ann. Intern. Med. 152, 315–323 (2010).
Lewis, J. R. et al. The effects of calcium supplementation on verified coronary heart disease hospitalization and death in postmenopausal women: a collaborative meta-analysis of randomized controlled trials. J. Bone Miner. Res. 30, 165–175 (2015).
Wallace, R. B. et al. Urinary tract stone occurrence in the Women's Health Initiative (WHI) randomized clinical trial of calcium and vitamin D supplements. Am. J. Clin. Nutr. 94, 270–277 (2011).
Serio, A. & Fraioli, A. Epidemiology of nephrolithiasis. Nephron 81 (Suppl. 1), 26–30 (1999).
Bischoff-Ferrari, H. A. et al. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: a meta-analysis of randomized controlled trials. Arch. Intern. Med. 169, 551–561 (2009).
Avenell, A., Mak, J. C. S. & O'Connell, D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst. Rev. 4, CD000227 (2014). This paper stresses that, although vitamin D alone may not prevent fracture, when given along with calcium, it may prevent fracture.
Ceglia, L. Vitamin D and its role in skeletal muscle. Curr. Opin. Clin. Nutr. Metab. Care 12, 628–633 (2009).
Cao, J. J. & Nielsen, F. H. Acid diet (high-meat protein) effects on calcium metabolism and bone health. Curr. Opin. Clin. Nutr. Metab. Care 13, 698–702 (2010).
Byberg, L., Bellavia, A., Orsini, N., Wolk, A. & Michaëlsson, K. Fruit and vegetable intake and risk of hip fracture: a cohort study of Swedish men and women. J. Bone Miner. Res. 30, 976–984 (2015).
Fenton, T. R., Eliasziw, M., Lyon, A. W., Tough, S. C. & Hanley, D. A. Meta-analysis of the quantity of calcium excretion associated with the net acid excretion of the modern diet under the acid-ash diet hypothesis. Am. J. Clin. Nutr. 88, 1159–1166 (2008).
Dawson-Hughes, B. & Harris, S. S. Calcium intake influences the association of protein intake with rates of bone loss in elderly men and women. Am. J. Clin. Nutr. 75, 773–779 (2002).
Sahni, S. et al. Protective effect of high protein and calcium intake on the risk of hip fracture in the Framingham offspring cohort. J. Bone Miner. Res. 25, 2770–2776 (2010).
Ensrud, K. E., Cauley, J., Lipschutz, R. & Cummings, S. R. Weight change and fractures in older women. Study of Osteoporotic Fractures Research Group. Arch. Intern. Med. 157, 857–863 (1997).
Cummings, S. R. et al. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N. Engl. J. Med. 332, 767–773 (1995).
Crandall, C. J. et al. Postmenopausal weight change and incidence of fracture: post hoc findings from Women's Health Initiative Observational Study and Clinical Trials. BMJ 350, h25 (2015).
Clynes, M. A. et al. Definitions of sarcopenia: associations with previous falls and fracture in a population sample. Calcif. Tissue Int. 97, 445–452 (2015).
Lang, T. et al. Cortical and trabecular bone mineral loss from the spine and hip in long-duration spaceflight. J. Bone Miner. Res. 19, 1006–1012 (2004).
Heinonen, A. et al. Bone mineral density in female athletes representing sports with different loading characteristics of the skeleton. Bone 17, 197–203 (1995).
Leichter, I. et al. Gain in mass density of bone following strenuous physical activity. J. Orthop. Res. 7, 86–90 (1989).
McKay, H. A. et al. “Bounce at the Bell”: a novel program of short bouts of exercise improves proximal femur bone mass in early pubertal children. Br. J. Sports Med. 39, 521–526 (2005).
Colberg, S. R. et al. Exercise and type 2 diabetes: the American College of Sports Medicine and the American Diabetes Association: joint position statement executive summary. Diabetes Care 33, 2692–2696 (2010).
Rubin, C. T., Bain, S. D. & McLeod, K. J. Suppression of the osteogenic response in the aging skeleton. Calcif. Tissue Int. 50, 306–313 (1992).
Iwamoto, J., Takeda, T. & Ichimura, S. Effect of exercise training and detraining on bone mineral density in postmenopausal women with osteoporosis. J. Orthop. Res. 6, 128–132 (2001).
Bolton, K. L. et al. Effects of exercise on bone density and falls risk factors in post-menopausal women with osteopenia: a randomised controlled trial. J. Sci. Med. Sport 15, 102–109 (2012).
Kemmler, W. et al. Exercise and fractures in postmenopausal women: 12-year results of the Erlangen Fitness and Osteoporosis Prevention Study (EFOPS). Osteoporos Int. 23, 1267–1276 (2012).
von Stengel, S., Kemmler, W., Engelke, K. & Kalender, W. A. Effects of whole body vibration on bone mineral density and falls: results of the randomized controlled ELVIS study with postmenopausal women. Osteoporos Int. 22, 317–325 (2011).
Leung, K. S. et al. Effects of 18-month low-magnitude high-frequency vibration on fall rate and fracture risks in 710 community elderly — a cluster-randomized controlled trial. Osteoporos Int. 25, 1785–1795 (2014).
Kanis, J. A. et al. Smoking and fracture risk: a meta-analysis. Osteoporos Int. 16, 155–162 (2005).
Krall, E. A. & Dawson-Hughes, B. Smoking increases bone loss and decreases intestinal calcium absorption. J. Bone Miner. Res. 14, 215–220 (1999).
Jensen, J., Christiansen, C. & Rødbro, P. Cigarette smoking, serum estrogens, and bone loss during hormone-replacement therapy early after menopause. N. Engl. J. Med. 313, 973–975 (1985).
Oncken, C. et al. Impact of smoking cessation on bone mineral density in postmenopausal women. J. Womens Health (Larchmt) 15, 1141–1150 (2006).
Schwartz, A. V., Nevitt, M. C., Brown, B. W. & Kelsey, J. L. Increased falling as a risk factor for fracture among older women: the study of osteoporotic fractures. Am. J. Epidemiol. 161, 180–185 (2005).
Oliver, D. et al. Strategies to prevent falls and fractures in hospitals and care homes and effect of cognitive impairment: systematic review and meta-analyses. BMJ 334, 82–82 (2007).
Harwood, R. H. et al. Falls and health status in elderly women following first eye cataract surgery: a randomised controlled trial. Br. J. Ophthalmol. 89, 53–59 (2005).
Bischoff-Ferrari, H. A. et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ 339, b3692 (2009).
Panel on Prevention of Falls in Older Persons, American Geriatrics Society and British Geriatrics Society. Summary of the Updated American Geriatrics Society/British Geriatrics Society clinical practice guideline for prevention of falls in older persons. J. Am. Geriatr. Soc. 59, 148–157 (2011).
Institute of Medicine. Dietary Reference Ranges for Calcium and Vitamin D (National Academies Press, 2011).
Michael, Y. L. et al. Primary care-relevant interventions to prevent falling in older adults: a systematic evidence review for the U. S. Preventive Services Task Force. Ann. Intern. Med. 153, 815–825 (2010).
Dawson-Hughes, B. et al. IOF position statement: vitamin D recommendations for older adults. Osteoporos Int. 21, 1151–1154 (2010).
Garvan Institute. Fracture Risk Calculator. Garvan Institutehttp://www.garvan.org.au/promotions/bone-fracture-risk/calculator/ (2016).
Billington, E. O., Gamble, G. D. & Reid, I. R. Reasons for discrepancies in hip fracture risk estimates using FRAX and Garvan calculators. Maturitas 85, 11–18 (2016).
Dawson-Hughes, B. et al. Implications of absolute fracture risk assessment for osteoporosis practice guidelines in the USA. Osteoporos Int. 19, 449–458 (2008).
Reid, I. R. Efficacy, effectiveness and side effects of medications used to prevent fractures. J. Intern. Med. 277, 690–706 (2015).
Ebetino, F. H. et al. The relationship between the chemistry and biological activity of the bisphosphonates. Bone 49, 20–33 (2011).
Murad, M. H. et al. Comparative effectiveness of drug treatments to prevent fragility fractures: a systematic review and network meta-analysis. J. Clin. Endocrinol. Metab. 97, 1871–1880 (2012).
Black, D. M. et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N. Engl. J. Med. 356, 1809–1822 (2007).
Khosla, S. et al. Benefits and risks of bisphosphonate therapy for osteoporosis. J. Clin. Endocrinol. Metab. 97, 2272–2282 (2012).
Cartsos, V. M., Zhu, S. & Zavras, A. I. Bisphosphonate use and the risk of adverse jaw outcomes. J. Am. Dent. Assoc. 139, 23–30 (2008).
Pazianas, M., Blumentals, W. A. & Miller, P. D. Lack of association between oral bisphosphonates and osteonecrosis using jaw surgery as a surrogate marker. Osteoporos Int. 19, 773–779 (2008).
Lin, T. C., Yang, C. Y., Kao Yang, Y. H. & Lin, S. J. Incidence and risk of osteonecrosis of the jaw among the Taiwan osteoporosis population. Osteoporos Int. 25, 1503–1511 (2014).
Schilcher, J., Michaelsson, K. & Aspenberg, P. Bisphosphonate use and atypical fractures of the femoral shaft. N. Engl. J. Med. 364, 1728–1737 (2011).
Shane, E. et al. Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J. Bone Miner. Res. 29, 1–23 (2014).
Schilcher, J., Koeppen, V., Aspenberg, P. & Michaëlsson, K. Risk of atypical femoral fracture during and after bisphosphonate use. N. Engl. J. Med. 371, 974–976 (2014).
Dell, R. M. et al. Incidence of atypical nontraumatic diaphyseal fractures of the femur. J. Bone Miner. Res. 27, 2544–2550 (2012).
Cummings, S. R. et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 361, 756–765 (2009).
Papapoulos, S. et al. The effect of 8 or 5 years of denosumab treatment in postmenopausal women with osteoporosis: results from the FREEDOM Extension study. Osteoporos Int. 26, 2773–2783 (2015).
Miller, P. D. et al. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized blinded phase 2 clinical trial. Bone 43, 222–229 (2008).
Aubry-Rozier, B., Gonzalez-Rodriguez, E., Stoll, D. & Lamy, O. Severe spontaneous vertebral fractures after denosumab discontinuation: three case reports. Osteoporos Int. 27, 1923–1925 (2016).
Leder, B. Z. et al. Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA Switch study): extension of a randomised controlled trial. Lancet 386, 1147–1155 (2015).
McClung, M. R. et al. Opposite bone remodeling effects of teriparatide and alendronate in increasing bone mass. Arch. Intern. Med. 165, 1762–1768 (2005).
Neer, R. M. et al. Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N. Engl. J. Med. 344, 1434–1441 (2001).
Nakamura, T. et al. Randomized teriparatide human parathyroid hormone (PTH) 1–34 once-weekly efficacy research (TOWER) trial for examining the reduction in new vertebral fractures in subjects with primary osteoporosis and high fracture risk. J. Clin. Endocrinol. Metab. 97, 3097–3106 (2012).
Bang, U. C., Hyldstrup, L. & Jensen, J. E. B. The impact of recombinant parathyroid hormone on malignancies and mortality: 7 years of experience based on nationwide Danish registers. Osteoporos Int. 25, 639–644 (2014).
Cosman, F. et al. Effects of intravenous zoledronic acid plus subcutaneous teriparatide [rhPTH(1–34)] in postmenopausal osteoporosis. J. Bone Miner. Res. 26, 503–511 (2011).
Cosman, F. et al. Effects of teriparatide in postmenopausal women with osteoporosis on prior alendronate or raloxifene: differences between stopping and continuing the antiresorptive agent. J. Clin. Endocrinol. Metab. 94, 3772–3780 (2009).
Recker, R. R. et al. Comparative effects of teriparatide and strontium ranelate on bone biopsies and biochemical markers of bone turnover in postmenopausal women with osteoporosis. J. Bone Miner. Res. 24, 1358–1368 (2009).
Chavassieux, P. et al. Bone histomorphometry of transiliac paired bone biopsies after 6 or 12 months of treatment with oral strontium ranelate in 387 osteoporotic women: randomized comparison to alendronate. J. Bone Miner. Res. 29, 618–628 (2014).
Meunier, P. J. et al. The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N. Engl. J. Med. 350, 459–468 (2004).
Reginster, J. Y. et al. Strontium ranelate reduces the risk of nonvertebral fractures in postmenopausal women with osteoporosis: Treatment of Peripheral Osteoporosis (TROPOS) study. J. Clin. Endocrinol. Metab. 90, 2816–2822 (2005).
Tan, K. W., Wang, Y. S. & Tay, Y. K. Stevens–Johnson syndrome due to strontium ranelate. Ann. Acad. Med. Singapore 40, 510–511 (2011).
Medicines and Healthcare Products Regulatory Agency (MHRA). Strontium ranelate (Protelos): risk of serious cardiac disorders — restricted indications, new contraindications, and warnings. Drug Safety Updatehttp://pad.res360.net/Content/Documents/Strontium%20Safety%20Alert%20April%202013.pdf (2013).
European Medicines Agency. Protelos/Osseor to remain available but with further restrictions. EMAhttp://www.ema.europa.eu/docs/en_GB/document_library/Referrals_document/Protelos_and_Osseor/European_Commission_final_decision/WC500173034.pdf (2014).
Lindsay, R., Hart, D. M., Forrest, C. & Baird, C. Prevention of spinal osteoporosis in oophorectomised women. Lancet 2, 1151–1153 (1980).
Rossouw, J. E. et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women — principal results from the Women's Health Initiative randomized controlled trial. JAMA 288, 321–333 (2002).
Anderson, G. L. et al. Effects of conjugated, equine estrogen in postmenopausal women with hysterectomy — the Women's Health Initiative randomized controlled trial. JAMA 291, 1701–1712 (2004).
Rossouw, J. E. et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA 297, 1465–1477 (2007).
Reid, I. R. et al. A comparison of the effects of raloxifene and conjugated equine estrogen on bone and lipids in healthy postmenopausal women. Arch. Intern. Med. 164, 871–879 (2004).
Ettinger, B. et al. Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene — results from a 3-year randomized clinical trial. JAMA 282, 637–645 (1999).
Barrett-Connor, E. et al. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N. Engl. J. Med. 355, 125–137 (2006).
Sharifi, M. & Lewiecki, E. M. Conjugated estrogens combined with bazedoxifene: the first approved tissue selective estrogen complex therapy. Expert Rev. Clin. Pharmacol. 7, 281–291 (2014).
Mirkin, S., Ryan, K. A., Chandran, A. B. & Komm, B. S. Bazedoxifene/conjugated estrogens for managing the burden of estrogen deficiency symptoms. Maturitas 77, 24–31 (2014).
Lindsay, R., Gallagher, J. C., Kagan, R., Pickar, J. H. & Constantine, G. Efficacy of tissue-selective estrogen complex of bazedoxifene/conjugated estrogens for osteoporosis prevention in at-risk postmenopausal women. Fertil. Steril. 92, 1045–1052 (2009).
Silverman, S. L. et al. Efficacy of bazedoxifene in reducing new vertebral fracture risk in postmenopausal women with osteoporosis: results from a 3-year, randomized, placebo-, and active-controlled clinical trial. J. Bone Miner. Res. 23, 1923–1934 (2008).
Cummings, S. R. et al. Lasofoxifene in postmenopausal women with osteoporosis. N. Engl. J. Med. 362, 686–696 (2010).
Lips, P. & van Schoor, N. M. Quality of life in patients with osteoporosis. Osteoporos Int. 16, 447–455 (2005).
Gold, D. T. The clinical impact of vertebral fractures: quality of life in women with osteoporosis. Bone 18, 185S–189S (1996).
Cauley, J. A., Thompson, D. E., Ensrud, K. C., Scott, J. C. & Black, D. Risk of mortality following clinical fractures. Osteoporos Int. 11, 556–561 (2000). This study, using data from the Fracture Intervention Trial, was one of the first to examine mortality resulting from major osteoporotic fractures and the first to identify excess mortality associated with clinical vertebral fractures.
Randell, A. G. et al. Deterioration in quality of life following hip fracture: a prospective study. Osteoporos Int. 11, 460–466 (2000).
Papaioannou, A. et al. Determinants of health-related quality of life in women with vertebral fractures. Osteoporos Int. 17, 355–363 (2006).
Bhattacharya, R., Shen, C. & Sambamoorthi, U. Excess risk of chronic physical conditions associated with depression and anxiety. BMC Psychiatry 14, 10 (2014).
Silverman, S. L. Quality-of-life issues in osteoporosis. Curr. Rheumatol. Rep. 7, 39–45 (2005).
Pike, C. et al. Direct and indirect costs of non-vertebral fracture patients with osteoporosis in the US. PharmacoEconomics 28, 395–409 (2010).
Eekman, D. A. et al. Indirect costs account for half of the total costs of an osteoporotic fracture: a prospective evaluation. Osteoporos Int. 25, 195–204 (2014).
Gold, D. T. & Silverman, S. L. The Downward Spiral of Vertebral Osteoporosis: Consequences (Cedars-Sinai Medical Center, 2003).
Ballane, G., Cauley, J. A., Luckey, M. M. & Fuleihan Gel, H. Secular trends in hip fractures worldwide: opposing trends East versus West. J. Bone Miner. Res. 29, 1745–1755 (2014).
Brauer, C. A., Coca-Perraillon, M., Cutler, D. M. & Rosen, A. B. Incidence and mortality of hip fractures in the United States. JAMA 302, 1573–1579 (2009).
Hiligsmann, M. et al. Trends in hip fracture incidence and in the prescription of antiosteoporosis medications during the same time period in Belgium (2000–2007). Arthritis Care Res. (Hoboken) 64, 744–750 (2012).
Wright, N. C. et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J. Bone Miner. Res. 29, 2520–2526 (2014).
Looker, A. C., Melton, L. J. 3rd, Borrud, L. G. & Shepherd, J. A. Changes in femur neck bone density in US adults between 1988–1994 and 2005–2008: demographic patterns and possible determinants. Osteoporos Int. 23, 771–780 (2012).
Jha, S., Wang, Z., Laucis, N. & Bhattacharyya, T. Trends in media reports, oral bisphosphonate prescriptions, and hip fractures 1996–2012: an ecological analysis. J. Bone Miner. Res. 30, 2179–2187 (2015). This paper shows the recent trend for fewer prescriptions of oral bisphosphonates and relates this to media announcements.
Eisman, J. A. et al. Making the first fracture the last fracture: ASBMR Task Force report on secondary fracture prevention. J. Bone Miner. Res. 27, 2039–2046 (2012).
McClung, M. R. et al. Romosozumab in postmenopausal women with low bone mineral density. N. Engl. J. Med. 370, 412–420 (2014).
Fierce Biotech. Amgen and UCB announce positive top-line results from the phase 3 study of Romosozumab in postmenopausal women with osteoporosis. Fierce Biotechhttp://www.fiercebiotech.com/press-releases/amgen-and-ucb-announce-positive-top-line-results-phase-3-study-romosozumab (2016).
Leder, B. Z. et al. Effects of abaloparatide, a human parathyroid hormone-related peptide analog, on bone mineral density in postmenopausal women with osteoporosis. J. Clin. Endocrinol. Metab. 100, 697–706 (2015).
Tucker, M. E. Novel agent abaloparatide reduces fractures, including wrist. Medscapehttp://www.medscape.com/viewarticle/841015 (2015).
Rizzoli, R. et al. Continuous treatment with odanacatib for up to 8 years in postmenopausal women with low bone mineral density: a phase 2 study. Osteoporos Int. 27, 2099–2107 (2016).
[No authors listed.] Merck announces data from pivotal phase 3 fracture outcomes study for odanacatib, an investigational oral, once-weekly treatment for osteoporosis. Business Wirehttp://www.businesswire.com/news/home/20140915006286/en/Merck-Announces-Data-Pivotal-Phase-3-Fracture (2014).
[No authors listed.] Merck provides update on odanacatib development program. Business Wirehttp://www.businesswire.com/news/home/20160902005107/en/Merck-Update-Odanacatib-Development-Program (2016).
Cosman, F. et al. Clinician's guide to prevention and treatment of osteoporosis. Osteoporos Int. 25, 2359–2381 (2014). This is a recent guide for clinicians who manage osteoporosis.
Compston, J. et al. Diagnosis and management of osteoporosis in postmenopausal women and older men in the UK: National Osteoporosis Guideline Group (NOGG) update 2013. Maturitas 75, 392–396 (2013).
Kanis, J. A. et al. Goal-directed treatment of osteoporosis in Europe. Osteoporos Int. 25, 2533–2543 (2014).
International Osteoporosis Foundation. Guideline references. IOFhttps://www.iofbonehealth.org/guideline-references (accessed 12 Sep 2016).
Cummings, S. R. et al. Goal-directed treatment of osteoporosis. J. Bone Miner. Res. 28, 433–438 (2013).
Austin, M. et al. Relationship between bone mineral density changes with denosumab treatment and risk reduction for vertebral and nonvertebral fractures. J. Bone Miner. Res. 27, 687–693 (2012).
Jacques, R. M. et al. Relationship of changes in total hip bone mineral density to vertebral and nonvertebral fracture risk in women with postmenopausal osteoporosis treated with once-yearly zoledronic acid 5 mg: the HORIZON-Pivotal Fracture Trial (PFT). J. Bone Miner. Res. 27, 1627–1634 (2012).
Felsenberg, D. et al. Incidence of vertebral fracture in Europe: results from the European Prospective Osteoporosis Study (EPOS). J. Bone Miner. Res. 17, 716–724 (2002).
Curtis, E. M. et al. Epidemiology of fractures in the United Kingdom 1988–2012: variation with age, sex, geography, ethnicity and socioeconomic status. Bone 87, 19–26 (2016).
McClung, M. et al. Odanacatib anti-fracture efficacy and safety in postmenopausal women with osteoporosis: results from the phase III long-term odanacatib fracture trial. ACRhttp://acrabstracts.org/abstract/odanacatib-anti-fracture-efficacy-and-safety-in-postmenopausal-women-with-osteoporosis-results-from-the-phase-iii-long-term-odanacatib-fracture-trial/ (2014).
Greenspan, S. L. et al. Effect of recombinant human parathyroid hormone (1–84) on vertebral fracture and bone mineral density in postmenopausal women with osteoporosis — a randomized trial. Ann. Intern. Med. 146, 326–339 (2007).
Reid, I. R. Short-term and long-term effects of osteoporosis therapies. Nat. Rev. Endocrinol. 11, 418–428 (2015).
Acknowledgements
R.E. was supported by a Senior Investigator Award from the National Institute of Health Research. L.C.H. was funded by Deutsche Forschungsgemeinschaft, SFB-655 and Transregio-67.
Author information
Authors and Affiliations
Contributions
Introduction (R.E.); Epidemiology (T.W.O.); Mechanisms/pathophysiology (L.C.H.); Diagnosis, screening and prevention (B.L.); Management (I.R.R.); Quality of life (D.T.G.); Outlook (S.R.C.); Overview of Primer (R.E.).
Corresponding author
Ethics declarations
Competing interests
R.E. has received consulting fees from Amgen, AstraZeneca, GlaxoSmithKline (GSK), Immunodiagnostic Systems, Ono Pharma, Lilly and Roche Diagnostics, and grant support from Amgen, Immunodiagnostic Systems, Lilly and AstraZeneca. L.C.H. has received honoraria for serving on the advisory board and for giving lectures from Amgen, Eli Lilly, Merck, Novartis and UCB. B.L. has received consulting fees from Amgen, Merck, Eli Lilly and UCB, and grant support from Novo Nordisk, Eli Lilly and Orkla Health. I.R.R. has received research funding and honoraria from Merck, Amgen and Novartis. S.R.C. serves as a consultant to Amgen, Radius, Merck and Eli Lilly about the design of studies and interpretation of results, and has received grant support from Amgen for systematic review of medical risk factors for hip fracture. He has not received funds for lectures or other promotional activities. D.T.G. and T.W.O. have no conflicts of interest or competing interests to report.
Rights and permissions
About this article
Cite this article
Eastell, R., O'Neill, T., Hofbauer, L. et al. Postmenopausal osteoporosis. Nat Rev Dis Primers 2, 16069 (2016). https://doi.org/10.1038/nrdp.2016.69
Published:
DOI: https://doi.org/10.1038/nrdp.2016.69
This article is cited by
-
The impact of diabetes, anemia, and renal function in the relationship between osteoporosis and fasting blood glucose among Taiwanese women: a cross-sectional study
BMC Women's Health (2024)
-
The serine synthesis pathway drives osteoclast differentiation through epigenetic regulation of NFATc1 expression
Nature Metabolism (2024)
-
Transcriptional reprogramming during human osteoclast differentiation identifies regulators of osteoclast activity
Bone Research (2024)
-
Effects of stepwise administration of osteoprotegerin and parathyroid hormone-related peptide DNA vectors on bone formation in ovariectomized rat model
Scientific Reports (2024)
-
Mind Gaps and Bone Snaps: Exploring the Connection Between Alzheimer’s Disease and Osteoporosis
Current Osteoporosis Reports (2024)